Abstract
Background
Therapeutic drug monitoring of ciclosporin A (CsA) and tacrolimus is traditionally performed using venous whole blood sampling. A number of reports have described development of ultra high-performance liquid chromatography tandem mass spectrometry (UPLC-MS/MS) methods for the quantitation of CsA and tacrolimus from dried blood spots (DBS), which may offer a convenient alternative. As yet, no such reports have validated this methodology using fingerprick capillary DBS samples collected from transplant patients.
Methods
Capillary fingerprick DBS were collected from heart and lung transplant patients in a specialist cardiothoracic transplant centre. We utilized our previously published method for the extraction and simultaneous quantitation of CsA and tacrolimus from DBS using UPLC-MS/MS. Drug concentrations measured from DBS were compared to concentrations measured in venous whole blood by our routine clinical UPLC-MS/MS assay.
Results
In total, 91 heart or lung transplant patients were enrolled onto the study; 46 patients were on CsA therapy and 45 on tacrolimus therapy. Passing–Bablock analysis demonstrated excellent agreement between capillary fingerprick DBS samples and venous whole blood samples. There was a mean positive bias of 2.6
Conclusions
We demonstrate utility of DBS for serial monitoring of CsA and tacrolimus using UPLC-MS/MS in heart and lung transplant patients. This may offer significant advantages for these patients including the ability to take capillary DBS samples in the community prior to clinic visits.
Keywords
Introduction
Ciclosporin A (CsA) and tacrolimus are immunosuppressant drugs principally used in solid organ transplants. Due to variable pharmacokinetics and narrow therapeutic ranges, therapeutic drug monitoring (TDM) of both drugs is essential to prevent nephrotoxicity associated with overdosage and graft rejection from underdosage.1,2 CsA and tacrolimus are routinely measured in venous whole blood samples and requirement for frequent TDM necessitates regular patient visits to hospital or community phlebotomy services. Fingerprick capillary blood sampling onto dried blood spots (DBS) offers several advantages. Patients can collect samples at home and post DBS cards to the laboratory for analysis thus reducing the frequency of hospital visits and improving patient–physician contact time. DBS are also advantageous for CsA C2 measurements which requires precise 2 h post-dose blood sampling and is considered a better measure of drug exposure than pre-dose trough sampling.3,4 Additionally, many analytes exhibit enhanced stability when dried on the blood spot card compared to stability in venous blood. 5
We have previously reported the development of an ultra high-performance liquid chromatography tandem mass spectrometry (UPLC-MS/MS) method for the simultaneous quantitation of CsA and tacrolimus from DBS which, for convenience, were prepared from venous whole blood samples. 6 If used in routine clinical practice, measurement of CsA and tacrolimus from DBS would involve the use of fingerprick capillary blood sampling. The composition of capillary blood is thought to be a mixture of arterial and venous blood, a factor which theoretically could significantly affect measured drug concentrations. As yet, no reports have validated DBS measurements of immunosuppressants using fingerprick capillary DBS collected from transplant patients. We aimed to validate DBS sampling for routine TDM of CsA and tacrolimus compared to venous whole blood sampling.
Materials and methods
Fingerprick capillary samples were collected onto Whatman 903 DBS cards from a cohort of 91 heart and lung transplant patients during outpatient consultations in a specialist cardiothoracic transplant centre over a period of six months. Venous whole blood samples for routine immunosuppressant TDM analysis were collected concurrently. Dosages received by patients were dependent upon the time post-transplantation and renal function. For CsA the average dose was 100–125 mg bd (range 25–225 mg bd). For tacrolimus the average dose was 3–4 mg bd (range 0.5–4.5 mg bd). Ethical approval for the study was obtained from the local ethics review board.
The UPLC-MS/MS method described previously for the measurement of CsA and tacrolimus from DBS was used in this study.
6
Briefly, CsA and tacrolimus DBS calibrators and quality control (QC) samples were prepared by spiking pooled venous whole blood with stock solutions of pure drug. Calibrators and QCs (25
Concentrations of CsA and tacrolimus measured from fingerprick capillary DBS using the UPLC-MS/MS method described above were compared to those obtained from venous samples using the routine UPLC-MS/MS clinical assay. This whole blood assay is calibrated using commercially available calibrators and QCs (Chromsystems, Munich, Germany). Concentrations determined by each method were compared using Analyse-It™ statistical analysis software (Analyse-It Software Ltd, Leeds, UK).
Results and discussion
In total, 46 patients on CsA therapy enrolled onto the study (32 male). The median age was 60.0, inter-quartile range (IQR) 14.0 (50.0–64.0), 19 were heart transplant patients and 27 lung transplant patients. Of the DBS samples collected from these patients, one was deemed too poor quality to analyse (small, overlaid spots) leaving 45 patients with acceptable DBS collections. A total of 45 patients on tacrolimus therapy enrolled on the study (38 male), the median age was 50.0, IQR 15.3 (43.7–59.0), 20 were heart transplant patients and 25 lung transplant patients. Of the DBS samples collected from these patients, three were deemed too poor quality to analyse leaving 42 patients with acceptable DBS collections.
Drug concentrations measured from DBS were compared to the concentrations measured in venous whole blood samples collected at the same time as the DBS. For CsA, Passing–Bablock analysis yielded the equation: Capillary DBS = 1.0 (venous blood) +0.2, Method comparison data for CsA and tacrolimus between the capillary fingerprick DBS and venous whole blood LC-MS/MS methods: (a) Passing–Bablock analysis for CsA, (b) Bland–Altman analysis for CsA, (c) Passing–Bablock analysis for tacrolimus and (d) Bland–Altman analysis for tacrolimus. Dashed lines on Passing–Bablock and Bland–Altman plots indicate 95% confidence intervals, dotted line on Bland–Altman plots indicates mean bias.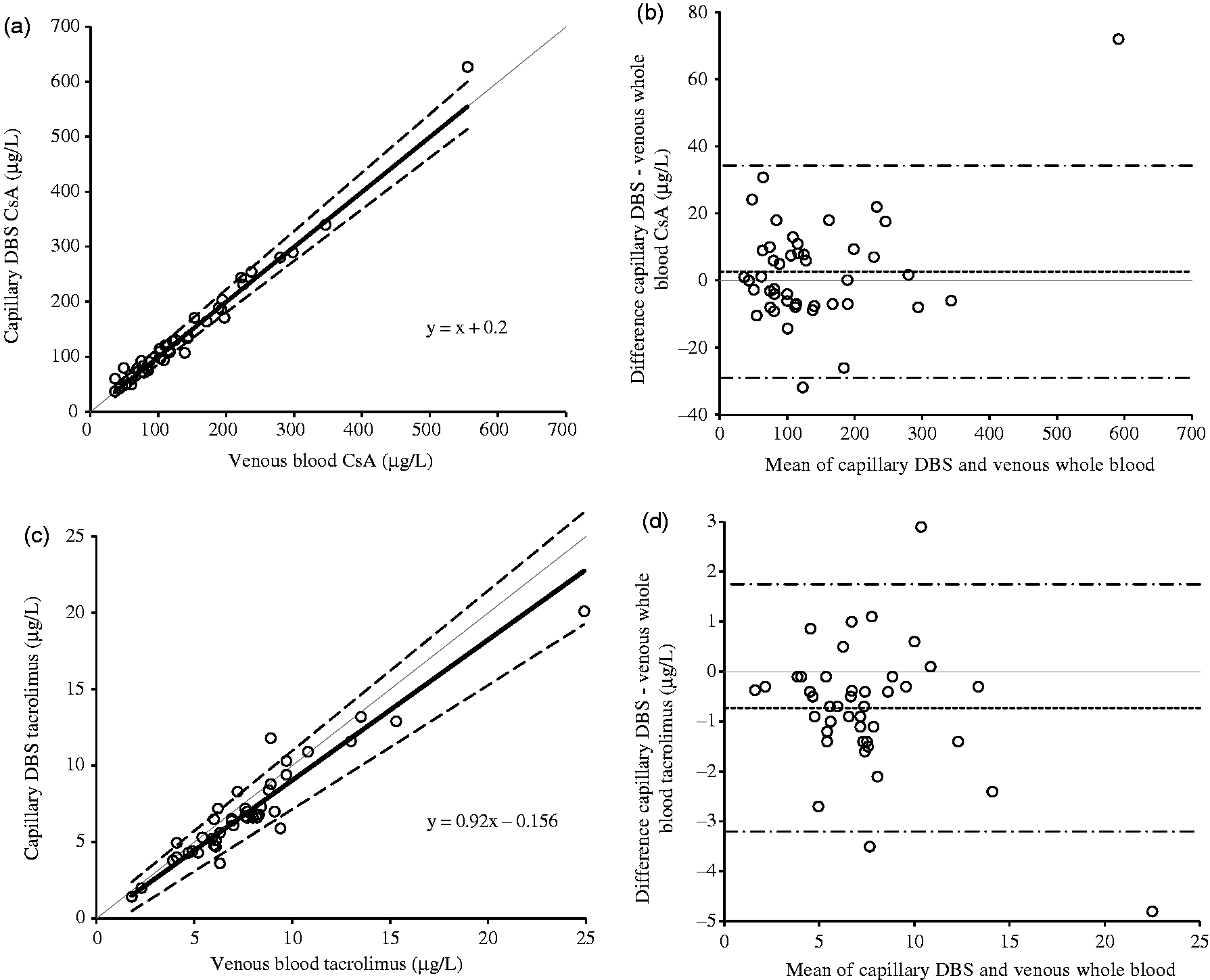
Conclusions
Previous reports describing development of LC-MS/MS methods for quantification of CsA and tacrolimus from DBS have thus far not been formally validated using capillary DBS samples collected from a cohort of transplant patients. Herein, we compared TDM of CsA and tacrolimus using DBS sampling and our previously described UPLC-MS/MS method for DBS quantitation, with that of our routine venous whole blood UPLC-MS/MS TDM assay. Comparison of 45 and 42 patient samples each for CsA and tacrolimus, respectively, demonstrated very good agreement between the two methods as demonstrated by Passing–Bablock and Bland–Altman analysis.
DBS samples were collected from a cardiothoracic outpatient clinic and analysed in batches weekly. Although we did not formally assess home collection of DBS we have previously demonstrated stability of CsA and tacrolimus in DBS for up to 14 days storage at room temperature. 6 This stability data allow ample time for DBS samples to be collected by patients at home or at a community clinic and subsequent postage to the laboratory for analysis.
Practically, DBS calibrators and QC sample preparation was performed in-house which is technically demanding and time consuming. In future, it would be advantageous for manufacturers to consider production of CsA and tacrolimus DBS calibrators, QCs and external QC material. This would avoid inter-laboratory variation arising from calibration preparation, allowing more widespread use of this sampling technique.
To conclude, we demonstrate utility of DBS for serial monitoring of CsA and tacrolimus in heart and lung transplant patients using a novel UPLC-MS/MS method. This may offer significant advantages for transplant patients, including the ability to take capillary DBS samples in the community prior to clinic visits.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
None.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Ethical approval for the study was obtained from the University Hospital of South Manchester Ethics Review Committee, Ref. 09/H1013/53 and 10/H1016/35.
Guarantor
BK.
Contributorship
BK was involved in study design and gaining ethical approval. JF and AR obtained informed consent, enrolled patients and collected all DBS samples. EH analysed all samples, performed the data analysis and wrote the first draft of the manuscript. All authors reviewed, edited and approved the final draft of the manuscript.
