Abstract
Background
This article describes three patients in whom measured serum testosterone concentrations were found to be artifactually high due to interference from norethisterone medication. This interference was investigated further by distributing samples containing norethisterone through an external quality assessment scheme.
Methods
Serum samples containing different concentrations of norethisterone were distributed to participants in the UK external quality assessment scheme (UK NEQAS) for female testosterone in order to assess the degree of interference from norethisterone in different commercially available immunoassay and liquid chromatography-tandem mass spectrometry methods for measurement of testosterone.
Results
The results have shown that apparent serum testosterone concentrations in excess of 5 nmol/L may be obtained using the Roche E170 Modular immunoassay method for samples collected from patients taking norethisterone medication and this interference can be reproduced by adding norethisterone to serum samples.
Conclusions
Although the biggest interference is seen with the Roche system, there is a small effect in the Siemens ADVIA Centaur assay and some of the other immunoassays may also be affected to a much lesser extent. Norethisterone does not interfere in testosterone measurements obtained by liquid chromatography-tandem mass spectrometry.
Introduction
Reports describing interferences in immunoassays for female concentrations of testosterone are not uncommon.1–7 Abnormal results can come to light when results are considered in relation to the clinical and biochemical picture and discussed with clinicians. This paper describes a series of three case reports where falsely high testosterone concentrations were reported for patients on norethisterone medication and an evaluation of this interference in different testosterone assays.
Case reports
The first patient was a 15-year-old girl who presented to her GP with disordered menstrual bleeding and amenorrhoea and was noted to have had testosterone results of 1.1 nmol/L and 0.8 nmol/L on two previous occasions (adult reference interval 0.3–1.7 nmol/L). She had a body mass index of 22.9 kg/m2 with a height of 166 cm and had recently gained weight. The patient was initially prescribed a dose of norethisterone of 10 mg twice a day which was subsequently modified to 5 mg three times a day. Measurement of testosterone on the Roche Modular system, whilst the patient was taking norethisterone, produced a result of 15.3 nmol/L; the result on the same sample obtained by tandem mass spectrometry was <0.2 nmol/L (<1.9 nmol/L). The gonadotrophin results on this sample were an LH of <0.1 IU/L (reference intervals for follicular phase 2.4–12.6 IU/L and luteal phase 1.0–11.4 IU/L) and an FSH of 0.4 IU/L (reference intervals for follicular phase 3.5–12.5 IU/L and luteal phase 1.7–7.7 IU/L) with an SHBG of 15.9 nmol/L (adult female reference interval 25–122 nmol/L). Norethisterone medication was discontinued whilst the possibility of interference in the testosterone assay was being investigated and a serum sample collected from the patient six weeks later showed a testosterone concentration of 1.2 nmol/L with an SHBG of 13.1 nmol/L, LH of 3.6 IU/L and FSH of 8.0 IU/L all measured using Roche assays.
The second patient was a 24-year-old woman noted previously to have a history of iron deficiency anaemia, low folate and infertility characterized by absent ovulation. A serum testosterone measured at an undefined point in her menstrual cycle produced a result of 0.6 nmol/L with an SHBG of 110.2 nmol/L, LH of 14.3 IU/L and FSH of 13.5 IU/L using the Roche modular assays. The patient was given a 10-day course of norethisterone at a dose of 5 mg three times a day. A serum sample was received in the laboratory on the following day for measurement of testosterone and the result was 5.8 nmol/L with an SHBG of 120.6 nmol/L, LH of 13.0 IU/L and FSH of 9.3 IU/L. This sample was referred for measurement of testosterone using tandem mass spectrometry and the result was 0.5 nmol/L. A second sample received 10 days later also showed a raised testosterone concentration of 5.3 nmol/L on the Roche assay. The possibility of heterophillic antibody interference was investigated in this sample using antibody blocking tubes and the testosterone result, after treatment of the sample using these tubes, was unchanged at 5.5 nmol/L. After withdrawal of norethisterone medication, the Roche serum testosterone result for the patient, on a sample collected two months later, was 1.4 nmol/L.
The third patient was aged 32 years and was taking norethisterone at a dose of 5 mg three times a day for irregular periods, when a laboratory testosterone result of 7.1 nmol/L was reported using the Roche assay (reference interval 0.3–1.9 nmol/L) together with an SHBG (Immulite) of 6 nmol/L (reference interval 30–120 nmol/L), LH (Abbott Architect) of <1 IU/L, FSH (Abbott Architect) of <1 IU/L, prolactin (Abbott Architect) of 1018 mU/L (reference interval 25–630 mU/L), 17 OHP (in-house radioimmunoassay Glasgow, RIA) of 3.0 nmol/L (reference interval 0–13 nmol/L), DHEAS (Immulite) of 5.7 µmol/L (reference interval 2–11 µmol/L) and androstenedione (in-house RIA Glasgow) of 4.1 nmol/L (reference interval 0.6–8.8 nmol/L). The presence of macroprolactin was excluded. Measurement of testosterone using an extraction procedure followed by an immunoassay (Abbott Architect) produced a result of 0.9 nmol/L and analysis by tandem mass spectrometry gave a result of 0.4 nmol/L. A repeat sample collected from the patient six days later gave a measured testosterone result of 7.8 nmol/L using the Roche Modular assay.
Materials and methods
One 5 mg tablet of norethisterone was obtained from the hospital pharmacy department, crushed using a pestle and mortar and dissolved in mass spectrometry grade methanol. Insoluble material was removed using centrifugation and the supernatant solution was added to a base pool of serum to produce a norethisterone concentration of 30 µg/L (100 nmol/L). An aliquot of this solution was mixed in equal proportions with the base pool of serum to produce a concentration of 15 µg/L (50 nmol/L). All three samples were distributed to participants registered with UK NEQAS for the measurement of testosterone in female samples. Heterophillic antibody blocking tubes were supplied by Skybio limited, Bedfordshire, UK.
Results
Concentrations of testosterone obtained for patients taking norethisterone using different assays.
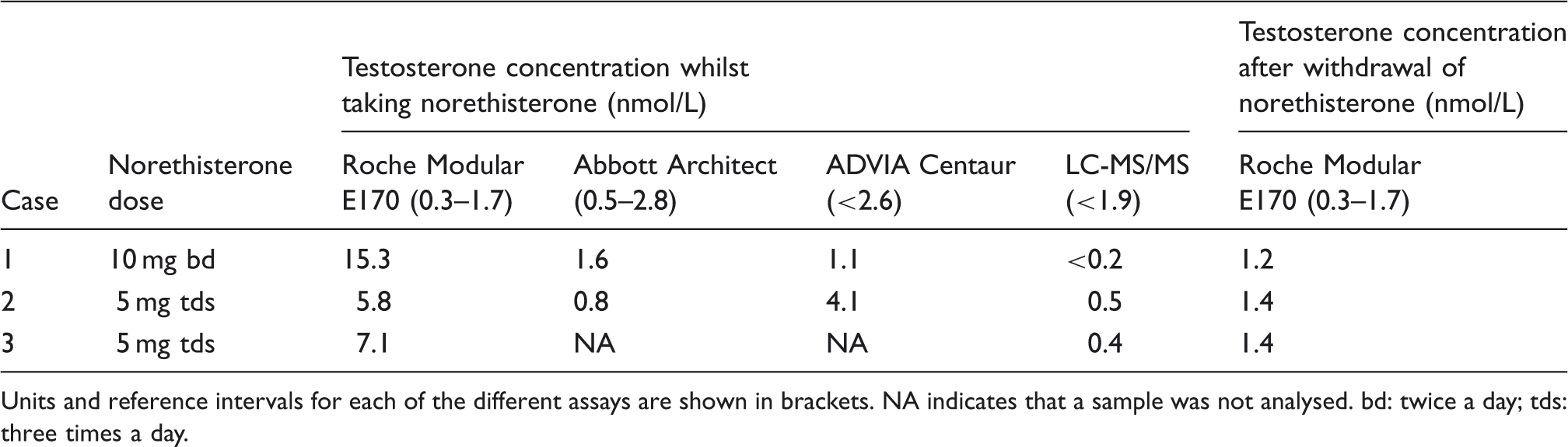
Units and reference intervals for each of the different assays are shown in brackets. NA indicates that a sample was not analysed. bd: twice a day; tds: three times a day.
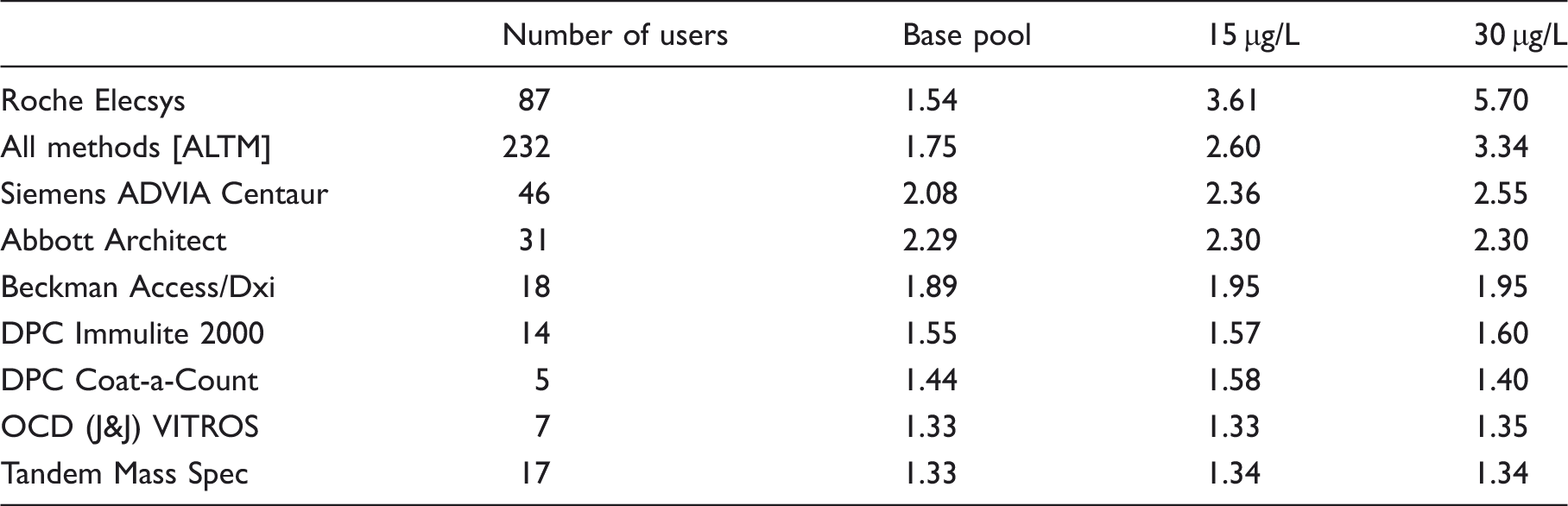
Method mean results for testosterone (nmol/L) obtained from the UK NEQAS external quality assessment scheme for a base pool of serum and the same serum containing either 15 µg/L or 30 µg/L norethisterone for the overall consensus mean (All methods [ALTM]) and the most commonly used methods.
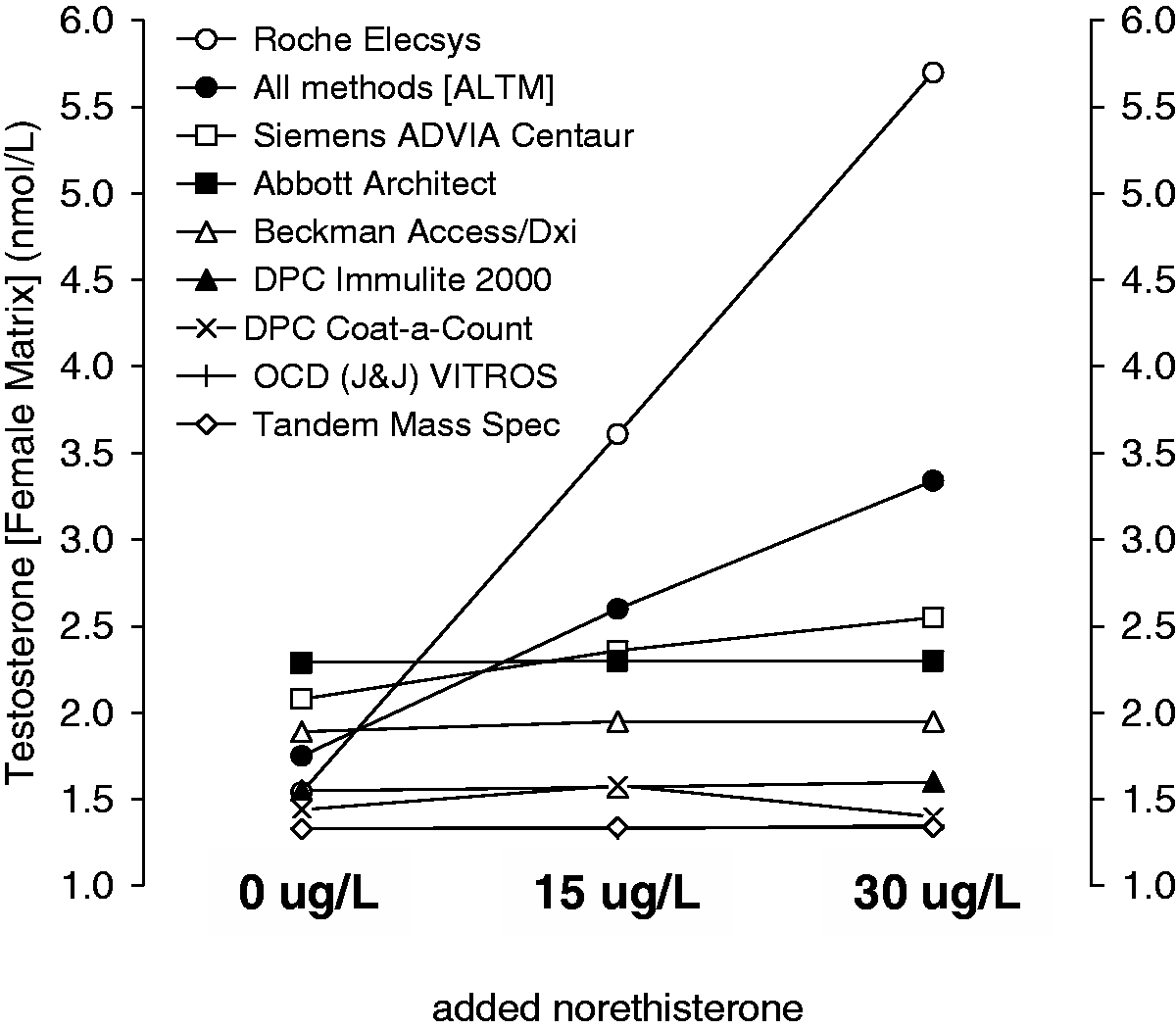
Distribution of serum testosterone concentrations obtained with different commercial immunoassays for pooled female serum samples containing added norethisterone at concentrations of 0, 15 and 30 µg/L.
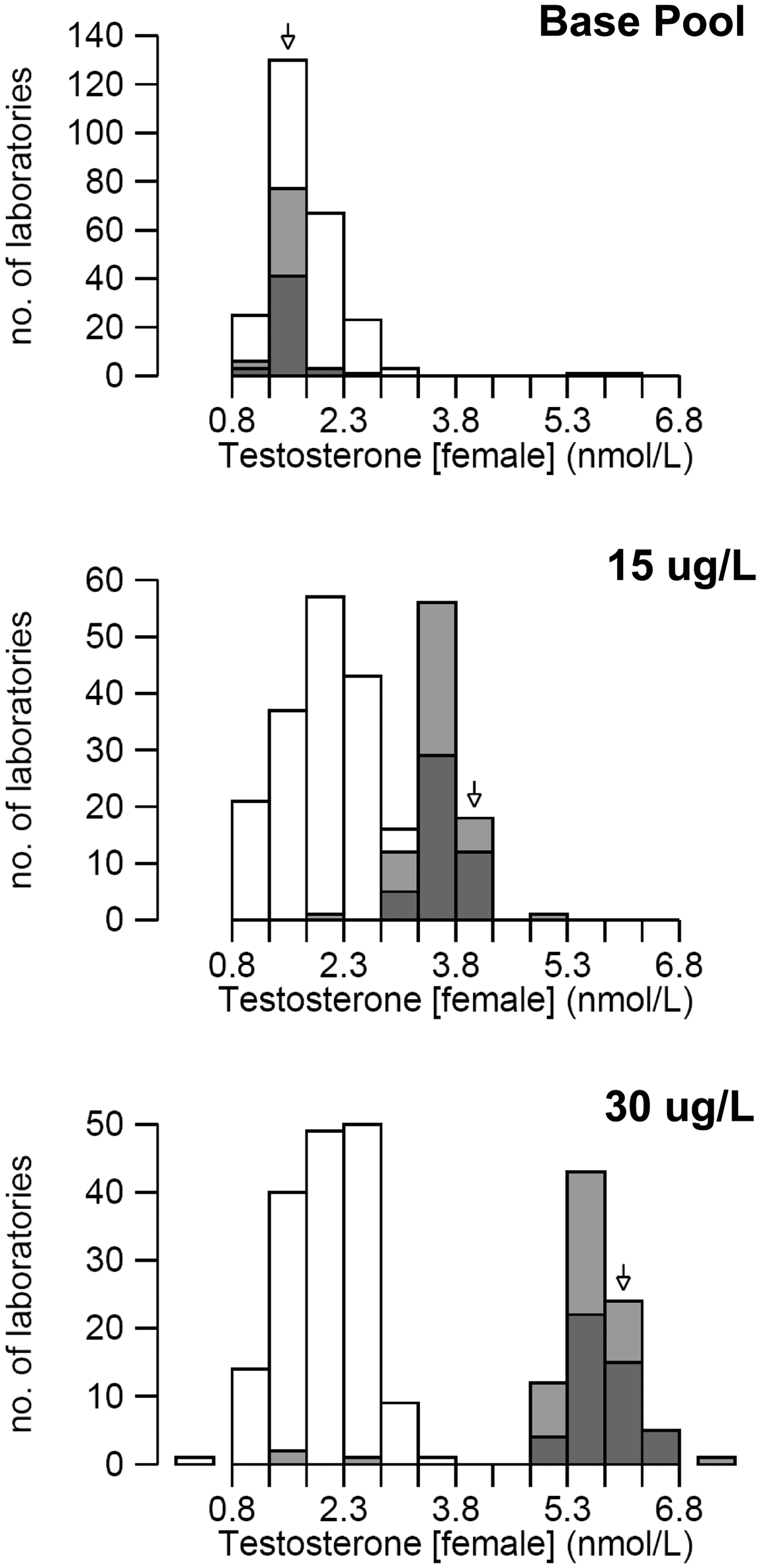
Histograms showing the data obtained for all laboratories subscribing to the UK NEQAS EQA scheme for pooled female serum samples containing added norethisterone at concentrations of 0, 15 and 30 µg/L. The shaded bars show the data obtained with the Roche method, and the open bars show the data obtained with the other methods.
Discussion
Norethisterone (19-nor-17α-ethynyltestosterone) is classified pharmacologically as a progestagen without oestrogen action and is a component of a number of commonly prescribed combined and progestogen only contraceptive pills and implants, for example Primolut, Utovlan, Micronor, Noriday, Loestrin, Brevinor, Ovysmen, Norimin, Norinyl-1, BiNovum, TriNovum, Synphase and Evorel. Norethisterone exerts a local influence on the endometrium leading to the cessation of dysfunctional bleeding by conditioning the endometrium into a state typical of that found in the final stages of the luteal phase. In adequately oestrogenized women, withdrawal bleeding then occurs 2–4 days after discontinuation of norethisterone. The drug has an inhibitory effect on gonadotrophin secretion and affects the regulation of basal body temperature. Peak serum concentrations of norethisterone are achieved within about 1.5 h of oral administration. Due to a marked first pass effect through the liver, the bioavailability of norethisterone after an oral dose is quoted to be 64%.
9
Norethisterone is over 95% bound to a combination of albumin and sex hormone-binding globulin (SHBG) in serum with approximately 35% of the drug bound to SHBG and 61% bound to albumin. Norethisterone is mainly metabolized by saturation of the double bond in the terminal 6 carbon ring (Figure 3) and the reduction of the 3-keto group to a hydroxyl group, followed by conjugation to sulphates and glucuronides. The drug is excreted in both urine and faeces. Norethisterone has a molecular mass of 298.4 g/mol.
Molecular structures of testosterone and norethisterone.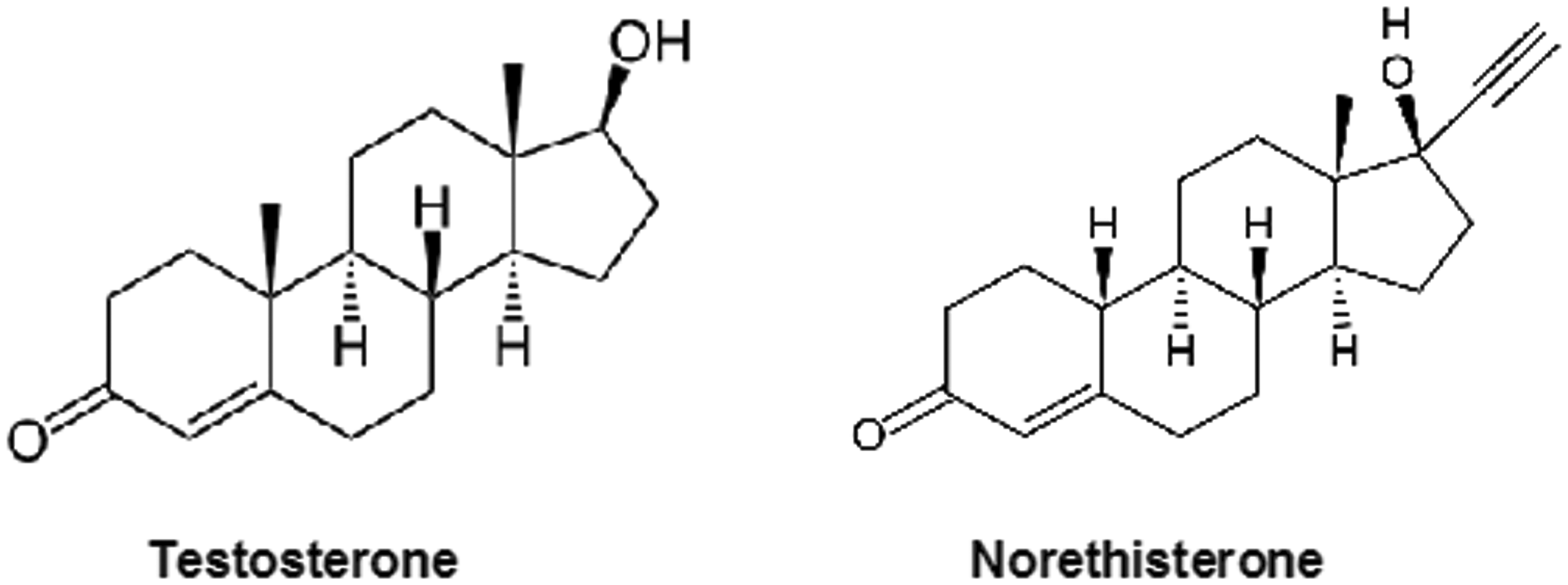
During multiple-dose daily administration of norethisterone, an accumulation of the parent compound is unlikely because of the relatively short half-life of the drug. Some of the metabolites of norethisterone however have long half-lives, up to 67 h in plasma. During long-term treatment with oral administration of norethisterone, there is a possibility that some of these metabolites may accumulate. Norethisterone is partly metabolized to ethinyloestradiol and although this is unlikely to cause interference in commercially available oestradiol assays, it may have been the cause of the suppressed gonadotrophin results observed in some of the patients. Concentrations of norethisterone may be higher in serum if other drugs such as oestrogens are co-administered to affect SHBG concentrations. It is interesting to note that there appears to be no correlation between measured testosterone concentration and the concentration of SHBG in serum collected from the patients described above. When norethisterone is prescribed as an oral contraceptive, it is administered at a lower dose, typically 0.5–1.5 mg per day. In addition to the administration of norethisterone as a parent compound, drugs such as lynestrenol are rapidly converted to norethisterone in vivo. 10
The potential for norethisterone interference in the testosterone assay was confirmed by staff in the technical services department at Roche who provided the following additional information, ‘norethisterone is structurally similar to the anabolic steroid nandrolone which is known to have significant cross-reactivity in the testosterone assay. A 5% cross reactivity rate is quoted with norethisterone up to concentrations of 10 µg/L. Serum concentrations of norethisterone up to 15 µg/L can occur after administration of 5 mg norethisterone which would be expected to produce a measured testosterone of approximately 2.6 nmol/L’. The higher concentrations of testosterone shown for the Roche method in Table 2 would be due to the endogenous testosterone present in the base serum pool.
Conclusion
Cross reactivity of norethisterone in some commercial immunoassays may produce falsely raised testosterone results for samples collected from patients taking norethisterone medication. For these patients, an elevated testosterone can be confirmed using a more specific assay such as liquid chromatography-tandem mass spectrometry.
Footnotes
Acknowledgements
The authors thank staff in the laboratories at the Royal Liverpool University Hospital and the Leeds SAS Steroid hormone centre in Leeds General Infirmary for LC-MS/MS testosterone results; Andy Brain in Newport and Hazel Borthwick in Durham for preliminary results on other immunoassay platforms; staff in Treliske Hospital for measurement of DHEAs; staff in St Bart’s Hospital and Glasgow Royal Infirmary for measurement of 17-hydroxyprogesterone, androstenedione and testosterone following ether extraction; and finally, Sara Oxford and Louise Simmons from Roche for their technical support and advice. Norethisterone was donated by Derriford Hospital Pharmacy Department.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not required.
Guarantor
JJ.
Contributorship
JJ and GB identified and investigated the index cases, FM and LP proposed the project, FM organized the distribution of samples through the external quality assessment scheme and all authors have contributed to the draft and final manuscript.
