Abstract
Background
The clinical usefulness of the serum-free light chain assays has expanded since their first description, and further applications other than plasma cell dyscrasia are emerging. Currently, we have the ability to perform the measurements with two certified methods: the Freelite™ assay (The Binding Site Ltd, Birmingham, UK) and the new N Latex free-light chain assay (Siemens, Germany). In the present study, we investigated the impact of free light chain concentrations and structures on their quantification, performed with both tests.
Methods
A total of 524 serum samples from 497 patients from our routine laboratory were analysed with the Freelite™ and the N Latex free light chain assay. The results were compared in two subgroups: with or without monoclonal component. Twenty-four samples were subsequently investigated for the presence of dimeric and monomeric free light chain with sodium dodecyl sulphate polyacrylamide gel electrophoresis and densitometric quantification.
Results
Methods comparison showed that the Pearson rank correlation coefficients were 0.90 for polyclonal k and 0.91 for polyclonal λ free light chain. Conversely for monoclonal immunoglobulins, the Pearson rank correlation coefficient was lower with 0.82 for kM >500 mg/L and 0.56 for λM >500 mg/L. Furthermore, densitometric quantification of the involved monoclonal free light chains showed that both assays do not reflect the Coomassie-stained protein mass.
Conclusion
Samples containing high amounts of a single pathologic free light chain may not be considered like a sample containing a sum of different polyclonal free light chains. Indeed, free light chain dimerization leads to different scatter efficiency of macromolecular complexes.
Introduction
Immunoglobulin-free light chains (FLCs) have long been considered a by-product of plasma cells, but in the last five years they have become most popular by being a useful marker in plasma cell dyscrasia (PCDs). To evaluate the presence of a monoclonal component in a serum sample, 1 serum protein electrophoresis (PEL), serum immunofixation (IFE) and serum immunoglobulin FLC assay are at present the gold standards. In 2001, The Binding Site developed the FLC quantitative assay (the Freelite™ Assay, The Binding Site Ltd, Birmingham, UK) and from 2009 this assay is recommended by the International Myeloma Working Group for the evaluation and management of multiple myeloma (MM) and related clonal PCDs. 2 This test uses sheep polyclonal antibodies directed against the ‘hidden’ epitopes of the FLC, recognizing only FLC that are untied to the heavy chains. 3 It detects low concentrations of FLCs and can measure the k FLC to λ FLC ratio. The k/λ ratio plays an important role in screening, diagnosis, monitoring and prognosis of clonal PCDs potentially reducing the amount of bone marrow biopsies.1,4 Recently, the increase of polyclonal FLC has also been linked with immunoglobulin imbalance associated with diseases: elevated polyclonal FLC concentrations might result from non-B-cell malignancy increased production or alternatively from reduced clearance due to renal, hepatic or reticulo-endothelial dysfunction.2,5,6 The Binding Site assay is based on the use of sheep polyclonal antisera, able to recognize polyclonal FLC and a broad spectrum of paraproteins. Recently, a new assay has been marketed from Siemens, the N Latex FLC assay (N Latex FLC, Siemens, Camberley, Surrey, UK) based on the mixture of several monoclonal antibodies, also raised against the FLC hidden portion. 7 First evidences suggest that the Freelite™ assay and the N Latex assay are both useful; however, many aspects need to be studied. 8 The difference in the formulation of the two assays is a polyclonal serum versus a monoclonal cocktail. It is not known how many clonal antibodies are present in the N Latex cocktail and how the sheep are chosen for immunization. In our experience, both assays have advantages and disadvantages: monoclonal antibodies are useful for inter assay reproducibility but the broad spectrum of potential paraproteins could escape detection; polyclonal antibodies are probably composed of a wider range of paraprotein-recognizing antibodies but their inter assay reproducibility could be more variable.
The FLC assay may be the only mean of detecting a disease marker in some disorders such as non-secretory MM, 9 amyloid light-chain (AL) amyloidosis since low monoclonal protein (M-protein) concentrations may not be detected with conventional techniques and light chain MM, in which M-protein consists only of FLC. 10 The concept of low monoclonal concentrations of FLC paraproteins is hard to discuss because FLC multimers might not be visualized as a monoclonal band with PEL and quantified with the immunometric assays. Immunonephelometric assays do not use standardized controls that can distinguish between FLC monomers and polymers and these two structures can confound the interpretation of serum FLC assay. 11 Hence, we investigated the incidence of monoclonal k- and λ FLC on the correlation between the two assays and evaluated how the presence of dimers could affect the FLC nephelometric measurements in discordant samples.
Materials and methods
Patients
The institutional review board of Azienda Ospedaliera Spedali Civili of Brescia (AOSCB) approved our study in adherence with the Declaration of Helsinki. All traceable identifiers from collected serum samples were removed before freezing at −80℃ to protect patient confidentiality. A total of 524 serum samples from 497 patients were tested for routine laboratory screening or follow-up of monoclonal gammopathy in the Protein Profiling Laboratory Unit from the Laboratory of Biochemical Chemistry. Analysis comprised serum PEL and/or urine PEL with additional IFE and/or urine IFE and FLC quantification. 7 Samples were collected either from patients with diagnosed monoclonal gammopathy (MM, AL amyloidosis, monoclonal gammopathy of undetermined significance and Waldenstrom’s macroglobulinernia) or from patients with a polyclonal increase of immunoglobulins (increased IgG, IgA or IgM) such as patients with renal impairment and patients without a specific diagnosis.
FLC assays
The FLC measurements were performed on a BN II (immunonephelometer, Siemens AG) with the Freelite™ (The Binding Site Ltd, Birmingham, UK) and the new N Latex FLC assay (Siemens, Germany). Commercial kits were used following the manufacturer’s recommendations.
We used reference ranges indicated by each company: N Latex FLC k 6.7–22.4 mg/L and Freelite™ k 3.3–19.4 mg/L. N Latex FLC λ 8.3–27 mg/L and Freelite™ λ 5.7–26.3 mg/L. k/λ N Latex FLC ratio 0.31–1.56 and Freelite™ ratio 0.26–1.65.12,13 Reference FLC ratio was adjusted (0.37–3.1) for patients with renal impairment (serum creatinine concentration >1.2 mg/dL).
Statistics
All values were normalized with a logarithmic function.
The linear regression, the normalized median differences between the two methods, Bland–Altman Test, Pearson rank correlation coefficients and concordance correlation coefficient (CCC) were estimated. Concordance analysis of results was performed on the absolute value of k FLC and λ FLC and the k/λ ratios. The evaluations were also performed on two selected groups: ‘with’ (monoclonal) or ‘without’ monoclonal gammopathy (polyclonal). The ‘Freelite™’ method was considered as reference test. Monoclonal samples were divided into two groups: FLCs <500 mg/L and FLCs >500 mg/L. All statistic analyses were performed using GraphPad Prism 6 (©2013 GraphPad Software, Inc. All rights reserved) and Microsoft Excel (Microsoft Corporation).
Sodium dodecyl sulphate polyacrylamide gel electrophoresis, immunoblotting and antibodies
We selected 14 monoclonal patients with high FLC serum concentrations, tested by PEL, IFE and presenting a high discrepancy in FLC quantification with the two methods. As controls we selected nine polyclonal patients. Serum samples from these patients were analysed to investigate the monoclonal peak of FLC. To visualize the FLC signal we performed electrophoresis under native conditions of patients’ serum samples, commercially available purified human k or λ FLC (Bethyl Laboratories, USA) and bovine serum albumin (BSA, Sigma): samples were resuspended in non-reducing sample buffer (50 mM Tris, 2% sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS), 10% glycerol, 0.1 Bromophenol Blue pH 6.8) and boiled for 5 min at 95℃. Afterwards, samples were run in a 12.5% acrylamide–bisacrylamide SDS 0.4% gel and stained with Coomassie brilliant blue 1 h at room temperature or transferred for 1 h onto a polyvinylidene difluoride (PVDF) membrane, blocked overnight with 5% fat-free milk, 0.05% Tween-20 in PBS 1X. The PVDF membrane was incubated with sheep anti-λ FLC and sheep anti-k FLC antibodies (Bethyl Laboratories, USA and the Binding Site, UK) for 2 h in PBS Tween 0.05% +1% fat-free milk. The membranes were washed 3 × for 10 min with PBS Tween 0.05% and incubated for 1 h with a donkey anti-sheep secondary antibody (Jackson Immuno Research). Blots were detected using Luminata Classic HRP western substrate (Millipore). Images were acquired using a G:Box Chemi XT Imaging system (Syngene, UK). For densitometric analysis, we took advantage of the Gene Tools (Syngene, UK) software to compare the protein quantification of monoclonal bands.
Serum PEL and IFE
All assays were performed according to the protocols in the Protein Profiling Laboratory Unit from AOSCB. PEL was performed with the serum PEL kit from Interlab on a G26 automated instrumentation following the manufacturer’s suggestions (Interlab, Rome). On the same instrument, IFE was also performed with the dedicated kit (Interlab, Rome). Analysis of densitograms was performed with the Elfolab software.
Results
Polyclonal versus monoclonal ratio
We tested 524 serum samples obtained from 497 patients admitted to the Protein Profiling Laboratory Unit from AOSCB for screening or follow-up of monoclonal gammopathy. In this study, we considered the published reference ranges for Freelite™ assay and the N Latex FLC assay.12,13 As shown in Figure 1(a), the majority of samples was collected from the Hematology Clinical Unit followed by the Nephrology Clinical Unit. The first analysis was to check discrepancies in pathologic ratios. The Binding Site ratio reference ranges were adjusted (0.37–3.1) in those patients with renal impairment to avoid monoclonality misinterpretations in subjects with slowered renal light chain clearance rather than PCDs.
14
We considered concordant all results with the same ratio interpretation (normal or suspicious monoclonality) and discordant all results for which divergent interpretation occurred. Two subgroups were identified as polyclonal or monoclonal after complete evaluation of screening tests. Figure 1(b) clearly shows that the concordance of polyclonal samples analysis was 97% while monoclonal samples presented a higher level of discrepancy (16%). These evidences suggested us to better investigate the incidence of monoclonal protein production on the assays’ comparison.
FLC ratio concordance. (a) Main clinical units that send samples to the Protein Profiling Laboratory Unit for monoclonal gammopathy screening/monitoring. (b) Concordance graphs for the FLC ratios between the N Latex FLC assays (Siemens) and the Freelite™ (The Binding Site) assays. The calculated ratios from 524 serum samples were considered concordant if both were pathological or normal and discordant if one was pathological and the other one normal.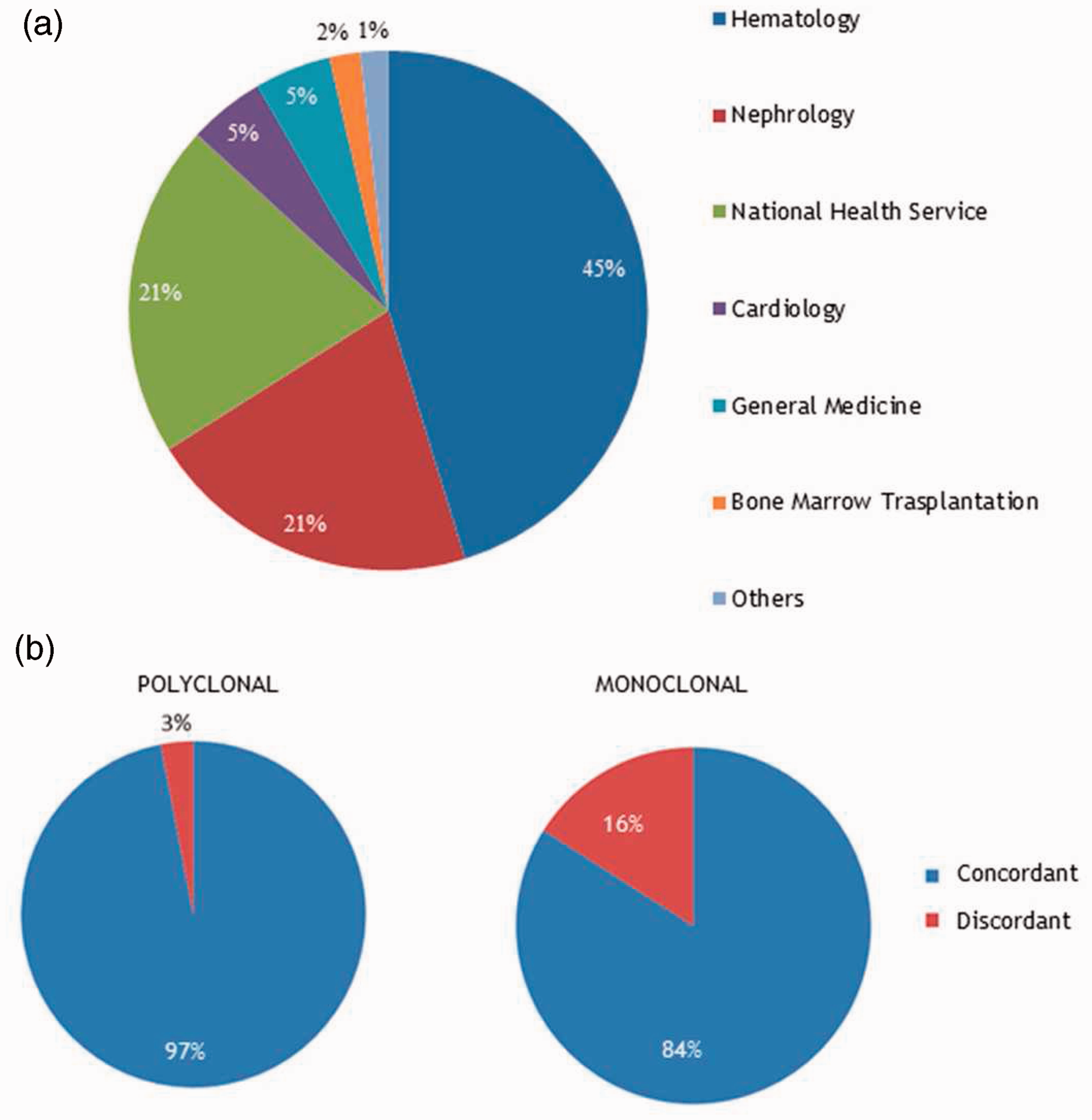
FLC absolute value comparison
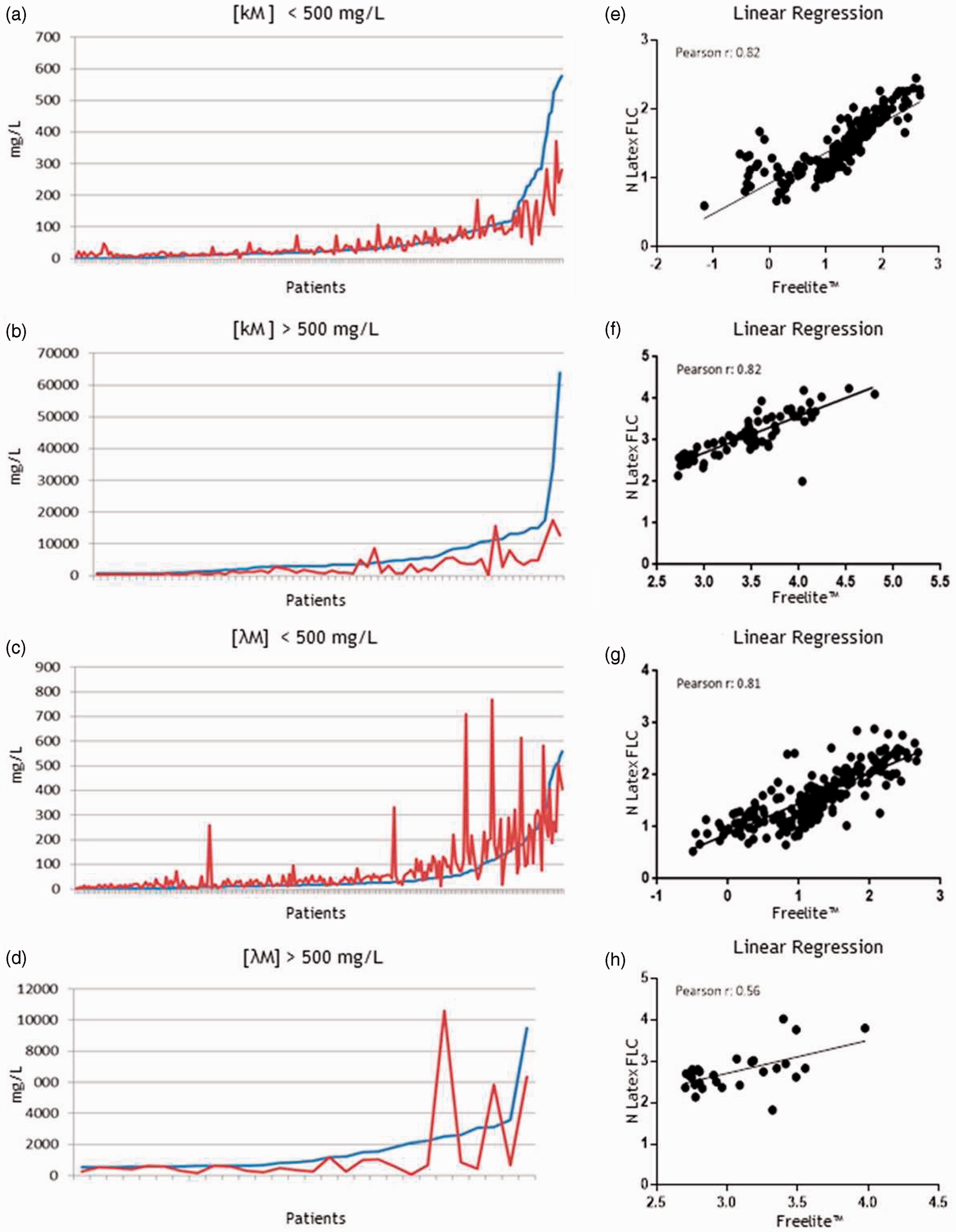
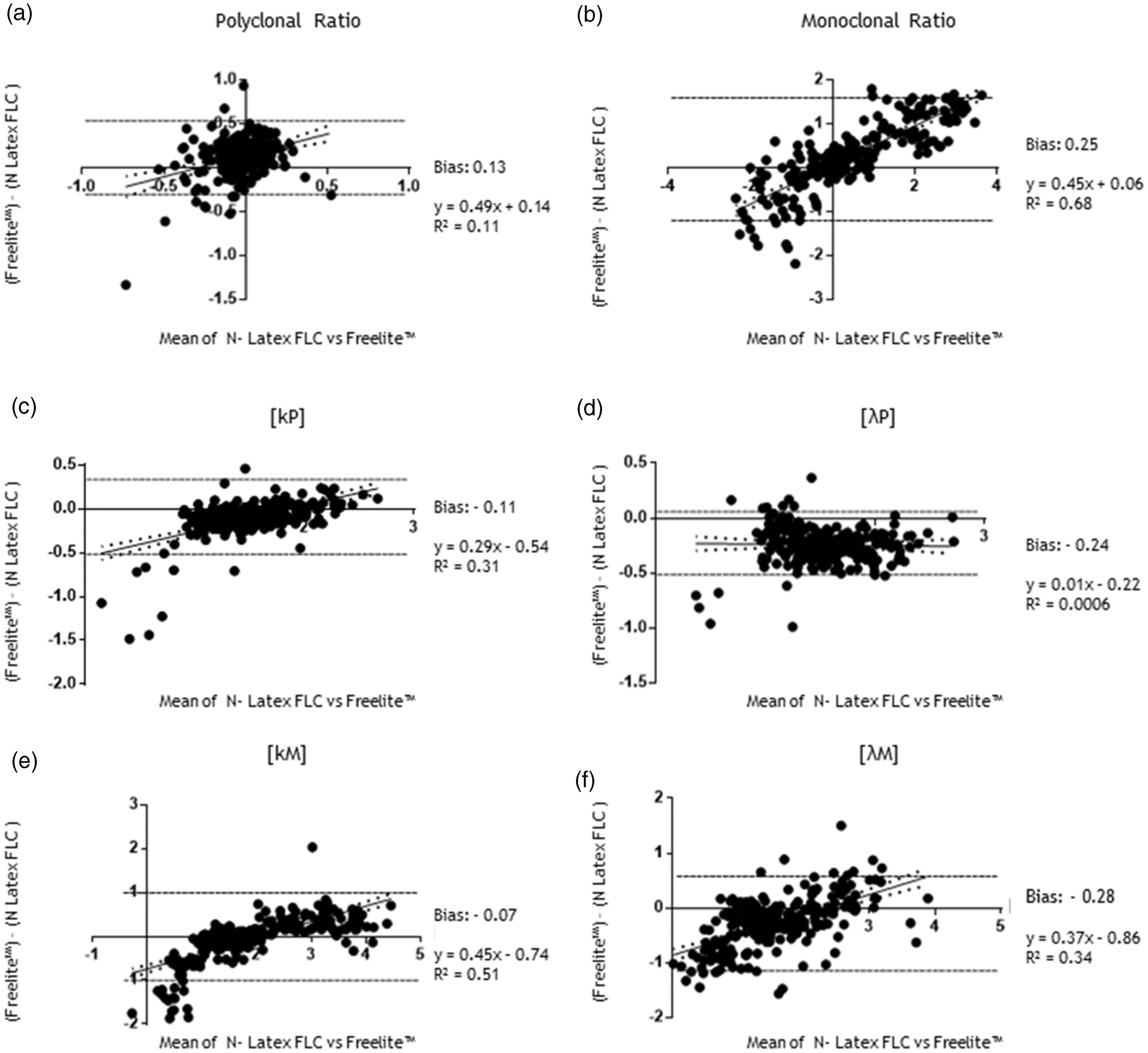
For absolute values comparison, the Freelite™ k FLC and λ FLC measurements were placed in ascending order and the corresponding values obtained with the N Latex FLC assay were also plotted in a linear graph (Figure 2(a) and (b); Figure 3(a) to (d)). The polyclonal immunoglobulin values (kP and λP) showed a good concordance between the two methods while values of monoclonal immunoglobulin (kM < and > 500 mg/L, λM < and > 500 mg/L) had lower concordance. Furthermore, the differences were mainly visible at higher concentration ranges. To further analyse the correlation between the two methods, the k- and λ FLC logarithmic values were plotted and analysed with linear regression (Figure 2(c) and (d); Figure 3(e) to (h)). Methods comparison showed that the Pearson rank correlation coefficients (r) were 0.90 for k polyclonal (kP) and 0.91 for λ polyclonal (λP) FLC. Thus, for polyclonal immunoglobulin, the antibodies provided with the two methods react quite similarly. Conversely, for monoclonal immunoglobulins the Pearson rank correlation coefficient was lower: for both kM <500 mg/L and >500 mg/L, r = 0.82, whereas for λM <500 mg/L, r = 0.81 and for λM >500 mg/L, r = 0.56. Thus, method comparison for kM and λM <500 mg/L showed a moderate correlation while a significant difference was observed for highest λ FLC concentrations. These data were also confirmed with the CCC, a statistical agreement index for two variables. The CCC for kP was 0.83 and for λP was 0.76. The CCC for kM FLC <500 mg/L was 0.61 while CCC for kM FLC >500 mg/L was 0.64. The CCC for λM <500 mg/L was 0.65 while the CCC for λM >500 mg/L was 0.41.The mean differences and 95% limits of agreement between ratios (k/λ FLC) and k-λ FLC absolute values were thus examined with Bland–Altman plots (Figure 4).
15
FLC values comparison of polyclonal samples. Graphs represent the trend of absolute (a) Polyclonal k values (kP) and (b) Polyclonal λ values (λP). Freelite™ absolute values are incrementally plotted with a blue line while the red line represents the corresponding N Latex FLC absolute values. Plots represent regression analysis of (c) kP and (d) λP logarithmic values. The Pearson correlation factor r was also calculated and represented. FLC values comparison of monoclonal samples. Graphs represent the trend of absolute values for monoclonal k ((a) [kM] < 500 mg/L or (b) [kM] > 500 mg/L) and monoclonal λ ((c) [λM] < 500 mg/L or (d) [λM] > 500 mg/L) values: Freelite™ absolute kM or λM values are incrementally plotted with a blue line while the red line represents the corresponding N Latex FLC absolute values. Plots show regression analysis of k ((e) [kM]< 500 mg/L; (f) [kM] > 500 mg/L) and λ ((g) [λM] <500 mg/L; (h) [λM] >500 mg/L) logarithmic values. The Pearson correlation factor r was also calculated and represented. Bland–Altman agreement test. Bland–Altman test plots of (a) Polyclonal and (b) Monoclonal FLC ratios; Bland–Altman test plots of (c) Polyclonal k (kP) and (d) λ (λP) FLC; Bland–Altman test plots of (e) Monoclonal k (kM) and (f) λ (λM) FLC. Linear regression equation, R2 and the bias of the average of the differences were calculated for every graph.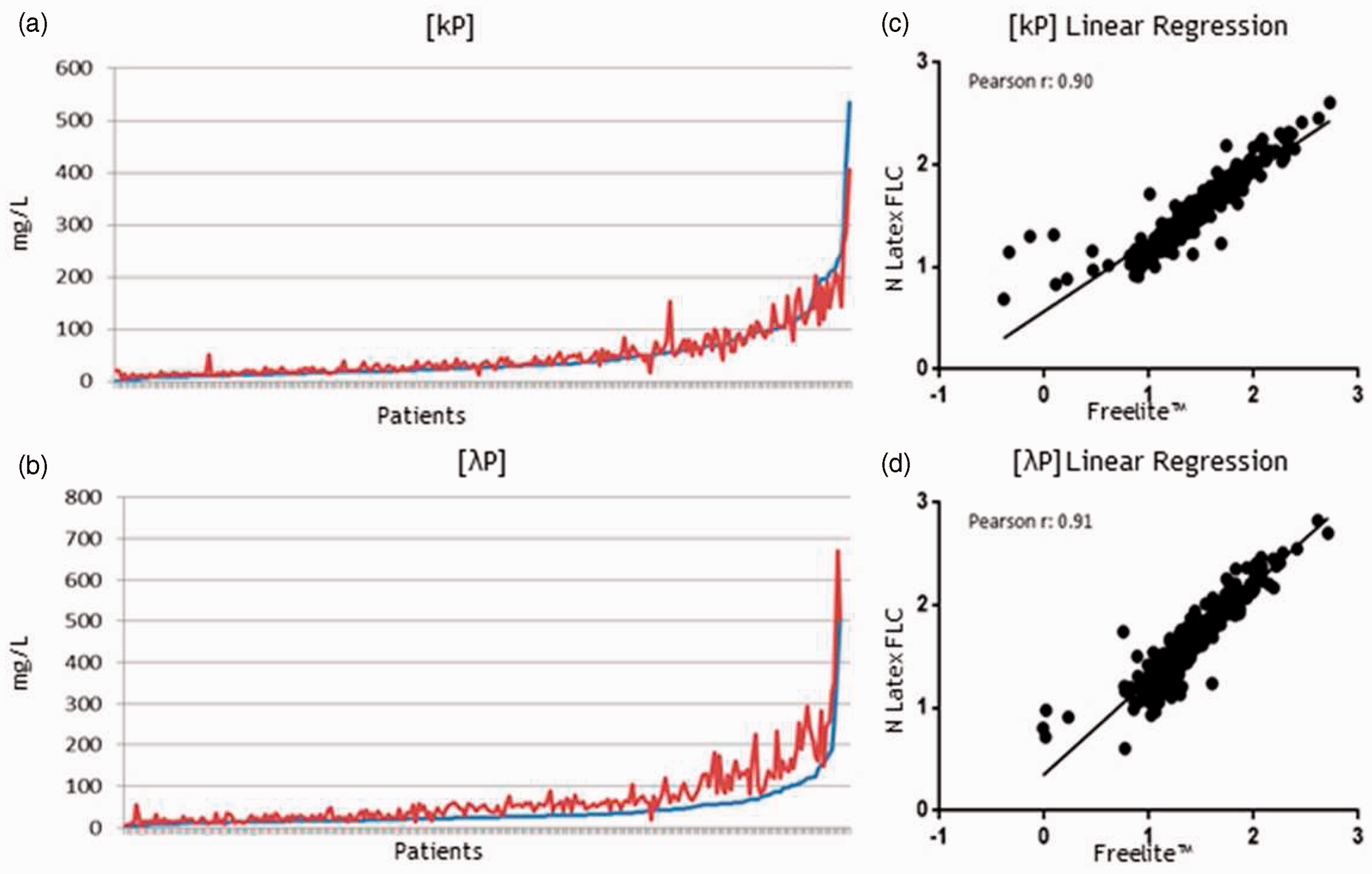
The resulting plots were analysed with linear regression to study their distribution: a quite good correlation between the two methods is represented by a straight line with a zero value slope (Figure 4).
Ratios from polyclonal immunoglobulins distributed well with 0.13 bias and R2 = 0.11, thus with good agreement between the two methods (Figure 4(a)). Lower agreement values with 0.25 bias and R2 = 0.68 were obtained for monoclonal ratios (Figure 4(b)). Concerning absolute values k FLC and λ FLC distributed differently among monoclonal (kM and λM) or polyclonal (kP and λP) samples. The N Latex FLC assay values are higher than the Freelite™ values (negative bias) but good correlations for kP FLC (0.29 slope and R2 = 0.31, Figure 4(c)) and for λP FLC (0.01 slope and R2 = 0.0006, Figure 4(d)) were obtained. The monoclonal FLC distribution shows higher discrepancies in the concentration range up to 300 mg/L (0.45 slope and R2 = 0.51 for kM, Figure 4(e); 0.37 slope and R2 = 0.34 for λM, Figure 4(f)). Altogether, our data highlight that the two methods behave differently in monoclonal samples, particularly at higher concentrations. This confirms that a monoclonal protein, depending on its biochemical–biophysical characteristics could present different stoichiometric interactions with different test antibodies.
True value of circulating FLC
FLC results can be difficult to be interpreted, and we are actually missing a reliable control for FLC measurements.
11
According to our laboratory experience, samples with high amounts of monoclonal k- or λ FLC sometimes behave in a bizarre way, such as no signal on PEL and high amounts on nephelometric assays or negative IFE and extremely abnormal FLC ratios. To better understand these events we selected samples with significant discrepancies in k- or λ FLC measurements, serum or urine IFE. In these samples, we did not find a quantitative correlation between k FLC and λ FLC measurement and PEL monoclonal band quantification.
16
We decided to show patients n. 38, 148, 531, 532 (Figures 5 and 6), n. 123, 231, 390, 128, 25, 76 (Supplementary Figure 1), n. 144, 155, 150, 58 (Supplementary Figure 2). We processed 9 k FLC, 5 λ FLC. As controls we analysed nine polyclonal patients’ serum: n. 394 c and n. 146, 284, 339, 147, 176, 126, 89, 314 (Figure 5; Supplementary Figure 3). The presence of either IgD or IgE was excluded with anti-IgD and IgE antiserum. All FLC measurements were performed with serial dilution steps (from 1:100 to 1:20,000) to exclude aberrant reactions due to antigen excess.
17
Samples were analysed with polyacrylamide gel electrophoresis under non-reducing conditions and Western blot (WB) assay using antibodies anti-k or -λ FLC to visualize the presence of FLC monomers and dimers in patients’ serum. Commercially available purified human k- or λ FLC was also loaded to better distinguish these two FLC structures. As shown in Figure 6(a), FLC monomers were identified in the region between 20 kDa and 25 kDa (white arrow), dimers between 37 kDa and 50 kDa (black arrow).
11
In our experimental conditions, we were not able to recognize trimers or higher molecular weight bands.
Monoclonal samples characterization. FLC quantification (with both the N Latex FLC and the Freelite™ assays), IgA, IgG and IgM quantification, serum (S-IFE) and urine (U-IFE) immunofixation, total protein concentration, monoclonal band densitometric quantification (MC) of patients n. 394c (polyclonal), 38, 148, 531, 532 (monoclonals). Patient’s 38, 148, 531, 532 PEL densitograms, S-IFE and U-IFE are also shown. Monomers and dimers analysis. (a) Western blot analysis of 0.2 µg of purified human k FLC or λ FLC and 15 µg of serum proteins (patient n. 349c (polyclonal), 38, 148, 531 and 532 (monoclonals)) was loaded in a 12.5% polyacrylamide gel and run under non-reducing conditions. All samples were stained with sheep anti-k FLC or sheep anti-λ FLC (The Binding Site) primary antibody. (b) Densitometric profiles of Coomassie brilliant blue stained proteins; 4 µg BSA, 4 µg purified human k FLC and 75 µg patients’ serum proteins were loaded as described. SDS-Page under non-reducing conditions was performed and bands were quantified using Gene Tools program, Syngene. (c) Istogram showing FLCs dimers (44 kDa, black arrow) and monomers (22 kDa, white arrow) quantification using Gene Tools program (Syngene) (green column) compared to Freelite™ assay (red column) and N Latex FLC assays (blue column) quantification.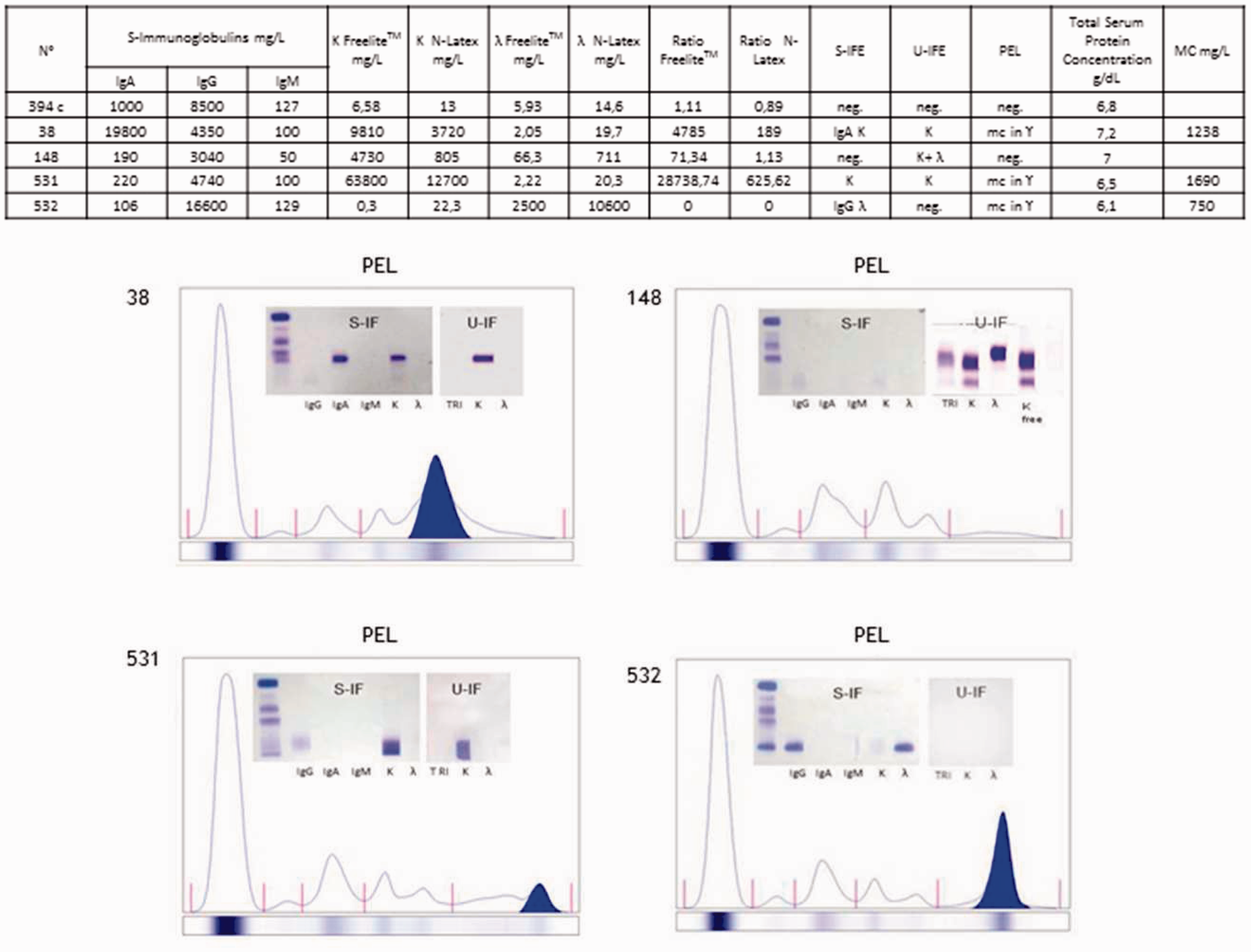
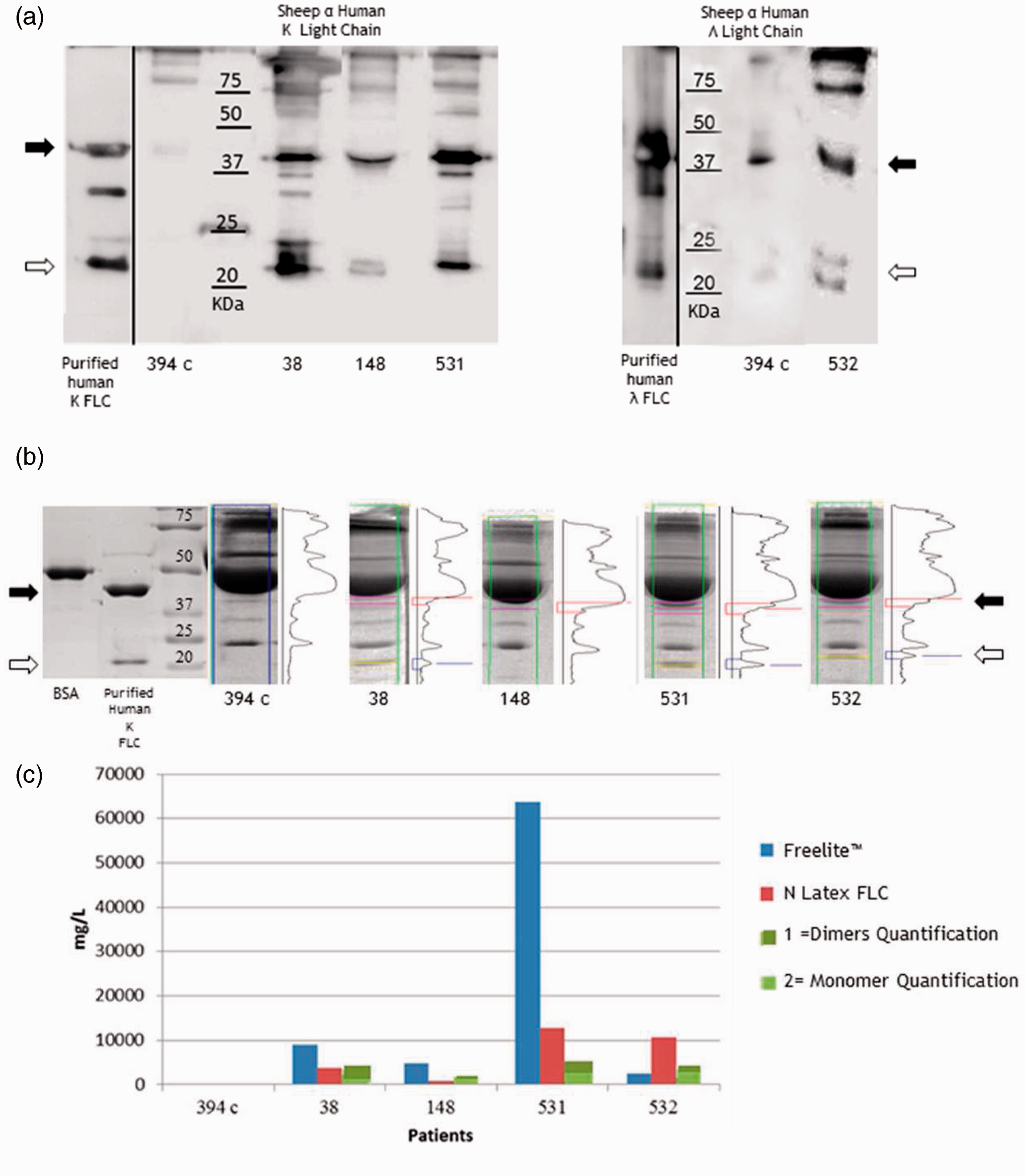
To quantify monomers and dimers, all samples were run in a 12.5% polyacrylamide gel and Coomassie stained in order to avoid antigen-antibody different reactivity that can occur in WB (Figure 6(b); Supplementary Figure 1(b), 2(b), 3(b)). Purified human k FLC confirmed the monomers and dimers FLC molecular weights while the thick band at 50 kDa has been identified as BSA (Figure 6(b)). Thus, we decided to investigate and quantify the 22 kDa and 44 kDa regions to better analyse the bands composition in comparison to polyclonal serum. All data were plotted together with the nephelometric results in Figure 6(c). Our results highlight that for patients producing paraproteins the antiserum could display different antigen–antibody interactions generating macromolecular immunocomplexes giving rise to different light absorption/diffraction properties affecting the analytical measurements.
Discussion
In the present study, we decided to investigate the differences in monoclonal versus polyclonal FLC measurements using the two available commercial kits and to investigate the impact of dimers on monoclonal measurements.
The FLC assay is the most sensitive method to detect monoclonal components in serum samples if not revealed in serum and urine PEL and IFE. 9 However, the recognition of a full range of monoclonal FLC could be hindered by the heterogeneity of FLC molecules, arising from genetic recombination and somatic hyper-mutation of the variable regions after antigen exposure. 18 Single mAb will be unable to detect the diverse range of monoclonal FLC. Hence, the Freelite™ assay 3 was developed using polyclonal antibodies raised in sheep, and the N latex FLC 7 assay contains several mouse monoclonal antibodies for recognition of a wide range of pathological FLC.
We tested the two methods in our routine laboratory work. We chose samples accepted for monoclonal component screening and/or monitoring and we compared the results in two different subpopulations: patients producing polyclonal antibodies and patients producing a single or double monoclonal component. We found a quite good agreement in polyclonal samples for k/λ ratios whereas monoclonal samples were more discordant. Bland–Altman comparison and Pearson index of the absolute values further confirmed these data. These evidences clearly indicate that samples containing high amounts of a single pathologic FLC may not be considered like a sample containing a sum of different polyclonal FLCs. Furthermore, we decided to investigate the effect of dimerization on immunonephelometric quantification.19,20 Multimeric complexes of FLC were detected in the serum from MM patients. 21 Indeed, shape, size and amounts of the epitopes may change after dimerization or trimerization, leading to different scatter efficiency of macromolecular complexes. 11 We hypothesized that monoclonal FLC dimerization could influence the avidity of both polyclonal and monoclonal antibodies and checked for dimers in samples with highly divergent quantifications by WB analysis. We found dimers in all samples showing significant differences between the two nephelometric FLC quantifications (21% of monoclonal samples). We processed some of them with SDS-Page under non-reducing conditions for monomers and dimers densitometric scanning and we decided to show 14 representative monoclonal patients and nine polyclonal patients. Densitometric quantification of monomers and dimers shows a discrepancy with both nephelometric assays. Since we did not consider trimers, we cannot exclude that these structures might influence FLC densitometric quantification. We indeed argue that FLC polymers influence the immunometric quantification, and we are currently working on the use of both reagents in kidney damage prediction. 22 It must be noticed that for monoclonal samples careful management must be performed. If therapies influence FLC dimerization, FLC quantification changes could occur without plasma cell decrease.
Footnotes
Acknowledgements
We would like to thank ‘The Binding Site Italy’ for providing us the anti-FLCs antibodies. We thank Eliana Avanzi for her assistance in this research.
Declaration of conflicting interests
The authors have declared that no competing interests exist.
Funding
This work was supported by the University of Brescia Research fund (ex 60%) and ‘Prima spes onlus’ foundation. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Ethical approval
The institutional review board of the Azienda Ospedaliera Spedali Civili of Brescia approved this study (REC number: SFLC01).
Guarantor
DR.
Contributorship
DR and GDN conceived and designed the experiments. GDN and EC performed the experiments. GDN, AD, EC and LP were involved in samples recruitment. DR, GDN, EC, AD, LP, LC and AR analysed the data. DR and GDN wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
