Abstract
Background
Gestational hypothyroidism has been shown to be associated with adverse pregnancy outcomes as well as adverse outcomes for the child. Thyroid hormones concentrations change in gestation, especially within the first trimester, so the results of thyroid function test often are outside non-pregnant reference ranges. The objective of this study was to establish the first trimester reference ranges for thyroid stimulating hormone (TSH) and free thyroxine (FT4) for pregnant women in Barcelona (Spain).
Methods
It was a prospective study in which 673 women were recruited during their first trimester of gestation (8–13 weeks). Serum TSH, FT4 and antithyroid peroxidase antibodies (TPOAb) were measured with Atellica® IM 1600 (Siemens Healthineers). After excluding 418 women, the reference ranges for TSH and FT4 were calculated by the 2.5th and 97.5th percentiles. Potential variables examined in this study were age, body mass index (BMI), ethnicity, iodine supplementation and smoking habit.
Results
The reference ranges established on the Atellica® IM 1600 for the first trimester pregnancy in our population were 0.111 to 4.291 mIU/L for TSH and 11.45 to 17.76 pmol/L for FT4. No significant differences were found in thyroid hormones concentrations regarding maternal age (≤30 years vs >30 years) (p = .117), iodine supplementation (p = .683) and smoking habit (p = .363). The prevalence of TPOAb was estimated at 10.0%.
Conclusions
We found that in our local population, the optimal TSH upper reference limit in the first trimester of gestation was 4.3 mIU/L, similar to that proposed by de ATA-2017 guideline (4.0 mIU/L).
Keywords
Introduction
Thyroid dysfunction is a common disorder in the general population. In Spain, its prevalence was investigated recently by the Di@bet.es Study and showed a high prevalence of thyroid dysfunction (around 10%) and a clear predominance in women compared with men. Moreover, the prevalence of thyroid disorders in women of childbearing age was of 2.0–4.5% for clinical hypothyroidism, 5.0–7.0% for subclinical hypothyroidism, 0.5–1.0% for hyperthyroidism and 5.0–10.0% for positive thyroid autoimmunity. 1
Hypothyroidism is the most common thyroid disorder and has been shown to be associated with infertility, adverse pregnancy outcomes as well as adverse outcomes for the child among many other disorders. 2 In Spain, universal screening for thyroid disease in the first trimester of pregnancy is recommended by the Spanish Society of Endocrinology and Nutrition (SEEN) and the Spanish Society of Gynecology and Obstetrics (SEGO), 3 but there is still no agreement between the clinical practice guidelines used in the different regions of the country.
Accurate assessment of maternal thyroid function during pregnancy remains difficult since pregnancy has clear effects on thyroid physiology. During the first trimester, human chorionic gonadotropin (hCG) and increased production of thyroxine-binding globulin (TBG) induce a transient increment in free thyroxine (FT4) concentrations resulting in lower maternal thyroid stimulating hormone (TSH) concentrations. 4 Therefore, the reference ranges of thyroid parameters used for the general population are not applicable to pregnant individuals and reference ranges for normal thyroid function during pregnancy are required for its use in clinical practice. 5
The latest American Thyroid Association guideline (ATA-2017) recommended trimester-specific reference ranges for serum TSH during pregnancy defined by each laboratory. The reference ranges should be defined in healthy negative thyroid peroxidase antibodies (TPOAb) pregnant women, with optimal iodine intake and without thyroid illness. If this approach is not feasible, laboratories can use pregnancy-specific TSH reference ranges obtained from similar populations assessed by comparable TSH assays or, alternatively, they can use a universal upper reference limit of 4.0 mIU/L. 6
Although guidelines agree on the need to obtain own reference ranges, according to the TYROGEST study, in Spain, most centres still use the universal upper reference limit proposed by ATA-2017 guideline or, even, reference ranges for the non-pregnant population provided from the manufacturer. 7 Although ATA-2017 guidelines state that for most assays, the upper reference limit represents a reduction in the non-pregnant TSH upper reference limit of 0.5 mIU/L; according to a recent study, the upper limit of TSH should not be extrapolated from the reference ranges of the non-pregnant population. 8
TSH and FT4 reference ranges differ mostly according to assay method, reference population characteristics (e.g. ethnicity), gestational age, thyroid autoimmunity, iodine nutritional status and statistical tests used.9,10 Several studies showed a marked variation for the upper limit of TSH due to differences in assay methods.8,11,12 To date, there are no studies carried out on the pregnant population to define reference ranges in diagnostic tests for thyroid dysfunction with Atellica® IM 1600 Analyzer (Siemens Healthineers), which is an immunoassay analyzer widely implemented since its release on the market in 2018.
There are also methodological differences between studies when choosing the exclusion criteria for the selection of the reference population. Some studies, such as this one, apply further exclusion criteria in addition to those recommended by the current ATA guidelines (known thyroid disease, use of thyroid interfering medication, multiple gestation, iodine deficiency and TPOAb positivity) in order to eliminate factors that may affect thyroid hormones concentrations. According to a recent meta-analysis, the application of additional exclusion criteria does not invalidate the results obtained (additional exclusion criteria had less than 5% of effect on the reference limits), but limits the precision of the results obtained by decreasing the number of participants included. 13
The aim of this study is to define population-based first trimester reference ranges for serum TSH and FT4 through assessment of local population data using a new immunoassay analyzer (Atellica® IM 1600, Siemens Healthineers).
Materials and methods
Patients
After obtaining the approval of the Ethics and Clinical Research Committee of Vall d’Hebron Universitary Hospital (VHUH), a total of 673 individuals were enrolled between July and October 2021. The participants consisted of healthy pregnant individuals seen at the Department of Obstetrics and Gynaecology of a public tertiary centre (VHUH, Barcelona, Spain) and their associated Primary Healthcare Centers (CAPs). A complete thyroid profile (TSH, FT4 and TPOAb) was determined in all participants between 8 and 13 weeks of gestation. The results were concealed to both participants and care providers.
The inclusion criteria were pregnant women in their first trimester of gestation. The exclusion criteria included positive TPOAb, history of thyroid diseases, multiple gestation, diabetes, hypertension, age below 18 years, pregnancies conceived by assisted reproductive techniques, pregnancy complications (placental abruption, premature rupture of membranes, preterm delivery, preeclampsia, gestational diabetes, miscarriage and cesarean delivery for any reason), neonatal complications (neonatal death, paediatric intensive care unit admission and low birthweight, defined as neonatal weight below the 10th percentile) 14 and treatments that may affect thyroid function (e.g. amiodarone and lithium). Lost to follow-up patients were also excluded.
Medical records were systematically reviewed in all participants and the following information was recorded in an electronic database: age, body mass index (BMI), smoking habit, ethnicity and intake of iodine supplements. BMI, calculated as weight in kilograms divided by the height in metres squared, categories: <18.5 kg/m2, underweight; 18.5–24.9 kg/m2, normal weight; 25.0–29.9 kg/m2, overweight; >30.0 kg/m2, obesity.
Laboratory measurements
Blood samples were centrifuged (2390 g, 10 min) to obtain serum. Serum TSH, FT4 and TPOAb concentrations were measured by automated chemiluminescent immunoassay system (CLIA) (Atellica® IM 1600, Siemens Healthineers) and were tested within 6 h. Functional sensitivity of the TSH assay was 0.008 mIU/L. Reference ranges for TSH and FT4 for non-pregnant population provided by the manufacturer were 0.555–4.780 mIU/L and 11.5–22.7 pmol/L, respectively. TPOAb value less than 60 IU/mL were considered negative as per the manufacture’s recommendation. Only women who were found to have serum TPOAb concentrations below this cut-off limit were included.
Internal quality controls (QC) materials were performed daily and employed to assess accuracy before samples analysis. Three levels of QC (Liquicheck Immunoassay Plus Control from Bio-Rad®) were performed for TSH and FT4 and two (Liquicheck Specialty Immunoassay Control from Bio-Rad®) for TPOAb. All values were within the acceptable range. The within-laboratory coefficients of variation (CVs) of TSH, FT4 and TPOAb assays obtained by the mentioned QC materials were less than 3.5%, 2.5% and 5.0%, respectively, for all levels. Additionally, monthly QC materials purchased from two external quality assessment programs (SEQCML and RIQAS) were used to verify accuracy. Bias is expressed as the Standard Deviation Index (SDI), being 0.0 the ideal target while values greater than 2.0 indicate an unacceptable performance. During the 4 months of the study, all SDI were less than 1.5 in all the magnitudes indicating good performance.
Statistical analysis
Our Laboratory Information System (Modulab®, Werfen) provided the data collected by Excel Microsoft. All statistical analyses were performed using Stata® software version 15 (StataCorp, College Station, Texas 77845 USA). Graphs were obtained using GraphPad® Prism version 8.0.2 for Windows. Values are reported as frequency and percentages or as median and interquartile range (IQR).
Before statistical analysis, 12 outliers of hormones concentrations were detected by Grubbs test. Normality assessment was verified using Kolmogorov–Smirnov test. Reference ranges were estimated by the 2.5th and 97.5th percentiles and the 95% confidence interval (CI) of the reference ranges was calculated wherever applicable.
Wilcoxon rank sum test or Kruskal–Wallis test (for more than two subgroups) were used to compare thyroid function test resultant between groups. The variables explored were maternal age (<30/≥30 years), smoking habit (yes/no), iodine supplementation (yes/no), BMI (<25/≥25.0 kg/m2) and ethnicity (Caucasian/South American/Central American/South Asian/Chinese/African/Arabian/Hindu/Others). We considered smokers those women who smoked at least one cigarette per day. All reported p values are two-tailed and statistical significance was defined as p < .05.
Results
Description of reasons for study exclusion.

Characteristics of the women included in the study.

Since differences in TSH concentrations by maternal age, iodine nutrition, smoking habit, ethnicity and maternal BMI have been described in the literature, 13 we investigated whether there were significant differences in TSH concentrations based on these parameters. Nevertheless, no significant differences were found for maternal age (≤30 years vs >30 years) (p = .117), iodine supplementation (p = .683), smoking habit (p = .363) or BMI (<25 kg/m2 vs ≥25.0 kg/m2) (p = .723). Thus, the reference ranges were calculated without being stratified by these variables. Due to an imbalance in the number of patients assigned in the BMI and ethnicity categories, these variables were not included in the study.
Reference ranges for serum thyroid stimulating hormone (TSH) and free thyroxine (FT4) during the first trimester of gestation.
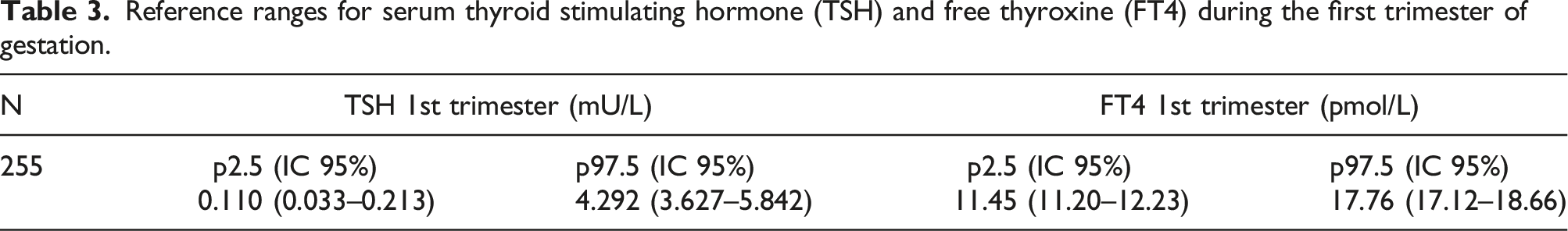
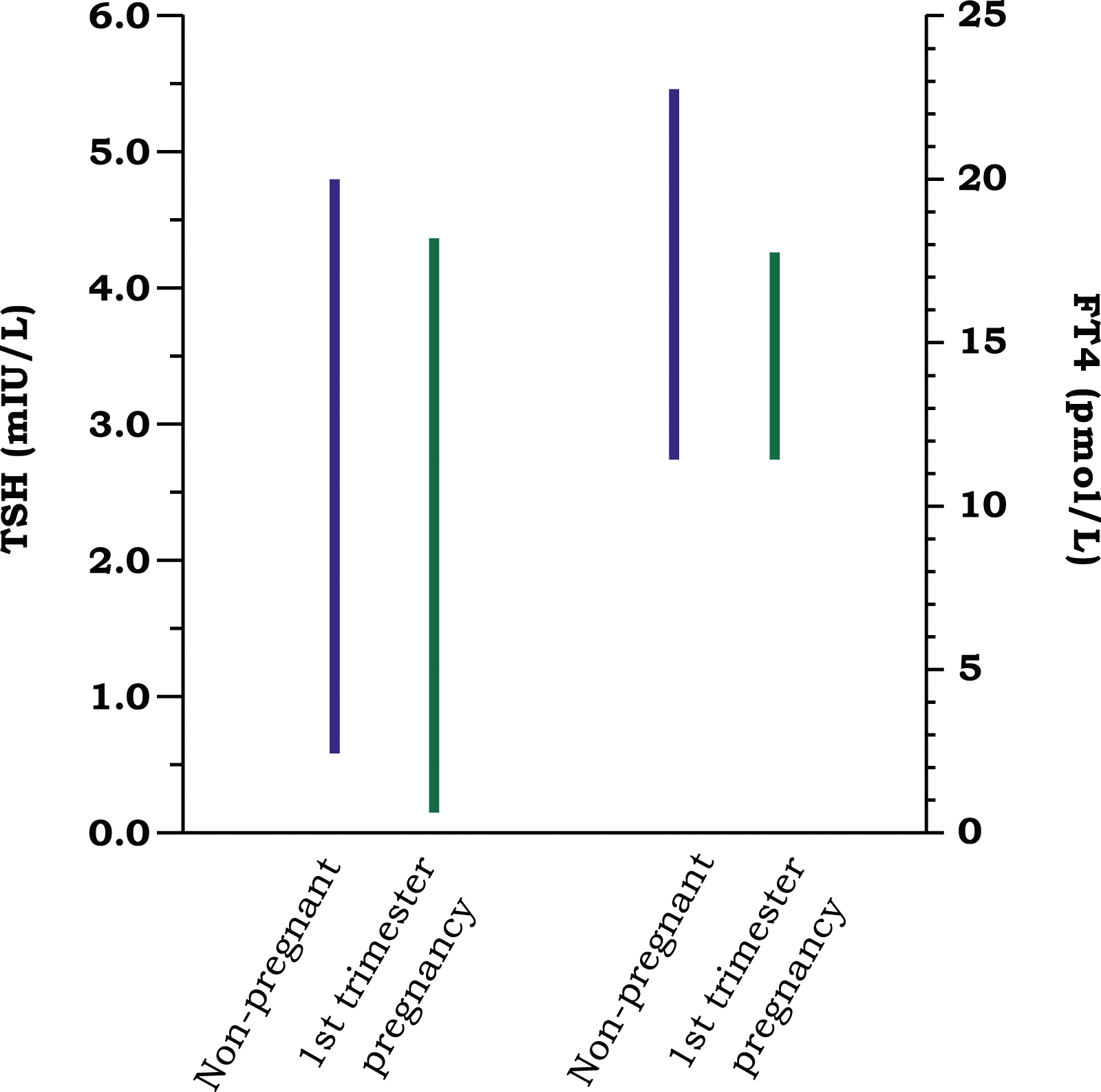
Differences between the manufacturer's reference ranges for thyroid stimulating hormone (TSH) and free thyroxine (FT4) in the non-pregnant population and those obtained in women in their first trimester of pregnancy (8–13 weeks of gestation).
Reference ranges were also calculated applying only the exclusion criteria recommended by the ATA guidelines, being 494 women the number of participants included in this second cohort. The 2.5th and 97.5th percentiles for TSH and FT4 were 0.122 and 4.157 mIU/L and 11.33 and 17.50 pmol/L, respectively. These results, obtained with a larger number of participants, were essentially the same to those obtained in the first cohort with additional exclusion criteria.
As recommended by the guidelines, the reference ranges described were calculated in pregnant women with negative TPOAb. Several studies reported higher baseline TSH concentrations in women with thyroid autoimmunity than women who were negative for antibodies.
15
In our TPOAb-positive subgroup (n = 66), TSH concentrations were significantly higher than in patients without antibodies (median 2.375 vs 1.584 mIU/L, p = .001) (Figure 2). Serum thyroid stimulating hormone (TSH) concentrations in pregnant women with and without TPOAb. The horizontal line in each box represents the median, the lower and upper boundaries of the boxes the interquartile range, the ends of the whisker lines the minimum and maximum values.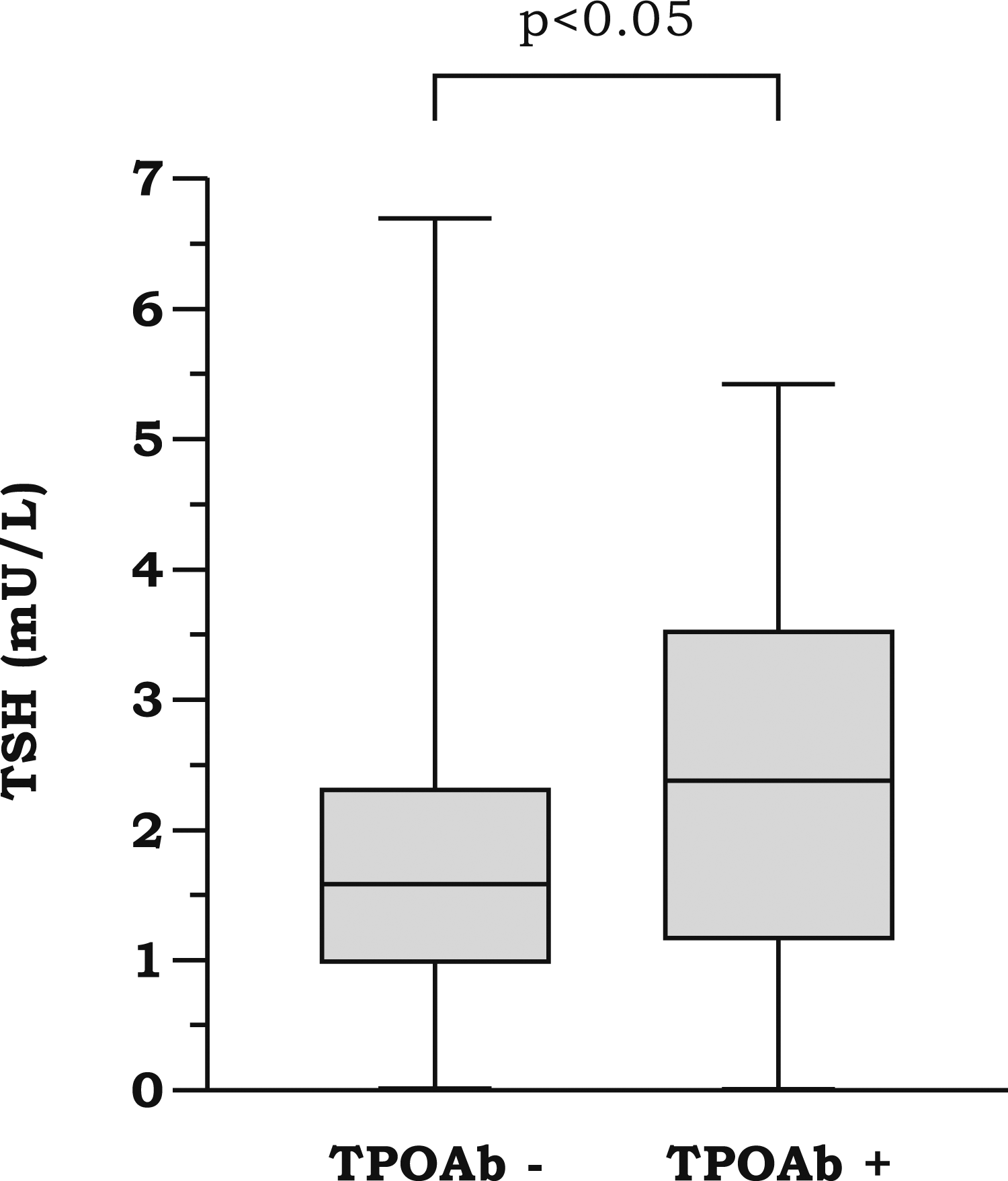
Discussion
Maternal thyroid dysfunction may have an adverse impact on both mother and child.5,16 Screening is assessed by measuring serum TSH concentrations, but due to physiological changes that occur in pregnant women, TSH normal values differ from non-pregnant population. Maternal subclinical hypothyroidism (elevated concentrations of TSH with normal concentrations of FT4) is the most common thyroid disorder 17 and its prevalence (5–15%) is determined by the cut-off value of TSH considered as the upper limit of normality.
Treatment effectiveness for women with subclinical hypothyroidism is controversial and there is no consensus among guidelines on when and how to treat this population. 18 ATA-2017 guideline recommended levothyroxine (LT4) therapy for TPOAb-positive women with a TSH greater than the upper limit of the reference range and considered LT4 therapy for TPOAb-negative women with TSH concentrations greater than the upper limit of the reference range. Therefore, reference ranges are importance for the correct assessment of maternal thyroid function.
In this prospective study, the local reference ranges of TSH (0.111–4.292 mIU/L) and FT4 (11.45–17.76 pmol/L) in the first trimester haven been established for pregnant women attending obstetric outpatient appointments in the catchment area of Vall d’Hebron Clinical Laboratories (Barcelona, Spain). To select an accurate reference population, maximum precautions were taken in our exclusion criteria. The TSH upper reference limit obtained in this study, 4.3 mIU/L, is similar to those published in other studies carried out with Spanish population and a similar measurement method (ADVIA Centaur®). 11 As TSH and FT4 immunoassays are not standardized, these reference ranges are specific for Atellica® IM 1600 analyzer.
Measuring FT4 is challenging and some FT4 automated immunoassays, as Atellica, overestimate FT4 concentrations in pregnant women due to binding-protein levels are altered (higher TBG and lower albumin concentrations). 19 This study, however, found a lower upper limit of FT4 in pregnant women (17.76 pmol/L) compared to the upper limit for non-pregnant adults provided by the manufacturer (22.70 pmol/L) and slightly lower compared to other studies with similar technology.11,20 Although there is a transient rise in FT4 in the first trimester due to the relatively high circulating hCG concentrations, most studies reported lower reference ranges in pregnant women in their first trimester.21,22 Therefore, it would also be advisable for the laboratory to verify the manufacturer's reference ranges for non-pregnant population.
Regarding the exclusion criteria, the results of this study show that it is not necessary to apply additional exclusion criteria of those recommended by ATA guidelines. The TSH upper reference limit changed from 4.3 to 4.2 mIU/L when no additional exclusion criteria were applied. So, factors as conception by assisted reproductive techniques, pregnancy complications or BMI seem not being determinant for TSH and FT4 concentrations.
Despite the recommendation of universal screening for gestational hypothyroidism (GH) by Hypothyroidism Working Group, 23 the inclusion of the TSH measurement in the first trimester analysis is not established in many protocols. The Catalan pregnancy monitoring protocol does not include GH screening; even so, in 2021, the 64.9% of the first trimester screenings received in our centre requested the measurement of TSH concentrations. According to the algorithm proposed by the guideline ATA 2017, reflex TPOAb should be performed if TSH is 2.5–10.0 mIU/L. Whenever possible, the laboratory should measure serum TPOAb automatically in the same blood sample and reflect the result in the report. This practice would help to prevent abortion, since a delay in the measurement of thyroid autoimmunity can cause a delay in obtaining the diagnosis and initiating treatment. In high-risk women, measurements of TSH should be performed as soon as pregnancy is confirmed, prior the first trimester screening.
The prevalence for TPOAb positivity in our pregnant women population during the first trimester of gestation was 9.8%, very similar to that found in another study of healthy pregnant women in the North of Spain using a similar analytical system. 24 TPOAb positivity may alter fertility or cause early miscarriages, 25 most of them occur before the 10th week of gestation. Therefore, the percentage of childbearing age women with thyroid autoimmunity could be higher. According to the guidelines, in these women, serum TSH concentration should be performed at time of pregnancy confirmation (if her autoimmune pathology is already known) and every 4 weeks throughout mid-pregnancy.
A limitation of this study was not measuring urinary iodine concentration in order to assess the nutritional iodine status of pregnant women. However, although Spain had been an iodine-deficient area historically, in 2004, the World Health Organization (WHO) included Spain among the countries with optimum iodine nutrition 26 and, according to Iodine Global network, a non-government organization for the sustainable elimination of iodine deficiency worldwide, the median urinary iodine concentration in Spain in 2017 was adequate (173 µg/L). The iodization of milk and the iodized salt have greatly contributed to the improvement of the situation in Spain.27,28
In conclusion, we found that in our local population, in the first trimester of gestation, the optimal TSH upper reference limit is 4.3 mIU/L, similar to that proposed by de ATA-2017 guideline (4.0 mIU/L). This value is lower than the upper reference limit of TSH defined by our methodology for non-pregnant population (4.7 mIU/L), but is much higher than the conservative cut-off recommended for different endocrine associations (2.5 mIU/L) in 2007. 29 The women included represent the typical population cared in our centre, a multiethnic population but mostly Caucasian. In addition, this study provides relevant information due to the fact that it is the first one carried out with the Atellica® IM 1600 Analyzer, the technological renewal of the widely employed ADVIA Centaur® Analyzer. Although the reagent formulations used in the Atellica® IM 1600 Analyzer are the same as in the ADVIA Centaur® system, results may slightly vary due to differences in analytical platform, incubation times, luminometer characteristics and analytical procedure.
Footnotes
Acknowledgements
The authors are grateful to the technicians from Clinical Laboratory of Vall d’Hebron University Hospital.
Data availability statement
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Research involving human subjects complied with all relevant national regulations, institutional policies and is in accordance with the tenets of the Helsinki Declaration (as revised in 2013), and has been approved by the Ethical Committee of the University Hospital Vall d’Hebron (PR [AG]215/2021), Barcelona, Spain.
Guarantor
Not applicable.
Contributorship
Marina Giralt and Roser Ferrer researched literature, conceived the study and obtained ethical approval. Immaculada Comas and Albert Blanco were involved in the recruitment of patients. Noelia Díaz was responsible for data analysis. Marina Giralt wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript. Furthermore, all authors have accepted responsibility for the entire content of this manuscript and approved its submission.
