Abstract
Background
Growth differentiation factor-15 (GDF-15) is a divergent member of transforming growth factor-beta super family. Under physiological states, it is weakly expressed in most tissues, but it is elevated in impaired kidney function. High concentrations of GDF-15 have been found in some haemoglobinopathies associated with suppressed concentration of hepcidin and iron overload. It is not clear whether the increased concentration of GDF-15 in chronic kidney disease influences iron metabolism.
Methods
The serum concentrations of GDF-15 and hepcidin, iron (Fe), ferritin, transferrin (Tf), total iron binding capacity, transferrin saturation, soluble transferrin receptor1 (sTfR1), erythropoietin and Hb were measured in 32 stable chronic kidney disease stage 5-dialysis (CKD5-D) patients and 24 healthy adults (controls) to investigate any relationship between GDF-15 and iron indices.
Results
GDF-15 was significantly elevated in the haemodialysis group (4840.6 ± 1520.5 ng/L) compared to control (472.8 ± 148.1 ng/L). There was a positive correlation between GDF-15 concentration and age in both groups. In the haemodialysis group, hepcidin was increased and correlated with serum ferritin, Tf, total iron binding capacity and sTfR1. There was no correlation between GDF-15 and hepcidin or other iron indices.
Conclusions
GDF-15 was significantly elevated in our haemodialysis patients but there was no correlation between GDF-15, hepcidin and various iron indices. In this small observational study, GDF-15 would not appear to be associated with iron metabolism in stable CKD5-D patients.
Introduction
Growth differentiation factor-15 (GDF-15) is a divergent member of transforming growth factor-beta (TGF-β) super family which shares 15–29% amino acid homology with other members. 1 It is expressed in most tissues at different concentrations but its roles in general and basic cellular function have not been elucidated adequately. GDF-15 has been shown to increase significantly in a number of haematopoietic disorders such as thalassaemia, congenital dyserythropoietic anaemia type 1, refractory anaemia with ring sideroblasts and pyruvate kinase deficiency 2 ; all of these conditions are associated with ineffective erythropoiesis. In thalassaemia syndrome, iron overload occurs as a result of multiple blood transfusions with the markedly elevated serum GDF-15 concentration inhibiting hepcidin mRNA expression in hepatocytes, thereby promoting intestinal iron absorption. 3 In the kidney, GDF-15 is mainly expressed in the renal tubules. The serum concentration of GDF-15 is elevated in patients with renal dysfunction. We aimed to study the role of GDF-15 in iron metabolism in a group of chronic kidney disease (CKD5-D) patients by investigating the relationship of GDF-15 serum concentration with hepcidin and other iron indices.
Materials and methods
Between September and October 2013, 32 consecutive CKD5-D patients were recruited. These patients were either receiving maintenance haemodialysis (HD) three times a week at the haemopurification center of the University of Hong Kong Shenzhen Hospital or at the Nanshan Affiliated Hospital of Guangdong Medical College in China. The inclusion criteria were: ≥18 years of age, receiving regular HD treatment for at least two months. The exclusion criteria were: history of myocardial infarction, episodes of angina pectoris or heart failure within the previous two months, history of malignancy, infections within the previous two weeks requiring antibiotics therapy, history of blood transfusion within the two months prior to recruitment. The control group comprised 24 randomly selected adults undergoing routine health checks at the health examination centre of the Nanshan Affiliated Hospital of Guangdong Medical College. These adults fulfilled the following criteria: ≥18 years of age, full blood count profile within the healthy Chinese population reference range (for haemoglobin [Hb], defined as 120–160 g/L [men], 110–150 g/L [women], no erythrocyte morphological abnormality, normal white cell and platelet counts), kidney and liver profile results within the healthy population reference range, negative serum hepatitis B surface antigen and antihepatitis C antibody, no history of cancer or ischaemic heart disease. The study was conducted in accordance with the ethical standards of the Helsinki Declaration, and was approved by ethics committees of the University of Hong Kong Shenzhen Hospital and the Nanshan Affiliated Hospital of Guangdong Medical College. In the HD group, all blood samples were taken in the morning following breakfast at the start of a dialysis session either via a fistula (30 cases) or a jugular vein permanent catheter (two cases). Of the 32 patients, 20 had blood samples taken two days after HD, whereas 12 patients had blood sample taken three days after HD. In the control group, a fasting blood sample was taken in the morning of visit by standard venepuncture.
The serum concentrations of GDF-15, hepcidin, Fe, serum ferritin (SF), Tf, total iron binding capacity (TIBC), transferrin saturation (TS), sTfR1, erythropoietin (EPO) and Hb were measured in both groups. Erythrocyte, white blood cells and platelet counts, Hb, Fe, SF, Tf, TIBC and TS were measured with automated analysers. Serum GDF-15, hepcidin, sTfR1, EPO concentrations were determined with ELISA by using the following kits: GDF-15 (DGD150 Human GDF-15 Quantikine ELISA, R&D Systems, MN, USA), for hepcidin (EIA-5258 ELISA, DRG, Marburg, Germany), sTfR1 (DTFR1 Human sTfR Quantikine IVD, R&D Systems, Minnesota, USA) and EPO (RE56011 Sandwich ELISA, IBL, Hamburg, Germany). All ELISA tests were performed according to the manufacturers’ instructions. All statistical analyses were performed using SPSS 15.0 for Windows (SPSS, Chicago, IL, USA). Normally distributed data are presented as mean ± SD values and compared by independent or paired sample t-tests. Non-normally distributed data are presented as median (25% percentile, 75% percentile) and compared by rank-sum test.
Results and discussion
The age and gender distributions were similar in the HD and control group. The causes of the end renal disease (ESRD) in the HD group were: chronic glomerulonephritis (N = 21), polycystic kidney disease (N = 2), diabetic nephropathy (N = 1), lupus nephritis (N = 1), chronic interstitial nephritis (N = 1), antineutrophil cytoplasmic antibody associated vasculitis (N = 1), renal tuberculosis (N = 1) and unknown cause (N = 4). HD was performed with bicarbonate-based dialysis solution for 4 h each session. All HD patients received intravenous recombinant erythropoietin (rEPO), ranging from 3000 to 10,000 IU per week, given at the end of an HD session aiming for a target Hb of 110–125 g/L. In addition, they received iron supplementation with different preparations (ferrous sulphate, polysaccharide–iron complex or iron sucrose) either orally or intravenously to maintain an SF concentration of 200–500 µg/L.
Participant demographic details and concentrations of GDF-15, hepcidin, iron indices and Hb in HD and healthy control groups.
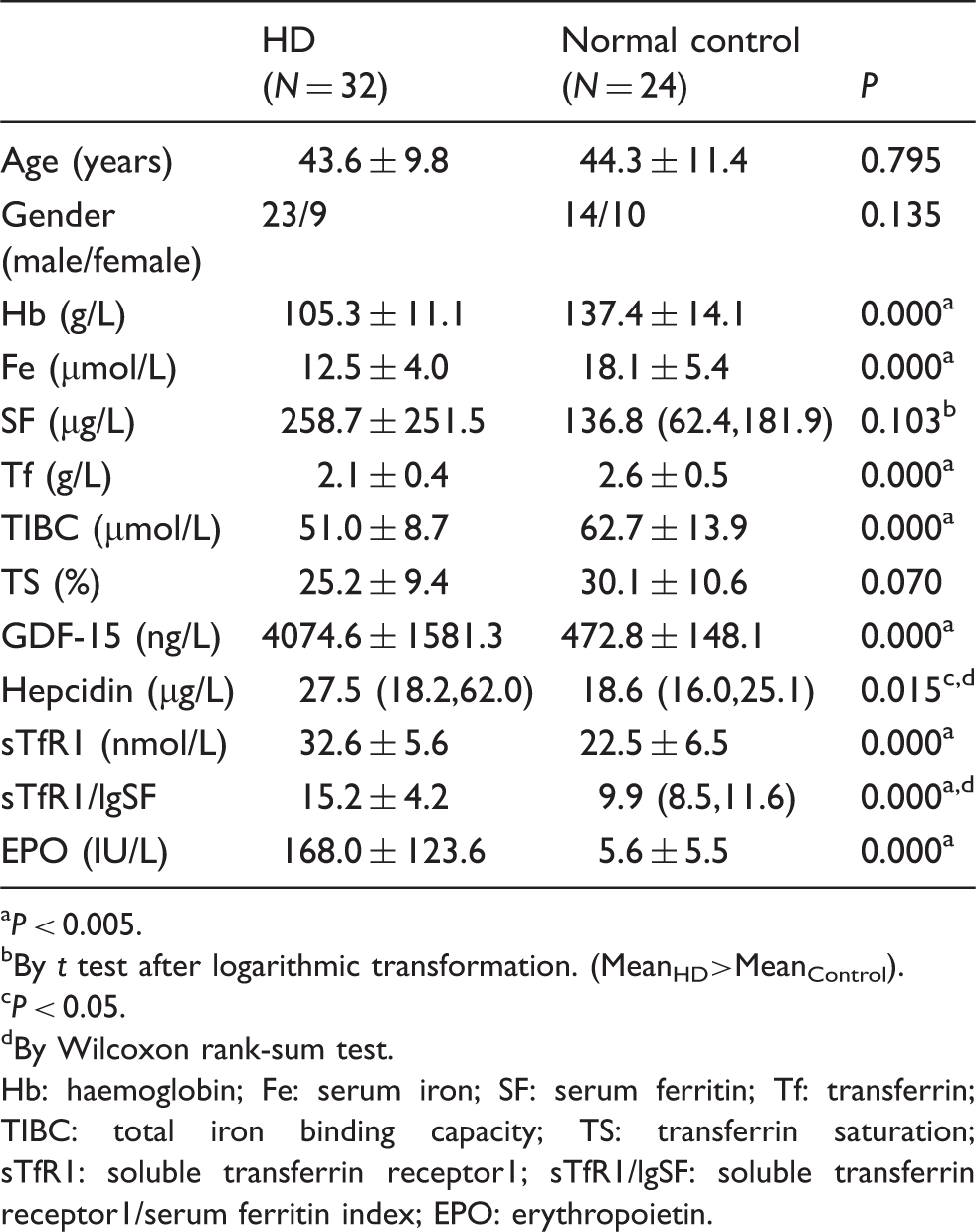
P < 0.005.
By t test after logarithmic transformation. (MeanHD>MeanControl).
P < 0.05.
By Wilcoxon rank-sum test.
Hb: haemoglobin; Fe: serum iron; SF: serum ferritin; Tf: transferrin; TIBC: total iron binding capacity; TS: transferrin saturation; sTfR1: soluble transferrin receptor1; sTfR1/lgSF: soluble transferrin receptor1/serum ferritin index; EPO: erythropoietin.
In both HD and control groups, the serum GDF-15 concentration correlated positively with age (Figure 1(a) and (b)). Using multiple linear stepwise regression analysis, age was found to be a predictive factor for GDF-15 in control groups (GDF-15 = 6.462 + 10.519 Age, R2 = 0.656, P = 0.000) but not in the HD group. In both groups, serum hepcidin concentration was found to be positively correlated with SF, but negatively with Tf, TIBC and sTfR1/lgSF. Using multiple linear stepwise regression studies, Tf and Fe were predictive of hepcidin concentration in the control group (lgHepc = 2.385–0.286Tf–0.017Fe, R2 = 0.38, P = 0.040). In the HD group, there was a negative correlation between hepcidin and Hb concentrations. Further analysis showed that high hepcidin concentration was predictive of anaemia (Hb = 133.381–15.608 lgHepc, R2 = 0.459, P = 0.002). Using both Pearson and Spearman correlation, no significant correlations between GDF-15 and hepcidin, GDF-15 and other iron indices, GDF-15 and Hb were found. HD patients were further divided into those with GDF-15 concentration <5000 ng/L (subgroup ‘a’, N = 24) and those with ≥5000 ng/L (subgroup ‘b’, N = 8) to determine whether there was an association with hepcidin or iron indices only at higher GDF-15 concentrations (≥5000 ng/L).
3
Within this subgroup, no correlation was observed. The results of our study showed an increase of GDF-15 in chronic HD patients but a lack of correlation between GDF-15, hepcidin and various iron indices.
Correlation of GDF-15 with age in the (a) HD group and (b) in the healthy control group.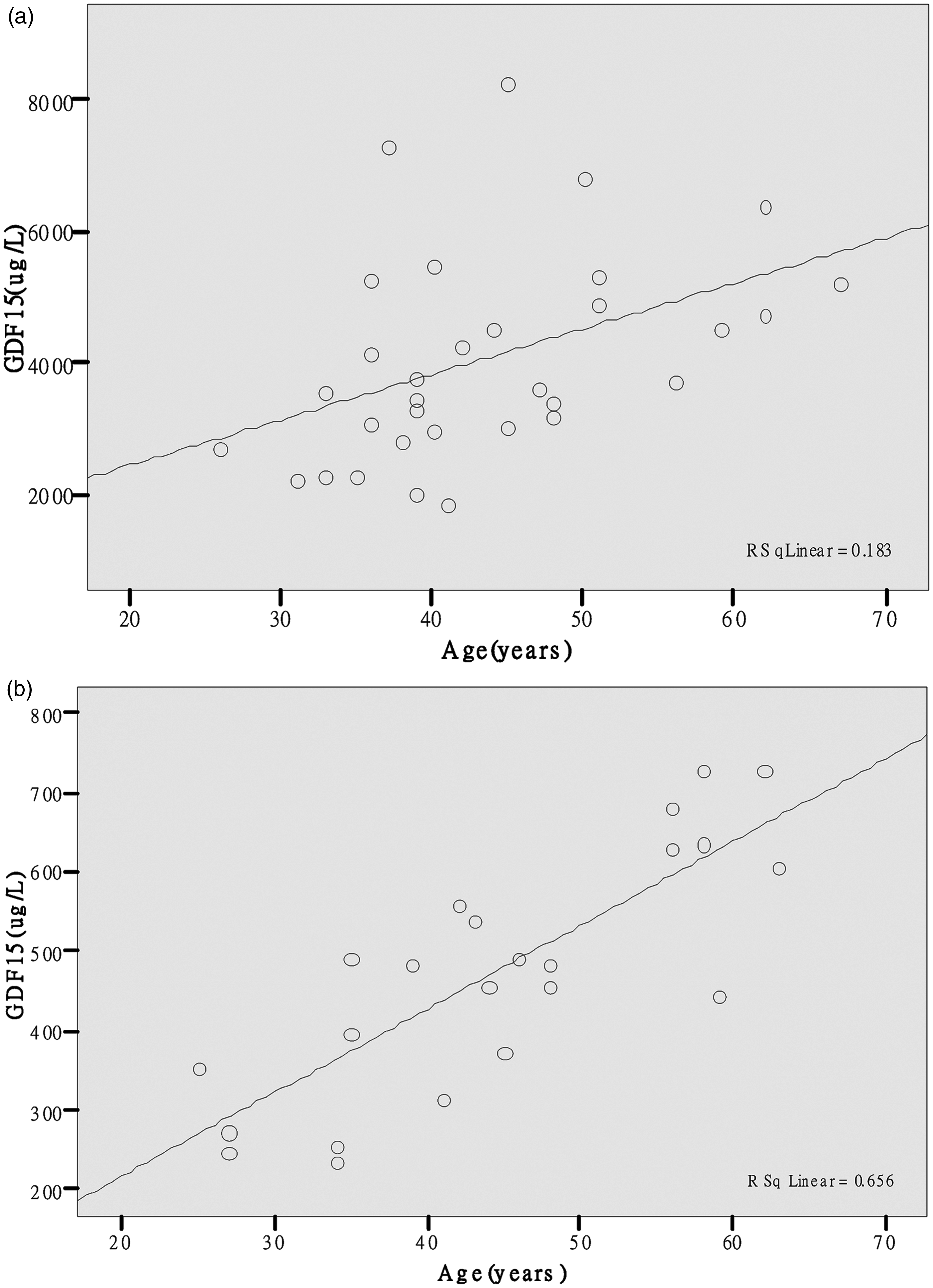
GDF-15 has been shown to correlate with age in healthy elderly individuals, 4 as well as patients with different stages of heart failure 5 and ESRD subjects. 6 These findings were replicated in our study in respect of age and ESRD, and furthermore demonstrated that age is predictive of GDF-15 concentration in apparently healthy adults. The mechanism underlying the increase in GDF-15 with age requires further investigation but is beyond the scope of this study. In our investigation, we have excluded heart failure or infection as a confounding factor. Tanno et al. 3 found an elevated GDF-15 concentration of 66,000 ± 9600 ng/L (range, 4800–248,000 ng/L) in a group of thalassaemia subjects. According to their ex vivo experiment, a GDF-15 concentration exceeding 5000 ng/L was required to suppress effectively the synthesis of hepcidin by hepatocytes. 3 We attempted to divide our HD groups into two subgroups based on the GDF-15 concentration but the patient number was too small. It is possible that the lack of correlation between GDF-15 and hepcidin may be related to the concentration of GDF-15 (4840.6 ± 1520.5, 1858.5–8287.8 ng/L) being too low to suppress hepcidin expression. A recent study of kidney allograft recipients (average glomerular filtration rate (eGFR) >50 mL/min using MDRD formula), revealed a correlation between GDF-15 and hepcidin but it was not possible to impute causation and exclude inflammation as a confounding factor. 7 The limitations of our study included the small sample size, the heterogeneity of the HD group regarding the aetiology of ESRD and the differing degrees of iron and rEPO supplementation.
In conclusion, our preliminary data would indicate a lack of correlation between serum GDF-15, hepcidin and other iron indices in a group of stable chronic HD patients. However, there were limitations to our experimental design.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Approved by ethics committees of the University of Hong Kong Shenzhen Hospital and the Nanshan Affiliated Hospital of Guangdong Medical College.
Guarantor
Pearl Pai.
Contributorship
X-YL conceived and designed the methodology; X-YL, JY, J-HL, S-LZ and JL performed the research. X-YL and PP analysed the data; JL contributed to reagents/materials/analysis tools; X-YL and PP did the writing up.
