Abstract
Background
Repeated freezing and thawing of plasma (or serum) may influence the stability of plasma (or serum) constituents. Despite the alarming warnings from commercial manuals that freeze–thaw cycles affect the stability of hormones in plasma (or serum), surprisingly little, consistent information about this concept is available in literature.
Methods
We studied the stability of 15 endocrine parameters (adrenocorticotropic hormone, osteocalcin, plasma renin activity, α-subunits, cortisol binding globulin, glucagon, inhibin B, fT4, TT4, TT3, rT3, TBG, TSH, chromogranin A and thyroglobulin upon repeated freeze–thaw cycles in plasma (or serum) samples from 10 volunteers. Blood was collected by venipuncture and after centrifugation and aliquoting, all samples were frozen at −20℃. Aliquots were thawed up to four times and changes in concentrations of endocrine parameters were compared to baseline condition.
Results
Repeated freeze–thaw cycling resulted in significant and relevant increases of plasma renin activity and a small decrease of adrenocorticotropic hormone.
Conclusions
For most of the analysed endocrine parameters, we found no effects of multiple freeze–thaw cycles despite alarming notifications in assay manuals. Plasma renin activity was the only endocrine parameter that showed significant and relevant changes following repeated freeze–thaw cycling.
Introduction
Repeated freezing and thawing of plasma may influence the stability of plasma (or serum) constituents. Results of analyses performed in plasma samples exposed to repeated freeze–thaw cycles might therefore differ from analyses performed in fresh, or only once thawed samples. Nowadays targeted laboratory diagnostics results in a reduction of initial blood sampling and laboratory requests. If necessary, add-ons are requested and thus valid information about freeze–thaw stability of plasma constituents is essential. Manuals of commercial assays for endocrine parameters often report limited freeze–thaw stability, despite lacking or inconsistent evidence. We investigated the influence of repeated freeze–thaw cycles on 15 endocrine parameters that are regularly requested in our laboratory and of which (consistent) information on freeze–thaw stability is lacking.
Methods
Ten volunteers (3 male, 7 female; age range 28–64 years, not fasted) donated 50 mL blood. The local Medical Ethical Committee of the Academic Medical Center confirmed that ethical approval was not required. Venous blood was collected in Vacutainer TM blood collection tubes (Becton Dickinson, Franklin Lakes, NJ, USA) with K2EDTA for adrenocorticotropic hormone (ACTH), osteocalcin and plasma renin activity (PRA) analyses, lithium heparin for α-subunits, cortisol binding globulin (CBG), glucagon, inhibin B, free thyroxine (fT4), total thyroxine (TT4), total triiodothyronine (TT3), reverse triiodothyronine (rT3), thyroxine-binding globulin (TBG), and thyroid stimulating hormone (TSH) analysis, and blank serum tubes for chromogranin A and thyroglobulin (TG). EDTA blood for ACTH measurements was stored on ice and quickly centrifuged (1559 g, 3000 r/min, 4℃) and aliquoted. All other tubes were quickly centrifuged at room temperature (RT) (1559 g, 3000 r/min). Serum or plasma was aliquoted into polystyrene tubes (within 2 h) and stored at −20℃ until further analysis (T0). Samples were thawed at RT during 1 h and frozen again at −20℃. This procedure was performed in respective aliquots once (T1), twice (T2) or three (T3) times on consecutive days. In addition, the 24 h-stability of hormones on ice in a transport box was evaluated to mimic transport conditions between laboratories. Frozen aliquoted samples were placed on an icepack in a Styrofoam transport box, which was left at RT for 24 h (T24h). After 24 h samples were frozen at −20℃ until further analysis.
ACTH was determined with a chemiluminescent immunometric assay (Immulite 2000, Siemens, Los Angeles, USA, interassay variation (CV) 9%). Chromogranin A (CgA II) was determined by TRACE technology on a Kryptor Compact Plus analyser (Thermofisher, Hennigsdorf, Germany, CV 2%). fT4 was determined using a fluoroimmunoassay and TSH using a fluoroimmunometric assay (Delfia, PerkinElmer Life and Analytical Sciences, Turku, Finland, CVs 4 and 6%). TT4, TT3 and rT3 were determined by an in-house 125−I RIA (CVs 5%, 8% and 9%). 1 TG was measured using a chemiluminescent immunometric assay (Lumitest Tg-PluS, Thermofisher, CV 5%). TBG (Thermofisher, CV 7%), glucagon (Millipore, Billerica, MA, USA, CV 9%) and CBG (Diasource, Nivelles, Belgium, CV 5%) were determined by commercial 125−I RIAs. α-subunits were determined by a commercial IRMA (Beckman Coulter, Prague, Czech Republic, CV 10%). PRA was determined with an assay based on the conversion of endogenous angiotensinogen into angiotensin I followed by an in-house RIA of angiotensin I (CV 12%). 2 Osteocalcin and inhibin B were measured using an ELISA assay (Osteocalcin Human Direct Elisa kit, BioSource, Nivelles, Belgium, CV 8% and Inhibine B Gen II, Beckman Coulter, Webster, Texas, USA, CV 8%). Analyses were performed within three months after blood collection, except for inhibin B and osteocalcin which were performed within five months. All aliquots of a sample from a single volunteer were assayed in the same run. Statistical analyses were performed using Prism Graphpad 5. Data were assessed for normality (d’Agustino-Pearson test) and outliers (Grubbs test). Changes in concentrations of hormones over time were analysed using repeated measures ANOVA following post hoc Bonferroni testing or paired t-tests.
Results and discussion
Effects of repeated freeze–thaw cycling and 24 h transport box condition on 15 endocrine parameters.
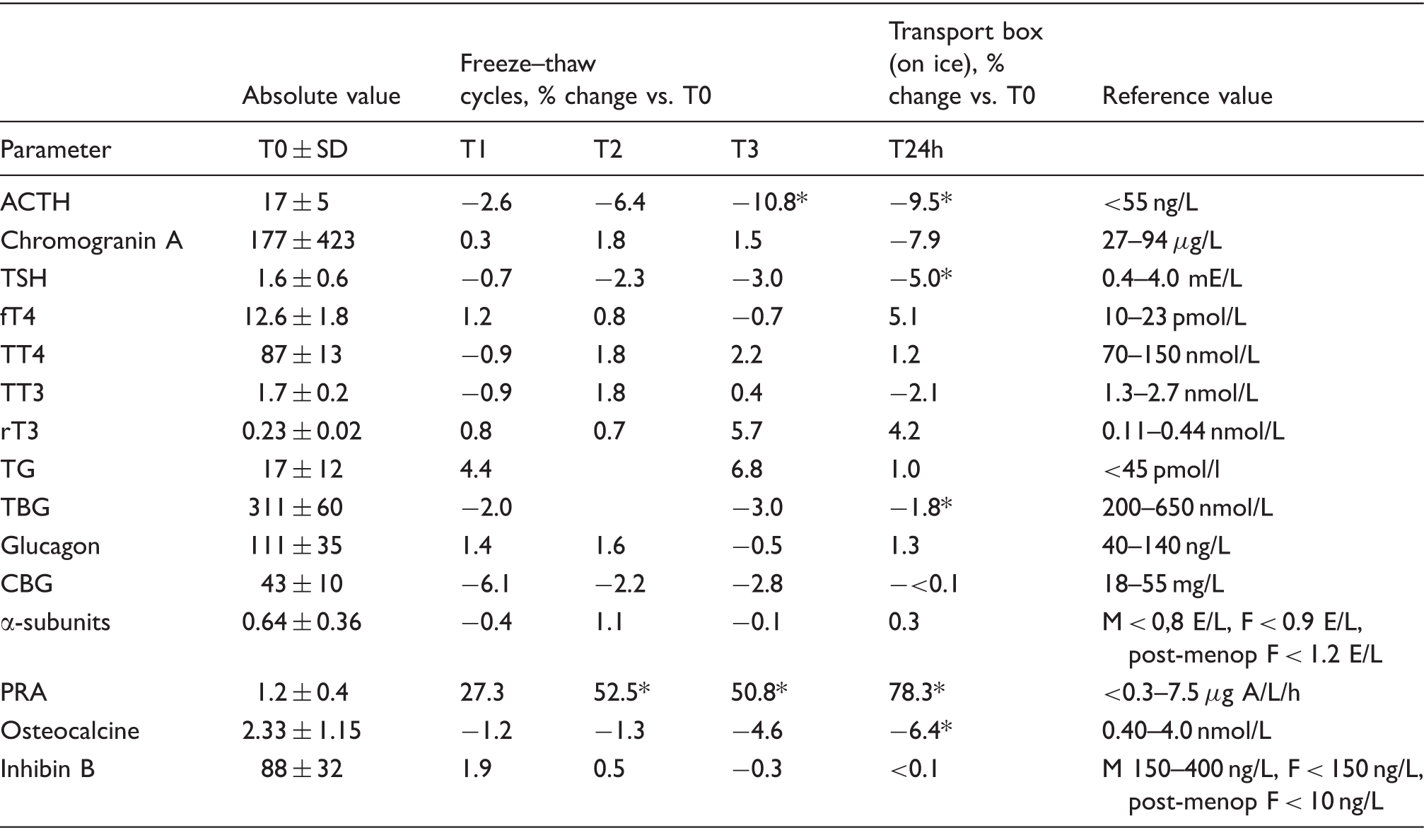
ACTH: adrenocorticotropic hormone; TG: thyroglobulin; PRA: plasma renin activity; CBG: cortisol binding globulin; TSH: thyroid stimulating hormone; TBG: thyroxine-binding globulin.
Note: Data were normally distributed except for chromogranin A and osteocalcin due to extreme values from two subjects. These values were not excluded because relative changes were not affected. For inhibin B: n = 6, because four subjects had values below LoQ (<10 ng/L). For PRA: n = 7, because two subjects had PRA values below LoQ (<0.3 μg Angiotensin 1/L/h) and one subject above the assays’ upper limit (>10 μg Angiotensin 1/L/h). T2 data for TBG and TG are missing due to erroneous aliquot distribution. *P < 0.05.
Increased PRA following repeated freeze–thaw cycling and upon storage on ice in a Styrofoam box for 24 h can be explained by unfolding of the prorenin segment and its cleavage by proteases (e.g. cryoactivation) at temperatures between −5℃ and 4℃. 3 The observed increases of PRA were larger than the interassay variation. PRA analysis thus enquires fresh or only once frozen samples. ACTH is notorious for its instability due to proteolytic degradation; however, effects of multiple freeze–thaw cycles on ACTH stability have not been frequently described. Given the interassay variation, the observed changes in ACTH concentrations should be considered of minimal clinical relevance. Data on freeze–thaw stability of osteocalcin in literature is inconsistent. This is probably due to assay differences as some assays measure osteocalcin fragments and others do not. 4 We used an ELISA measuring total osteocalcin and observed no changes in osteocalcin up to three freeze–thaw cycles, and a small, but significant, reduction in osteocalcin upon storage on ice in a Styrofoam box for 24 h. For TBG and TSH, we also found decreases upon storage on ice in a Styrofoam box for 24 h. Stability of TBG during different storage conditions has not been described in literature before. TSH stability was demonstrated at several storage conditions,5,6 however, TSH deterioration due to long-term cold storage has also been suggested. 7 In view of interassay variations, the observed changes in osteocalcin, TBG and TSH were considered of minimal (clinical) relevance.
Conclusion
For most analysed endocrine parameters, we found no effects of multiple freeze–thaw cycles despite alarming notifications in assay manuals. PRA was the only endocrine parameter that showed significant and relevant changes.
Footnotes
Acknowledgements
All volunteers, the Endocrine section of the Dutch Foundation for Quality Assessment in Medical Laboratories, and Thermofisher are thankfully acknowledged.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by the Endocrine section of the Dutch Foundation for Quality Assessment in Medical Laboratories. Thermofisher provided free CgA II kits.
Ethical approval
Not required.
Guarantor
JH.
Contributorship
All authors made a substantial contribution to the concept of the study. JH performed the study, analysed the results and drafted the article. AH and EE revised the article critically. All authors approved the article to be published.
