Abstract
Background
Pregnancy is a risk factor for venous thromboembolism (VTE) due to increased coagulation factor activity and decreased protein S activity. However, thrombosis markers for predicting VTE in pregnancy remain controversial. This study aimed to investigate the relationship between VTE risk and thrombosis markers in pregnant women and to identify markers related to VTE risk.
Methods
Archived plasma samples from 107 pregnant women were used in this study, and the concentrations of D-dimer, fibrin monomer complex (FMC), plasmin-plasmin inhibitor complex, prothrombin time, activated partial thromboplastin time, and fibrinogen were measured. VTE risk was scored according to the Royal College of Obstetricians and Gynaecologists green-top guidelines and the patients were divided into low- or high-risk groups.
Results
The median (range) of risk score for deep vein thrombosis was 2 (0–8), and we defined the high-risk group included those with a score of ≧3. D-dimer and FMC concentrations were significantly higher in the high-risk group than in the low-risk group (D-dimer 4.5 vs 2.6 μg/mL, p = 0.008; FMC 14.6 vs 3.4 μg/mL, p < 0.001). Although D-dimer concentration significantly increased with gestational age (Spearman’s correlation coefficient [rs] = 0.317, p < 0.001), FMC concentration did not (rs = −0.081, p = 0.409). The area under the receiver operating characteristic curve values of D-dimer, FMC, and both D-dimer and FMC for the high-risk group were 0.656, 0.713, and 0.738, respectively.
Conclusions
FMC may be a thrombosis marker related to VTE risk in pregnancy and is potentially preferable over D-dimer concentrations.
Introduction
The number of patients with venous thromboembolism (VTE) is increasing annually, and pregnancy is a risk factor for VTE. Pregnant women are five times more likely to develop VTE than non-pregnant women. 1 Pregnant women with a history of recurrent VTE, medical comorbidities, surgery, and emergency caesarean sections, among others, are particularly at a high risk of VTE.2–5 The Royal College of Obstetricians and Gynaecologists (RCOG) guidelines describe and score VTE risk factors in pregnancy. 6
Thrombin-antithrombin complex (TAT) and D-dimer concentrations are among the tests utilized clinically for predicting and diagnosing thrombosis and for diagnosing VTE in pregnancy. However, these thrombosis markers are not sufficient for VTE diagnosis during pregnancy. Reliable markers for predicting and diagnosing VTE are required to appropriately manage pregnant women.
TAT concentration is known to increase with inflammation, diabetes mellitus, inadequate phlebotomy, and pregnancy.7–9 D-dimer concentration also increases in various scenarios, including pregnancy, even in the absence of clinical thrombosis. 9 The specificity of these thrombosis markers is low; therefore, these tests are not appropriate for VTE diagnosis during pregnancy.
Fibrin monomer complex (FMC) is used to monitor the proteolytic cleavage of fibrinogen by thrombin and is therefore used as a marker of thrombin activity. Thrombin cleaves fibrinopeptides A and B from fibrinogen to create soluble fibrin monomers (SFMs). These SFMs can combine with fibrinogen to form non-covalently associated soluble fibrin monomer complexes (SFMCs). In the final step, SFMCs polymerize via factor XIIIa crosslinking and a thrombus is formed. As FMC concentration reflects thrombin activity and its concentration can be detected earlier than D-dimer concentration, FMC can be used as an alternative for assessing thrombosis. 10
FMC reportedly enhanced VTE detection in pregnancy in some studies, while other studies have found that it is not a useful marker. Thus, the thrombosis markers used to detect VTE in pregnant women remain controversial. Therefore, this study aimed to investigate the relationship between VTE risk and thrombosis markers in pregnant women and to identify markers related to thrombosis risk.
Materials and methods
Patients and clinical samples
This study included 107 pregnant women who were admitted to the obstetrics ward and the maternal fetal intensive care unit of Tottori University Hospital in japan between September 2019 and December 2021. All samples were collected using standard venipuncture blood collection tubes containing one-tenth volume of sodium citrate (3.2%/0.109 M) to attain a final citrate concentration of 0.32%/0.0109 M. Platelet-poor plasma was obtained after centrifugation of citrated whole blood for 15 min at 1500 g. All plasma samples were stored at −80°C and thawed at 37°C immediately prior to the assays. All samples were analysed within one month. This study was approved by the Ethics Committee of the Tottori University Faculty of Medicine (approval number: 19A056).
Risk classification based on RCOG green-top guidelines
Clinical characteristics of patients.
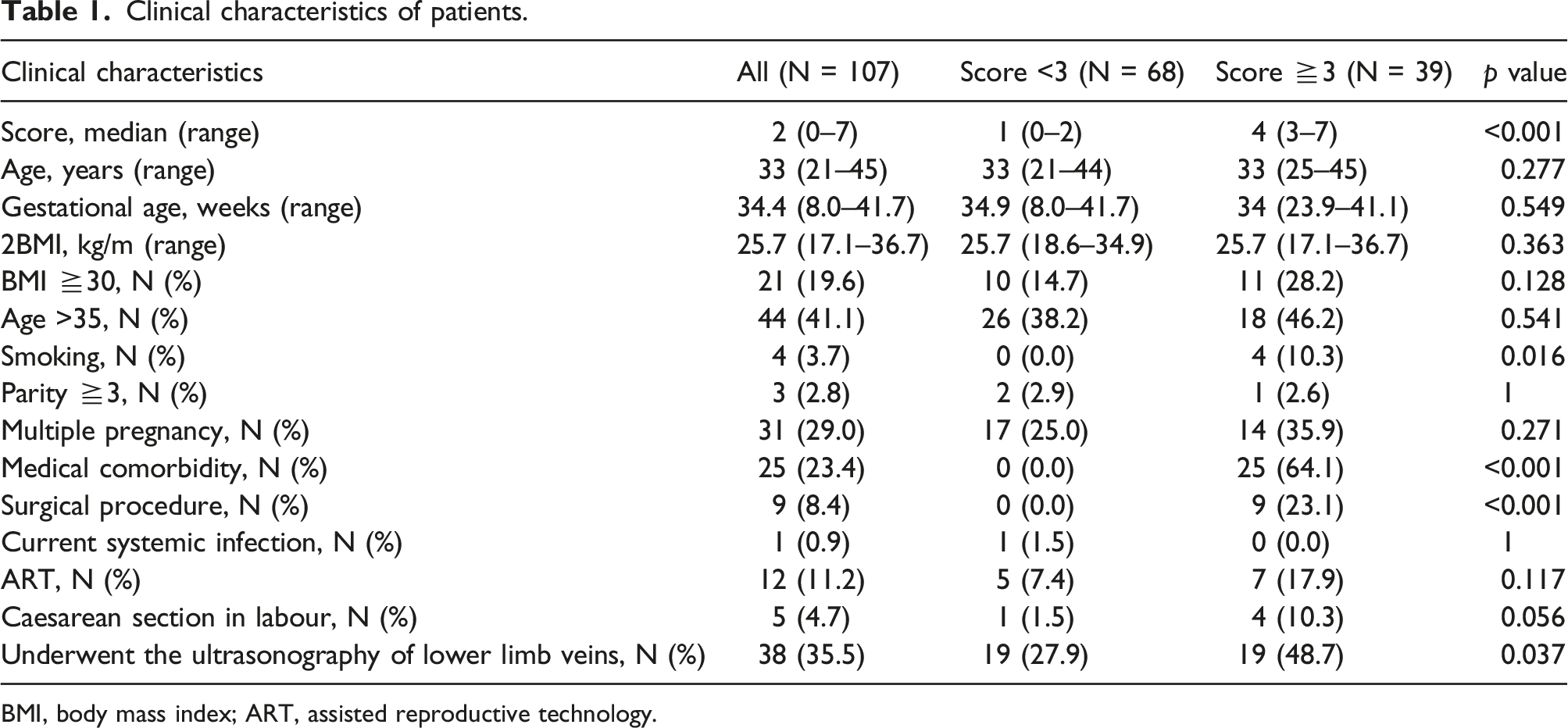
BMI, body mass index; ART, assisted reproductive technology.
Measurement of coagulation/fibrinolysis markers
Reagents and methodology used for study.

Analyses and statistics
All analyses were performed using EZR software (ver. 1.54, Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a Japanese user interface for R (ver. 4.0.3, The R Foundation for Statistical Computing, Vienna, Austria). 11 We compared differences in thrombosis markers using the Mann–Whitney U test. The correlation between gestational age and thrombolysis markers was calculated using Spearman’s correlation coefficient. We also investigated whether D-dimer or FMC concentrations were associated with a high risk for VTE via receiver operating characteristic (ROC) curve analysis. Cases with missing values were not excluded and were treated as missing values.
Results
The median deep vein thrombosis (DVT) risk score of the 107 pregnant women was 2 (range 0–8). The median (range) of age and gestational age were 33 (21–45) and 34.4 (8–41.7), respectively. Other clinical characteristics of the study population (107 pregnancies) are shown in Table 1. The surgical procedures were as follows: Shirodkar operation (n = 6), uterine artery embolization (n = 1), conization (n = 1), and fetal reduction surgery (n = 1). The medical comorbidities included myelodysplastic syndrome (n = 3), gestational diabetes (n = 7), hypothyroidism (n = 5), systemic lupus erythematosus (n = 4), hyperthyroidism (n = 4), and rheumatoid arthritis (n = 2). Thirty-eight pregnant women (35.5%) underwent lower-limb echocardiography and none of them developed thrombosis. The classification of the patient’s gestational age is before 16 weeks of gestation (n = 1); 16–27 weeks (n = 10); after 28 weeks (n = 96).
Distributions of FMC and D-dimer concentrations are shown in Figure 1. Distributions of fibrin monomer complex (FMC) and D-dimer concentrations in pregnant women. Reference intervals: D-dimer < 1.0 μg/mL, FMC < 7.0 μg/mL (reference intervals set by our hospital).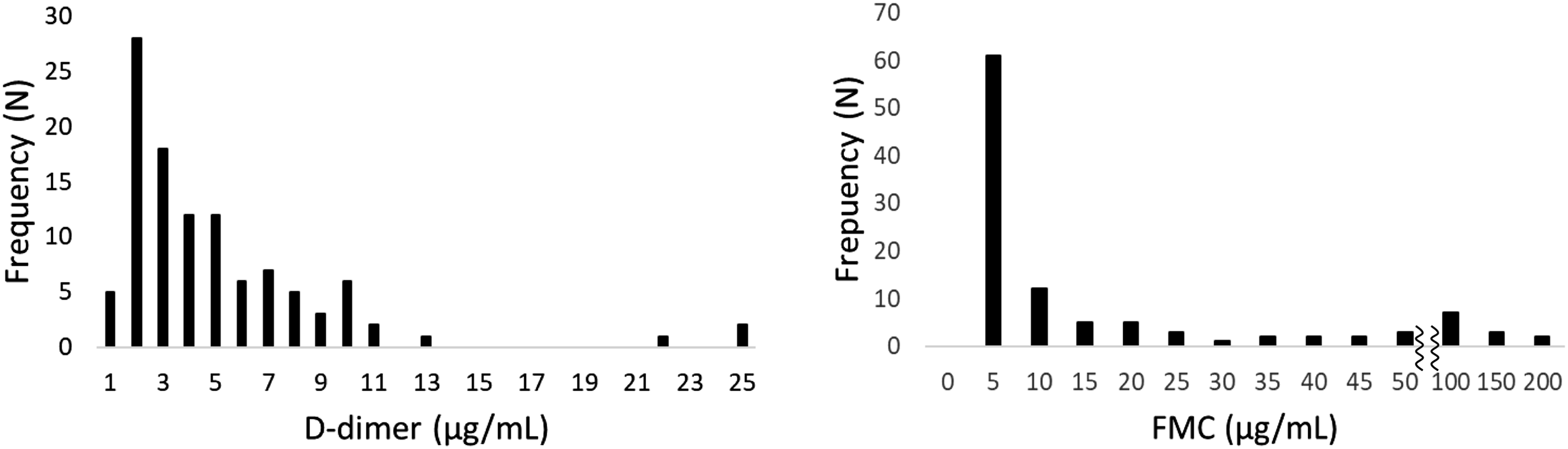
The median (range) concentrations of FMC and D-dimer were 4.0 μg/mL (0.6–200 μg/mL) and 3.3 μg/mL (0.7–25 μg/mL), respectively. Our hospital’s standard values are less than one for D-dimer and less than seven for FMC. FMC and D-dimer concentrations were significantly higher in the high-risk group than in the low-risk group (FMC 14.6 vs 3.4 μg/mL, p < 0.001; D-dimer 4.5 vs 2.6 μg/mL, p = 0.008). Other coagulation/fibrinolysis markers were not significantly different between two groups (Figure 2). Comparisons of thrombosis markers between high-risk and low-risk groups. Reference intervals: FMC < 7.0 μg/mL, D-dimer < 1.0 μg/mL, PIC < 0.8 μg/mL, PT 70∼130%, APTT 23.3∼38.2sec, Fbg 200∼400 mg/dL (reference intervals set by our hospital).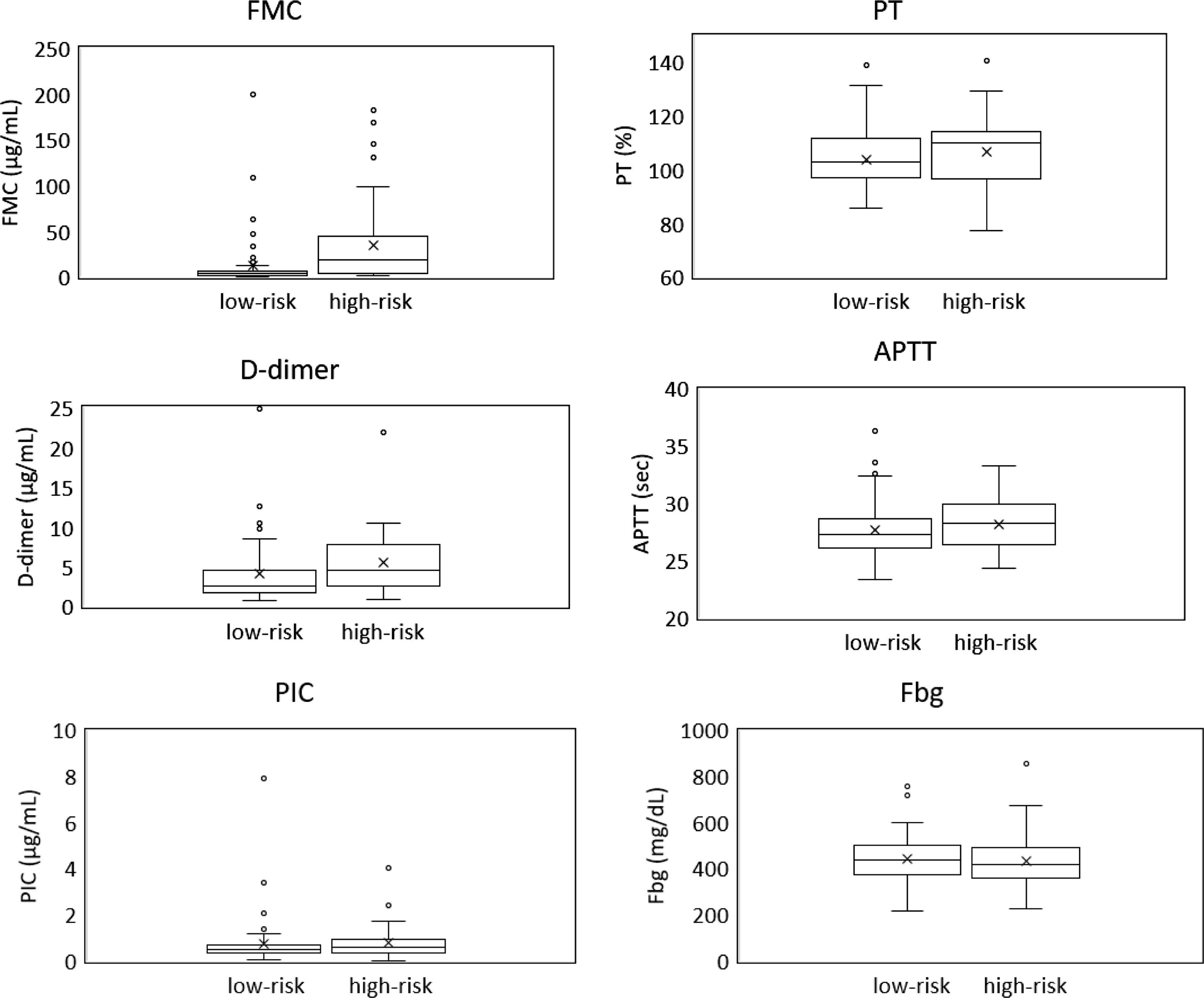
The relationship between gestational age and thrombosis markers was evaluated using Spearman’s correlation coefficient. D-dimer and PIC concentrations increased significantly throughout pregnancy (D-dimer; rs = 0.317, p < 0.001, PIC; rs = 0.248, p = 0.01); however, FMC, fibrinogen, PT, and APTT (FMC; rs = −0.081, p = 0.409, fibrinogen; rs = 0.181, p = 0.073, PT; rs = −0.031, p = 0.753, APTT; rs = 0.063, p = 0.524) were unaffected by gestational age (Figure 3). The relationship between gestational age and thrombosis markers.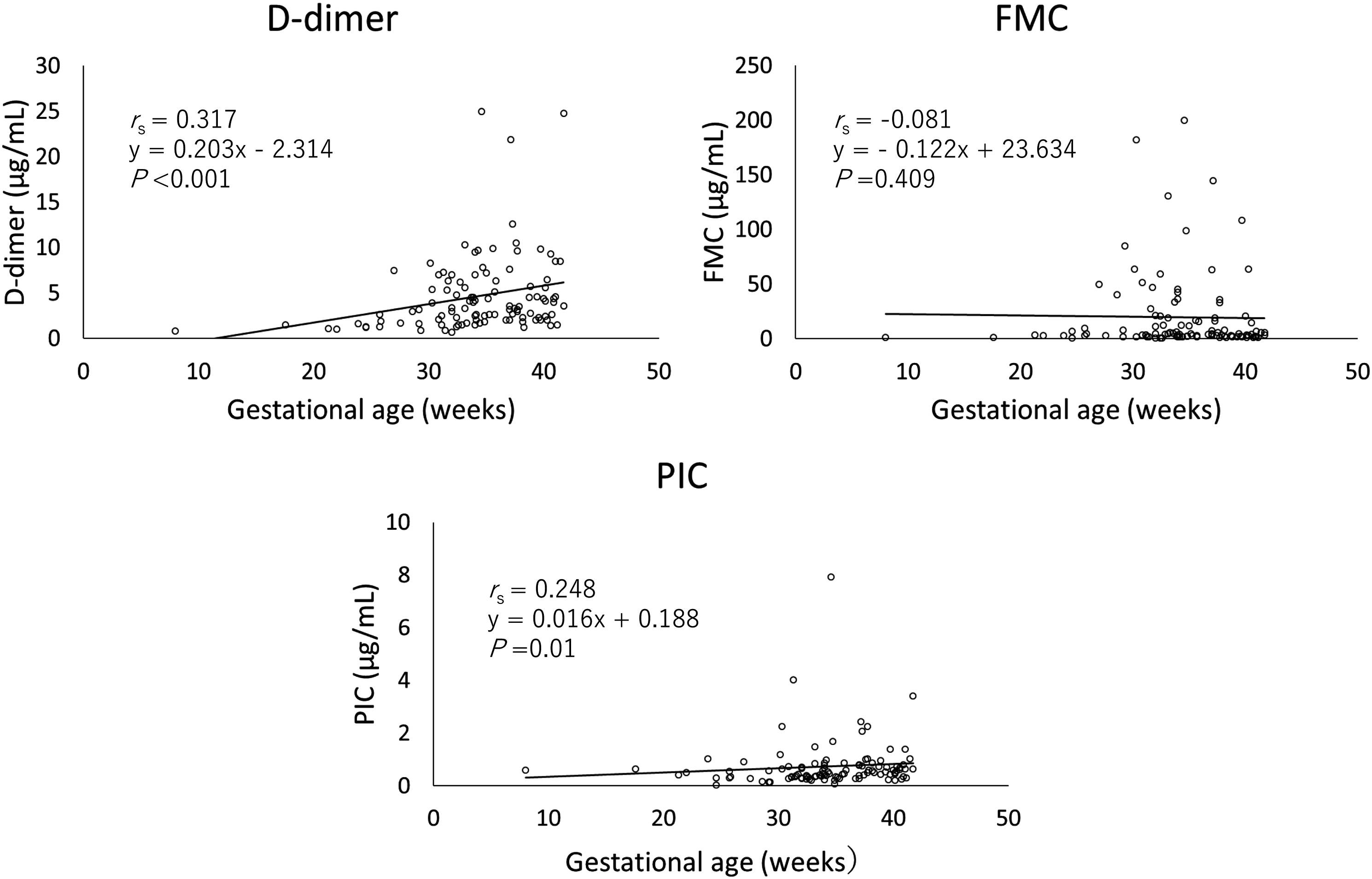
To evaluate the discriminative performance of FMC and D-dimer, we further applied ROC curve analysis (Figure 4). ROC analysis was performed with the high-risk group for VTE as the objective variable to compare D-dimer and FMC. The area under the curve, cut-off value, sensitivity, and specificity of D-dimer and FMC were 0.656 (95%CI; 0.546-0.766), 4.4 μg/mL, 0.590, and 0.721 and 0.713 (95%CI; 0.608-0.818), 5.8 μg/mL, 0.692, and 0.750, respectively. We also performed multivariate ROC analysis by combining D-dimer and FMC concentrations. The area under the curve of the multivariate ROC analysis was 0.738 (95%CI; 0.641-0.834). Discriminative performance of D-dimer, FMC, and both for the high-risk group.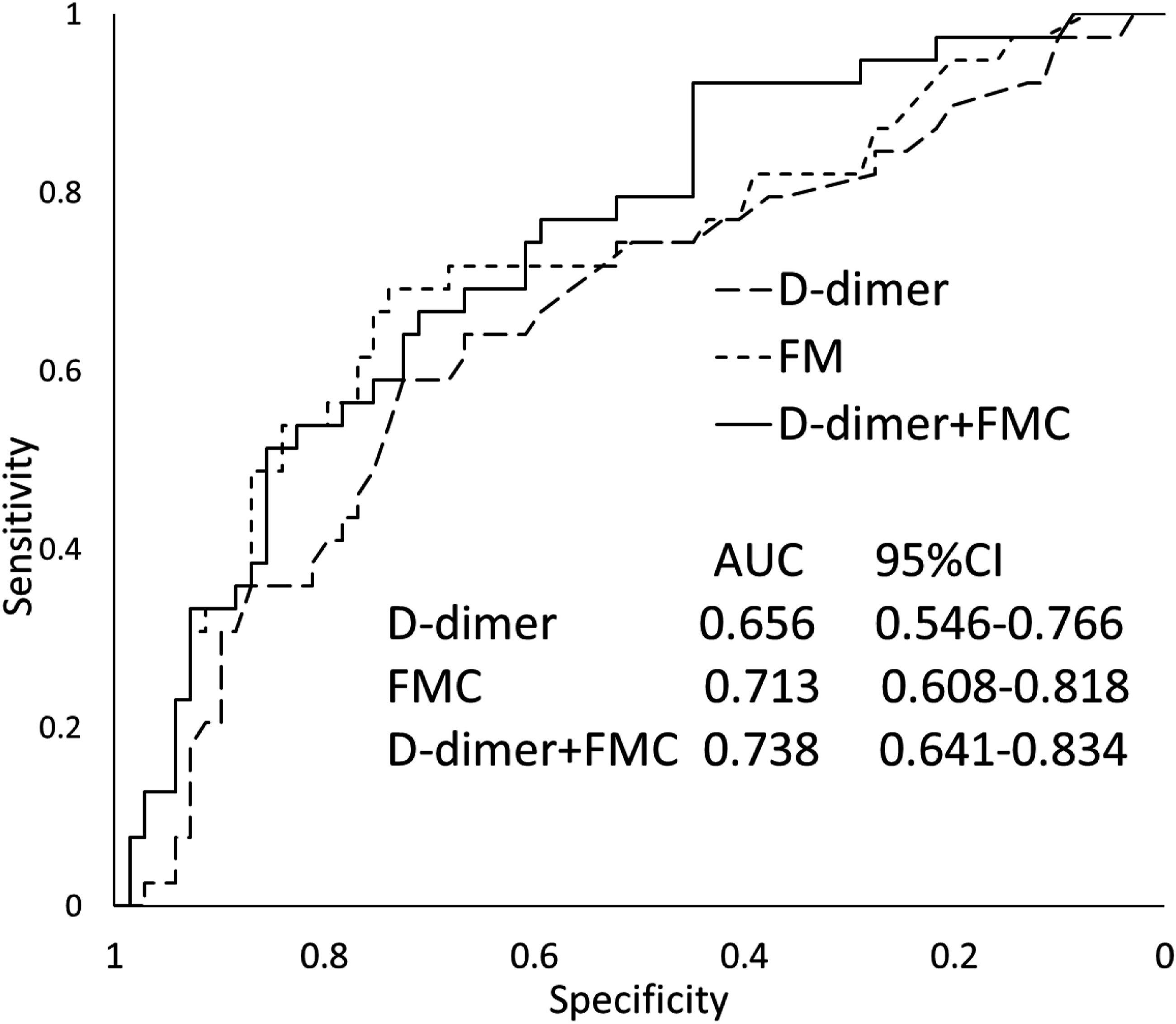
Discussion
This study demonstrated that D-dimer concentrations in the high-risk group were significantly higher than those in the low-risk group and this marker was significantly elevated during pregnancy. Although the reference value of D-dimer is often < 1 μg/mL, only four patients in our study had concentrations within the reference range. The median D-dimer concentration in this cohort was 3.3 μg/mL, and D-dimer concentrations were beyond the reference range in > 95% of the patients who were at a relatively high risk. Furthermore, several studies including healthy pregnant women reported an increase in D-dimer concentrations during late pregnancy.12,13 The RCOG guidelines also advise that D-dimer testing should not be performed in the investigation of acute VTE in pregnancy; 14 therefore, D-dimer is not recommended for managing VTE in pregnancy.
The present study also demonstrated that FMC concentration in the high-risk group was significantly higher than that in the low-risk group. Additionally, it was demonstrated that in contrast to PIC and D-dimer concentrations, FMC concentration was unaffected by gestational age. Although the reference value of FMC used in our hospital is < 7 μg/mL, sixty-eight patients (63.5%) in our study had concentrations within the reference range. Compared to DD, most patients were within the standard values. These findings suggest that FMC is a thrombosis marker that is preferable over PIC and D-dimer. There are three types of antibodies that recognize SF and FMC. IF-43 reacts with most of SF and J2-23 reacts with most of SF and FM. The F405 we used also reacts with SF, FM, fibrin degradation products (FMDP) activated by plasmin on SF and their complexes. Ieko et al. 15 reported there was a significant difference between SF and FMC measurements in DIC patients, but not in hypercoagulable states. From this, we believe that the results of this study will not change even if other measurement reagents are used.
None of the patients in this study developed DVT, even though this study included relatively high-risk pregnancies. Onishi et al. 13 reported the case of a patient who developed DVT with their FMC concentration increasing significantly up to 32.4 μg/mL. Subsequently, DVT improved and the FMC concentration gradually normalized. Since FMC remained relatively stable throughout pregnancy compared to D-dimer and PIC, FMC could be a specific thrombosis marker. There have been consistent reports on the usefulness of FMC measurement in pregnant women.13,16 FMC measurements potentially help in identifying pregnant women who could be at higher risk for perinatal complications.
This study suggests that it is better to assess FMC and D-dimer together than to consider either one of them in isolation. In practice, D-dimer is often used, whereas FMC is rarely used for managing VTE in pregnant women. The usefulness of a combination of thrombosis markers is yet to be investigated.
TAT measurements are often used to manage VTE in pregnant women. TAT is a good marker of coagulation activation since it is formed during the inactivation of thrombin by its inhibitor, antithrombin.7,17,18 However, there is a report that TAT is not useful in predicting the thrombosis risk in pregnant women because it does not correlate with a risk stratification assessed by clinical criteria. 7 TAT concentration is non-specifically elevated during pregnancy, particularly during late pregnancy.13,19 Therefore, TAT is not suitable for detection of DVT in pregnancy. FMC, reflecting fibrin formation, is induced when thrombin generation is high enough to form complexes and is thus more specific to fibrin formation than TAT.18,20,21
PIC is occasionally used as a marker of thrombosis. PIC is a fibrinolytic marker and an increase in PIC indicates fibrinolytic activity by plasmin production, which increases with endogenous fibrinolytic activation in pathological conditions in which a thrombus is formed in vivo. However, this study showed that PIC levels are also elevated significantly during pregnancy. Thus, PIC is not a good marker for VTE detection during pregnancy. It remains unknown why these markers increase significantly during pregnancy, whereas FMC concentrations reflecting fibrin formation remain relatively stable.
This study had some limitations. First, it was a single-center study with a relatively small number of participants. Second, none of the participants in this study developed VTE. Thus, we could not estimate the sensitivity or set a cut-off value for FMC for diagnosing VTE in pregnancy. Third, the study was retrospective and VTE surveillance was at the obstetrician’s discretion. A prospective study is required to determine a cut-off value for FMC, which would clarify the significance of FMC in VTE management in pregnant women.
In conclusion, we found that FMC was significantly higher in the high-risk group than in the low-risk group and was unaffected by gestational age. We also showed that the combination of FMC with D-dimer might be more associated with a high risk for thrombosis than each marker alone. These findings suggest that FMC is an effective marker for predicting thrombosis during pregnancy. Despite some limitations, this is the first report evaluating thrombosis markers using score of RCOG guidelines. Although our study could not set a cut-off value for diagnosing VTE, the FMC cut-off value determined in this study may be useful for initiating treatment in patients at high risk of VTE. Further studies with larger patient populations, including more cases of DVT, are required to confirm these findings.
Footnotes
Acknowledgements
I would like to thank Professor Toru Motokura and Mr. Yuki Hatayama for giving me appropriate advice throughout the writing of this thesis and for their careful guidance.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Ethics Committee of Tottori University Faculty of Medicine (approval number: 19A056).
Guarantor
T.I.
Contributorship
T.I. and Y.H. designed and performed the study; T.I wrote the first draft of the manuscript; H.N., N.K., T.H., N.Y., and H.I. acquired and analysed the data; T.F. and T.M. supervised the study and reviewed and edited the manuscript.
