Abstract
Background
Daratumumab (Darzalex) is a human IgG1 kappa monoclonal antibody targeting CD38 that has been recently approved for the treatment of refractory multiple myeloma. As it is a monoclonal protein, it can be detected on routine serum protein electrophoresis and by immunofixation.
Methods
Serum samples from four patients were analysed by serum protein electrophoresis immediately pre- and post-treatment with daratumumab.
Results
For all four patients, daratumumab was visible on serum protein electrophoresis as an additional small band (approximately 1 g/L) in the slow gamma region.
Conclusion
Diagnostic laboratories should be aware that daratumumab can be detected on routine serum protein electrophoresis of myeloma patients and should liaise closely with clinicians to ensure the presence of daratumumab is not misinterpreted as development of a new monoclonal protein.
Keywords
Introduction
Daratumumab is a human IgG1 kappa monoclonal antibody that binds with high affinity to CD38 molecules which are highly expressed on the surface of multiple myeloma cells. It is marketed by Janssen Pharmaceuticals as DARZALEX™ for the treatment of patients with multiple myeloma who are refractory to both proteasome inhibitors and immunomodulatory therapies. The modes of action include the induction of tumor cell death through apoptosis, complement-dependent cytotoxicity, antibody-dependent cellular phagocytosis, antibody-dependent cellular cytotoxicity, and a reduction in immune-suppressive myeloid derived suppressor cells (MDSCs), regulatory T cells (Tregs) and B cells (Bregs), which express CD38. 1
The recommended dose of daratumumab is 16 mg/kg body weight given as an intravenous infusion weekly for the first eight weeks then every two weeks until week 24 and then every four weeks as ‘maintenance treatment’. At this dosage, the monoclonal IgG kappa is seen as an additional paraprotein band on the serum protein electrophoresis and as an IgG kappa on serum immunofixation. The serum half-life of IgG1 is approximately 21 days so this small additional paraprotein band will remain visible during the patients’ treatment and for some weeks after the drug has been stopped. The IgG of the daratumumab will also contribute to the total IgG quantification measured immunochemically; this will be most noticeable in patients with low background IgG concentrations and non-IgG monoclonal proteins.
A monoclonal protein will always have the same electrophoretic mobility, but an individual patient’s electrophoretic pattern may change throughout their disease course. The paraprotein may disappear, new paraproteins may appear, e.g. retained Bence Jones protein, and there may be an ‘oligoclonal’ pattern seen secondary to infection or in ‘immune reconstitution’ following aggressive chemotherapy or peripheral blood stem cell transplantation. One study reported an oligoclonal humoral response (defined as the presence of a serum and/or urine monoclonal spike on immunofixation that was different from the original myeloma protein either in heavy and/or light chains as well as in electrophoretic migration) in 33.3% of their patients in complete remission following treatment for myeloma. 2 The disappearance of a monoclonal protein by protein electrophoresis or by immunofixation is part of the International Myeloma Working Group (IMWG) Uniform Response Criteria for Multiple Myeloma for very good partial remission and complete remission post treatment. 3 The correct identification of the reappearance of a paraprotein, of the appearance of a new monoclonal protein or of an ‘oligoclonal banding’ pattern are important findings that will contribute to the management of patients. Accurate identification and recording of monoclonal proteins are therefore essential for good patient management. Equally, it is important that the presence of daratumumab is not erroneously interpreted as the emergence of a new monoclone from the patient’s B-cell population.
Capillary zone electrophoresis (CZE) is an ideal tool for precise documentation of electrophoretic mobility of paraprotein bands. The X-axis co-ordinate can be used to record a peak's delimits and the position of the peak apex. Although there will be some differences between the capillaries and between instruments, a monoclonal protein will always migrate in a comparable position.
This short report describes the serum protein electrophoretic patterns of patients being treated with daratumumab and highlights the importance of detailed recording of the electrophoretic mobility of paraproteins.
Materials and methods
Details of patients being treated with daratumumab.
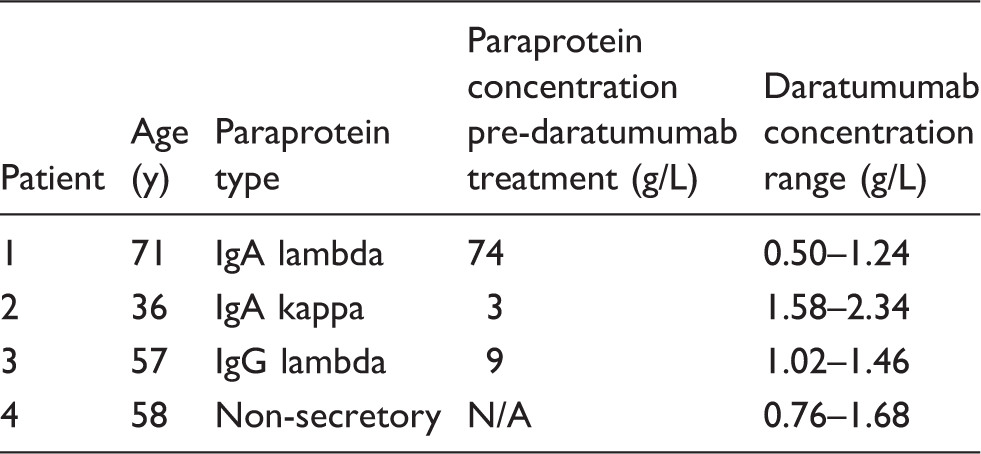
Note: The daratumumab concentration range, as calculated from capillary zone electrophoresis results, is shown for data from four (patients 1 and 2) or three (patients 3 and 4) months of treatment.
Results
Figure 1 shows the serum protein electrophoresis patterns for samples taken pre- and post-daratumumab treatment. Daratumumab migrates as a small distinct band with consistent electrophoretic mobility. The daratumumab concentration derived from the serum total protein and the area under the curve for the peak measured is approximately 1–2 g/L (the range in concentration for all samples received so far for all four patients is shown in Table 1).
Serum protein electrophoresis from four patients showing the pre-daratumumab separation with the monoclonal protein band shaded and indicated with a white arrow. Patient 4 did not have a visible monoclonal protein pre-daratumumab. The post-daratumumab separations show the additional band of daratumumab indicated with a black arrow. It is important to note that the peaks of daratumumab all show the same electrophoretic mobility.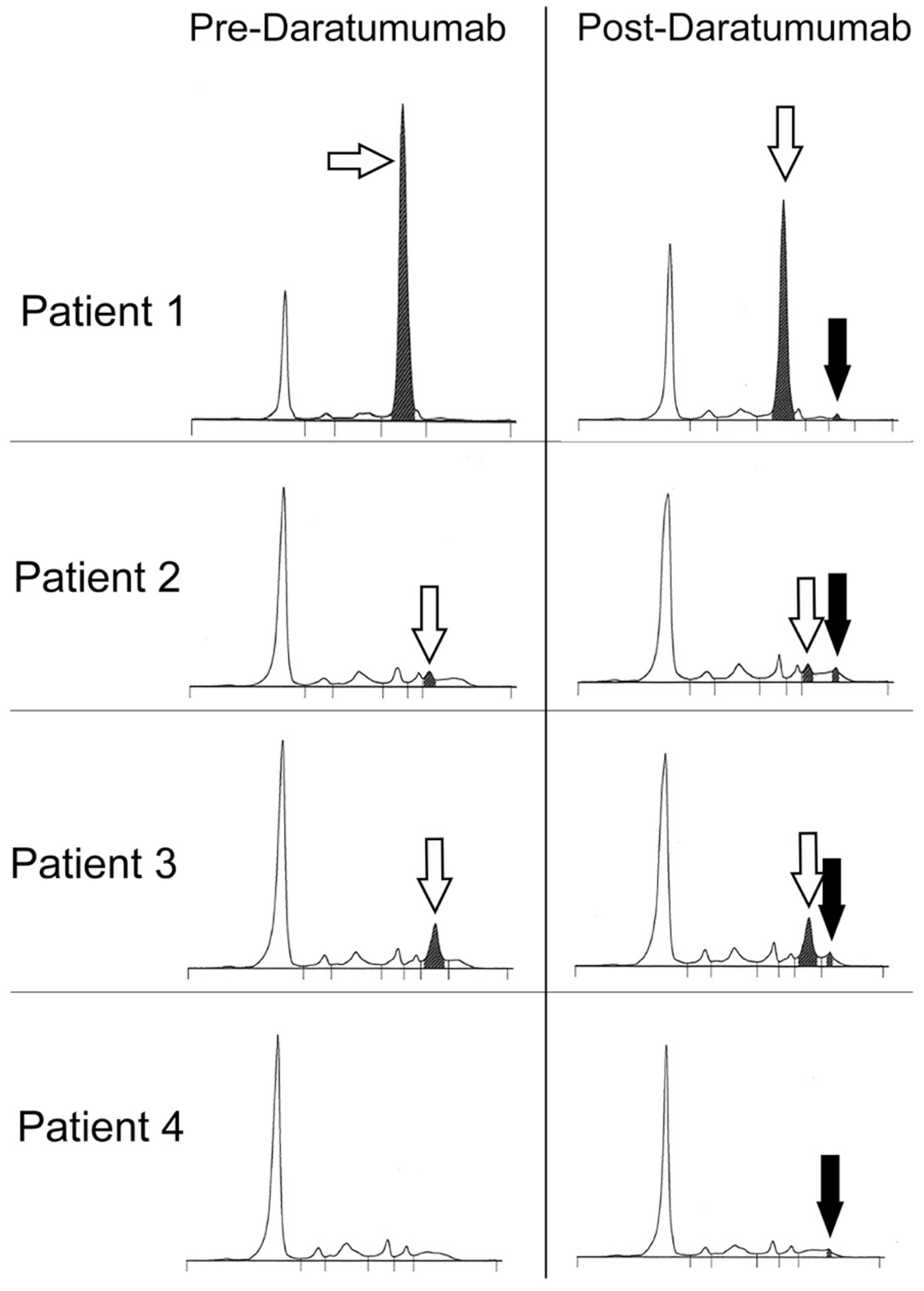
Discussion and conclusion
Biologics are increasingly prescribed and laboratories must be aware that the drug itself may be visible on serum protein electrophoresis. Careful evaluation of electrophoresis results is vital so that the appearance of a band due to daratumumab is not mistaken for the emergence of a new monoclonal immunoglobulin or of an oligoclonal response. To facilitate this, laboratories will need good communication with their Haematology team.
The patient results shown here are chosen to demonstrate the clear distinction between the patient's paraprotein and daratumumab. However, it is possible that in some cases, the patient’s paraprotein will have similar electrophoretic mobility to daratumumab and in this situation, no additional band will be seen.
This short report highlights the importance of detailed knowledge of serum protein electrophoresis and an understanding of the possibility that new treatment regimens may themselves contribute to the serum protein electrophoresis patterns.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
JS.
Contributorship
RP and JS conceived the work, JS and RDW analysed patient data. JS wrote the first draft, RDW created the figure and table. All edited and approved the manuscript.
