Abstract
Background
Diagnosis of subarachnoid haemorrhage, a neurosurgical emergency in patients with headache remains a logistical challenge. The rationale of the traditional pathway of cerebrospinal fluid xanthochromia analysis following negative computed tomography head scans to exclude subarachnoid haemorrhage has been challenged by the increasing accuracy of modern computed tomography scanners.
Aim
We set out to establish whether our xanthochromia service was adding value to the diagnostic pathway for subarachnoid haemorrhage or whether it was acting merely as a supportive test.
Method
A retrospective audit of all cerebrospinal fluid requests received since the inception of Xanthochromia service at a tertiary trauma centre. Cases interpreted as being consistent with subarachnoid haemorrhage based on cerebrospinal fluid xanthochromia analysis were selected for in-depth review from the total number of cases.
Results
In total 660 requests were received for cerebrospinal fluid xanthochromia between August 2009 and July 2012. A total 28 of these were interpreted as being consistent with subarachnoid haemorrhage. Only 18 (64.3%) of requests were deemed appropriate as the clinical presentation in the remaining 10 (35.7%) was strongly suggestive of other causes of headache. A final clinical diagnosis of subarachnoid haemorrhage was made in 11 of the 18 patients who had cerebrospinal fluid xanthochromia requested appropriately. From these 11, five (45%) were deemed initially computed tomography negative and cerebrospinal fluid analysis led to final correct diagnosis of subarachnoid haemorrhage and appropriate surgical management.
Conclusion
Despite improved computed tomography scanning technology, cerebrospinal fluid xanthochromia interpretation aids in the definitive diagnosis of subarachnoid haemorrhage. When requested appropriately cerebrospinal fluid xanthochromia analysis remains a vital service as results impact on clinical decision making, especially when computed tomography scan results are equivocal and is also important in later presenting patients when computed tomography accuracy decreases.
Introduction
Of all patients presenting to the emergency department (ED) with a headache, approximately 1% will have a subarachnoid haemorrhage (SAH). 1 SAH is a spontaneous intracranial bleed associated with high mortality and morbidity, with up to 10% of patients dying immediately without any warning symptoms. 2 In the first six months after the event, mortality rates can approach 60% and survivors are often left with permanent neurological deficits. 3 Urgent and accurate diagnosis is essential as some of these patients will have an identifiable cause for the SAH amenable to neurosurgical intervention which can increase survival and improve the outcome.
Headache is one of the most common presenting complaints in the ED 4 and it is important to differentiate SAH from other causes of headache such as migraine and meningitis. Clinical diagnosis is difficult because SAH has few distinguishing signs: up to 50% of patients with SAH have no neurological signs. 2 The exclusion of SAH in patients presenting to the ED with a headache therefore rely upon specialized radiological and biochemical investigations.
A computed tomography (CT) head scan is the first-line investigation when SAH is suspected. The CT head is positive in up to 98% of cases of SAH within the first 12 h of presentation but drops to 50% after a week. 5 Therefore, if SAH is suspected and the CT head is non-diagnostic, patients are required to undergo a lumbar puncture (LP) so that the cerebrospinal fluid (CSF) can be analysed for the presence of xanthochromia (yellowish discoloration of CSF). However, the reliability of xanthochromia analysis is also time dependent. The reliability of CSF xanthochromia detection after two weeks or more remains debatable.6,7 General consensus suggests that patients presenting two weeks after suspected SAH should be referred urgently to a neuroscience unit for consideration of further investigation 8 as xanthochromia is detected in only 70 and 40% of cases of SAH after three and four weeks, respectively. 1 However, if CT head and CSF analysis are both normal within two weeks of headache then SAH can be excluded.
Not all hospitals offer a 24-h CSF xanthochromia detection service. Therefore, patients are often admitted overnight or over the weekend to await results: this causes delay in the diagnosis of SAH or in the discharge of non-SAH patients. It has been proposed that, with new third-generation CT technology, a CT head completed within 6 h of the onset of symptoms suggestive of SAH has a sensitivity approaching 100%. 3 Studies suggest that with sensitivities this high, CT head alone could be considered a ‘rule out’ test for SAH without the need for a LP and analysis of the CSF for xanthochromia. 2 This would be of benefit to both patient and hospital. It would obviate the need for LP and its associated risks to the patient such as pain, headache and infection. It would also expedite patient discharge as there would be no need to wait for the mandatory 12 h after the onset of symptoms to perform the LP, nor would it be dependent upon a service that is infrequently available 24 h a day which often delays the result. 2
Our hospital is an acute care facility that serves a local inner London population of nearly 600,000 and has a tertiary neurosurgical unit that serves most of the southeast of England. Our CT scanners are third-generation multiscanners (4–16 slices per rotation). The current diagnostic algorithm for suspected SAH stipulates the need for a LP and CSF xanthochromia analysis following a non-diagnostic CT head. During the audit period, our clinical biochemistry department provided a CSF xanthochromia detection service during office hours (Monday to Friday 09:00 to 16:00) only. There was no service overnight or at weekends. Thus, patients with suspected SAH who had a CSF sample taken outside office hours were admitted for monitoring until the results were available.
Aims
To assess the effect of the CSF xanthochromia detection service on the management of patients with suspected SAH. Due to its limited availability, we wanted to assess if it was directing patient management or simply providing supportive evidence of a diagnosis of SAH that had already been made by a CT head. We also looked at the feasibility of offering a 24-h laboratory xanthochromia service.
Methods
We performed a baseline retrospective audit of all xanthochromia requests received by our laboratory since the inception of our xanthochromia service in August 2009 until July 2012. The audit was approved by the clinical effectiveness committee (reference number CASS 3028).
CSF xanthochromia was tested using a UV–Vis spectrophotometer (PerkinElmer Lamba 35, USA) with Winlab interpretation software (PerkinElmer) and reported in accordance with the 2008 revised national guidelines for analysis of CSF for xanthochromia. 6 We reviewed medical records of all patients whose CSF analysis was consistent with a diagnosis of SAH. The data collected included the time of onset of symptoms, the time of presentation to the ED, the clinical history and examination findings, the time that the initial CT head was performed, the time of the LP and the clinical details of the CSF xanthochromia request. We reviewed all specialist imaging performed for each patient, such as CT angiography and magnetic resonance imaging (MRI). We recorded the final clinical diagnosis from the medical records of each patient. We also reviewed whether patients underwent any interventional neurosurgical management and their 60-day mortality.
We determined the request for CSF xanthochromia analysis to be appropriate if the working or differential diagnosis included SAH. This was ascertained from the clinical notes and from the request form for xanthochromia analysis. If there was a high clinical suspicion of an alternative diagnosis such as meningitis, trauma or neuropathy and there was no reference to ‘suspected SAH’ in the clinical notes or the request forms, we determined these requests as inappropriate.
Results
During the 36-month period, there were a total of 660 requests for CSF xanthochromia analysis of which 28 were consistent with SAH, based on 2008 guidelines.
6
Figure 1 shows the follow of all 28 patients in study through radiology, CSF analysis and final clinical diagnosis.
Follow chart of patients in study through radiology, cerebral spinal fluid (CSF) results and final clinical diagnosis.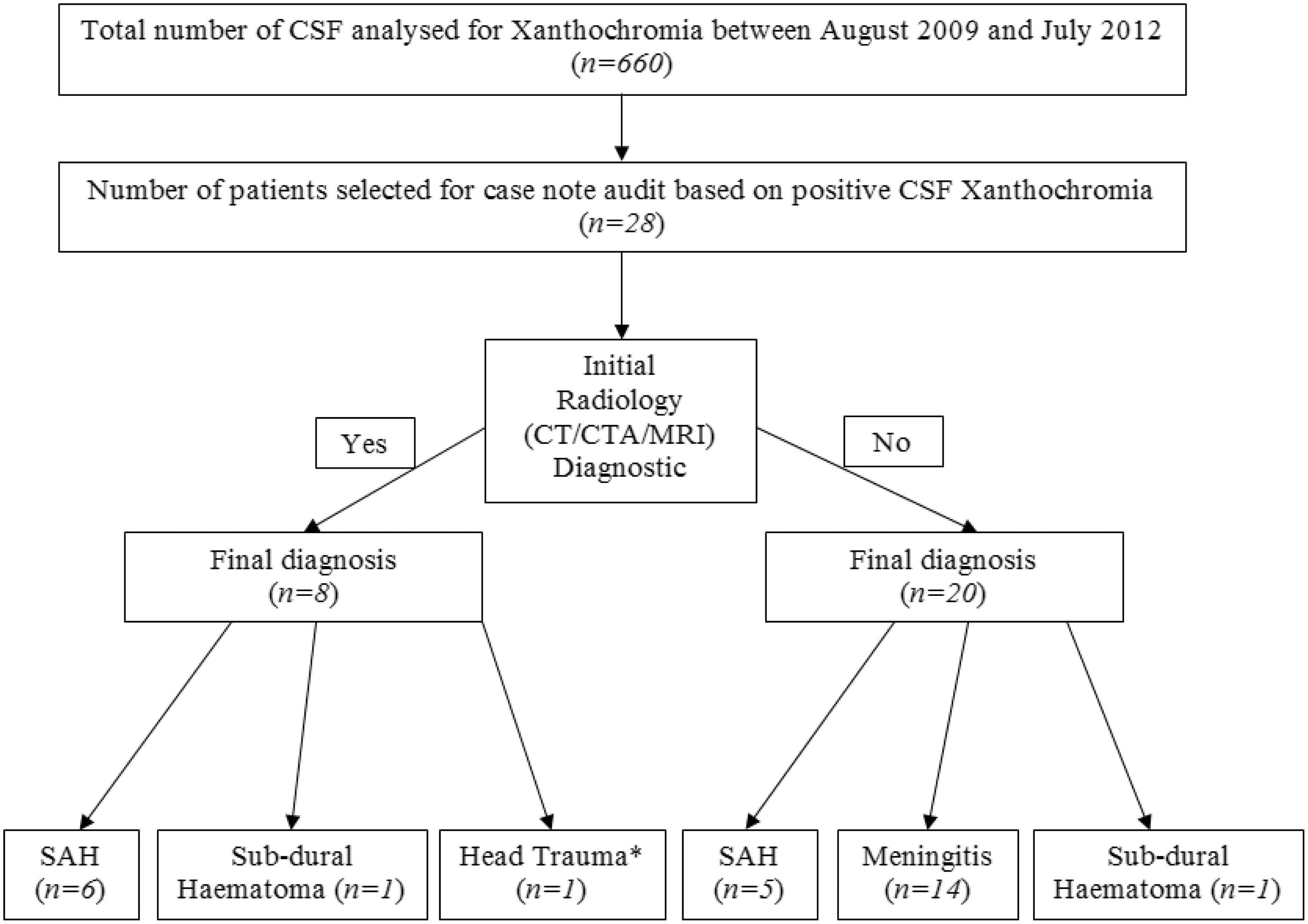
The time from onset of symptoms to presentation to the ED was 44 h (4 h to 21 days) mean (range). In 10 of the 28 patients (35.7%), the request for CSF xanthochromia was deemed inappropriate because there was a high clinical suspicion of meningitis (n = 8), sub-dural haematoma (n = 1) and trauma (n = 1), and SAH was not suspected in any of these patients.
We deemed that the remaining 18 requests appropriate, as at the time of presentation the diagnosis of SAH was among the differential diagnosis. From these 18 requests, the final clinical diagnosis of SAH was made in 11 patients in total. The remaining seven patients had other diagnosis including meningitis (n = 6) and sub-dural haematoma (n = 1). In all seven of these patients, the clinical presentation was of headache with vague accompanying symptoms, thus SAH was still amongst the possible diagnoses.
In the 11 patients with a final clinical diagnosis of SAH, six had positive radiological evidence of a bleed (either from CT head, CT angiogram or MRI) prior to the availability of CSF results. Therefore, CSF results in these six patients did not alter management. In the remaining five patients who had final clinical diagnosis of SAH, there was no radiological evidence of SAH prior the availability of CSF results. In one of these patients, the CT head was subsequently reviewed by a different radiologist in the light of CSF xanthochromia and the consequent CT report did note evidence of SAH. This patient proceeded to coil embolization. The other four patients, who were CT-head negative but CSF xanthochromia positive, underwent CT angiography that confirmed SAH secondary to cranial artery aneurysms. All of these patients were managed surgically.
The time from onset of symptoms to presentation of the four CT-negative patients with a final diagnosis of SAH was 62 h (4 h to 14 days) mean (range). This compared to a time of 27 h (6 h to 5 days) mean (range) for patients with SAH whom were CT head positive.
In all, eight of the 11 patients with a clinical diagnosis of SAH haemorrhage required surgical treatment. Six of these patients underwent coil embolization and two underwent clipping of the aneurysm. All eight patients improved following surgery and were discharged from hospital between one to three weeks following procedure. The remaining three were managed conservatively due to the delay in presentation and all made full recovery. To date no mortality has been reported in any of the patients diagnosed with SAH either managed conservatively or surgically.
Discussion
We have demonstrated that CSF xanthochromia detection plays a vital role in SAH diagnosis in spite of improved CT-head technology in a tertiary trauma referral centre. A number of recent multicentred studies have found that modern third-generation CT technology are extremely sensitive in detecting SAH when performed within 6 h, with some studies reporting sensitivities of up to 100%. 2 This has led to questions about the traditional model of excluding SAH with CSF xanthochromia detection. Only four of the 28 patients included in our study had initial CT head performed with 6 h of onset of headache. This was due to late presentation following the onset of headache. The time of presentations following onset of headache in those presenting after 6 h ranged from 24 h to two weeks. Only one of the 11 patients with SAH presented within 6 h.
Our results are in keeping with previous studies that showed the sensitivity of CT-head results to fall after 6 h as five out of 11 (45%) of our patients eventually diagnosed with SAH were not detected on initial CT head. In one of these patients, however, CT-head report was revisited by the Consultant Neurologist in light of the CSF xanthochromia findings who deemed along with a second Radiologist that there was in fact evidence of blood in the sylvian fissure. This particular patient had an initial CT performed 24 h after the onset of headache.
In four patients that were truly CT-head negative but positive for xanthochromia, the CSF result lead to further imaging by either CT angiograph or MRI that confirmed a diagnosis of SAH and appropriate management. Our results show that despite improving CT-head technology, the natural history of SAH means that CT-head sensitivities will remain low due to late presentation. Our results showed that patients with subarachnoid haemorrhage were more likely to be CT-head negative when presenting later. This adds to the argument that CSF xanthochromia is a vital service, especially when patients’ presentation is delayed. Interestingly, one of the patients in our study who had a normal CT head but was subsequently diagnosed with SAH based on CSF findings had the CT head performed within 6 h of onset of symptoms. This finding challenges the notion of using early CT head alone to exclude SAH. Our work also shows that patients in our inner city area are unlikely to present this early even though their symptoms may be severe. Patients almost invariably presented late meaning that there was selection bias as our study as it would have only included patients well enough to present to the ED and then undergo LP. This may also explain the 100% survival in our patients with SAH whether managed conservatively or surgically.
We found a large number of inappropriate requests. We deemed this was due to our computer-based requesting system which displays all tests available on CSF samples simply by entering those three letters. Thus, there may be an inclination for more junior medical staff to request CSF xanthochromia even in patients where meningitis is highly suspected. At the time of the audit there were no control measures in the laboratory for CSF xanthochromia requests, all requests samples were analysed on receipt. Following this audit, we have introduced measures to reduce inappropriate testing, whereby requests are reviewed by the Duty Biochemist. Any requests deemed inappropriate are cancelled after consultation with the requesting team.
Our audit highlighted a high rate of false positives, where xanthochromia was detected in CSF in the absence of SAH (60.7%). The final diagnosis in the majority of these cases was meningitis. CSF protein concentrations of at least 1.5 g/L as seen in many infectious and inflammatory conditions, as well as traumatic LP that contain more than 100,000 RBCs/mm3 will result in xanthochromia. 9 Oxyhaemoglobin alone without increased bilirubin can be detected on spectrophotometry in the absence of SAH as a result of traumatic LP and in vitro haemolysis. This potential analytical interference can be minimized by adhering to the revised guidelines, which recommends to collect sequential specimens when possible and use the least blood-stained CSF sample for xanthochromia analysis. 6 We often do not receive the third or fourth collection from the LP.
Finally, we also assessed if there would be an economic argument for providing a 24 h CSF xanthochromia service. In order to facilitate this service, it would require Clinical Biochemistry out of hour’s staff to be trained in the analysis and interpretation of CSF xanthochromia scans. We estimated the cost of providing this service to be approximately £11,000 per year. This cost analysis is based on the additional equipment and reagent cost envisaged as well as the cost to train the Clinical Biochemistry out of hour’s workforce in the analysis and interpretation of CSF xanthochromia scan. The cost of an acute in-patient bed per night at the trust is around £748. This means that we would need to analyse just 15 samples a year out of hours in order for one reduced overnight stay in order to make the service economically viable. At present our service analyses between 200 and 250 samples per year out of which approximately 100 requests are made out of hours therefore these criteria would be comfortably met. We have now successfully implemented a 24 h CSF xanthochromia screening service in our trust.
Conclusion
Our findings confirm that despite improving CT scanning technology, CSF xanthochromia detection remains a vital service for the diagnosis of SAH. CSF xanthochromia results impact clinical decision making, especially when CT scan results are equivocal. The non-availability of a 24 h CSF xanthochromia service does lead to increased hospital admission and length of hospital stay. Thus, there is a strong economic argument to implementing round-the-clock service in tertiary trauma centres.
Footnotes
Acknowledgements
We would like to thank Laura Gonzalez from the clinical biochemistry team at King’s College Hospital, for her administrative assistance in the present audit.
Declaration of conflicting interests
None declared.
Funding
Not applicable.
Ethical approval
Not applicable.
Guarantor
RPV.
Contributorship
The authors’ contributions were as follows: AG: collection of data, analyses and interpretation of data, drafting of the manuscript, critical review of the manuscript’s content, and approval of the final version submitted for publication; JOS: collection of data, analyses and interpretation of data; and approval of the final version submitted for publication. JM: interpretation of data, critical review of the manuscript’s content and approval of the final version submitted for publication; JK: original idea, design, critical review of the manuscript’s content and approval of the final version submitted for publication; RPV: design, interpretation of data, drafting of the manuscript, critical review of the manuscript’s content and approval of the final version submitted for publication.
