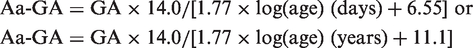Abstract
Background
We previously reported that glycated albumin (GA) levels increased in an age-dependent manner in infancy. In order to determine whether this phenomenon is true from infancy to adulthood, we investigated the GA levels in non-diabetic subjects of a wide range of age.
Methods
GA levels of 376 non-diabetic subjects [average age, 31.8 ± 23.8 years (4 days–78 years)] were determined. A relationship between GA and logarithmically transformed age [log(age)] was analysed.
Results
GA levels were significantly positively correlated with log(age) [R = 0.865, P < 0.0001, GA = 1.77 × log(day) + 6.55]. Based on a regression line, we established the formula for adjusting GA levels according to age.
Conclusion
We showed that GA increases with age from infancy to adulthood and that normal GA levels are demonstrated as a simple regression formula with log(age). This formula allowing us to use the adult reference range has the potential for treatment monitoring of diabetic patients regardless of age.
Introduction
Glycated albumin (GA) is one of the indexes of glycaemic control and reflects the status of glycaemic control changes for 2–4 weeks. 1 The principal advantage of GA against glycosylated haemoglobin (HbA1c), which is widely used as an indicator for glycaemic control, 2 is that GA is not affected by both haemoglobin metabolism and a variant haemoglobin. 1 On the other hand, GA has been reported to be inadequate to assess the glycaemic control of diabetic patients with albumin metabolism disorders. 1 For example, in nephrotic syndrome, hyperthyroidism and glucocorticoid therapy, which increases albumin metabolism, GA is apparently low. In contrast, GA is apparently high in liver cirrhosis and hypothyroidism, which decreases albumin metabolism. We previously reported that GA, but not HbA1c, reflects glycaemic control in patients with neonatal diabetes mellitus (NDM) whose disease onset is within six months of age, because in this period fetal haemoglobin is high and gradually decreases. 3 On the other hand, we have also found that GA in healthy infants within one year of age is low and correlated with logarithmically transformed age [log(age)]. 4 However, there is no study to investigate whether the phenomenon that GA is correlated with log(age) is true from infancy to adulthood so far. If there is a significant correlation between them, it is possible to calculate age-adjusted GA (Aa-GA), which enables us to use the adult reference value when assessing glycaemic control for patients at any age. Thus, we investigated the GA levels in non-diabetic subjects of a wide range of age.
Subjects and methods
Study subjects
A total of 376 non-diabetic subjects [236 males, 140 females; average age, 31.8 ± 23.8 years (4 days–78 years)] were selected. Of these 376 subjects, 78 were infants (58 cases were previously reported 4 ); 86, children and 212, adults. 5 For non-diabetic children, the subjects as follows were used: patients with growth hormone therapy, developmental disability or obesity, of which plasma glucose and HbA1c levels were within normal range. We excluded those participants with thyroid disease, liver disease, renal disease, anaemia or administration of glucocorticoid. The data of the infants and children were collected from October 2010 to November 2011 and from January 2012 to May 2012, respectively.
This study was approved by the Ethics Committee at Asahikawa Medical University. All subjects and/or subjects’ parents provided written informed consent.
Laboratory measurements
Serum GA was determined using Hitachi 7600 autoanalyzer (Hitachi Instruments Service Co., Tokyo, Japan) by enzymatic method using albumin-specific proteinase, ketoamine oxidase and albumin assay reagent (Lucica GA-L; Asahi Kasei Pharma Co., Tokyo, Japan). Inter-assay coefficient variations (CV) in the data collection for the infants were 4.55% and 5.56%, respectively, as determined in two representive samples. Inter-assay CV in the data collection for the children were 2.14% and 1.87%, respectively, as well. The adult reference value for GA is 14.0 ± 1.1% (range, 11.7–16.2%), where inter-assay CV were 1.38% and 1.32%, respectively as determined in two representive samples. 5
Statistical analysis
Simple regression analyses were employed to assess the association between continuous variables. On the basis of the regression lines of GA and log(age), Aa-GA was calculated: Aa-GA was derived from the equation of multiplying the adult mean value of GA (14.0% as described above) by that regression formula. All analyses were conducted using SPSS 20 at a significance level of P < 0.05.
Results
GA levels were significantly correlated with log(age) in 376 non-diabetic subjects (R = 0.865; P < 0.0001). Figure 1 shows the regression line and equation. By using this equation, we established the formula for Aa-GA by using the following equations.
Correlation between glycated albumin (GA) and logarithmically transformed age [log(age)] in 376 non-diabetic subjects (age range, 4 days–78 years) are depicted. The actual data were plotted on a lin-log graph (using a logarithmic scale on the x-axis). The grey column represents the reference range of GA for the adults (11.7–16.2%). The broken line represents the average GA value for the adults (14.0%).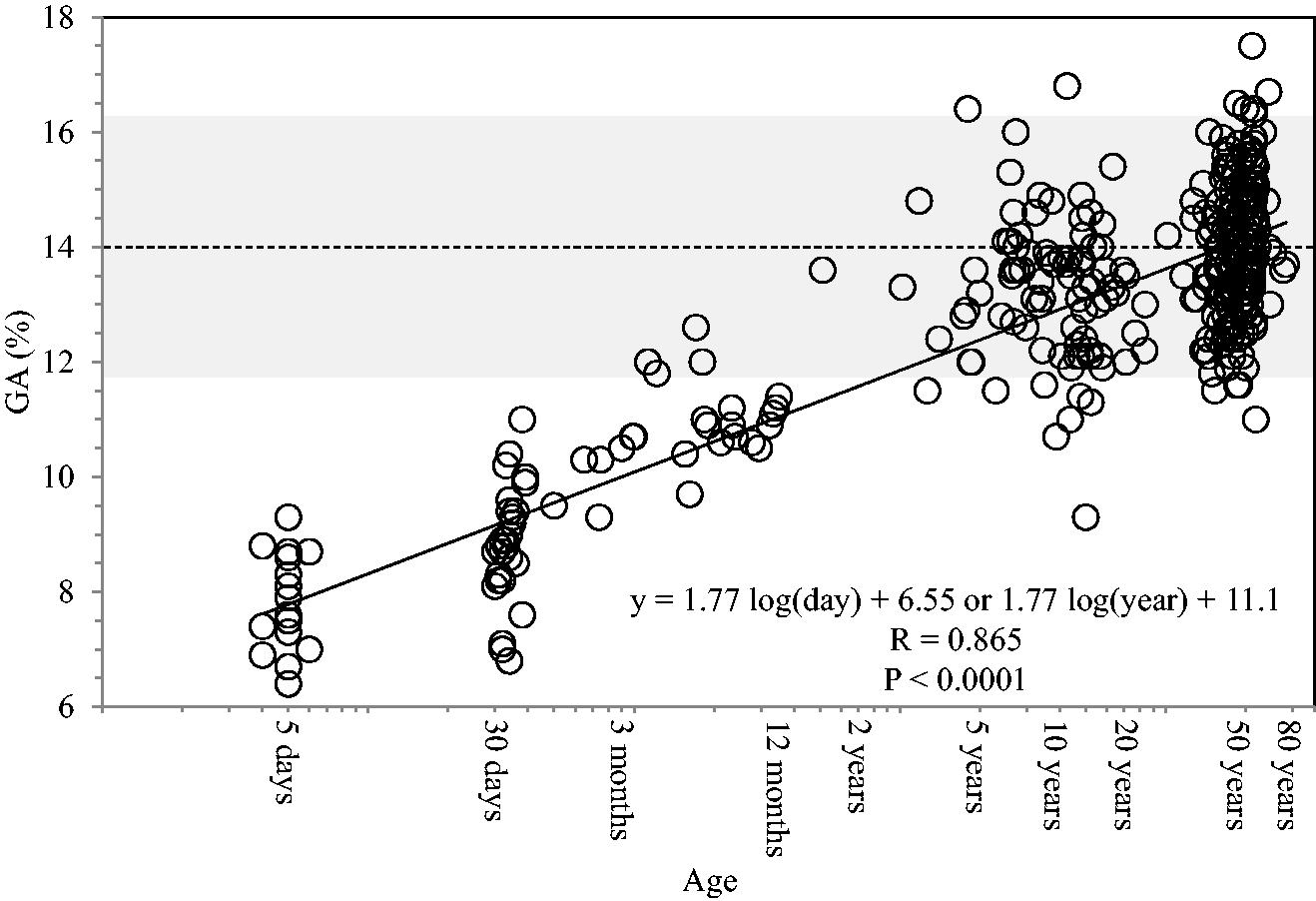
Discussion
We previously reported a significant positive correlation between GA and log(age) in healthy infants of up to one year of age. 4 Our current study broadened the analysis to include adults, and we found that a significant positive correlation exists between GA and log(age). It is well known that GA levels are influenced by albumin metabolism. 1 As GA is measured as a ratio of glycated to total albumin, it is not generally affected by the serum albumin levels. However, serum albumin was significantly positively correlated with GA or log(age) in healthy infants. 4 Both the low GA levels and positive correlation between serum albumin and GA in infants can be explained because of probably accelerated albumin metabolism. Increased albumin biosynthesis has been shown in a dynamic study using an isotope. 6 Protein synthesis rate was also reported to be more than twice as fast as that in adults. 7 During fetal and neonatal development, weight increases rapidly in parallel with protein gain, which results from a difference between protein synthesis and breakdown (i.e. protein turnover). 7 More rapid growth and protein gain were repoted to be associated with higher rates of protein turnover.7,8 Accordingly, albumin metabolism is speculated to have a negative correlation with log(age).
Many measured parameters show various patterns of gradual increase or decrease concurrent with growth from childhood. 9 Nevertheless, most studies for age-related change are limited in children. It might be interesting to determine whether changes similar to that of GA can be detected for some parameters from infancy to the aged.
There is a danger of assessing NDM patients with poor glycaemic control state as good if the adult reference range of GA is used because GA levels in infants are lower than in adults. Therefore, we have previously proposed that GA levels should be compared with age-dependent reference range in order to make accurate assessments regarding glycaemic control. 4 However, one problem with this method was that an age-dependent reference range is always necessary for judgment of the GA levels in NDM patients.
Because GA showed strong positive correlation with log(age), Aa-GA is well adapted for comparison with the adult reference range and can be a useful indicator for glycaemic control in NDM patients. One advantage of using Aa-GA levels is that it can be easily calculated from measured GA levels and age without using age-dependent reference range. Another advantage of using Aa-GA levels is that long-term trends of glycaemic control can be compared.
In conclusion, GA in non-diabetic subjects is strongly positively correlated with log(age). Consequently, Aa-GA allowing us to use the same adult reference range has the potential for treatment monitoring of diabetic patients, especially NDM patients, regardless of age.
Footnotes
Acknowledgements
We would like to thank the staff of the Department of Pediatrics who helped collect samples. This paper is dedicated to our dear mentor Professor Kenji Fujieda, who passed away on 19 March 2010.
Declarations of conflicting interests
None declared.
Funding
This study was supported in part by a Grant-in-Aid for Young Scientists (B) (24791036) provided by the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan.
Ethical approval
This study was approved by the Ethics Committee at Asahikawa Medical University (reference number, 790 and 1210).
Guarantor
SS.
Contributorship
SS and MK researched data, wrote manuscript, contributed to the discussion and reviewed/edited manuscript. NN and AF researched data, contributed to the discussion and reviewed/edited manuscript. KM, YT and HA contributed to the discussion and reviewed/edited manuscript.