Abstract
Multiple myeloma (MM) is a plasma cell dyscrasia characterized by the malignant proliferation of a plasma cell clone that produces a monoclonal immunoglobulin. Diagnosis and management of patients with monoclonal gammopathies depend on accurate identification and characterization of monoclonal proteins. We present a 67-year-old male patient with anaemia, weakness and weight loss for six months. His physical examination was normal with no fever, and no bone lesions were present in the imaging studies. Laboratory investigations revealed low haemoglobin and albumin concentrations with high total protein and beta 2-microglobulin concentrations. Capillary zone electrophoresis with immunosubtraction method revealed a triclonal pattern of M-protein (IgG κ + IgG λ + IgA κ) which was not prominent with immunofixation electrophoresis. After bone marrow biopsy, MM with triclonal gammopathy was diagnosed and autologous stem cell transplantation was performed. Six months later, again a triclonal M-protein was detected by immunosubtraction method, and a relapse was confirmed with a second bone marrow biopsy. The occurrence of monoclonal and biclonal gammopathies can often be seen upon diagnosis in plasma cell dyscrasias and lymphoproliferative disorders, but triclonal paraproteins are very rare and their clinical significance is unknown. In this particular patient, triclonality was detected by an alternative method called immunosubtraction by capillary electrophoresis. The patient was resistant to therapy suggesting that more than one monoclonal M protein may be a negative prognostic factor, and with new technologies and methods, the number of patients with different monoclonal patterns may increase.
Introduction
Gammopathies are of clinical and diagnostic significance for plasma cell dyscrasias and lymphoproliferative disorders. Multiple myeloma (MM) is a plasma cell dyscrasia characterized by the malignant proliferation of a plasma cell clone that produces a monoclonal immunoglobulin. The excessive production of a single gammaglobulin (M-protein) and proliferation of a single plasma cell clone result in extensive symptoms such as renal failure, lytic bone lesions and anaemia. If a monoclonal component in serum is detected in a patient by serum protein electrophoresis and immunofixation studies, further investigations are conducted such as bone marrow biopsy and imaging studies, for the exact diagnosis and prognosis determination. Early diagnosis and intervention still remain key for preventing irreversible renal injuries in patients with MM since renal function impairment is one of the prognostic factors together with beta 2-microglobulin concentrations, haemoglobin concentrations, hypercalcaemia, presence of circulating plasma cells, albumin concentrations and cytogenetic status. 1 Myeloma therapy has progressed with the introduction of autologous stem cell transplantation (ASCT) and the recent introduction of the novel agents, thalidomide, lenalidomide and bortezomib. 2 ASCT after initial chemotherapy is the essential component of therapy in patients with MM, who are younger than 70 years, and achievement of complete remission (CR) is the most important step for prolonged survival. 3 In addition, sensitivity to the initial chemotherapy, measured by M-protein concentrations at the time of transplantation, is the prominent predictor of CR after transplantation. 1 With conventional chemotherapy regimens, the pretransplant and post-transplant CR rates are reported to be 5%–10% and 35%, respectively, and the median survival is approximately six years. 3
Occurrence of monoclonal and biclonal gammopathies can often be seen at diagnosis, but triclonal paraproteins are very rare and clinical significance is still unknown.4,5 Although abnormal protein bands (APB) on serum electrophoresis are seen more often after ASCT and thought to be transient, still true triclonal gammopathies are very rare after therapy. We hereby present a relapsing MM patient with triclonal gammopathy (IgG κ + IgG λ + IgA κ) at diagnosis and after ASCT, detected by capillary zone electrophoresis (CZE)–immunosubtraction method.
Case report
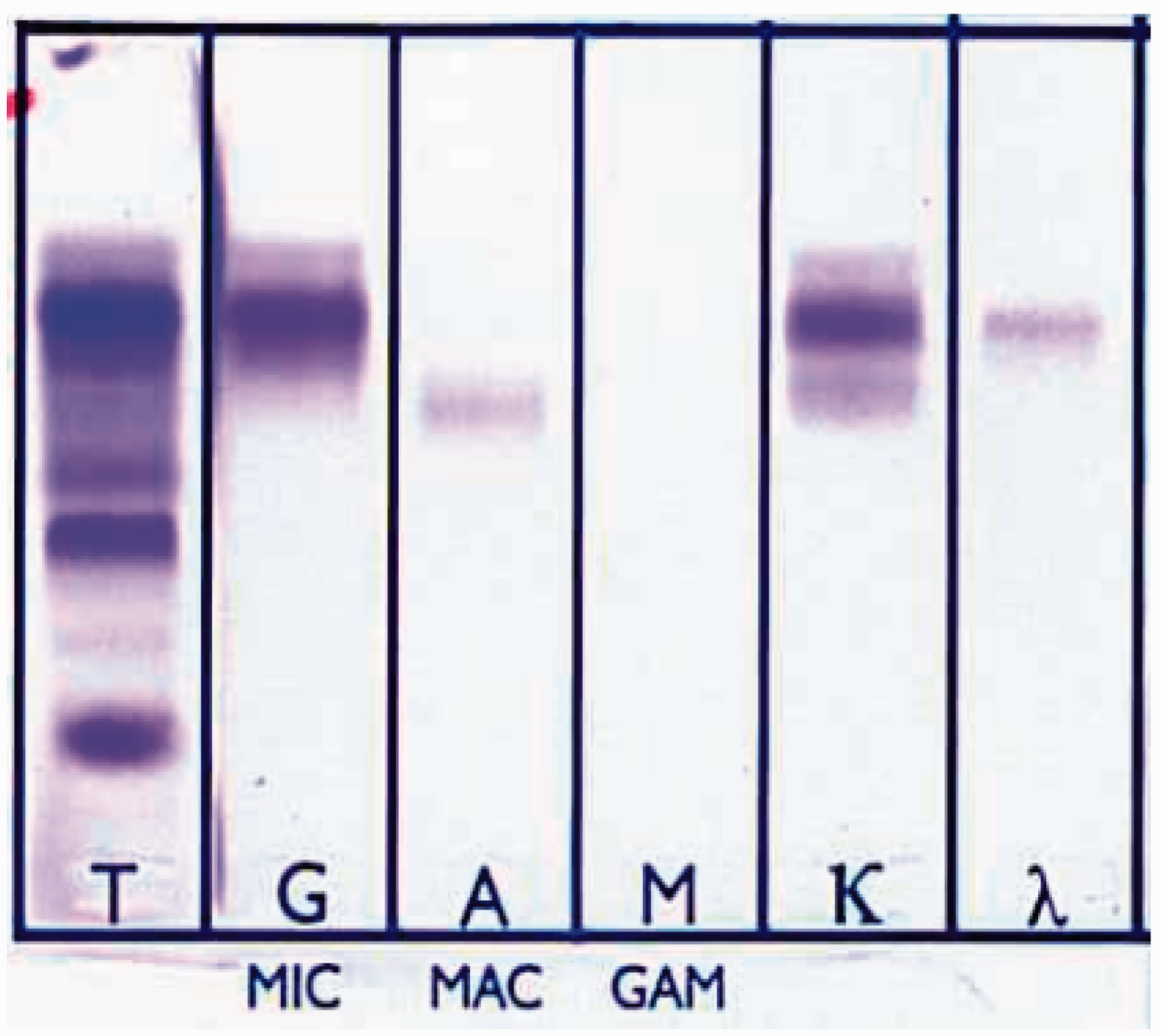
A 67-year-old male patient was referred for the evaluation of anaemia, weakness and weight loss for over the previous six months. He described a rapid weight loss of at least 8 kg in the last four months and obvious night sweats. His family and medical history were unremarkable. Physical examination was totally normal with no fever. He was 156 cm tall and his weight was 59 kg at the time he presented: his pulse rate was 88/min and arterial blood pressure was 120/80 mmHg. Laboratory work-up and imaging studies were conducted. Lytic bone lesions were not present in the skeletal tissue, evaluated by positron emission tomography. Laboratory findings were as follows (reference ranges are in parenthesis): serum calcium concentration (corrected for albumin) was 2.3 mmol/L (2.15–2.5 mmol/L); total protein, 94.1 g/L (64–83 g/L); albumin, 30 g/L (35–50 g/L); haemoglobin, 83 g/L (12–16); beta 2-microglobulin, 3.28 mg/L (1.09–2.53); lactate dehydrogenase, 316 U/L (10–250); erythrocyte sedimentation rate, 123 mm/h (0–20) and serologic studies were negative. Serum creatinine was (112.3 µmol/L) (61.9–106 µmol/L), estimated glomerular filtration rate (Cockroft–Gault equation) was 47.1 mL/min, and Bence Jones proteinuria was negative. Serum protein electrophoresis and CZE with immunosubtraction revealed a triclonal pattern of M-protein (IgG κ + IgG λ + IgA κ) which was not prominent with immunofixation electrophoresis (IFE) (Figures 1 and 2). Nephelometric quantitation of serum immunoglobulins was as follows: IgG 38.43 g/L (7–16), IgA 4.06 g/L (0.7–4), IgM 1.27 g/L (0.4–2.3), total kappa(tκ) 6.2 g/L (1.7–3.7), total lambda (tλ) 3.56 (0.9–2.1), free kappa (fκ) 21.18 mg/L (3.3–19.40), free lambda (fλ) 22.03 mg/L (5.71–26.3) and fκ / fλ 0.96 (0.26–1.65). Bone marrow trephine biopsy revealed 42% atypical plasma cell infiltration with CD38 and CD138 immunophenotypes and positive for IgG, IgA and kappa, lambda immunohistochemical stains. Patient was diagnosed to be MM, stage IIA (Durie and Salmon staging system), and in cytogenetic studies he was found to be negative for immunoglobulin heavy chain translocations t(11;14), t(14;16) and 17 p deletion. After initial chemotherapy with bortezomib/dexamethasone, ASCT was performed. Biochemical studies were performed by Roche Serum immunosubtraction report by using V8 automated clinical capillary electrophoresis (Helena, UK). Arrows indicate specifically subtracted parts of immunoglobulins which means those are monoclonal paraproteins. In this report, a triclonal paraprotein is present: IgG κ + IgG λ + IgA κ (report shows heavy and light chains seperately). Immunosubtraction method separates serum proteins following an incubation of serum in the presence of antisera for specific heavy and light chains, thus removing them and detection is based on their absence. The second line under the arrows shows subtracted parts. SPE: serum protein electrophoresis. Serum immunofixation electophoresis (IFE) with agarose gel (Helena, UK). A prominent gamma (G) heavy chain can be observed easily; however, monoclonal alpha (A) heavy chain is not distinct. T lane shows serum protein electrophoresis (SPE).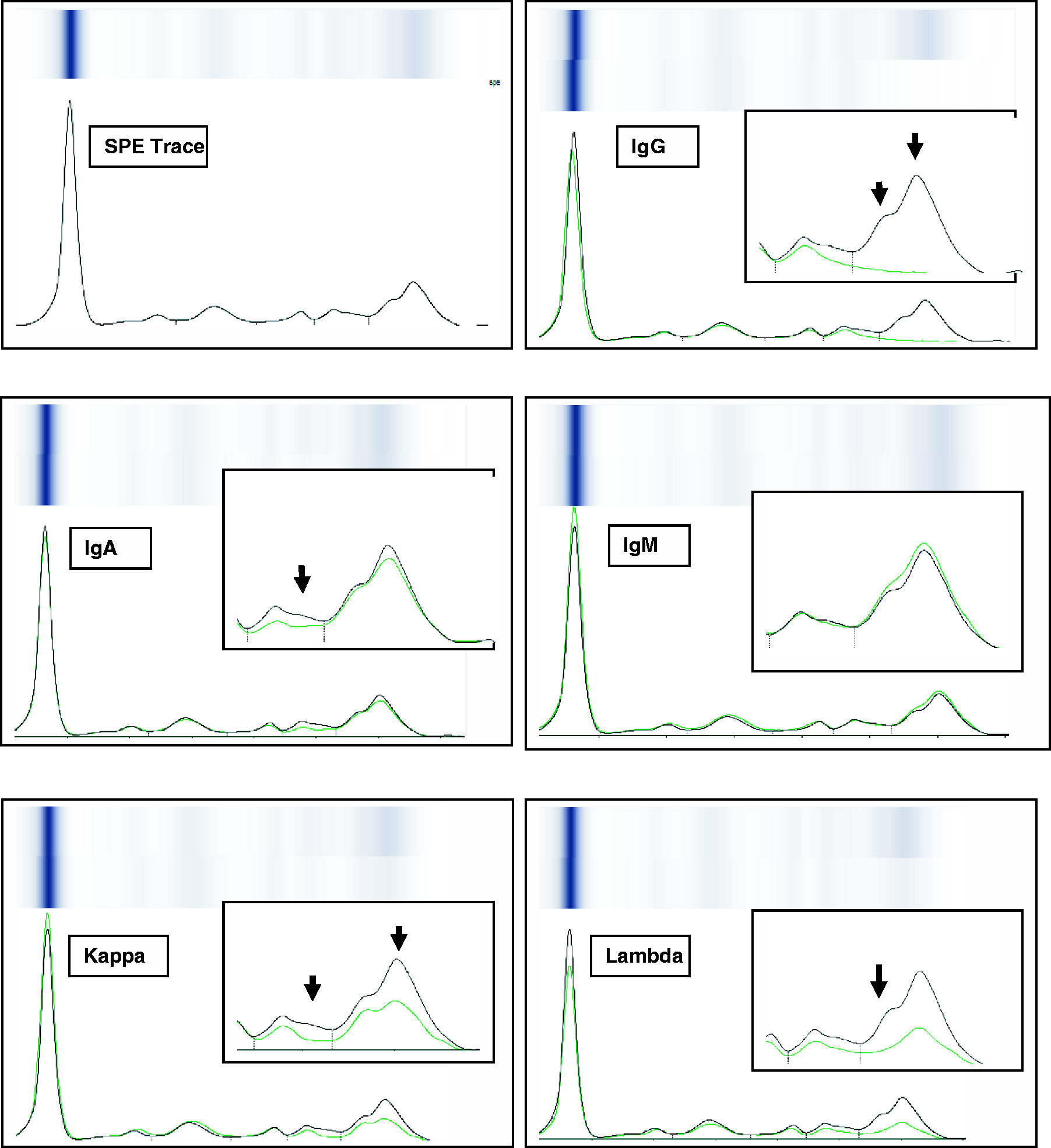
Six months after the ASCT, a triclonal pattern of M-protein (IgG κ + IgG λ + IgG κ) was again detected, but this time alpha (IgA) heavy chain was replaced by gamma (IgG) heavy chain. Nephelometric quantitation of serum immunoglobulins was as follows: IgG 19.41 g/L (7–16), IgA 0.25 g/L (0.7–4), IgM 0.13 g/L (0.4–2.3), tκ 4.2 g/L(1.7–3.7), tλ 1.56 (0.9–2.1), fκ 19.18 mg/L (3.3–19.40), fλ 12.03 mg/L (5.71–26.3) and fκ/ fλ 1.59 (0.26–1.65). Oligoclonal IgG evaluation was performed with isoelectric focusing–immunoblotting, and two monoclonal regions were detected with some other oligoclonal bands. Hence, a new bone marrow trephine biopsy was performed, and immunohistochemical studies revealed a 27% CD138 positive plasma cells with positive kappa and lambda stains. A disease relapse was confirmed, and a second chemotherapy and ASCT were planned although there are controversies about the effectiveness of the second ASCT in relapsing patients. 1
Discussion
Diagnosis and management of patients with monoclonal gammopathies depend on the accurate identification and characterization of monoclonal proteins. MM is characterized by the clonal expansion of malignant bone marrow cells engaged in the production of a monoclonal immunoglobulin. In addition to monoclonal pattern, biclonal or triclonal paraproteins are occasionally present at diagnosis and also switching of paraprotein can occur during disease relapse. 2 Accurate disease monitoring is critical after therapy in MM, to enable prompt detection of ineffective therapy, to confirm achievement of CR or to detect early relapse before the occurrence of an organ damage. We present this patient in the light of the rarity of reported cases of triclonal gammopathies at diagnosis and after ASCT; furthermore, paraprotein detection in this particular patient was performed with an alternative method – CZE–immunosubtraction method. Although classical agarose gel IFE shown in Figure 2 suggested the presence of three monoclonal proteins, triclonality was not prominent and it was difficult to interpret accurately.
Triclonal gammopathies were shown to be associated with other malignancies and inflammatory pathologies in some case reports,4–7 but there was no accompanying disease in our patient. Previous case reports with bi- and triclonal gammopathies conclude that there was no significant impact on prognosis.4,8 However, our patient was resistant to therapy suggesting that more than one monoclonal M-protein may be a negative prognostic factor which must be confirmed with more case reports.
A triclonal pattern continued after the therapy in our patient with a shift from IgA κ to IgG κ in heavy chains. Although appearance of monoclonal components after stem cell transplantation does not always reflect a neoplastic process, 9 determination is still important for monitoring the response to therapy and to protect the kidneys from excess free light chain excretion. However, appearance of such different bands must be followed cautiously as they may represent a true isotype switching, as was the case in our patient, in order to foresee a potential relapse. In a study by Hall et al., 10 it is demonstrated that 48% of patients had a relapse after 15 months after ASCT. APB after ASCT can be oligoclonal or monoclonal of origin. These bands are thought to be due to transient dysregulation of the regenerating B cell compartment during the post-transplant period. The appearance of APB can cause problems for the laboratory since they may potentially represent a change in the antibody production of the previous plasma cell clone or the emergence of a new malignant clone which is very difficult to discriminate in the first 3–6 months. Moreover, excessive production of monoclonal proteins in relapsing MM patients can result in renal failure due to precipitation of light chains within the distal and collecting tubules as cast formation (cast nephropathy). Free light chain interaction with proximal tubule cells can also activate inflammatory cascades that lead to tubulointerstitial fibrosis which is a major feature of myeloma kidney. Hence, early diagnosis and intervention is very important in preventing renal injuries in relapsing MM cases, too. Serum immunoassays such as IFE, immunosubtraction method with CZE and immunonephelometric quantitation of heavy and light chains of immunoglobulins provide valuable information about the type of the monoclonal paraprotein. Renal toxicity is shown to be variable among the different light chains and also dose dependent, suggesting that both free light chain structure and concentration determine cellular toxicity in cast nephropathy. 11 Hence, cases with multiple monoclonal proteins, such as the present case, must be carefully monitored for potential renal injury, and it should be kept in mind that the APBs after ASCT can display the actual relapse.
In addition to the clinical importance of triclonality, detection of this particular pathology is diagnostically important. IFE by agarose gel and immunosubtraction method with capillary electrophoresis can be used alternatively in detecting type of paraproteins. 12 In IFE, specific antibodies are overlaid after electrophoresis and the corresponding immunoglobulin heavy and light chains are bound and stained. IFE is a highly sensitive and specific method to classify monoclonal immunoglobulin. 13 With the development of CZE, an alternative method combined with CZE for identifying monoclonal immunglobulins has emerged into the field: immunosubtraction method, which is based on the studies in 1980s. 14 Immunosubtraction method – also called immunotyping or immunodisplacement – separates serum proteins following an incubation of serum in the presence of antisera for heavy and light chains thus removing them, and detection is based on their absence when compared to serum protein electrophoresis. In CZE, the sample runs through the narrow capillary tubes, and direct protein detection is performed by a measurement at 200 nm, eliminating the need for staining. Quick and automated reporting and easy interpretation are advantages of CZE–immunosubtraction method over gel electrophoresis. Although according to the comparison studies and recommendations for standardized reporting of protein electrophoresis, the detection limit for paraprotein of immunosubtraction method is reported to be higher than IFE 15 and non-IgG paraproteins are poorly detected, 12 for this particular patient, we have detected the triclonality and especially IgA κ by immunosubtraction method using CZE which was not obvious in IFE. Currently, triclonal gammopathy incidence and importance is unknown, but with the emergence of new technologies and methods, patients with different monoclonal patterns may be diagnosed accurately leading us to a more precise approach to these patients.
Footnotes
Acknowledgements
We would like to thank to our colleagues Yasemin Doventas and Sema Gogus for their valuable contributions.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
The written informed consent to publication has been obtained from the patient.
Guarantor
FBA.
Contributorship
FBA, MA and MS conceived and gathered data for the case report. MA obtained written informed consent from the patient. AC and IU were involved in literature search and data analysis. FBA wrote the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
