Abstract
Diagnosis and management of pancreatic cyst lesions is challenging as there is currently no investigation that offers both high diagnostic sensitivity and high specificity for the identification of potentially malignant lesions. Accurate classification of these lesions is vital in order to avoid unnecessary treatment of benign lesions and missed opportunities for early treatment of lesions that are malignant/pre-malignant. Pancreatic cyst fluid analysis has an important role in diagnosis, although all currently available investigations based on fluid analysis have significant limitations. Cytological analysis can reveal features that are specific for a certain class of cyst, but offers limited sensitivity in detecting malignant/pre-malignant cysts. Measurement of tumour markers, particularly carcinoembryonic antigen can also be informative. Concentrations of cyst fluid carcinoembryonic antigen tend to be higher in malignant/pre-malignant cysts, although there is a wide overlap between the various classes of cyst. A number of studies have suggested that diagnostic carcinoembryonic antigen cut-offs can be chosen that provide a high degree of specificity but limited sensitivity. Studies of the analytical validity of tumour marker assays in pancreatic cyst fluid analysis have highlighted discrepancies in some fluid specimens, which require further investigation. DNA analysis also has a role. In particular, K-Ras-2 mutational analysis appears to provide high specificity for detection of malignant/pre-malignant lesions. A number of diagnostic algorithms have been published, integrating use of available investigations in order to achieve the optimum discrimination of benign and potentially malignant cysts. Research into new biochemical markers and optimal use of available pancreatic cyst fluid analyses is ongoing.
Introduction
Pancreatic cyst lesions are common in the general population and in hospitalized patients. The reported prevalence varies between studies 1 but the likely incidence of asymptomatic pancreatic cysts in the general population is between 1% and 3%. 2 Many of these cysts are asymptomatic and are discovered incidentally when the patient is being investigated for an unrelated problem. 3 Awareness of these lesions has increased in recent years, in part due to widespread use of high resolution abdominal imaging. Diagnosis and management of pancreatic cysts can be challenging although the understanding of the natural history and optimal management of the various types of cyst is improving. The classification of pancreatic cysts is complex but the most important differentiation is between non-neoplastic and neoplastic cysts. Benign cysts that are asymptomatic may be managed conservatively, while cysts with malignant potential will generally be managed surgically if possible.1,4 Accurate classification is therefore required if malignant/pre-malignant cysts are to be identified and treated appropriately and also if patients with benign cysts are to avoid unnecessary exposure to the risks of surgery.
Analysis of pancreatic cyst fluid has become an important part of the investigation and classification of pancreatic cysts. Analyses include cytological, biochemical and DNA analysis. This article will summarize the background and clinical significance of various pancreatic cysts, review the investigation of these cysts, focusing particularly on analysis of pancreatic cyst fluid and describe how the various investigations may be brought together in unifying diagnostic and management pathways. Finally, some of the analytical issues that laboratories face when analysing pancreatic cyst fluid samples will be discussed.
Classification of pancreatic cysts
Typical characteristics of pancreatic cystic lesions.
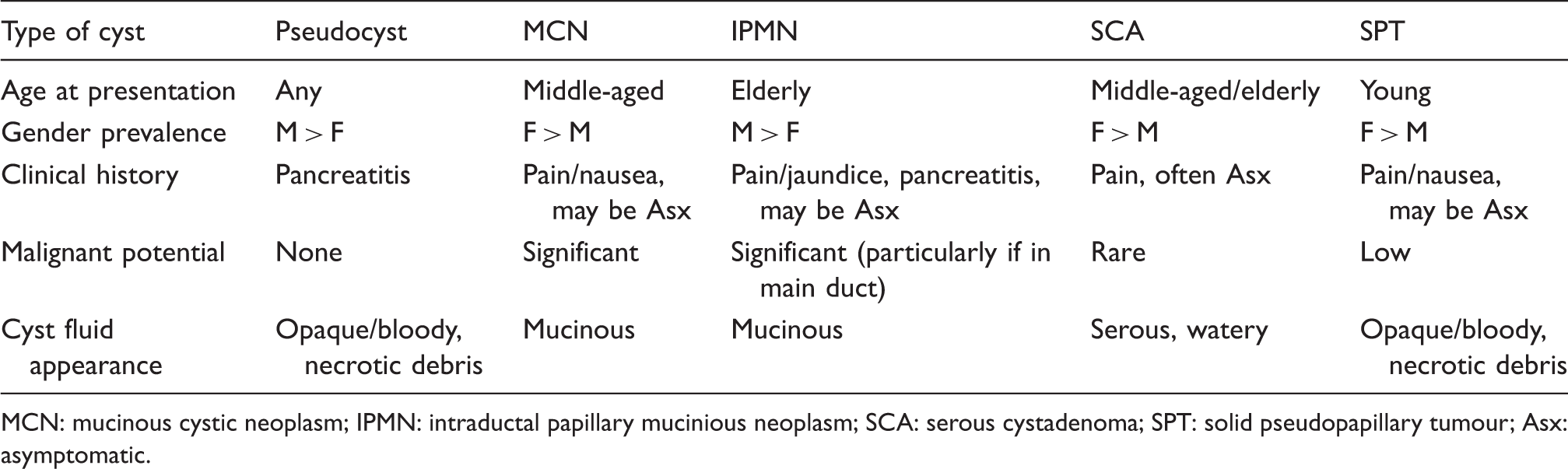
MCN: mucinous cystic neoplasm; IPMN: intraductal papillary mucinious neoplasm; SCA: serous cystadenoma; SPT: solid pseudopapillary tumour; Asx: asymptomatic.
Pancreatic pseudocyst
Pseudocysts are one of the more common types of pancreatic cyst. They are inflammatory in nature, being collections originating from around the pancreas and have no malignant potential. Pseudocysts may be differentiated morphologically from cystic tumours in that they do not have an epithelial lining.3,5 They arise following acute/chronic pancreatitis or pancreatic trauma. Pseudocysts typically contain necrotic tissue including necrotic fat, neutrophils and are surrounded by granulation tissue forming a fibrotic wall. 6 They may be located anywhere in the pancreas and may have communication with pancreatic ducts. Radiology will typically reveal a single, unilocular cystic lesion. Aspirated cyst fluid is dark in colour due to the presence of pancreatic juice and blood. The fluid often contains visible necrotic debris. 1
Pseudocysts can present with abdominal pain, obstructive jaundice or sepsis secondary to infection. 7 Haemorrhage complicates only a minority of pseudocysts (approximately 5%) but carries a high risk of mortality (40%). 8 Diagnosis of pancreatic pseudocysts is often straightforward as there is usually a history of acute or chronic pancreatitis or pancreatic trauma. However, certain neoplastic pancreatic lesions may mimic pseudocysts in terms of presentation and appearance9–13 so detailed investigation is required to minimize the potential for misdiagnosis. Pseudocysts are often managed conservatively. Surgical drainage may be performed in the case of symptomatic lesions or if there is increased risk of complications such as infection or haemorrhage 14 (e.g. where cysts are large or persistent).
Serous cystadenoma
SCAs are considered to be benign lesions, which may occur anywhere in the pancreas. A small number of cases of malignant SCA have been described, but the risk of malignancy is considered to be less than 3%. 15 They are multilocular, often appearing as a number of small cysts packed together and are usually less than 5 cm in diameter. 3 Fluid aspirated from SCAs is usually non-viscous, colourless and free of mucin. SCAs are frequently asymptomatic and so are often discovered incidentally. Where symptoms do occur they usually consist of abdominal pain or discomfort. 16 Particularly large SCAs may cause compression of the bile duct and obstructive jaundice. Small, asymptomatic SCAs can generally be managed conservatively with long-term surveillance, as long as a mucinous cystic tumour has been excluded through appropriate investigation.1,16 Symptomatic patients with larger SCAs would usually be considered for surgical resection.
Mucinous cystic neoplasm
MCNs are usually solitary lesions and can be found in the head, body or the tail of the pancreas. 17 The cysts are typically septated with thick walls and septae. MCNs may also harbour solid areas containing invasive carcinoma.18,19 A thickened wall, calcification and vascular involvement are suggestive of malignant changes. Aspirated fluid is viscous and typically clear, but may be blood-stained or contain necrotic material.
The majority of MCNs occur in females, presenting with abdominal discomfort or pain.17,19–21 Weight loss and anorexia may also feature. MCNs are considered to be pre-malignant with a recent series showing 11% to be invasive at the time of diagnosis. 22 Excision of the lesion is therefore usually the treatment of choice if the patient is fit enough to withstand pancreatic surgery. Surgical resection is usually curative unless invasive carcinoma is present. Invasive carcinoma from MCNs typically follows a less aggressive course than classical ductal pancreatic adenocarcinoma but is still associated with a poor prognosis and low five-year survival rates.19,21
Intraductal papillary mucinous neoplasm
Intraductal papillary mucinous neoplasms (IPMNs) are mucinous cystic tumours which are considered pre-malignant and may be frankly malignant.23–25 IPMNs are typically associated with the main pancreatic duct but may also affect the side branches. IPMNs affecting the main duct and side branches may be considered separate subtypes that follow different clinical courses. ‘Branch-type’ IPMNs (those affecting the side branches rather than the main pancreatic duct) typically occur in younger patients and are less likely to become malignant.24–26 IPMNs are histologically heterogeneous with the various histological subtypes having different malignant potential and prognosis. 27 Fluid aspirated from IPMNs is typically viscous, mucinous and colourless.
IPMNs are more likely than most neoplastic pancreatic cysts to be symptomatic at the time of discovery. Patients typically suffer from epigastric pain which may be exacerbated by eating. Excess mucin produced by these cystic lesions may cause blockage of the pancreatic duct. Weight loss, fever and jaundice may also feature. 24 Risk factors for malignancy include size, association with the main duct and jaundice. 28 The lower risk of malignancy in small (<3 cm) side-branch IPMNs has led to more conservative management of these cysts with observational follow-up becoming routine for asymptomatic patients. 29 Due to the higher risk of malignancy in main-duct IPMNs surgery is usually recommended if the patient is fit enough to be considered a candidate for pancreatic resection.30,31
Solid pseudopapillary tumour
SPTs are rarer than the lesions discussed above, and occur almost exclusively in young females. 32 They are solid tumours which can undergo a degenerative process giving rise to a cystic appearance (imaging often reveals alternating areas of solid and cystic appearance). The cystic areas contain blood and necrotic material. Patients typically present with abdominal pain that may be associated with weight loss and anorexia. SPTs are usually considered relatively indolent tumours, although local and distant metastasis can occur.32,33 Resection is usually considered due to the risk of metastatic disease.
Other cystic lesions
Other, rarer pancreatic cystic lesions include acinar cell carcinoma, lymphoepithelial cysts and cystic endocrine pancreatic carcinomas. Ductal adenocarcinomas can, rarely, undergo cystic degeneration.34,35
Investigation of pancreatic cysts
The aim of the investigation of pancreatic cysts is to identify the type of lesion present so that an appropriate management plan can be formulated. Depending on the potential for malignancy and the presence of symptoms, optimal management may include resection (or cyst ablation if the patient is not fit for surgery), cyst drainage or simple surveillance.3,4,36–40 It is important that cystic lesions with malignant potential are not misdiagnosed as benign when there may be an opportunity for curative surgery. It is equally important to correctly identify benign cysts so that asymptomatic patients at low risk of long-term complications are not exposed unnecessarily to the risks of pancreatic surgery. Differentiating pancreatic cysts can be difficult and a definitive pre-surgical diagnosis may not always be reached. Clinical history and presentation can be important indicators (e.g. a clear history of pancreatitis would lead to strong suspicion of a pseudocyst). A range of investigations including imaging, cytology and biochemical cyst fluid analysis are also important. The optimal investigation and management of pancreatic cysts remains an area of much active research.
Imaging of pancreatic cysts
The purpose of this article is to focus on the role of pancreatic cyst fluid analysis, so the use of imaging is only dealt with briefly here (a number of more comprehensive reviews have been published).41–44 The non-invasive imaging modalities of computed tomography (CT) and magnetic resonance imaging (MRI) are widely used to evaluate pancreatic cysts.4,45 CT scan (with IV contrast) is frequently used to evaluate the pancreas as it is relatively straight-forward and inexpensive, as is MRI with magnetic resonance cholangiopancreatography (MRCP) which may better identify communication of cysts with the pancreatic ducts. 46 The ability of these imaging techniques to differentiate benign and neoplastic cysts however, is limited. The reported diagnostic accuracy varies widely between 20% and 80%.47,48 It has also been reported that using CT alone provides limited assistance in the differentiation between serous and mucinous neoplasms. 49
The invasive technique of endoscopic retrograde cholangiopancreatography (ERCP) has been used in the evaluation of IPMNs as it is useful in defining the communication of the lesion with the pancreatic ductal system50 as well as providing an opportunity for tissue sampling. As ERCP is associated with a risk of pancreatitis, endoscopic ultrasound (EUS) is increasingly being used as the invasive imaging technique of choice. Since the pancreas lies directly adjacent to the stomach, an EUS transducer can be placed in close proximity to the pancreas, and the entire gland can be readily imaged. EUS allows detailed evaluation of the wall and contents of pancreatic cysts and can distinguish internal septae and solid areas within the cysts. Certain features identified by EUS may provide a high degree of diagnostic specificity, for example microcystic appearance in SCA 51 or a cyst without septations or solid areas in a pancreas with parenchymal abnormalities in pseudocyst. One study of 34 patients with a surgical diagnosis found that the presence of any one of four features had a sensitivity of 91% and specificity of 60% for detecting a lesion with malignant potential. These features were: cyst-wall thickness greater than 3 mm, intracystic compartments larger than 10 mm, intramural masses or cystic dilatation of the main pancreatic duct. 52 However, a large multicentre study found that the accuracy of the diagnosis of mucinous versus non-mucinous cysts using EUS was only 51%. 53 One of the limitations of EUS may be variations in interpretation between endosonographers. A study found the accuracy for detecting lesions with malignant potential varied from 40% to 93% between eight different endosonographers interpreting the same EUS procedures. 54
Another important aspect of EUS is that it allows fine-needle aspiration (FNA) of cysts and recovery of pancreatic cyst fluid for further investigation. Analysis (cytological, biochemical and molecular) of the pancreatic cyst fluid sampled via FNA during EUS can significantly improve diagnostic accuracy.4,14,53 The improved diagnostic performance of EUS-FNA must be balanced against the increased risks compared with non-invasive imaging. There is an increased risk of infection, haemorrhage and pancreatitis with EUS-FNA, although most studies indicate that the benefits outweigh the risks.55,56
Utility of pancreatic cyst fluid analysis in differentiation of cysts
Analysis of pancreatic cyst fluid obtained via EUS-FNA can greatly improve the diagnostic accuracy of the investigation of pancreatic cysts and can be particularly useful in helping to determine malignant potential. Analysis of pancreatic cyst fluid is multidisciplinary with cytological, biochemical and genetic analyses playing a role. Fluid samples obtained from pancreatic cysts can vary greatly in volume, viscosity, colour and cellular content. The nature of the cyst fluid obtained may limit the analysis that can be performed (e.g. due to small volumes, high viscosity or lack of cellular material). Estimates of the diagnostic accuracy of the various investigations that can be performed on cyst fluid vary widely, but it is clear that fluid analysis significantly improves the accuracy of the differentiation of pancreatic cysts. The majority of studies determine diagnostic performance in terms of differentiation between mucinous cysts (MCN, IPMN, frankly malignant cysts) and non-mucinous (SCA, pseudocysts, etc.). Determining malignant potential is usually the key consideration when deciding upon optimal treatment. Limitations of the published studies include small population sizes in some cases and a bias towards larger cysts, as smaller cysts may not provide sufficient fluid for analysis. Also, assessment of the diagnostic accuracy of cyst fluid analyses requires a definitive diagnosis for each patient included in a study, which can only be made by histological examination of a surgical specimen. 57 There is therefore a bias in published studies towards cyst lesions that are symptomatic or suspected to be mucinous or malignant and have therefore been removed surgically. This means that the relative abundance of the various types of cyst lesion present in study populations may not be representative of that found in patient populations investigated in clinical practice.
Cytological investigations
Pancreatic cyst fluid obtained by EUS-FNA routinely undergoes cytological analysis. This analysis usually includes microscopic examination of smears allowing morphological characterisation of any cellular material. Certain cytological findings are suggestive of different types of cyst. Fluid from pseudocysts typically contains inflammatory cells such as neutrophils, macrophages and histiocytes, 58 an epithelial lining is absent. Cyst fluid from IPMNs and MCNs may contain mucinous columnar cells with variable presence of atypical cells.59,60 Staining of the fluid for mucin antigens with stains such as periodic acid-Schiff stain, Alcian blue or mucicarmine is also a useful tool.60,61 Mucin staining may be positive in cases of mucinous tumours such as MCN and IPMN but is usually negative in pseudocyst and SCA. The presence of cuboidal epithelial cells which stain positive for glycogen is strongly suggestive of an SCA, but this finding is present in a minority of cases. 62
The diagnostic performance of cytological analysis of pancreatic cyst fluid obtained via EUS-FNA varies between studies. A large multicentre study involving 112 patients found that when considered alone, cytological analysis yielded a diagnostic accuracy of 59% with a higher specificity than sensitivity (83% versus 34.5%) for the detection of mucinous/malignant cysts. 53 The sensitivity for the detection of malignancy in cases of mucinous cysts was 22%. In a study of 78 patients, columnar mucinous cells were seen in cyst aspirates from 31% of mucinous tumours with 100% specificity, while staining for mucin gave a sensitivity of 55% and specificity of 100% for detection of mucinous/malignant cysts. 63 A pooled analysis combining data from 12 separate studies (data from 450 patients in total) determined that cytological investigation provides a sensitivity of 35% and specificity of 83% for diagnosing mucinous/malignant lesions. 64 In summary, the majority of studies of pancreatic cyst fluid cytology and guidelines for the diagnosis of pancreatic cystic lesions agree that cytological analysis offers limited sensitivity in the detection of mucinous/malignant lesions. Certain findings offer high specificity for the identification of the type of cyst however.4,14 One of the factors limiting the sensitivity of cytological analysis of cyst fluid is that a significant proportion of fluid samples recovered from pancreatic cysts do not yield sufficient cellular material. This problem is most likely to affect smaller cysts due to the small volumes of fluid recovered. The use of cellular brushing of the cyst wall during EUS-FNA may increase cytological yield, although the relative improvement in cytological diagnosis associated with brushing and cyst aspiration versus cyst aspiration alone varies between studies.65–67
Biochemical investigations
Tumour markers
Diagnostic sensitivity and specificity of cyst fluid CEA for detection of mucinous pancreatic cysts as determined in a selection of published studies.
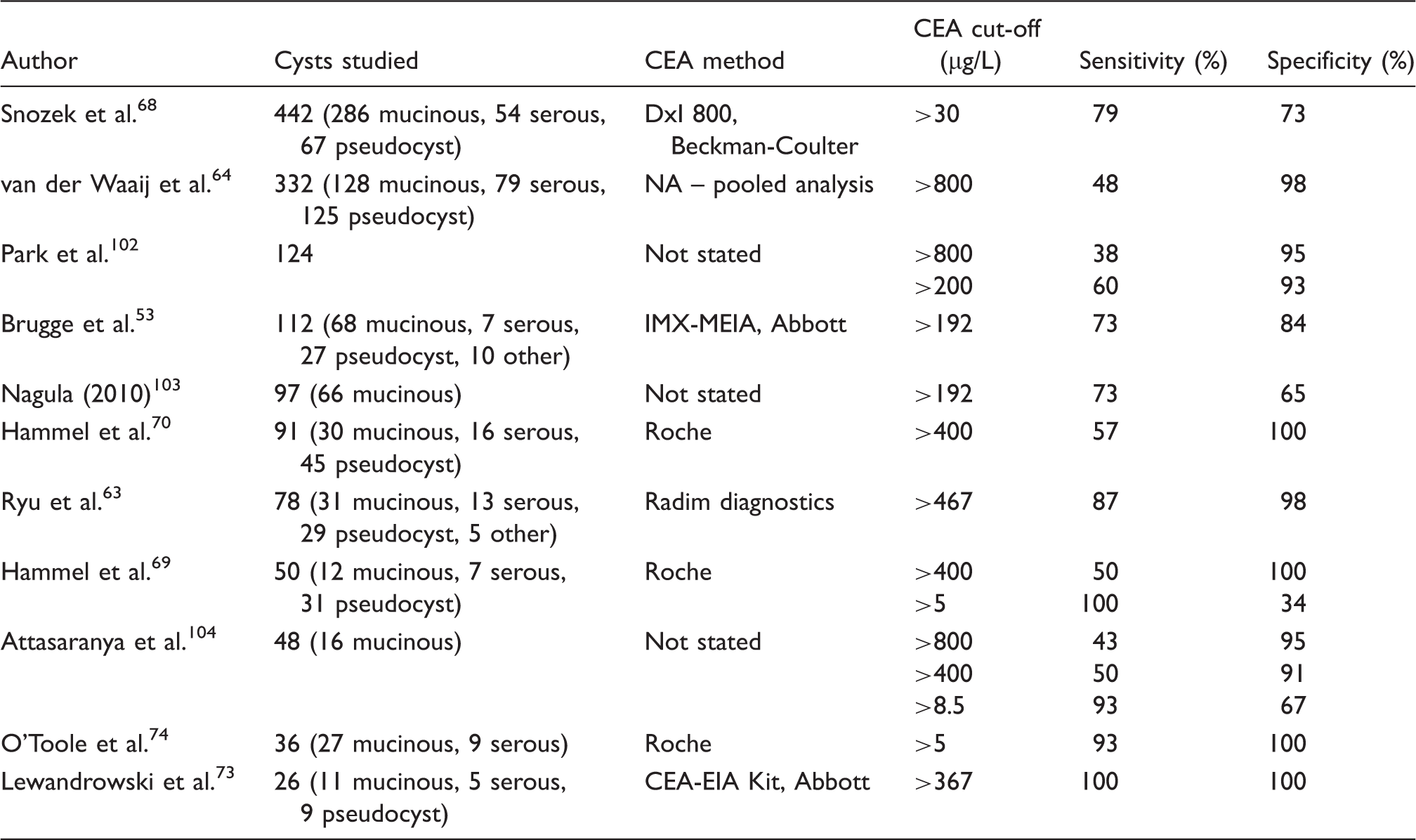
Diagnostic sensitivity and specificity of cyst fluid CA 19-9 for detection of mucinous pancreatic cysts as determined in a selection of published studies.

Sensitivity and specificity for detection of SCA or pseudocyst.
The diagnostic performance of other tumour markers such as CA 125, CA 15-3 and CA 72-4 has also been studied, although not as widely as that of CEA and CA 19-9. A study of 112 cysts found a CA 15-3 cut-off of 121
Enzymes
The role of amylase and lipase activity in pancreatic cyst fluid in the differentiation of pancreatic cyst lesions has also been studied (amylase more widely than lipase). There is significant overlap between pancreatic cyst fluid amylase and lipase activity in benign and mucinous/malignant cystic lesions to the extent that measurement of amylase/lipase is not useful in the detection of cysts with malignant potential.64,68,69 However, amylase and lipase activity are almost always relatively high in pseudocysts. Therefore, amylase/lipase cut-offs that may reliably exclude pseudocyst have been determined in a number of studies. For example, in a pooled study of 155 pancreatic cysts, 98% of pseudocysts had a cyst fluid amylase activity of greater than 250 IU/L. 64 A study of 442 pancreatic cysts found that 91% of pseudocysts had an amylase activity above 8500 IU/L and 85% of SCAs had an amylase activity below 350 IU/L. 68 In another study, an amylase activity of greater than 5000 IU/L was found in 94% of pseudocysts and lipase activity greater than 2000 IU/L in 100%. 69 In the same study, an amylase activity of less than 5000 IU/L was found in all SCAs and lipase activity less than 2000 IU/L in 86%.
DNA analysis
Studies of pancreatic cancer precursor lesions (i.e. non-cystic pancreatic neoplasia) have demonstrated certain molecular changes including mutations in specific genes, deletions and chromosomal losses during pancreatic carcinogenesis.75,76 Investigations of cystic lesions have revealed that similar molecular changes parallel the histological changes seen in the progression of mucinous pancreatic cysts.77–80 These molecular changes include K-ras mutation, p53 over-expression and loss of expression of p16 and SMAD4. Identification of these genetic changes during progression of mucinous pancreatic cyst lesions has informed studies evaluating the role of DNA analysis of pancreatic cyst fluid in the differentiation of mucinous from non-mucinous cysts. Molecular analyses studied include DNA quantity, K-Ras gene point mutation and loss of heterozygosity (LOH). The K-Ras point mutation present in pancreatic carcinomas can be detected through sequencing of the first exon (mutations are present at codon 12) of the K-Ras-2 gene. 81 LOH of tumour suppressor genes is determined by analysis of closely linked polymorphic microsatellites. 76 A microsatellite is a region of DNA with a sequence of 1–4 bases that are repeated over a short distance. The number of repeats at a given locus is often variable between alleles so that each chromosome can be identified (as there will usually be a shorter and a longer microsatellite). In LOH analysis, highly heterogeneous microsatellites are selected that serve as chromosomal markers at loci that immediately flank tumour suppressor genes or other genes of interest. LOH (for example, either the shorter or longer microsatellite is missing) suggests that one of the two chromosomal arms has been lost. Genes that have been targeted in LOH analysis in pancreatic carcinoma include retinoblastoma interacting zinc-finger (RIZ), VHL, adenomatous polyposis coli (APC), CDKN2A/p16, MX11 and Tp53.76,82 A number of published studies of the diagnostic application of LOH analysis in differentiation of pancreatic cysts have used Pathfinder TG® analysis (provided by RedPath Integrated Pathology, Inc., Pittsburgh, PA, USA). The DNA markers used in Pathfinder TG are proprietary and so are not divulged.
A pilot study involving 36 pancreatic cysts with confirmed histology (11 malignant, 15 pre-malignant and 10 benign) found that K-Ras-2 mutation followed by LOH analysis allowed sensitive and specific detection of malignant cysts (91% sensitivity and 93% specificity).
83
A later, larger study by the same group analysed cysts from 113 patients with a histological diagnosis (25 non-mucinous, 88 mucinous, 40 of which were malignant).
84
K-Ras-2 mutation analysis was found to have a sensitivity of 45% and a specificity of 96% for the detection of mucinous cysts, while LOH analysis had a sensitivity of 65% and specificity of 67%. The presence of K-Ras-2 mutation was not significantly different between malignant and non-malignant mucinous cysts. The authors concluded that using DNA analysis can increase the diagnostic yield for mucinous pancreatic cysts compared with using CEA and cytological analysis alone. For example, cytological analysis was negative for 10 of the 40 malignant cysts included in this study, while DNA analysis gave positive results for all 10 of these cysts. A smaller study of 35 cysts (6 malignant, 15 mucinous, 14 benign) with ‘clinical consensus’ diagnoses found that K-Ras-2 mutation gave a sensitivity of 57% and specificity of 100%, while LOH (as performed as part of Pathfinder TG analysis) gave a sensitivity of 43% and specificity of 90%.
85
In a further study, patients with pancreatic cysts less than 3 cm (
New markers in pancreatic cyst fluid
None of the currently available diagnostic tests based on analysis of pancreatic cyst fluid provide both high specificity and high sensitivity for the detection of mucinous or malignant pancreatic cysts. There is therefore a requirement for further research into new biomarkers in pancreatic cyst fluid that may offer improved differentiation of pancreatic cysts. The application of array and proteomic techniques to identify potentially useful markers has been investigated. One approach has involved the use of an antibody-lectin microarray method to detect proteins including CEA, mucin (MUC)1, MUC5AC, MUC16 and specific glycosylation variants of these proteins. 88 A study of 53 cyst fluid samples (17 MCN, 15 IPMN, 12 SCA and 9 pseudocysts) found that a MUC5AC bearing a specific glycan variant discriminated mucinous cysts (MCN and IPMN) from benign cysts (SCA and pseudocysts) with a sensitivity of 78% and specificity of 80%, suggesting that this approach could lead to discovery of new markers with the potential to improve the diagnosis of mucinous pancreatic cysts. A further study used an antibody array method to measure a panel of 54 proteins in pancreatic cyst fluid from 59 patients. 89 This panel included growth factors, immune modulators, cytokines, tumour antigens and hormones. The only proteins clearly over-expressed in IPMN and MCN compared with SCA were CEA and CA 72-4. A number of proteins were found to be down regulated in IPMN and MCN relative to SCA. As might be expected CEA was the best individual marker for discrimination of IPMN/MCN, but using a panel of 14 of these proteins appeared to offer improved discrimination compared with CEA alone. For example, the error rate for CEA alone in discriminating between SCA and IPMN was 14% compared with 8% for the multimarker panel. In another study, a proteomic approach to discovery of potential biomarkers in pancreatic cyst fluid highlighted a number of proteins as potentially useful biomarkers in the discrimination of mucinous pancreatic cysts. 90 A combination of matrix-assisted laser-desorption-ionization time-of-flight mass spectrometry peptidomics, 2D gel electrophoresis and tryptic digestion of proteins fractionated by SDS-PAGE with identification by LC-MS/MS highlighted 14 potential markers (2 homologues of amylase, 4 soluble mucin molecules, 4 CEA-related adhesion molecules and 4 S100 homologues). Further clinical studies would be required to characterize the diagnostic performance of these markers individually or as a panel. Plectin-1 has also been investigated as a potential marker of malignancy in IPMN. 91 This molecule is of the plakin family of proteins which are involved in the mechanical stabilization of almost all mammalian cells. Plectin-1 expression was determined in histology samples from 31 malignant IPMNs and 6 benign IPMNs using immunostaining. The specificity of Plectin-1 in distinguishing malignant IPMN from benign IPMN was 83% and its sensitivity was 84%. Furthermore, all (four out of four) cyst fluids from malignant IPMNs, but none from three benign IPMNs, were Plectin-1 positive. These results suggest that Plectin-1 expression in pancreatic cyst fluid may be a useful marker in differentiation of malignant IPMN, although further study is required.
Diagnostic pathways for pancreatic cystic lesions
The above review of the available imaging modalities and pancreatic cyst fluid analyses illustrates that there is a number of investigations that may be useful in the differentiation of pancreatic cysts, but that no single test offers absolute sensitivity and/or specificity for the identification of mucinous or malignant cysts. Therefore, these investigations must be used in a coordinated fashion to achieve the most accurate discrimination of mucinous/malignant and benign cysts possible. In order to optimize the diagnosis of pancreatic cyst lesions, evidence-based unifying diagnostic algorithms are required. The design of these algorithms is challenging due to the inherent limitations of the published studies of the diagnostic performance of the available investigations (for example, selected study populations and small populations). There are also difficulties involved in determining optimal management for the various types of cyst. An illustrative diagnostic/treatment algorithm summarizing recommendations in a number of published algorithms is shown in Figure 1 (NB this algorithm is not intended for use as a guideline but to reflect advice published in the literature). The majority of published algorithms suggest that all pancreatic cyst lesions associated with significant symptoms should be considered for surgery or cyst ablation, depending on the fitness of the patient.92–96 In the case of pseudocysts, diagnosis can often be made on the basis of a history of pancreatitis, although it may be important to exclude a neoplastic cyst lesion as IPMNs may also present with pancreatitis. Pseudocysts may be managed conservatively or with surgical/endoscopic drainage depending on factors such as size and severity of symptoms.
14
Where pancreatic cyst lesions are asymptomatic and/or discovered incidentally, the decision of whether to treat conservatively or aggressively is dependent on whether there is any evidence that the lesion is mucinous or malignant.4,92–96 Published guidelines and diagnostic algorithms agree that CT or MRI of the pancreas is unable to differentiate the type of pancreatic cyst lesion in most cases. EUS with aspiration of cyst fluid for cytological and biochemical analysis is therefore recommended. As discussed above, certain findings upon cytological analysis are specific for mucinous/malignant cysts and there are also features specific for SCA, although cytology has limited sensitivity. A high CEA concentration is suggestive of a mucinous or malignant lesion. Available data on the diagnostic performance of CEA suggest that a ‘high’ cut-off can offer a high degree of diagnostic specificity for mucinous/malignant cysts, while a ‘low’ cut-off can offer a high degree of diagnostic sensitivity (the exact limits used would be dependent on the desired diagnostic specificity/sensitivity, CEA assay used and the population being investigated). Investigations of DNA analysis of pancreatic cyst fluid indicate that the presence of K-Ras-2 mutation yields a high diagnostic specificity for mucinous/malignant cysts, with limited sensitivity. Therefore, cyst fluid analysis could be used to stratify pancreatic cysts into the following groups: likely mucinous/malignant cyst, likely benign cyst and uncertain aetiology (Figure 1). This assessment would then inform treatment, with cysts likely to be mucinous/malignant considered for resection, asymptomatic benign cysts managed conservatively and those of uncertain aetiology regularly monitored or considered for intervention if the cyst is large or showing evidence of increasing size. The various algorithms and guidelines available recommend the use of different diagnostic cut-offs for CEA concentration (or do not mention specific cut-offs) but there is agreement that asymptomatic cysts may be classified as likely mucinous/malignant, likely benign or indeterminate using a combination of imaging, cytology and biochemical analysis.
An illustrative diagnostic algorithm demonstrating the role of cyst fluid analysis in the diagnosis of pancreatic cyst lesions (NB this algorithm is not intended for use as a guideline but to reflect advice published in the literature). 1Intraductal papillary mucinous neoplasm (IPMN) can present with pancreatitis so it may be important to exclude a mucinous lesion before diagnosing pseudocyst. 2Fitness for surgery, life expectancy and risk/benefit ratio to be considered. 3Pseudocyst to be excluded before proceeding with pancreatic resection. 4‘High’ CEA cut-off chosen to provide high specificity for mucinous/malignant lesion (CEA cut-offs may be assay and population dependent). 5’Low’ CEA cut-off chosen to provide high sensitivity for mucinous/malignant cyst. 6CEA concentration between ‘high’ and ‘low’ cut-offs.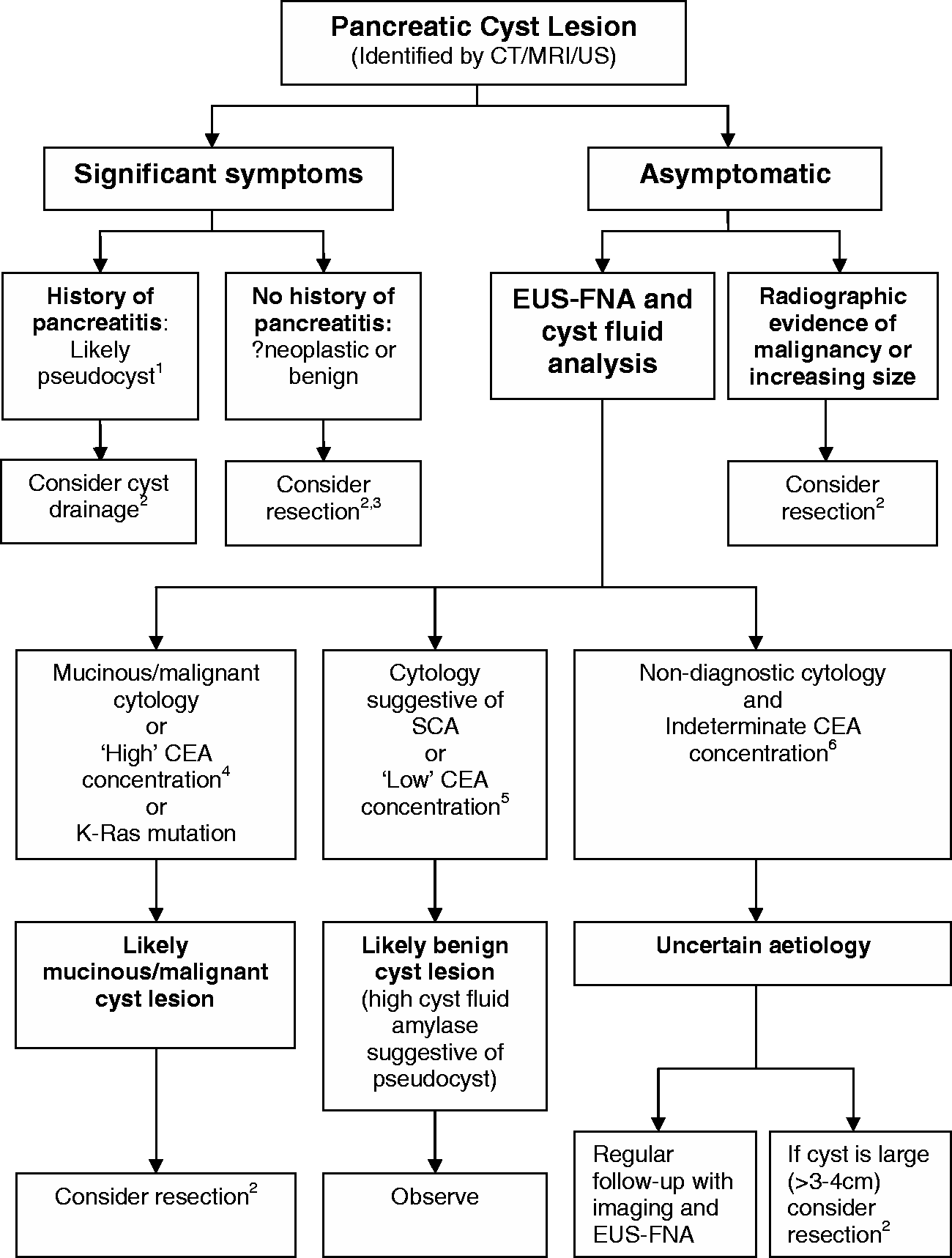
Analytical aspects of the biochemical analysis of pancreatic cyst fluid
The analytical validity and practicality of the biochemical analysis of pancreatic cyst fluid specimens are not discussed in detail in the majority of publications describing studies of the diagnostic performance of pancreatic cyst fluid analysis. These are important topics to consider in detail, as commercially available methods for the measurement of enzymes and tumour markers are validated for the analysis of serum but not cyst fluids. The discussion here is limited to tumour markers (e.g. CEA and CA 19-9) and enzymes (e.g. amylase) but the same considerations also apply to any emerging markers measured in pancreatic cyst fluid.
Physical properties of pancreatic cyst fluid specimens
Pancreatic cyst fluid varies widely in its physical properties. Specimens can vary in viscosity from a serous, watery fluid to being mucinous and quite viscous. Pancreatic cyst fluid can also be a range of colours from clear to dark brown/green. Specimens may be stained with fresh blood if there has been any haemorrhage during aspiration of the fluid. Solid material is present in many pancreatic cyst fluid specimens. The volume of fluid obtained upon cyst aspiration varies hugely from several microlitres (or none at all from small cysts or cysts containing fluid too viscous for FNA) to several millilitres. High viscosity can introduce analytical problems, as in the case of a particularly viscous sample it may not be possible to accurately sample a specific volume (vital in any quantitative analysis) either manually or via sampling probes in an automated analyser. There may also be a risk of blockage of probes or fluidics in automated analysers. The presence of solid material in many pancreatic cyst fluid samples means that it is important to centrifuge specimens prior to analysis in order to ensure particulates are not sampled during analysis.
Measurement of tumour markers in pancreatic cyst fluid
An important consideration in tumour marker analysis is that there can be significant variation in the results obtained with different assays. Data produced by external quality assessment (EQA) schemes demonstrate that significantly different results are often obtained for the same serum specimen when different assays for the same tumour marker are used. For example, a German EQA scheme found that different CEA assays in clinical use gave mean results differing by up to 85% for the same pooled specimens. 97 Variation between CA 19-9 assays was even more pronounced, with mean results differing by up to 140% and variation between CA 72-4 assays was up to 500%. These interassay differences for CEA and CA 19-9 are also evident in reports produced by United Kingdom National External Quality Assurance Scheme. It is important to account for these differences when applying diagnostic cut-offs for tumour marker concentrations as these cut-offs are not necessarily transferable between different assays. Ideally, assay-specific diagnostic limits should be determined and it is important that the methods used for tumour marker analysis are clearly stated in all reports of studies of pancreatic cyst fluid tumour marker analysis.
Commercially available assays for tumour markers such as CEA and CA 19-9 have been validated for use in the analysis of serum or plasma but not alternative matrices such as pancreatic cyst fluid. The analytical validity of applying serum tumour marker assays to the analysis of pancreatic cyst fluid does not appear to have been investigated in the majority of published studies investigating the diagnostic performance of pancreatic cyst fluid tumour marker analysis. A study investigating the reproducibility of Roche Elecsys and Bayer (now Siemens) Centaur assays for CEA in cyst fluid found intra-assay variability to be low for both assay systems (coefficient of variation was less than 5% in triplicate analyses of 40 samples), but there were significant differences between the two assays, with the Bayer assay giving values around 50% of those from the Roche assay. 98 Linearity and recovery were assessed for three samples in this study through serial dilution and addition of known concentrations of CEA. Recovery of added CEA varied between 49% and 111%, while analysis of diluted samples gave results within ±20% of the values obtained analysing neat samples. An investigation of the validity of analysis of pancreatic cyst fluid using the Roche Elecsys CEA and CA 19-9 assays evaluated the linearity of 21 specimens on dilution. 99 In the case of CEA, seven specimens exhibited non-linearity upon dilution with a progressive increase in the apparent CEA concentration as the dilution factor increased (3-fold in the most extreme case). The same pattern of non-linearity was observed for CA 19-9 in seven specimens (60-fold increase in apparent CA 19-9 concentration in the most extreme case). The cause of the non-linearity in these samples was not clear, but suggests the presence of an interference or matrix effect for some pancreatic cyst fluid specimens (although for one specimen a falsely low CA 19-9 concentration was clearly due to a hook-effect). A recent study of the analytical performance of an immunoradiometric assay of gastric M1/MUC5AC mucin also found an apparent increase in the concentration of the analyte upon dilution of around 10% of pancreatic cyst fluid specimens. 100 Western blot analysis revealed evidence of degradation of both the mucin analyte and the monoclonal antibodies used in the assay in the affected pancreatic cyst fluid specimens. It was suggested that the non-linearity and mucin/antibody degradation observed could be due to the presence of proteases in these specimens.
The analytical discrepancies found in these studies could have significant implications for the interpretation of pancreatic cyst fluid tumour marker concentrations. More research is required into the underlying causes of these discrepancies and how they can be accounted for or eliminated. Future developments in tumour marker analysis (such as development of mass spectrometry techniques allowing specific detection of protein markers or peptide fragments of these markers) may overcome some of the analytical problems discussed here.
Measurement of enzymes in pancreatic cyst fluid
Most of the analytical aspects discussed above with regard to measurement of tumour markers in pancreatic cyst fluid also apply to enzymes such as amylase and lipase. Although amylase is widely measured routinely in body fluids such as drain fluids and pleural fluid, commercially available assays have been validated for serum/plasma (and sometimes urine) only. Matrix-specific effects may therefore affect enzyme measurement in pancreatic cyst fluid, although between assay comparability is generally better for amylase and lipase than for the tumour markers discussed above. Differentiation of mucinous or malignant pancreatic cysts does not depend on diagnostic limits for amylase/lipase as it does in the case of tumour markers, particularly CEA. Amylase or lipase concentrations can be supportive of the diagnosis of pseudocyst or IPMN where the activity of these enzymes are typically very high, usually greater than 10 times the upper limit of the reference range for serum.64,68,69
Summary of the analytical aspects of biochemical analysis of pancreatic cyst fluid
As discussed above, the use of commercially available assays for tumour markers and enzymes have not been formally validated for the analysis of pancreatic cyst fluid. Existing guidelines on body fluid analysis do not provide detailed guidance on the validity of specific biochemistry tests for different fluid types. Guidelines from the Association of Clinical Biochemistry in Ireland give the general advice that although method validation for fluids other than blood and urine has usually not been carried out, valuable information may still be obtained for individual patients. 101 These guidelines also highlight that quality control may be an issue as internal quality control material and EQA material is usually serum or urine based. They suggest that exchange of fluid specimens between sites may be useful in the absence of matrix-specific EQA schemes.
Despite the highlighted concerns of matrix-specific interference effects and the lack of formal validation for tumour marker (and enzyme) assays, many studies demonstrate that these assays provide useful tests for the differentiation of pancreatic cyst lesions. In particular, a number of studies have found that cyst fluid CEA is one of the most useful tests for diagnosing mucinous/malignant cysts.10,53,63,64,68,87 It is therefore recommended that requests for biochemical analysis of pancreatic cyst fluid specimens from hepatobiliary surgery and radiology colleagues are carefully considered by pathology departments. Cooperation between clinicians involved in managing patients with pancreatic cysts and laboratory medicine departments is important. This can improve understanding of the clinical need for these tests in laboratories and also improve understanding of the analytical issues involved in cyst fluid analysis for clinicians. This cooperation should lead to agreement on what tests are required and what services should be available. Laboratories should help to ensure that analytical parameters such as linearity and precision have been investigated for the methods being used and the potential ramifications of any analytical discrepancies identified should be discussed with the relevant clinical teams. It is vital that results from the biochemical analysis of pancreatic cyst fluid are interpreted in conjunction with imaging, cytology and clinical history as no biochemical test offers absolute diagnostic accuracy. It is also recommended that laboratories offering analysis of pancreatic cyst fluid collaborate with clinicians to monitor the clinical usefulness and diagnostic performance of the tests offered. This can be challenging as a definitive diagnosis is often not available for patients that have undergone investigation for a pancreatic cyst lesion.
Conclusions
The detection rate of pancreatic cyst lesions has increased in recent years. Many of these lesions are asymptomatic and discovered incidentally. Detection of a pancreatic cyst lesion can offer a diagnostic and management challenge as the aetiology and potential for malignancy is often unclear. Biochemical analysis of pancreatic cyst fluid obtained via EUS-FNA is an important part of the investigation of these cysts in conjunction with cytological analysis and a variety of imaging techniques. Measurement of cyst fluid CEA concentrations has proven to be particularly useful in helping to differentiate benign and mucinous or malignant cysts. However, care must be taken in the interpretation of cyst fluid CEA concentrations as there is a large overlap between benign and mucinous/malignant lesions. High concentration cut-offs may be used to diagnose a mucinous cyst with high diagnostic specificity, while a low cut-off can provide a high diagnostic sensitivity for mucinous cysts so that CEA concentrations below this cut-off are likely to be benign. Diagnostic limits for CEA should be assay-specific and determined for the population being investigated. A number of studies have found that DNA analysis has a role in the investigation of pancreatic cysts. In particular, K-Ras-2 mutational analysis appears to offer a high diagnostic specificity for mucinous or malignant cysts albeit with limited sensitivity. A number of studies, some using multiplex analytical techniques, have identified further markers in pancreatic cyst fluid which could potentially be useful in differentiation of cysts. Further research is required to define the diagnostic performance of these emerging markers. There are a limited number of published studies examining the analytical validity of applying serum tumour marker assays to pancreatic cyst fluid analysis. Some analytical discrepancies have been identified, particularly non-linearity on dilution of some pancreatic cyst fluid specimens. Further study is required to determine the cause of these discrepancies and the impact they may have on clinical interpretation. Collaboration between relevant clinical groups and laboratories is important in establishing and maintaining appropriate pathology services for the investigation of pancreatic cyst lesions.
Footnotes
Acknowledgements
The author would like to thank Dr Penny Clark, Dr Kirsty Gordon, Dr Brinder Mahon and Mr Simon Bramhall for discussions on the subject of pancreatic cyst fluid analysis.
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
CB.
Contributorship
CB compiled and wrote this review without assistance.
