Abstract
Background
Glycated albumin is a useful glycaemic control indicator for neonatal diabetes mellitus. However, glycated albumin concentrations in infants are lower than those in adults and increase in an age-dependent manner. Based on our investigation of non-diabetic subjects, we proposed the possibility that the reference range for adults may be used regardless of age, provided that age-adjusted glycated albumin is employed. In the present study, we evaluate the usefulness of age-adjusted glycated albumin in neonatal diabetes mellitus patients.
Methods
Six neonatal diabetes mellitus patients (four patients with permanent neonatal diabetes mellitus and two patients with transient neonatal diabetes mellitus) were included. Measured glycated albumin or age-adjusted glycated albumin was compared to calculated glycated albumin, which was determined using calculation formulae we had reported based on past blood glucose over the 50 days before measurement of glycated albumin.
Results
Measured glycated albumin was significantly lower than calculated glycated albumin (20.5 ± 4.9% versus 28.2 ± 6.1%; p < 0.0001), whereas age-adjusted glycated albumin was equivalent to calculated glycated albumin, showing no significant difference (27.5 ± 6.8% versus 28.2 ± 6.1%). Measured glycated albumin concentrations in patients with transient neonatal diabetes mellitus in remission were lower than the reference range for adults, whereas age-adjusted glycated albumin concentrations were within the reference range for adults.
Conclusion
We demonstrated that age-adjusted glycated albumin concentrations were consistent with calculated glycated albumin. Age-adjusted glycated albumin is therefore a useful glycaemic control indicator for neonatal diabetes mellitus patients.
Introduction
Glycaemic control indicators, whose values reflect middle- to long-term glycaemia but are not generally affected by every meal and short range exercise, are monitored for diabetic patients in clinical practice. Glycation of various proteins is increased in diabetic patients, and it has been suggested that part of this phenomenon is involved in the development or progression of chronic diabetic complications. 1 HbA1c is one of these glycated protein and widely used as an indicator of glycaemic control status. 2 However, the presence of diseases in which lifespan of erythrocytes is shortened such as hemolytic anaemia and variant haemoglobin affect HbA1c measurement. 3 A glycaemic control indicator other than HbA1c is therefore required for these conditions.
Neonatal diabetes mellitus (NDM) is a type of diabetes mellitus that is caused by a single-gene abnormality, in which an insulin-dependent state develops within six months after birth. 4 Depending on its clinical course, NDM is classified into two major categories: transient NDM (TNDM), in which treatment will no longer be necessary within several months after onset and permanent NDM (PNDM) in which lifelong treatment is necessary. Therefore, appropriate glycaemic control is necessary for determining the acceptability of continued treatment and for lifelong monitoring of the treatment course. However, during this period, fetal haemoglobin (HbF) is replaced by adult haemoglobin (HbA). Patients with a high concentration of HbF tend to show a low concentration of HbA1c. 5 Therefore, in NDM patients who show a low concentration of HbA1c because of the influence of HbF, HbA1c cannot be used as a glycaemic control indicator. 6
Similar to HbA1c, glycated albumin (GA) is used as a glycaemic control indicator. 3 We have reported that GA is useful for monitoring glycaemic control in NDM patients because GA is not affected by high concentrations of HbF. 6 However, we found that GA is lower relative to blood glucose in NDM patients when compared with patients with child-onset type 1 diabetes mellitus (T1DM). 7 We also provided evidence that GA concentrations in healthy infants are lower than the reference range for adults and are positively correlated with logarithmically transformed age. 8 Therefore, for more accurate evaluation of glycaemic control, it is necessary to use an age-dependent reference range. 8 On the other hand, from the investigation of non-diabetic subjects from neonates to adults, GA showed a strong positive correlation with logarithmically transformed age. 9 Therefore, we proposed a possibility that the reference range for adults can be used regardless of age if age-adjusted GA (Aa-GA) is used. 9 However, it has not been investigated to date whether Aa-GA accurately reflects blood glucose in diabetic patients.
We previously created calculation formulae to estimate HbA1c and GA from past blood glucose concentrations based on the fact that HbA1c and GA are affected by the weighted mean of past blood glucose concentrations in diabetic patients and reported that calculated values in patients with child-onset T1DM were similar to actually measured values. 10 In the present study, we compared measured GA and Aa-GA with calculated GA (cGA) to investigate whether Aa-GA accurately reflects blood glucose in NDM patients.
Patients and methods
Patients
Clinical characteristics of study patients.
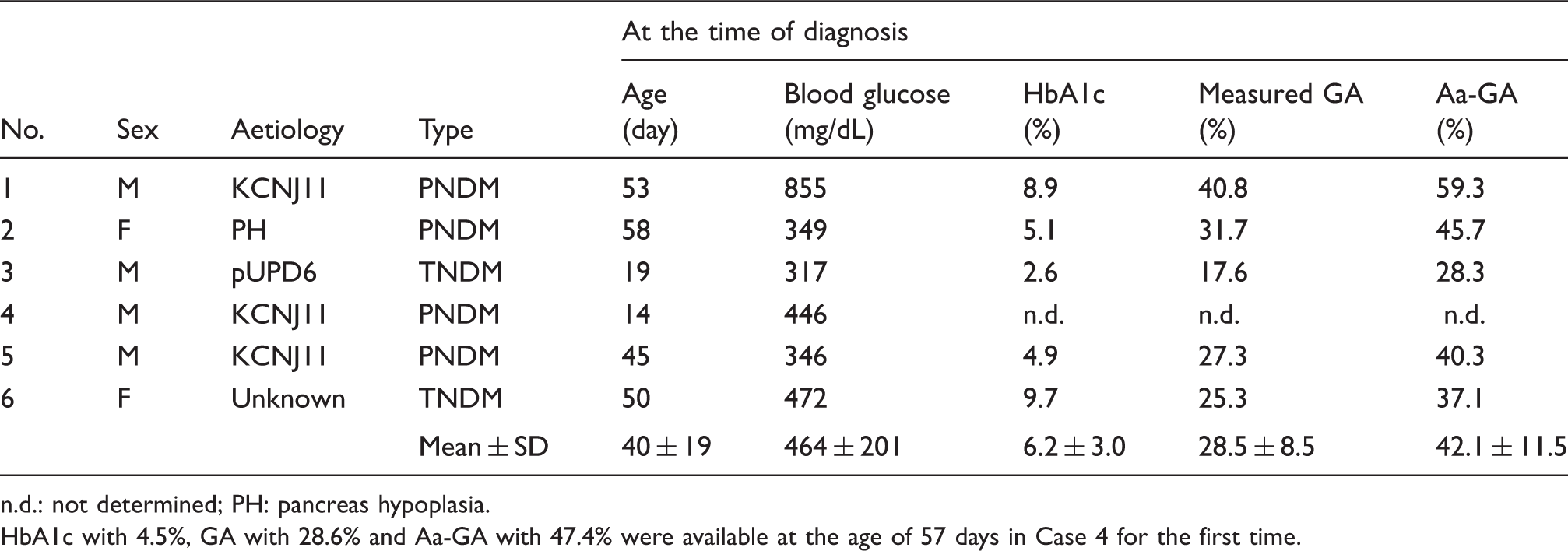
n.d.: not determined; PH: pancreas hypoplasia.
HbA1c with 4.5%, GA with 28.6% and Aa-GA with 47.4% were available at the age of 57 days in Case 4 for the first time.
Laboratory methods
HbA1c was measured by cation-exchange high performance liquid chromatography with an HLC-723G8 (Tosoh Co., Tokyo, Japan). HbA1c were converted to National Glycohemoglobin Standardization Program equivalent values in accordance with the official equation.
11
GA was determined using a Hitachi 7600 auto analyser (Hitachi Instruments Service Co., Tokyo, Japan), employing an enzymatic method using albumin-specific proteinase, ketoamine oxidase and albumin assay reagent (Lucica GA-L; Asahi Kasei Pharma Co., Tokyo, Japan).
12
The reference range was 4.6–6.2% for HbA1c
11
and 11.7–16.0% for GA.
13
The data from SMBG were obtained from the blood glucose meter data. Aa-GA was calculated using formula (1), according to a previous report
9
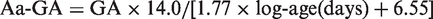
Estimated GA concentrations at t day (eGAt), which is an estimated GA concentration that supposing the MBG was not change for 50 days, were calculated from MBG at t day (MBGt) using formula (2).10,14 t is the number of days before measurement of GA; data during the period from 1 to 50 days before measurement of GA were used
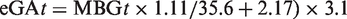
cGA was calculated using formula (3), which is the modified formula according to a previous report.
10
In the previous report, MBG during the past 20 weeks was used to calculate HbA1c with a long half-life; in the present study, MBG during the past 50 days was used because it was only necessary to calculate GA with a short half-life
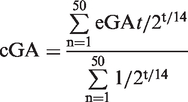
Statistical analysis
Results are expressed as mean ± SD. For statistical analyses, the paired t test was used to compare the various GAs. Simple regression analyses were employed to assess the association between continuous variables. Tests of comparison of correlations were performed using analysis of covariance. All analyses were performed using SPSS 20, with p < 0.05 regarded as significant.
Results
Of the six NDM patients included in the present study, four patients had PNDM, all of whom received insulin therapy. The remaining two patients had TNDM in which insulin treatment is no longer required at five months old. At the time of diagnosis, age was 40 ± 19 days, blood glucose was 464 ± 201 mg/dL, HbA1c was 6.2 ± 3.0%, measured GA was 28.5 ± 8.5%, Aa-GA was 42.1 ± 11.5% (Table 1).
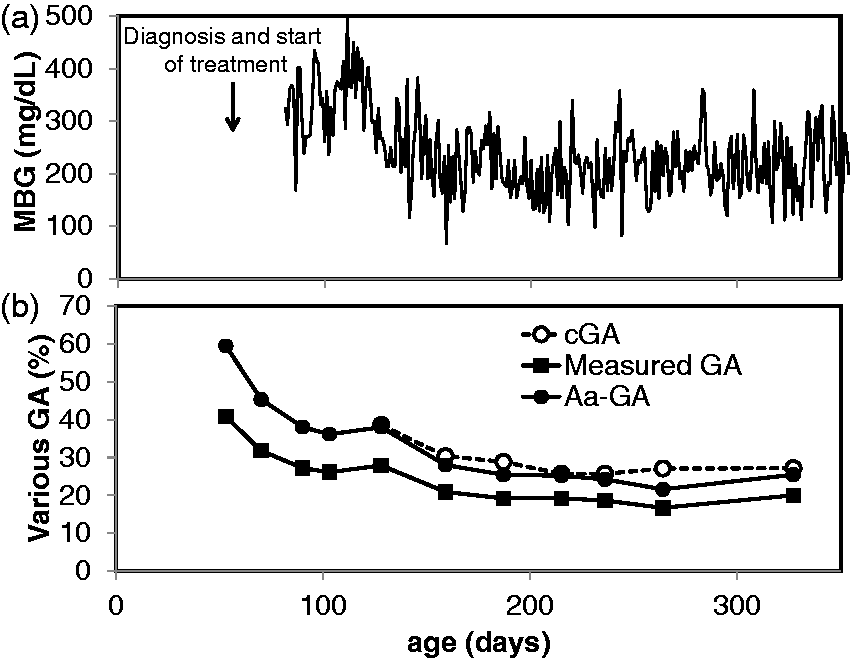
The time course of glycaemic control marker of Case 1, in whom long-term follow-up was feasible, is shown in Figure 1. In Case 1, PNDM developed with diabetic ketoacidosis; insulin therapy was started immediately after diagnosis.
7
In the beginning of initiating therapy, hyperglycaemia persisted, with MBG ranging from 300 to 400 mg/dL. Subsequently, MBG decreased to around 200 mg/dL with marked day-to-day variation. Although measured GA decreased gradually from the value at the time of diagnosis (40.8%), it remained around 20% after 150 days old. On the other hand, cGA, which could be calculated after 130 days old, was higher than measured GA and its concentrations were similar to those of Aa-GA. Aa-GA at the time of diagnosis was markedly high at 59.3%, suggesting measured GA concentrations underestimate the extent of hyperglycaemia.
Time courses of MBG (a) and various GA concentrations (b) in Patient 1. In Case 1, PNDM developed at 53 days old with diabetic ketoacidosis; insulin therapy was begun immediately.
7
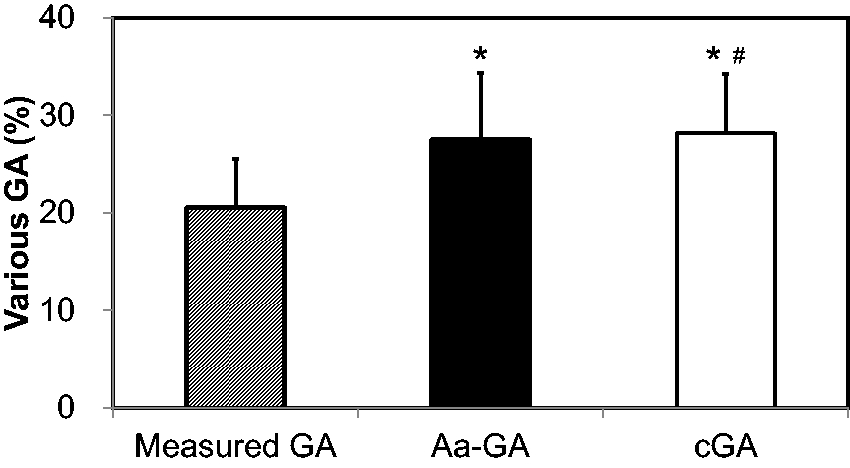
cGA was obtained from the six NDM patients at a total of 19 time points, and then GA or Aa-GA was compared with cGA. Measured GA showed a significant correlation with cGA (R = 0.899, p < 0.0001), but the regression line (y = 0.73x–0.13) was shifted downward relative to the straight line of y = x (Figure 2(a)). Aa-GA showed a significant correlation with cGA (R = 0.914, p < 0.0001), and the regression line (y = 1.03x – 1.6) was equivalent to y = x (Figure 2(b)). Measured GA was significantly lower than cGA (20.5 ± 4.9% versus 28.2 ± 6.1%; p < 0.0001), whereas Aa-GA was equivalent to cGA, showing no significant difference (27.5 ± 6.8% versus 28.2 ± 6.1%; p = 0.3268, Figure 3).
Correlation between cGA and measured GA (a) and correlation between cGA and Aa-GA (b) in the six NDM patients. The regression line between cGA and measured GA and the regression line between cGA and Aa-GA are shown as solid line, and the formula of y = x is shown as a dotted line, respectively. Comparisons among various GA concentrations. This figure shows comparisons among measured GA (hatched column), Aa-GA (closed column) and cGA (open column) of the six NDM patients.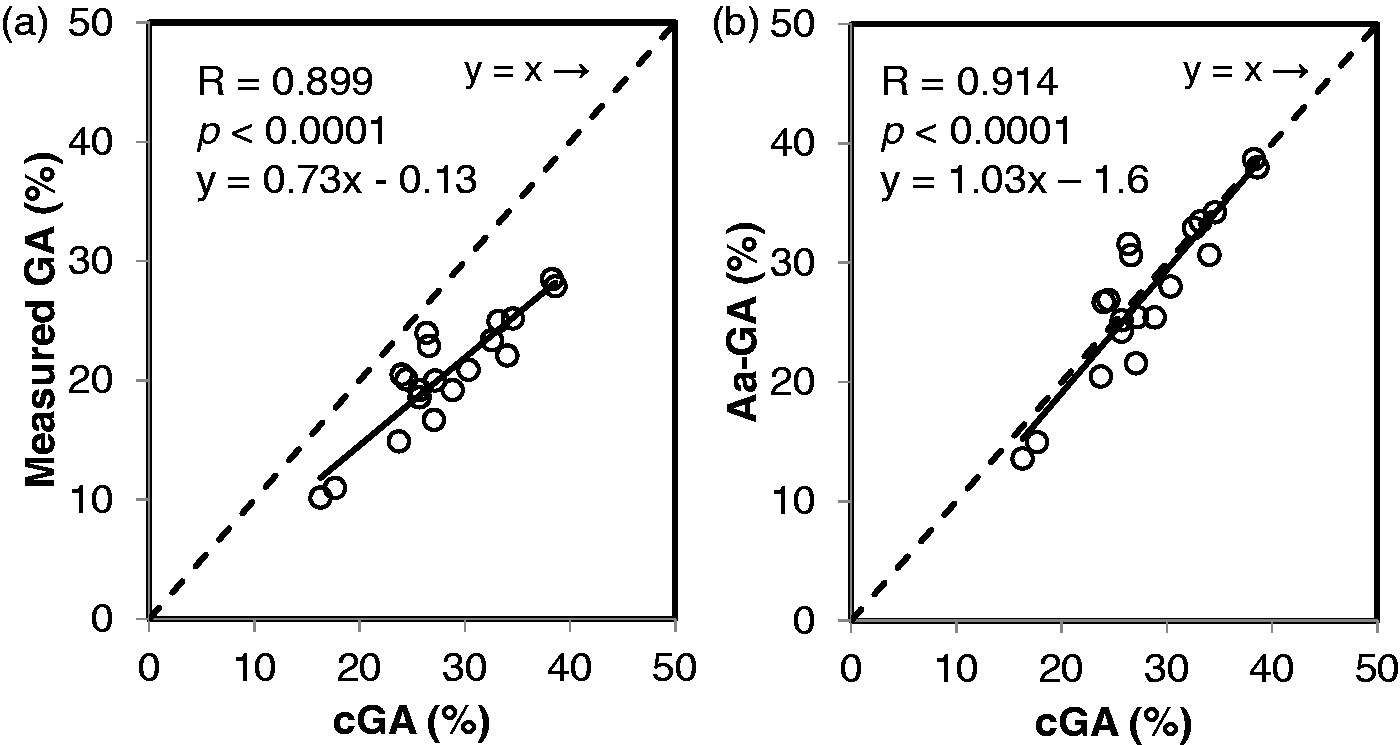
The time courses of measured GA and Aa-GA concentrations in the six NDM patients are shown in Figure 4. High measured GA observed in the NDM patients at the time of diagnosis decreased in response to treatment for diabetes mellitus in all the patients (Figure 4(a)). Throughout the treatment course from the time of onset, Aa-GA remained at a higher concentration relative to measured GA (Figure 4(b)). Patient 3 and Patient 6 had TNDM, in which insulin treatment is no longer required at five months old. In both patients, during and after the remission period, measured GA concentrations in these patients were lower or low normal relative to the reference range of GA for adults, whereas their Aa-GA concentrations remained within the reference range of GA for adults.
Time courses of measured GA and Aa-GA concentrations in the six NDM patients. Patients with TNDM and patients with PNDM are represented by closed symbols and open symbols, respectively. The reference range of measured GA for adults is shown in half-tone dot meshing.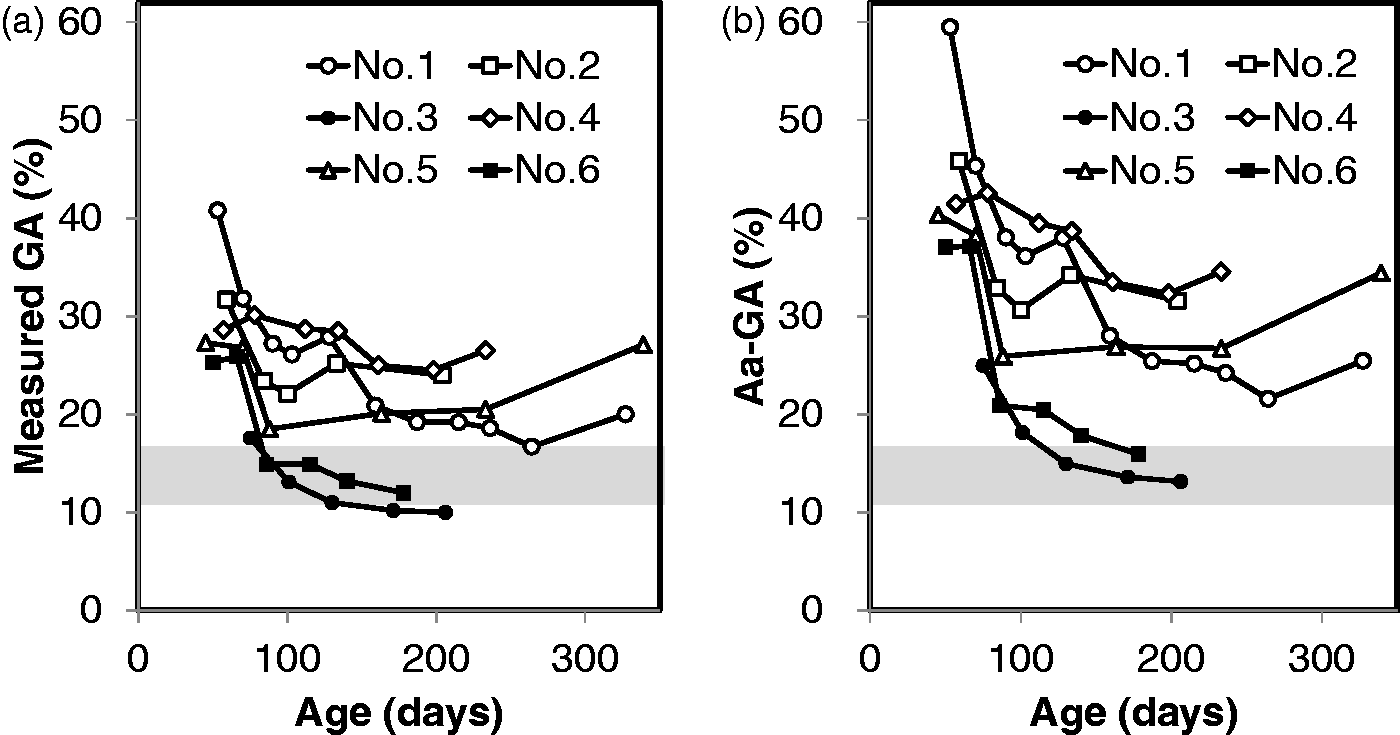
Discussion
Abnormal GA concentrations are observed in conditions associated with abnormal albumin metabolism, such as thyroid disorders, nephrotic syndrome, liver cirrhosis and during treatment with glucocorticoids. 3 It was found that because albumin metabolism is increased in neonates, neonates show an apparently low concentration of GA.7,8 Therefore, for accurate evaluation of glycaemic control, it was necessary to use an age-dependent reference range. 8 On the other hand, we demonstrated that GA shows a positive correlation with logarithmically transformed age from the neonatal period to adulthood. We therefore proposed that the reference range for adults is applicable to all ages if Aa-GA is used. 9 To confirm this for NDM patients in this study, we evaluated Aa-GA by comparing Aa-GA with cGA, which was a calculated value based on the fact that past blood glucose concentrations affect HbA1c and GA as a weighting function. 10 We already reported that these calculated values show an excellent correlation with actually measured values in patients with T1DM. 10
As a result, measured GA showed a significant correlation with cGA. However, measured GA was significantly lower relative to cGA, which was exemplified by an apparently consistent low concentration of measured GA in Patient 1, reflecting abnormal albumin metabolism during the neonatal period (Figure 1). Even when the six NDM patients were evaluated, measured GA was significantly lower relative to cGA. On the other hand, Aa-GA was significantly correlated with cGA and was equivalent to cGA, showing no significant difference (Figure 2), demonstrating that Aa-GA is an accurate glycaemic control indicator in NDM patients. The benefit of usage of Aa-GA instead of measured GA is that we do not need age-specific reference ranges for evaluating its value. Aa-GA enables us to evaluate blood glucose control status among different age groups. In addition, Aa-GA enables us to understand serial changes of diabetic control in each patient intuitively and easily because we can use the same reference values (adult reference range).
The calculation formula for Aa-GA shows that measured GA will tend to be lower by 15% for children at three years of age and by 10% for children at seven years of age than that of adults. When measured GA is used as a glycaemic control indicator for paediatric patients with T1DM, glycaemic control may be slightly underestimated. Therefore, glycaemic control can be evaluated more accurately in paediatric patients as well by using Aa-GA rather than measured GA.
We reported that HbA1c cannot be used during the neonatal period but that it can be used as a glycaemic control indicator at and after five months old. 15 However, because GA has various advantages over HbA1c as described below, we believe it desirable to keep using Aa-GA as a glycaemic control indicator even after NDM patients have grown into childhood: (i) NDM patients can be followed up using the same glycaemic control indicator (Aa-GA) from the time of onset. (ii) GA would be a better glycaemic control marker especially for T1DM due to reflecting the postprandial hyperglycaemia. 16 GA reflects not only MBG but also postprandial blood glucose whereas HbA1c mainly reflect MBG;17,18 therefore, we believe that measurement of GA is also desirable from the viewpoint of prevention of diabetic complications. (iii) Iron deficiency anaemia, which results in an apparently high concentration of HbA1c,19,20 can develop during the growth period. Therefore, HbA1c may thus not accurately reflect blood glucose in such patients.
Importantly, to establish the usefulness of Aa-GA in NDM patients, this study needs longitudinal follow-up of these patients to document the validity of these formulas.
There are several limitations to the present study. First, the data were obtained from only a small number of patients. Although NDM is a rare disease, it will be necessary to confirm the findings obtained in the present study, using a large number of NDM patients. Second, in most patients, SMBG was performed mainly before meals and before bedtime, with the result that the number of SMBG measurements per day was limited to about 3–4. Ideally, blood glucose should be measured seven times per day, including before and after meals. On the other hand, in the previous investigation of patients with T1DM, we found that calculated HbA1c and cGA from blood glucose measured only four times per day (before meals and before bedtime) in most patients were strongly correlated with the actually measured values. If long-term continuous glucose monitoring (CGM) is used for assessing the relationship between Aa-GA and MBG in NDM patients, we may solidify the findings in the present study because CGM can be measured at high precision.
In conclusion, we showed that Aa-GA is a useful glycaemic control indicator in NDM patients. By means of Aa-GA, we should investigate whether maintaining strict glycaemic control prevents the onset and progression of diabetic complications in NDM patients.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
None.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
This study was approved by the Ethics Committee at Asahikawa Medical University (reference number 266).
Guarantor
SS.
Contributorship
SS and AF contributed equally to this work. SS: performed genetic analysis, data analysis and prepared manuscript. AF: performed genetic analysis and data analysis. MK: performed data analysis and prepared manuscript. MO, SA, AN, KW, KO, SH and AI: performed practical aspects. All authors contributed to the study conception and edited and approved the manuscript.
