Abstract
Introduction
Heart-type fatty acid binding protein (HT FABP) is an emerging biomarker of ischaemic myocardial necrosis. While previous studies have demonstrated its additive value when compared to contemporary troponin assays in the diagnosis of acute myocardial infarction (AMI), its utility in the era of high-sensitivity troponin (hsTn) assays remains undetermined.
Methodology
A systematic review and meta-analysis of relevant studies was performed comparing the diagnostic performance of HT FABP both alone and in conjunction with hsTn in the early diagnosis and exclusion of AMI.
Results
A systematic literature search yielded eight eligible studies including 3395 patients. Of these, 716 patients (21.1%) were eventually diagnosed with AMI. The pooled sensitivity and specificity for hsTn on admission was 82.5% (95% confidence interval [CI]: 79.8–85.0%) and 89.7% (95% CI: 88.7–90.6%), respectively, while the area under the curve (AUC) for the summary receiver operating characteristics (ROC) curve is 0.92 (SE 0.02). While the pooled specificity (84.6%, 95% CI: 83.2–85.9%) of admission HT FABP is similar to hsTn for the early diagnosis of AMI (P = 0.07), its pooled sensitivity (63.5%, 95% CI: 59.9–67.1%, P < 0.001) is significantly worse. Accordingly, the AUC of the summary ROC curve for HT FABP (0.79, SE 0.03) is inferior to hsTn (P < 0.0001). The addition of HT FABP to hsTn resulted in no improvement in the sensitivity (P = 0.058) and worsened the specificity (P = 0.001) in the early diagnosis of AMI compared to hsTn alone.
Conclusion
HT FABP does not appear to improve the diagnostic accuracy of hsTn, and consequently its routine use currently cannot not be recommended.
Introduction
The timely diagnosis and exclusion of an acute myocardial infarction (AMI) is critical in the management of patients presenting to hospitals with a suspected acute coronary syndrome (ACS), as the associated clinical and economic implications can be considerable. Current diagnostic algorithms relying on contemporary troponin assays are limited by their relatively low sensitivity and therefore negative predictive value (NPV). Consequently, early exclusion of AMI and risk stratification with contemporary troponins remain a clinical challenge, prompting the search for an alternative marker of ischaemic myocardial necrosis which may accelerate the diagnostic process of AMI and facilitate timely delivery of care.
Over the last decade, the heart-type fatty acid binding protein (HT FABP) has emerged as a potential early biomarker of ischaemia-related myocardial necrosis. The HT FABP is a relatively small (15 kDa) intracellular protein which participates in the fatty acid metabolism by transporting fatty acids from the cell membrane to mitochondria for oxidation. 1 Its potential role in the diagnosis of AMI was first demonstrated by Tsuji et al. 2 who showed a significant and early elevation of HT FABP concentrations within 6 h of symptoms of an AMI. Since then, many studies have been dedicated to develop HT FABP as an alternative or adjunctive tool in the diagnosis of AMI in patients presenting with clinically suspected ACS. Others have also demonstrated its prognostic value in the determination of long-term outcome.3,4
Thus far, the evidence surrounding the utility of HT FABP as an early diagnostic marker of AMI has been mixed. In particular, as a standalone test, HT FABP seems to perform poorly compared to contemporary troponin assays.5,6 Carroll et al. 6 performed a systemic review and meta-analysis of 17 studies evaluating the role of HT FABP at presentation in the early diagnosis of AMI. They reported a pooled sensitivity and specificity of 81% (95% confidence intervals [CI]: 50–95%) and 80% (26–98%), respectively, for the quantitative assays, and 68% (11–97%) and 92% (20–100%), respectively, for the qualitative assays. Compared to an earlier meta-analysis, 7 only studies using the Universal Definition 8 of myocardial infarction were included in this analysis, although there remained significant heterogeneity in the data presented due to variation in the inclusion and exclusion criteria, timing of sample collection in relation to symptom onset, reference troponin assays and diagnostic threshold among included studies. The addition of HT FABP to contemporary troponins in four studies included in this analysis improved the overall sensitivity (42–75% vs. 76–97%) in the detection of AMI, although specificity was significantly compromised (94–100% vs. 65–93%). The authors concluded that the role of HT FABP in the early diagnosis of AMI remains uncertain.
In another recent meta-analysis performed by Lippi et al., 5 the diagnostic performance of HT FABP was directly compared to contemporary troponin across eight studies. HT FABP appeared to be a more sensitive (80% vs. 73%, P = 0.02) but less specific (83% vs. 94%, P < 0.001) predictor of AMI, while its overall diagnostic performance was significantly worse when compared with troponin alone (area under the curve (AUC) 0.784 vs. 0.820, P < 0.001). Similar to the findings by Carroll et al., the combination of HT FABP and contemporary troponin yielded higher sensitivity than troponin alone in the early diagnosis of AMI, although only at the expense of the overall specificity. The overall performance of the combination of biomarkers appeared to be better than troponin alone however (P < 0.001).
Comparisons so far between HT FABP and troponins have been limited to the contemporary troponin I and T. The contemporary troponin assays have superior sensitivity and specificity when compared with Creatine Kinase MB 9 in the diagnosis of AMI and are better predictors of prognosis. However, the sensitivity and NPV of contemporary troponin on admission are suboptimal,10,11 and its ability to detect small variations in the concentrations is further restricted by their imprecision with changes at or below the 99th percentile level. 12
In recent years, high-sensitive troponin (hsTn) assays have superseded contemporary assays in clinical practice due to their superior sensitivity. Its use has been shown to improve the diagnosis of AMI 13 and improve patient outcomes, 14 notwithstanding a minor decline in assay specificity. Accordingly, hsTn has now been adopted by many around the world as the biomarker of choice in the diagnosis of AMI.
As the primary strength of HT FABP over contemporary troponin assays appears to be the improved sensitivity in the early diagnosis of AMI, it is not clear if it would remain a useful adjunct in the era of high-sensitivity troponin (hsTn). Herein, we present a review of the comparison between hsTn and HT FABP in the early diagnosis of AMI based on their sensitivity, specificity, NPV and positive predictive value (PPV), and the overall diagnostic accuracy using the area under the receiver operating characteristics (ROC) curve (AUC).
Methodology
A systematic literature search was performed in July 2014 using Ovid Medline, Embase and Cochrane Database with Systematic Reviews with no date restriction. The aim was to determine the most optimal early diagnostic strategy for patients presenting to hospitals with suspected AMI. A combined search strategy was employed using keywords including: ‘heart type fatty acid binding protein’ or ‘HT FABP’, ‘high sensitivity troponin’, ‘troponin’, ‘myocardial infarction’, ‘chest pain’, ‘acute coronary syndrome’, ‘ischemic heart disease’ and ‘diagnosis’. Further, the reference list of relevant studies was reviewed for additional studies addressing this subject. Only studies published in English language were included for analysis.
Studies meeting the following criteria were eligible for inclusion: randomized control trials or diagnostic cohort study where AMI were diagnosed based exclusively on the universal definition of the European Society of Cardiology (ESC)/American College of Cardiology (ACC) guidelines with either troponin T or I exceeding the prespecified 99% cut-off; 8 adults (>18 years) presenting to hospital with clinical suspicion of AMI who had blood drawn on admission and assayed both for HT FABP (index test) and hsTnT or hsTnI either prospectively or retrospectively; studies containing raw data for retrieving directly or permitting indirect derivation of sensitivity, specificity, PPV, NPV, AUC, as well as the associated 95% CI and standard errors (SE). Studies were excluded if incomplete data were reported or if the patients included were not representative of the cohort of interests.
The quality of the retrieved citations was assessed against the Quality Assessment of Diagnostic Accuracy Studies (QUADAS) checklist criteria. The data were extracted independently by two of the co-authors (KL and SH) and summarized into 2 × 2 tables. Any differences in data collected were resolved by detailed discussion and review of the literature. Pooled sensitivity and specificity for the assays as well as their respective 95% CI were derived based on bi-variate random effect model. The pooled AUC generated were weighted derivations from the summary ROC curve (sROC). The assessment of interstudy heterogeneity was performed using the I-square statistics. Statistical analysis was performed and cross-checked with Review Manager ver 5.2.9 (Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration), MedCalc ver 12.2.1.0 (MedCalc Software, Mariakerke, Belgium) and Comprehensive Meta Analysis ver 2.2.064 (Biostat Inc, USA).
Results
The systematic search performed yielded a total of 245 records from the three electronic databases as well as from review of the study citations. One hundred and fifty-seven records remained after duplications were excluded. Among these, 148 studies were eliminated after a review of abstracts and/or full text based on established criteria. The remaining nine studies were subjected to detailed review of their methodology and quality of the data, and one was further excluded due to insufficient data for derivation of test sensitivity and specificity (Figure 1). Consequently, eight studies (Table 1) were included for quantitative analysis.15–22
PRISMA flow chart of the study selection process. Summary of studies included in the meta-analysis. Mean. AMI: acute myocardial infarction; ACS: acute coronary syndrome; ED: emergency department; NR: not reported.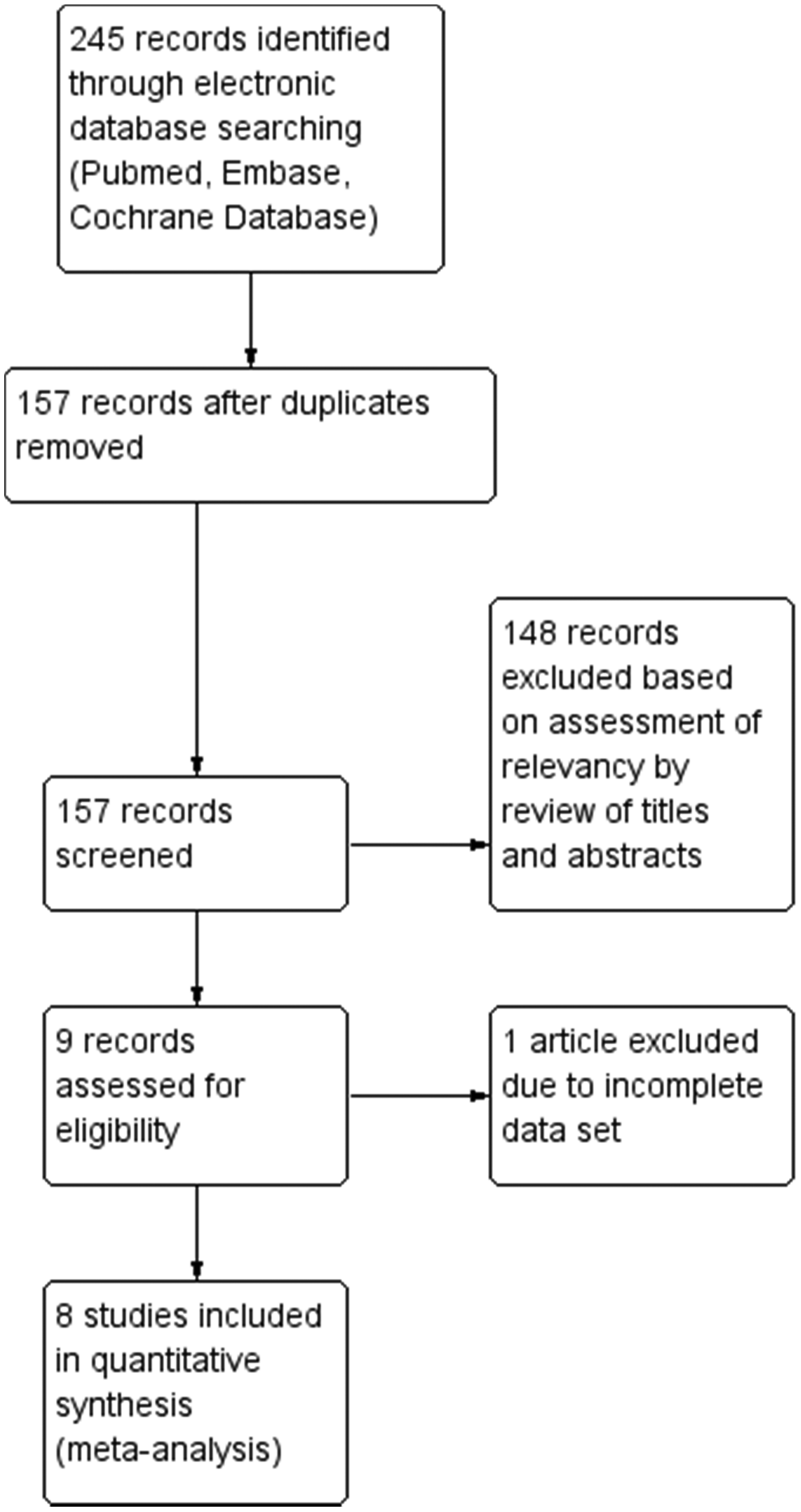

Summary of the diagnostic performance of admission hsTn, HT FABP and combined HT FABP/hsTn for acute myocardial infarction.
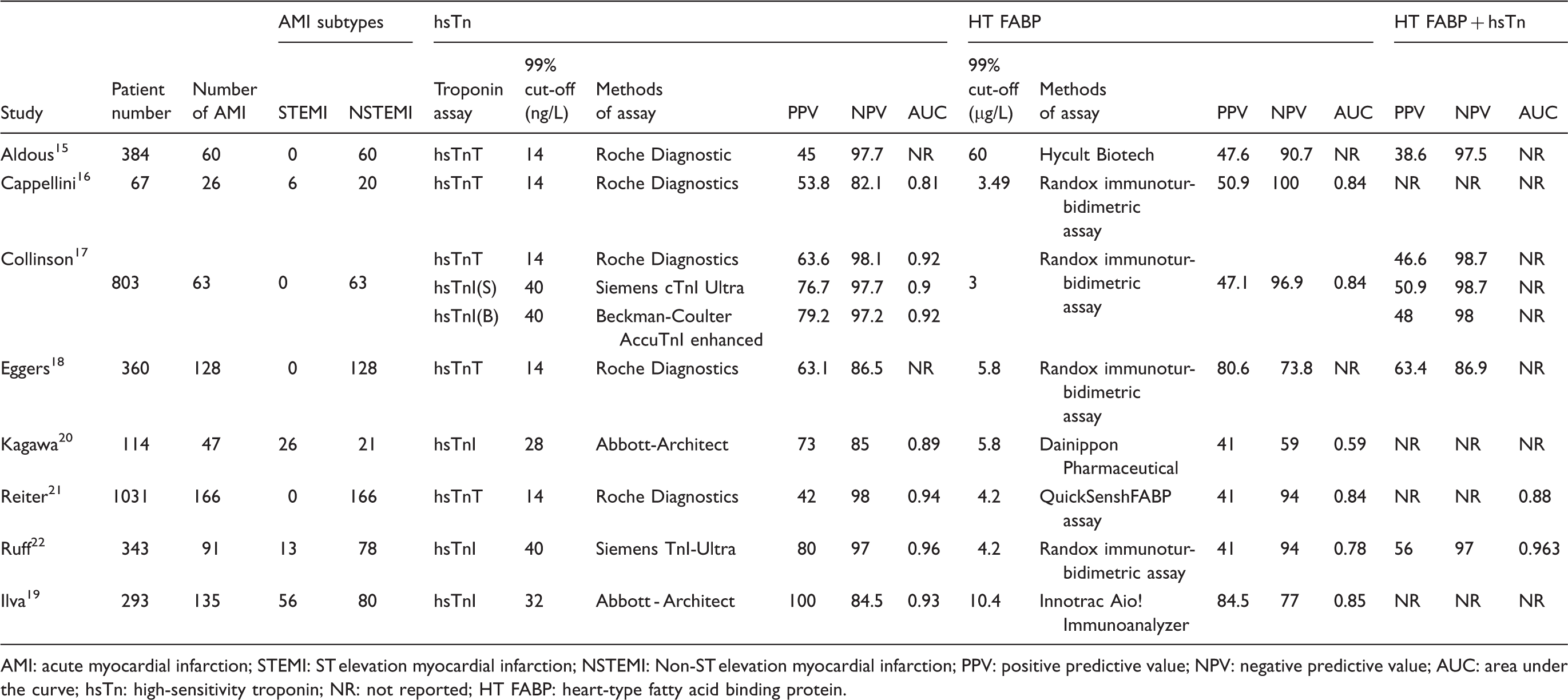
AMI: acute myocardial infarction; STEMI: ST elevation myocardial infarction; NSTEMI: Non-ST elevation myocardial infarction; PPV: positive predictive value; NPV: negative predictive value; AUC: area under the curve; hsTn: high-sensitivity troponin; NR: not reported; HT FABP: heart-type fatty acid binding protein.
High-sensitivity troponin (Figure 2)
Overall, the studies were highly heterogeneous in relation to the diagnostic performance of admission hsTn assays (I2 = 78.1% and 98.1% for sensitivity and specificity, respectively, P < 0.001). The pooled sensitivity and specificity for hsTn was 82.5% (95% CI: 79.8–85.0%) and 89.7% (95% CI: 88.7–90.6%), respectively, while the AUC for the sROC is 0.92 (SE 0.02) (Figure 3).
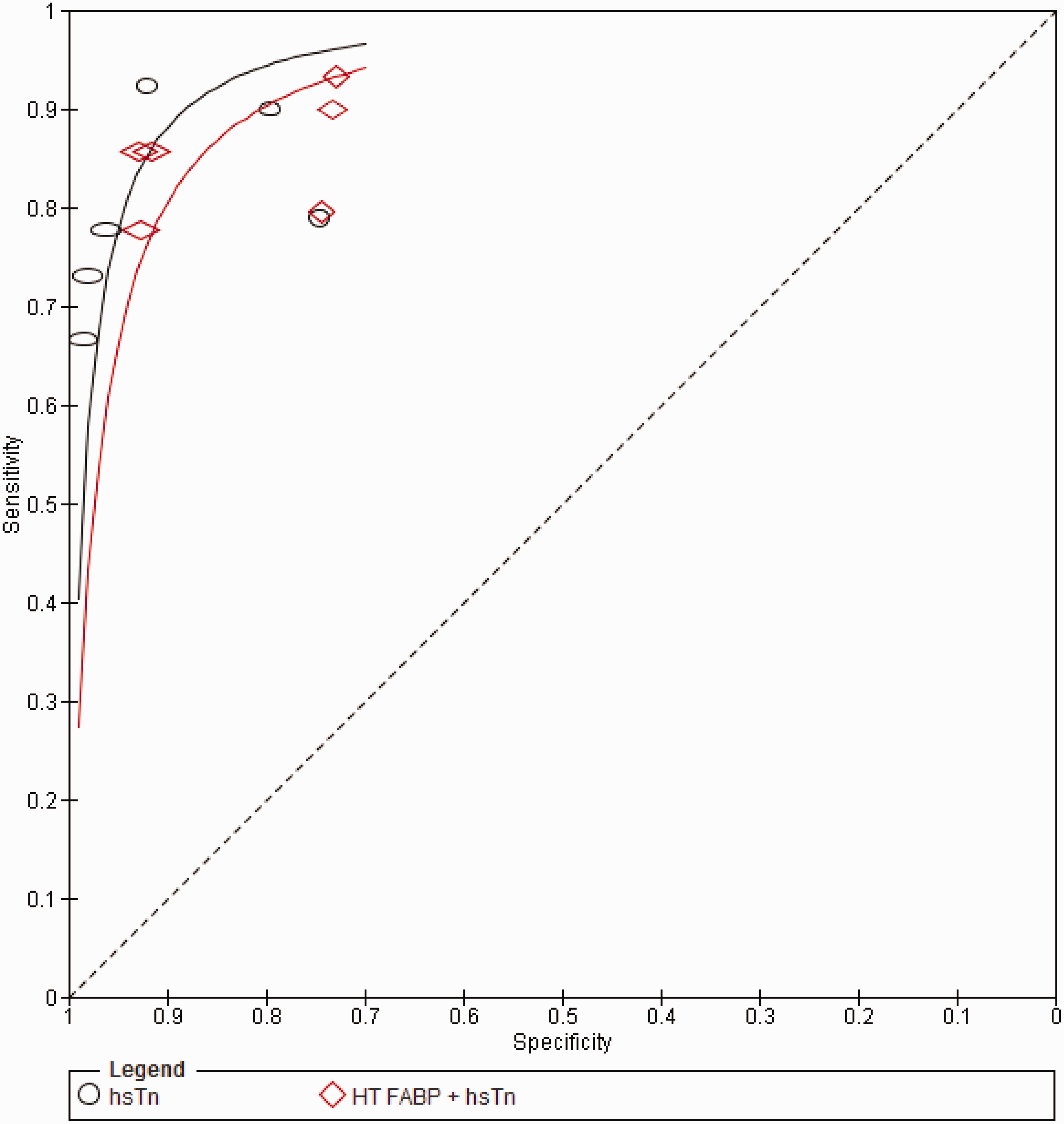
Summary of the diagnostic performances of HT FABP and hsTn. Summary ROC (sROC) for HT FABP vs. hsTn.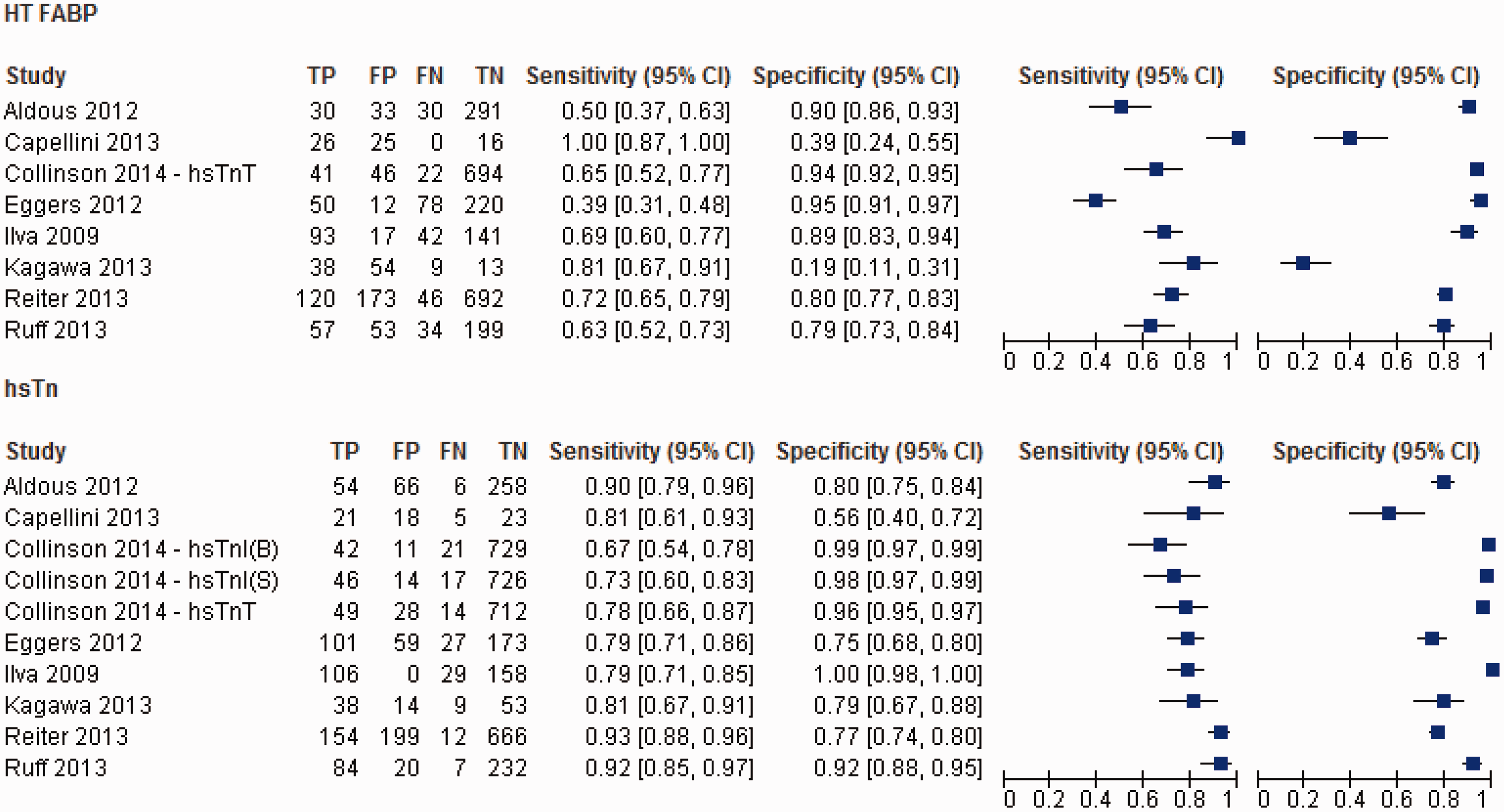
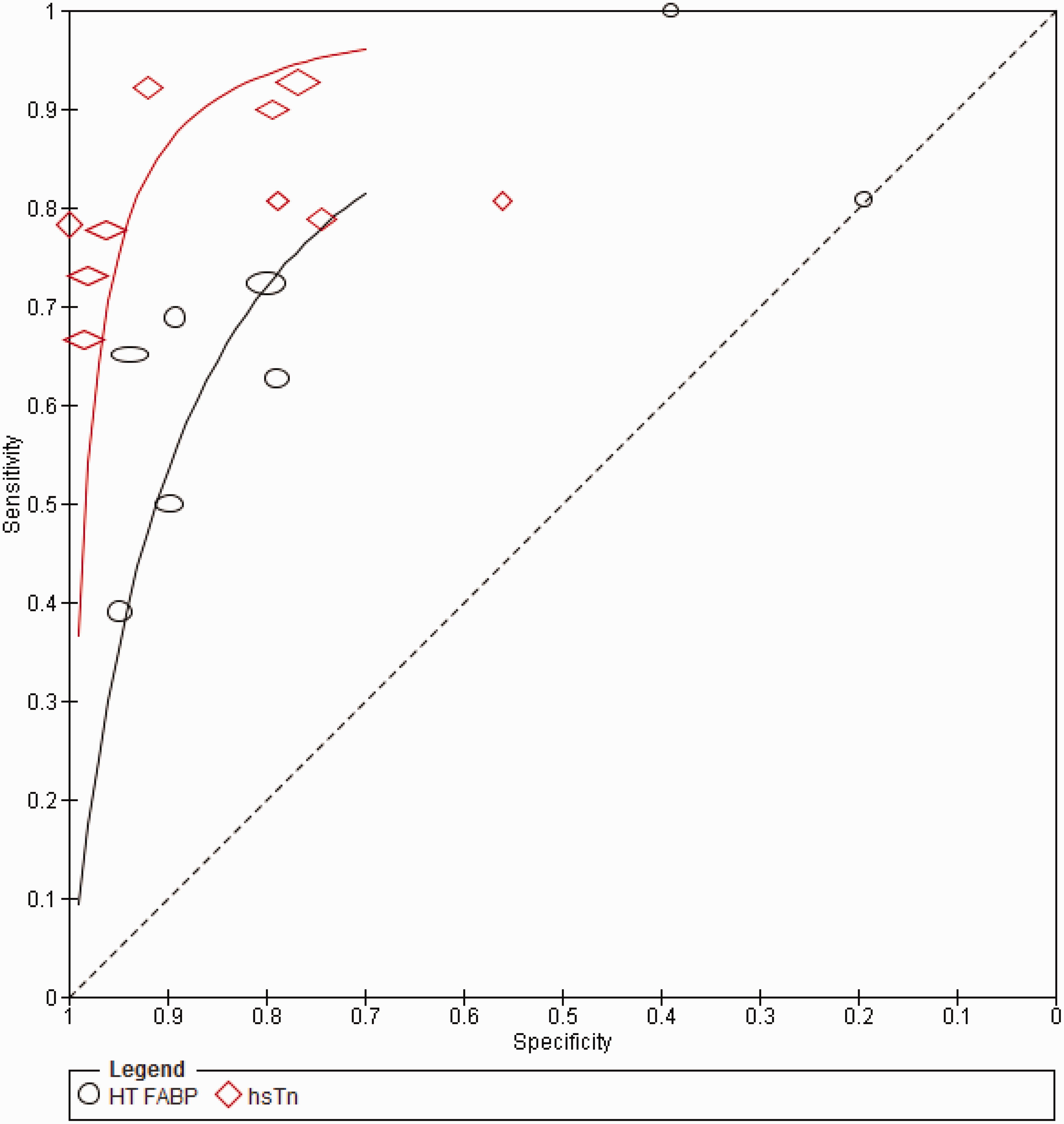
When hsTnT is considered in isolation, the pooled sensitivity and specificity was 85.6% (95% CI: 81.9–88.7%) and 83.2% (95% CI 81.6–84.7%). The AUC was 0.91 (SE 0.03). With hsTnI, the pooled sensitivity and specificity were 79.2% (95%CI: 74.9–83.1%) and 97.0% (95% CI 96.1–97.8%) respectively, while the AUC was 0.93 (SE 0.03).
Heart-type fatty acid binding protein (Figure 2)
The studies were equally heterogeneous with respect to the diagnostic performance of admission HT FABP (I2 = 90.5 and 97.6 for sensitivity and specificity, respectively, P < 0.001). The pooled sensitivity (63.5%, 95% CI: 59.9–67.1%) for AMI was significantly worse compared to hsTn overall (P = 0.001), while the pooled specificity was numerically inferior (84.6%, 95% CI: 83.2–85.9) though did not reach statistical significance (P = 0.07). The pooled AUC for HT FABP (0.79, SE 0.03) was significantly worse compared to hsTn overall (P < 0.0001) (Figure 3).
When HT FABP was compared specifically with hsTnT,15–18,21 the pooled sensitivity (60.3%, 95% CI: 55.5–64.9%) was much worse (P = 0.001) though the specificity (86.9%, 95% CI: 85.4–88.3%) did not differ significantly (P = 0.34). The AUC of 0.86 (SE 0.04) is significantly worse than hsTnT (P < 0.01). In contrast, HT FABP performs similarly to hsTnI19,20,22 in terms of its pooled sensitivity (68.2%, 95% CI: 62.9–73.1%; P = 0.068), although hsTnI significantly outperforms HT FABP in terms of its pooled specificity in the early diagnosis of AMI (86.0%, 95% CI: 84.0–87.9%; P < 0.001). Accordingly, the pooled AUC (0.77, SE 0.03) of HT FABP was significantly worse compared to hsTnI (P < 0.0001).
HT FABP + hsTn vs. hsTn (Figure 4)
Four studies15,17,18,22 assessed the diagnostic performance of a multibiomarker strategy on admission by adding HT FABP to hsTn in the early diagnosis of AMI. A positive test in this instance is defined as elevation of at least one of the two biomarkers. This yielded a pooled sensitivity and specificity of 85.0% (95% CI: 81.5–88.2%) and 87.5% (95% CI: 86.2–88.6%), respectively, and an AUC of 0.92 (SE 0.02). The addition of HT FABP, however, resulted in no improvement on the sensitivity (P = 0.058) and slightly worsened the specificity (P = 0.001) compared with hsTn alone. The overall difference in the diagnostic accuracy by AUC remained statistically insignificant (P = 0.68) (Figure 5).
Summary of the diagnostic performance of hsTn and the multibiomarker strategy with hsTn/HT FABP in four included studies. Summary ROC (sROC) for HT FABP vs. HT FABP+ hsTn.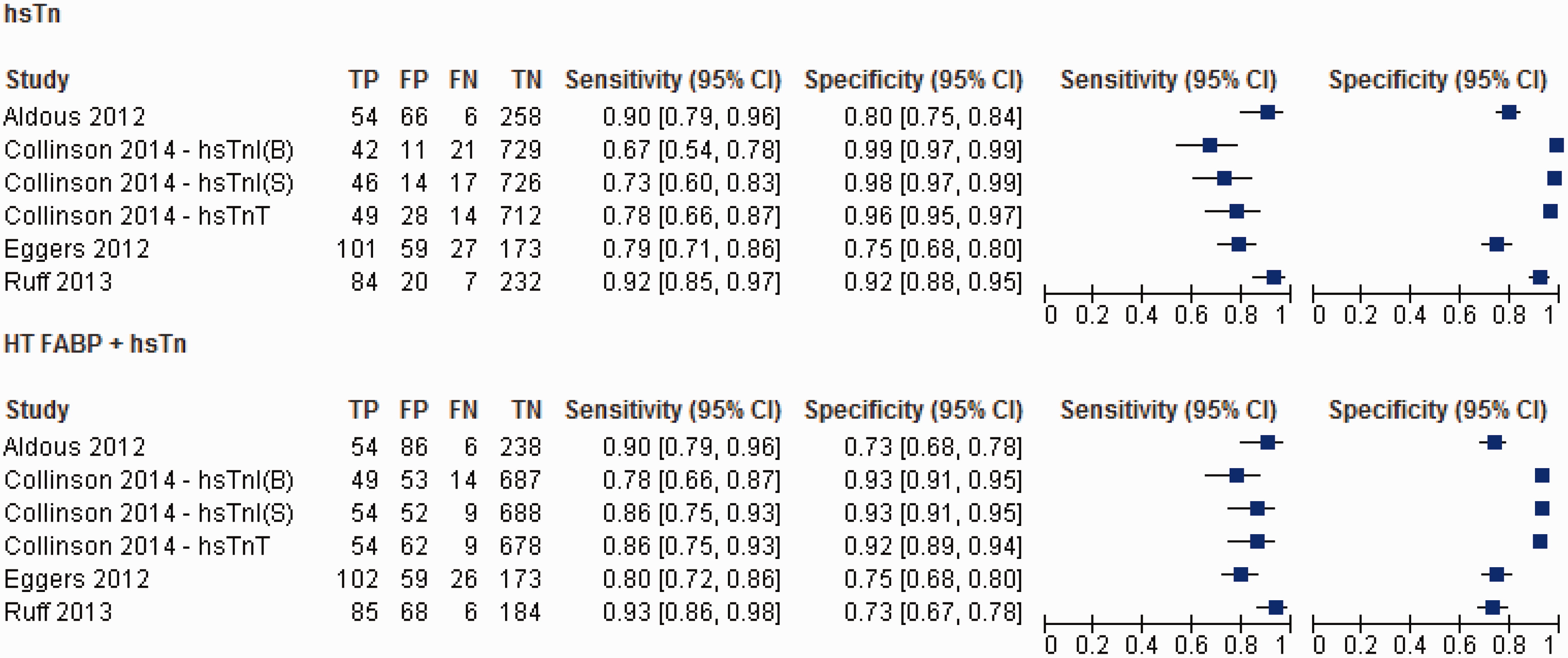
Three studies15,17,18 assessed the value of adding HT FABP to hsTnT. The pooled sensitivity and specificity were 83.7% (95% CI: 78.5–88.0%) and 84.0% (95% CI: 81.9–86.0%), respectively, while the AUC was 0.91 (SE 0.04). Compared to hsTnT alone, the sensitivity and specificity of the combination of HT FABP/hsTnT did not improve significantly (P = 0.49 and 0.082, respectively), while the difference in the overall diagnostic accuracy (AUC) also did not reach statistical significance (vs. 0.91, SE 0.04, P = 0.69).
Two studies17,22 assessed the value of adding HT FABP to hsTnI. The pooled sensitivity and specificity was 86.6% (95% CI: 81.4–90.9%) and 90.0% (95% CI: 88.5–91.4%). The combination did improve the sensitivity of hsTnI alone (P = 0.038), although only at the expense of specificity (P < 0.0001). Overall, the AUC for the combined HT FABP/hsTnI was statistically inferior to hsTnI alone (AUC 0.93, SE 0.01 vs. AUC = 0.97, SE 0.01, P = 0.02).
Discussion
The present meta-analysis demonstrates that, overall, the admission hsTn is a more sensitive and equally specific predictor of AMI compared to HT FABP. Specifically, HT FABP is less sensitive and equally specific when compared with hsTnT, while it is equally sensitive but less specific compared with hsTnI. The combination of HT FABP and hsTn does not provide additional diagnostic value when compared with hsTn alone, although the addition of HT FABP to hsTnI may improve its sensitivity but only at the expense of specificity. On the whole, HT FABP does not appear to provide any added value in the early diagnosis and exclusion of AMI in the era of hsTn.
While the Universal Definition of myocardial infarction 8 provides a consistent and uniform means of patient characterization in clinical practice and scientific trials, its role in the early exclusion of AMI is not clear. This in part is related to the need for serial biomarker measurements, which in turn is a result of the relatively low sensitivity of contemporary biomarkers for AMI particularly among early presenters. The value of any emerging biomarker of cardiac injury therefore resides in their ability to improve the existing diagnostic algorithm particularly surrounding the early phase of admission for patients with suspected AMI. Accordingly, the novel biomarker needs to be both tissue and mechanism specific and has a favourable release kinetics.
Among all the emerging biomarkers, HT FABP in particular has been extensively investigated in both an experimental and clinical setting. While it has been detected in small quantities in the skeletal muscles (173 ug/g), brain (16–40 ug/g) and the intestinal system (2.7–4.9 ug/g), the HT FABP is found predominantly within cardiac tissue (540–600–ug/g), thus conferring its cardiac tissue specificity. 23 Because of its small size, HT FABP is released very early following the onset of ischaemia and becomes detectable in the circulation within 20 min of the onset of ischaemia. Levels then peak at 3–4 h and return to normal within 24 h. 1
Our meta-analysis, however, suggests that when HT FABP is compared with hsTn, the overall diagnostic performance was at best equivalent if not worse when interpreted in isolation. When HT FABP was combined with hsTn, there was also no incremental value in the diagnostic performance of hsTn. Compared to previous reports, 5 the introduction of hsTn in our analyses has diminished the observed advantage of HT FABP over contemporary troponin in terms of its early sensitivity for AMI, while preserving its overall specificity. Similarly, the benefit observed by previous authors5,6 with the combination of HT FABP and contemporary troponin in terms of its improved sensitivity and diagnostic performance has been neutralized by the use of hsTn.
Several factors merit consideration. It has been postulated by many that cardiac troponin exists in two cellular compartments. Troponin that becomes detectable following MI has been traditionally believed to originate from the structural or the muscular pool. It signals cell death and is responsible for the persistence of troponin in the circulation despite its short half-life. 24 Based on studies on non-ischaemic causes of troponin elevation, 25 however, it has been speculated that troponin also exists within the cytosol, which leaks into the lymphatics and blood circulation when the permeability of the cellular membrane is increased by physiological or pathological stress. 26 Since both troponin and HT FABP exist within the same cellular compartment, their release following the onset of ischaemia may parallel each other. Consequently, as the sensitivity of the modern troponin assays increases, the diagnostic advantage offered by HT FABP may be lost.
Secondly, the release kinetics vary between troponin and HT FABP, and the variability in the time of blood sampling in relation to the onset of symptoms among trials included may also confound our results. While the diagnostic performance of hsTn improves with serial sampling,17,19,21 the diagnostic performance of HT FABP seems to decline with time. 21 This is because HT FABP concentration decreases with serial sampling 21 and normalizes quickly due to rapid renal clearance. 27 Accordingly, the sensitivity of HT FABP begins to drop 6 h after symptom onset while the overall diagnostic performance also decreases in parallel. 19 A significant proportion of patients in this study presented more than 6 h after their symptom onset. As a result, the sensitivity of HT FABP in these patients in comparison to hsTn may have been misrepresented.
Given its release kinetics, it may be argued that the primary role for HT FABP is in the diagnosis of AMI in early presenters. In comparison to hsTn, however, the diagnostic performance of HT FABP appears consistently poor irrespective of the time delay between symptom onset and presentation. Collinson et al., 17 Reiter et al. 21 and Ruff et al. 22 all demonstrated poor performance of HT FABP against hsTn even in early presenters (<3–6 h), while Eggers et al., 18 Kagawa et al. 20 and Ilva et al. 19 found the diagnostic performance of HT FABP against hsTn to be equally poor between early and late presenters. The interaction between the timing of sampling and combined HT FABP/hsTn appears more complex. In one study, 22 the addition of HT FABP significantly improved the NPV in early presenters (<6 h) compared with hsTn alone, although the overall diagnostic performance did not change. Another study, 18 however, demonstrated no improvement in the diagnostic performance (sensitivity or specificity) over hsTn alone in either early (<4 h) or late presenters (≥4 h) with the combination strategy.
The interpretation of our results may be further confounded by other sources of heterogeneity among the studies included. Specifically, not all studies included patients with STEMI. Indeed, one study exclusively enrolled patients at low baseline cardiovascular risk. 17 While HT FABP is a fairly specific marker of myocardial damage, it is unclear if its concentrations may be affected by critical illness or by the systemic effects of STEMI independent of cardiac ischaemia. Renal dysfunction, which falsely elevates both the troponin and HT FABP concentrations, was only cited by three studies as an exclusion criterion. An uneven distribution of renal function among patients included may influence the specificity of troponin assays in relation to HT FABP. It is similarly difficult to control for heterogeneity in HT FABP assay methodologies across studies and the cut-off thresholds. Indeed, a trend exists between a lower cut-off threshold and a higher NPV and lower PPV, and it would be difficult to know if the overall results would remain the same if the cut-off points were standardized across the board. By the same token, the inclusion of both hsTnI and hsTnT in pooled analyses may also influence the results as there appears to be inherent differences in their diagnostic performance. 13 Efforts have been made, however, to ensure separate analyses were performed with hsTnI and hsTnT, although the number of studies and subjects are reduced and the results may not be robust.
Further consideration needs to be given to the confounding effects of unreported variables in the included studies. While factors such as age, sex, hypertension, body mass index, previous bypass surgery and arrhythmia16,22,28 have been shown to influence the concentration of HT FABP, no studies have incorporated these in a multivariate model. The mode of sample storage and the thawing process, as well as its impact on assay accuracy for both hsTn and HT FABP is also unclear. Finally, the results of the analyses may be further skewed by the presence of numerical outliers. It is important to note that the outlying results belong to the two smallest included studies16,20 and the possibility of statistical error need to be taken into consideration.
Conclusion
HT FABP both alone and in conjunction with hsTn does not appear to improve the overall diagnostic performance of hsTn in the early diagnosis and exclusion of AMI. As the evidence is currently influenced by the considerable heterogeneity among the studies, the results of this systematic review need to be interpreted with caution. Nonetheless, the currently available data do not support the routine use of HT FABP in the era of hsTn as an early risk stratification strategy in patients presenting with suspected AMI.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
As this was a review and meta-analysis, no ethics approval was sought.
Guarantor
SO.
Contributorship
KL conceived the idea. KL and SH performed the literature search and extracted the data independently from relevant studies. KL performed the statistical analysis and wrote the first draft of the manuscript. SO provided overall supervision. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
