Abstract
Aim
A digital rectal examination (DRE) during routine assessment for patients with abdominal symptoms provides an opportunity to obtain faeces from the glove for faecal immunochemical testing (FIT). Here, we compared sampling via DRE to the standard faecal sampling by patients.
Method
Patients were recruited to a prospective observational cohort study between July 2019 and March 2020. Patients provided a sample for the FOB Gold Wide® which was compared to a further sample taken at clinic via DRE. Clinicians reported whether they obtained a ‘good’ sample filling all the grooves, a ‘poor’ sample filling some of the grooves or no faecal sample. Cohen’s kappa was used to compare percentage agreement around a negative threshold of <10 μg haemoglobin/g of faeces. Sensitivity for serious bowel disease (SBD) was calculated.
Results
Of 596 patients who underwent attempted DRE sampling, there were 258 (43.3%) ‘good’ samples, 117 (19.6%) ‘poor’ samples and 221 (37.1%) with no sample to wipe in the grooves. Cohen’s kappa dropped from 0.70 to 0.30 for the ‘good’ and ‘poor’ samples, respectively. Of those with DRE samples and definitive diagnostic outcomes, the sensitivity for SBD dropped significantly from 76.0% to 41.7% between ‘good’ and ‘poor’ samples, respectively (p = 0.041).
Conclusions
A ‘good’ sample obtained by DRE provides comparable results to samples obtained by patients. This creates potential benefit in speed and ease of testing for patients. However, not all DRE sampling attempts are successful, and the clinician must be satisfied that enough faeces is obtained to wipe adequately into all grooves.
Introduction
Speed of obtaining a faecal haemoglobin (f-Hb) result could be improved by the use of point-of-care (POC) devices or the use of high-speed laboratory-based devices with online reporting, but this does not solve the initial difficulty of obtaining the faecal sample. Some patients may find collecting a sample for faecal immunochemical testing (FIT) to be challenging. Previously identified reasons for non-compliance in faecal sampling include embarrassment, fear of results, concerns around hygiene and contamination, discretion and privacy, and lack of information. 1 Return rates for symptomatic patients reported by Chapman et al. for a primary care pathway were reported as 91.4%, and 1.3% of samples were unable to be analysed. 2 Furthermore, there is potential for delay as the patient needs to obtain the FIT sample collection device, collect their faeces and deliver it back to the requesting clinician or laboratory. A faecal sample obtained by digital rectal examination (DRE) has the potential to address these issues.
Two studies have reported the diagnostic accuracy of obtaining faeces via DRE and wiping the faeces from the glove onto the FIT sampling stick.3,4 A DRE is recognized as a routine part of the examination of patients with bowel symptoms, so the process of obtaining the sample does not subject the patient to any extra steps. The responsibility is moved from the patient to clinician and can be taken at first consultation. However, obtaining faeces via DRE has not been recommended by the manufacturers of the FIT analysers. Previous studies have compared the diagnostic accuracy of DRE sampling to patient-collected samples with guaiac faecal occult blood testing in screening patients. These have shown differing levels of accuracy and completion rates.5–7 There are no comparative studies of paired samples for FIT.
The aims of this study were to determine the feasibility of using DRE as a sampling method for FIT and to compare the f-Hb results and diagnostic accuracy with the standardized method of sampling. The objective was to obtain paired samples of a FIT faecal sample via DRE in clinic with comparison to a home collected sample that the patient brought to the clinic.
Methods
Design and intervention
A prospective observational cohort study was designed in line with the updated STARD checklist for reporting diagnostic accuracy studies. 8 Recruitment took place between July 2019 and March 2020 from symptomatic patients referred to the Royal Surrey NHS Foundation Trust (RSFT). These patients were invited to the ‘POC FIT’ study (REC: 19/LO/0889) by post and asked to provide two samples from the same bowel motion/faecal sample. One sample was analysed with the POC QuikRead go® (QRG), the other with the laboratory-based FOB Gold Wide® (Sentinel Diagnostics, Italy). The diagnostic accuracy of the QRG and its comparison to laboratory-based testing has previously been reported for this study.9,10 A third sample was obtained via DRE and also analysed with the laboratory-based FOB Gold Wide® to allow direct comparison with the sample collected by the patient.
The postage invitation supplied to the patients contained the specialized collection devices suitable for both the QRG and FOB Gold Wide®. The patient information leaflet explained the technique for home faecal sampling and the plan to obtain a third sample via DRE in clinic. They were asked to bring their samples to their clinic appointment, where written consent was obtained if they agreed to enrol in the observational study.
Inclusion and exclusion
All patients ≥18 years of age referred on the two-week wait (TWW) pathway were eligible. This pathway consisted of patients referred from primary care to colorectal surgery at RSFT with ‘red flag’ bowel symptoms or anaemia according to NG12 guidance. 11 Patients had to have capacity to consent and have provided their samples from fresh faeces and not a stoma bag. Patients who had collected their FIT samples more than 10 days prior to clinic appointment were excluded – based upon sample stability recommendations by the manufacturer.
Specimen collection and analysis
Three colorectal doctors were involved in the recruitment from their clinics and collected faecal samples via DRE from those that consented to the study. Faeces from the glove was wiped directly onto the grooves of the FIT sample stick of the collection device for the FOB Gold Wide®. Clinicians made a judgement on whether any faecal matter was obtained via DRE or not. If none was obtained, a sample was not sent. If a sample was obtained, this was categorized into: ‘good’ sample, filling all the grooves of the sampling stick; or ‘poor’ sample, filling some of the grooves. Patients and clinicians were blinded to f-Hb results in order not to influence decision-making for standard diagnostic tests. All FOB Gold Wide® samples were analysed in the research laboratory based at the Bowel Cancer Screening Southern Hub (BCSH) in Guildford. All laboratory analysis was overseen by a state-registered biomedical scientist. The FOB Gold Wide® at the BCSH is currently used for research purposes and not routine clinical use and therefore is not accredited, however, it has undergone thorough validation. All assays that are routinely used at RSFT bowel cancer screening hub and biochemistry department are UKAS accredited. Further details on FIT methods for this study can be found in the FITTER checklist as supplied in a supplementary file. 12
Result interpretation and statistical analysis
The limit of detection and upper limit of the measurement range for the FOB Gold Wide® is <3 μg of haemoglobin per gram of faeces (μg/g) and >1700 μg/g. For comparison of paired samples, results outside these ranges were excluded, and subsequent non-paired data points were also excluded. The relationship of these pairs was demonstrated by a Bland–Altman plot. Results where f-Hb <10 μg/g was considered a negative result. The proportion of results that showed f-Hb <10 μg/g was compared between the two systems using Chi squared testing. Percentage agreement at this threshold was calculated and assessed with Cohen’s kappa coefficient. 13
Diagnostic accuracy was determined by comparing f-Hb results with eventual definitive diagnostic outcomes through colonoscopy or CT colonography. Patients undergoing flexible sigmoidoscopy were only included if they had presented with perianal symptoms or anorectal bleeding (bright red blood seen separate to the faeces in the pan or on the paper). Sensitivity and specificity for CRC and serious bowel disease (SBD) were determined using the threshold of 10 μg/g. SBD was defined as either a diagnosis of CRC, inflammatory bowel disease or high-risk adenoma (HRA). Patients were defined as having HRAs if they were found to have an advanced adenoma (≥10 mm, any sessile serrated lesion or adenomas that contained high-risk dysplasia), or to have ≥5 polyps, as per the British Society of Gastroenterology guidelines for surveillance. 14 All CRC diagnoses were confirmed by histology reports.
Data
A secure web-based clinical database, associated with RSFT and approved by the local information governance team, was developed to audit colorectal patients on the urgent referral pathway. Recruited participants for the study were assigned a unique trial number and their FIT results were entered into the database in a pseudonymised fashion. Access to this data was restricted to members of the research team.
Results
There were 629 patients consented and included for the POC FIT study. There were 596 patients where DRE sampling was attempted. Figure 1 shows the patient pathway for inclusion and categorization for those that went on for DRE sampling. Patient pathway and comparative results of digital rectal examination with normal patient sampling technique.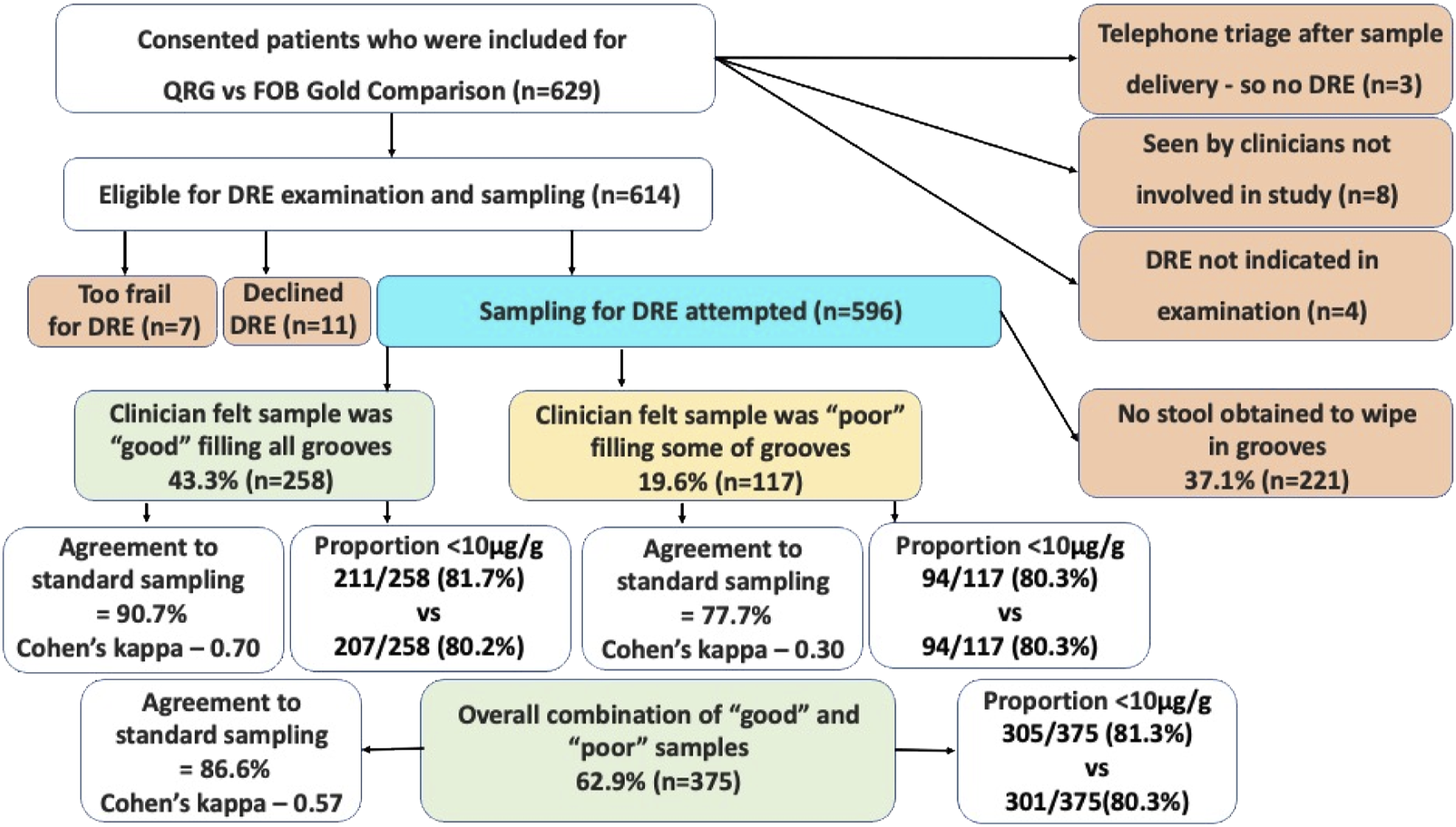
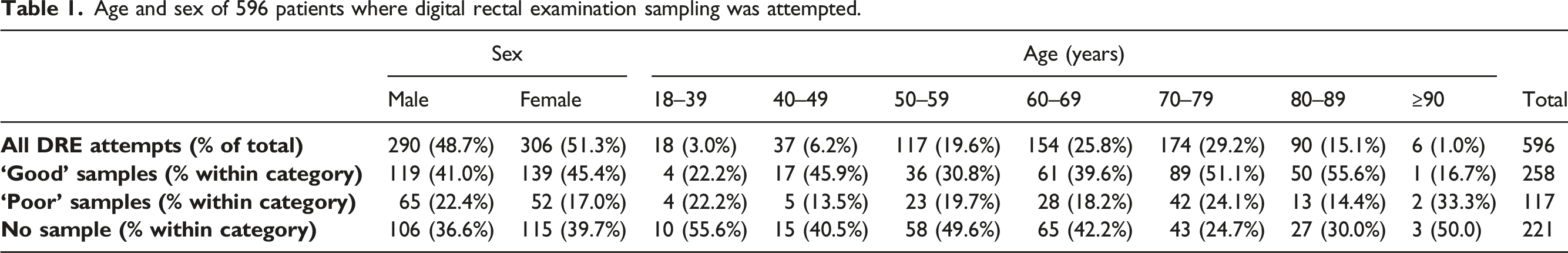
Age and sex of 596 patients where digital rectal examination sampling was attempted.
Of all the DRE samples obtained, the proportion that gave a f-Hb result of <10 μg/g was 81.3% (305/375), this compares to 80.3% (301/375) from the standard sampling method by the patient and the difference was not significant (p = .71). There were 211/258 (81.7%) ‘good’ samples with a f-Hb <10 μg/g vs 207/258 (80.2%) from their paired standard samples which was not statistically different (p = .65). There were 94/117 (80.3%) ‘poor’ samples with a f-Hb <10 μg/g, which was the same proportion as seen from their paired standard samples.
The agreement at a threshold of <10 μg/g for all the paired samples was 86.6%, and Cohen’s kappa coefficient was 0.57, demonstrating moderate agreement. However, the agreement was 90.7% for the ‘good’ samples with a Cohen’s kappa coefficient of 0.70 – demonstrating substantial agreement. The agreement was 77.7% for the ‘poor’ samples with a Cohen’s kappa coefficient of 0.30 for the ‘poor’ samples demonstrating fair agreement.
After exclusion of pairs with a result below the limit of detection and above the upper limit of the measurement range, the f-Hb comparisons are demonstrated in Figure 2 as a Bland–Altman plot. In this graph, there was a total of 69 paired f-Hb samples, these are plotted using the mean of the f-Hb values versus the percentage difference. The bias was −43.1 indicating that the DRE sample tended to give a lower f-Hb concentration. The dotted lines represent the upper and lower limits of agreement (153.0 and −239.3, respectively), and the filled line is the bias. The graph uses a log-scale for the x-axis. Bland–Altman plot to show the relationship between the 69 paired samples for both ‘good’ and ‘poor’ digital rectal examination samples versus patient own sampling.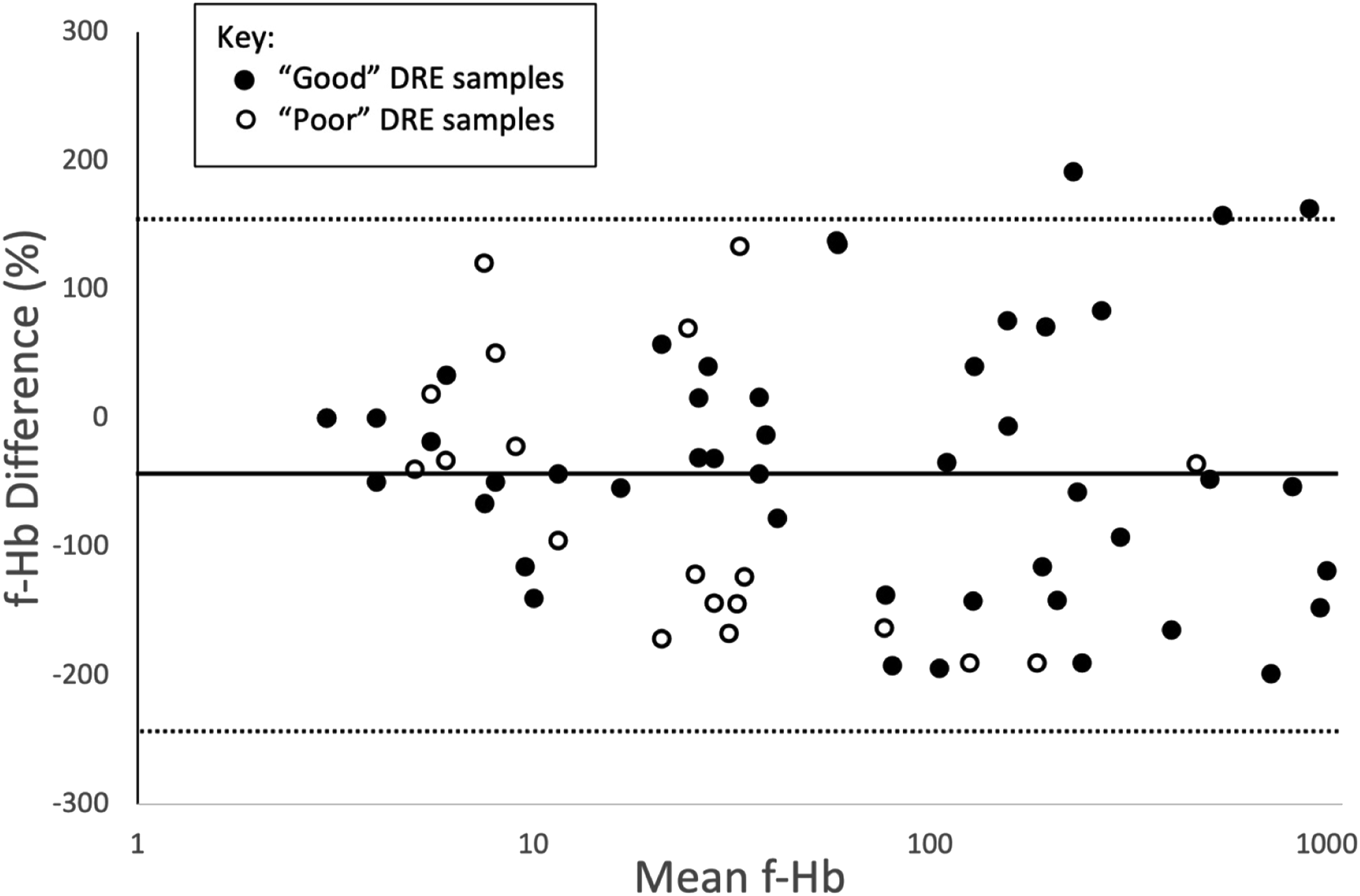
Diagnostic accuracy
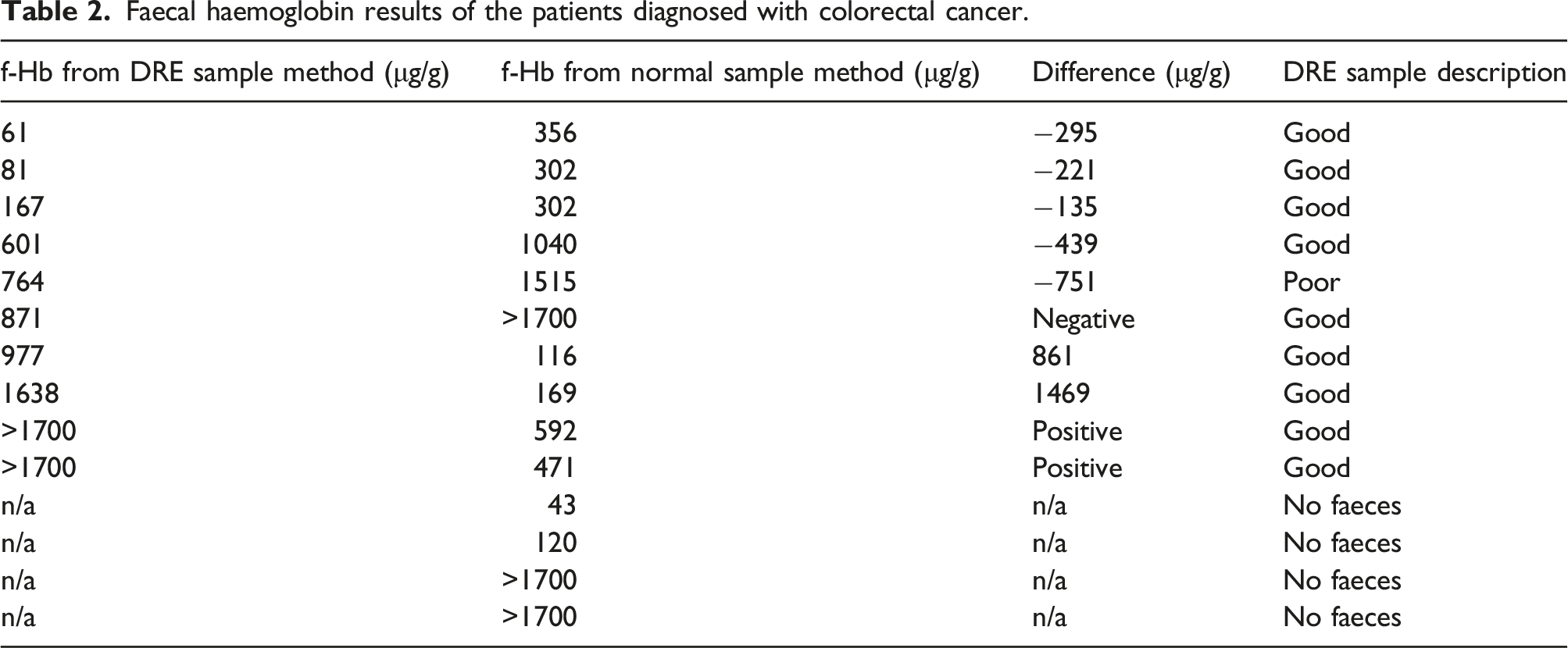
Faecal haemoglobin results of the patients diagnosed with colorectal cancer.
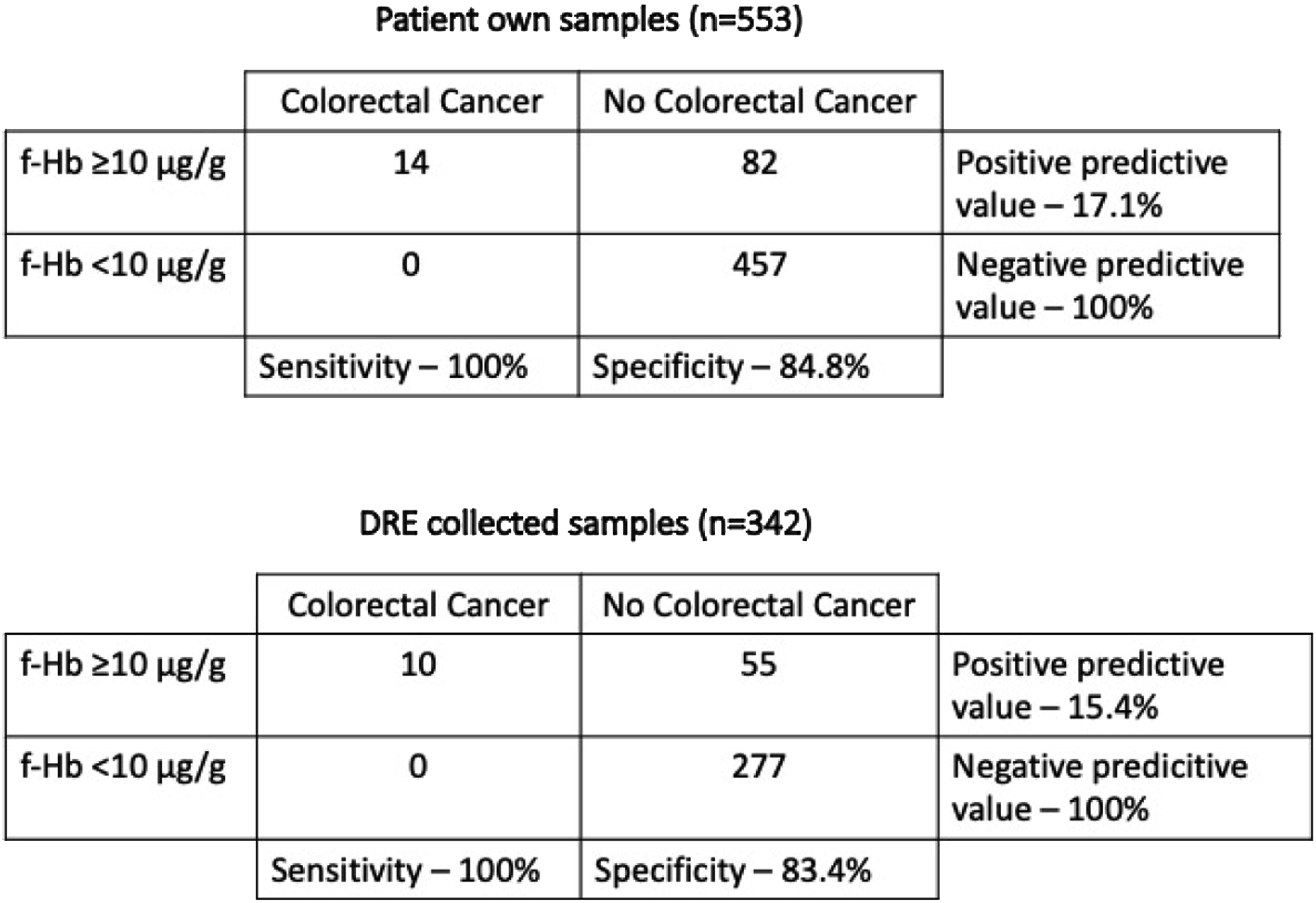
2X2 tables to compare the diagnostic accuracies for colorectal cancer with patient own collected samples versus digital rectal examination collected samples.
There were 52 diagnoses of other SBDs (CRC, inflammatory bowel disease or HRA). For samples taken by the patient, the sensitivity and specificity for SBD were 65.4% and 87.6%, respectively. 37/52 patients had a DRE where a faecal sample was obtained. Comparison in diagnostic accuracy between the two sampling methods for SBD is displayed in Figure 4. Of the 37 patients with a DRE faecal sample, 25 were described as ‘good’ and 12 were described as ‘poor’. Of the ‘good’ samples, 6/25 were <10 μg/g (sensitivity 76.0%) and of the ‘poor’ samples, 7/12 were <10 μg/g (sensitivity – 41.7%). Using Chi squared to test differences in proportions, this difference in sensitivity between ‘good’ and ‘poor’ samples was significant (p = .041). The specificities for the ‘good’ and ‘poor’ samples were 88.5% and 82.3%, respectively (p = .139). 2X2 tables to compare the diagnostic accuracies for serious bowel disease with patient own collected samples versus digital rectal examination collected samples.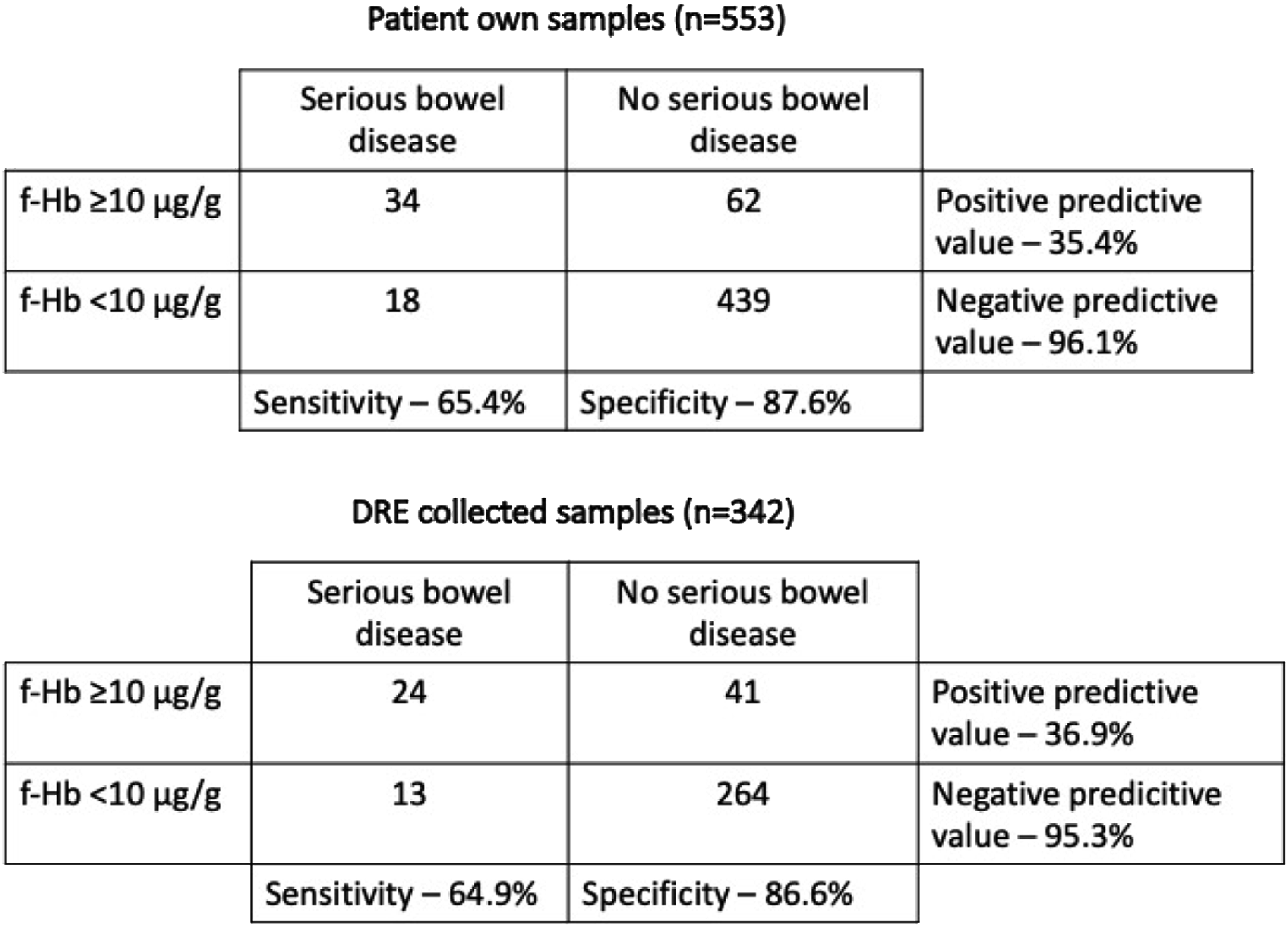
Discussion
This is the first patient comparative study of paired f-Hb results through the different sampling techniques for FIT. When the clinician was satisfied there was a good volume of faeces to wipe in the grooves of the FOB Gold Wide® sample collection device, the results were reliable showing substantial agreement between the sampling methods (Cohen’s kappa 0.70) and high sensitivity for SBD (76.0% from DRE sampling vs 65.4% for standard patient sampling technique). However, if the clinician was not satisfied with the sample, the agreement was mild (Cohen’s kappa 0.30) and the sensitivity for SBD significantly dropped to only 41.7%. In addition, the overall success of obtaining any form of sample was achieved in only 62.9% of cases. In patients with CRC and for whom the clinician had obtained faeces for sampling by DRE, none out of the ten had a false negative result using a threshold of 10 μg/g. However, in these cases, a ‘good’ sample was obtained in all but one of the patients.
For patients referred on the TWW in the United Kingdom, there is a requirement for patients to be seen by a specialist within two weeks from referral and for a diagnosis to be established within 28 days. If DRE were to be combined with POC testing, which has been shown to be comparable in sensitivity to laboratory-based testing, 9 then there is potential for a clinician to obtain a sample and have a result all within an initial consultation. This could better aid decision-making regarding referral or investigation choice. It would also help patient avoid the need to obtain the sample at home and the potential anxiety of waiting for results.
The quick turnaround of the DRE obtained sample can channel a more rapid decision for colonic imaging by endoscopy or CT colonography by avoiding delay in waiting for return of a sample by the patient. The high specificities seen within our cohort via both forms of sampling show that FIT is a reliable means to triage patients. Obtaining f-Hb results quicker and with better compliance through DRE may increase the use of FIT prior to referral and thus impact decision-making to better prioritize those with f-Hb ≥ 10 μg/g. This will help fast-track more at-risk patients, whilst minimising those undergoing unnecessary invasive colonic investigations creating a more cost-effective pathway. However, if the application of FIT was applied to a population with low prevalence of the condition, the positive predictive value of the test would necessarily fall, and greater numbers of patients would be referred on to the TWW pathway with false positive results. This may result in increased patient anxiety and increased resource pressures.
This data on DRE sampling for FIT has shown that faeces collected in this way is comparable to patient own sampling in a symptomatic cohort provided that the clinician feels that they have obtained an adequate sample. As there was a significant drop in sensitivity for SBD when a sample was deemed ‘poor’, we would only recommend processing ‘good’ samples for clinical application. A survey from Boston showed that DRE sampling would be acceptable to the majority of both patients and clinicians, but this was dependent on completion rates. 15 Initially, they found that 54% of care providers would routinely offer DRE sampling for FIT, but this would go up to 88% if they found the results to be comparable to patient collected samples and if the completion rate was 75%. 15 Our study found only 258 of the 596 DRE sampling attempts obtained were ‘good’ and therefore this practice cannot be relied upon for most cases. However, if DRE and POC FIT were to be used at first consultation and a FIT sample collection device given to all patients where a good sample was not obtained, then this would accelerate the pathway for over 40% of patients where immediate decisions could be made at first primary care consultation.
FIT has rapidly taken off for symptomatic patients and the application is not solely for low-risk patients as a rule in investigation as per the NICE guidance from 2017. 16 The results of the NICE FIT study demonstrated the high sensitivity of FIT for high-risk patients in primary care as a rule out investigation, 17 and FIT has been shown to be safe and effective for TWW patients in the coronavirus pandemic to better allocate diminished endoscopy capacity.18,19 Therefore, we suggest that FIT via DRE could also be applied in primary care as a rule out investigation for potential TWW patients. Furthermore, there is interest growing for repeat FIT to further improve sensitivity20–22 – in this situation, DRE sampling may also be the method of choice to obtain the additional test. If a routine referral to secondary care has been made due to a negative FIT from primary care, the specialist may find a second sample via DRE helpful to further determine the need for invasive colonic investigation.
Study limitations
There are different sample collection devices for the different FIT systems and therefore a larger study is required to assess the comparison of results and diagnostic accuracy of all these. The design of the sampling stick for the FOB Gold Wide® and the QRG is similar with circular grooves at the tip to collect the faeces. One of the DRE sampling diagnostic accuracy studies used the HM-JACKarc system. 3 The sampling stick for this has two small dimples and therefore may be more appropriate to wipe the smaller amounts of faeces than the multiple circumferential grooves of the FOB Gold Wide® and QRG.
The evidence presented here is based on DREs performed by only three clinicians and the patient cohort was from a single centre. The ability to obtain faeces from a DRE may be different according to experience from other colorectal clinicians or general practitioners in primary care.
Conclusions
DRE sampling produces FIT results comparable to patient own sampling when the clinician can obtain a ‘good’ sample. This creates potential benefits in terms of speed and ease of testing which could accelerate the patient pathway. However, not all DRE attempts are successful, and the clinician must be sensitive to the possibility of poor sampling and ensure that in such cases patients are offered standard home testing with FIT.
Supplemental Material
Supplemental material - Efficacy and accuracy of faecal sampling by a digital rectal examination for faecal immunochemical testing
Supplemental material for Efficacy and accuracy of faecal sampling by a digital rectal examination for faecal immunochemical testing by William Maclean, Sally C Benton, Martin B Whyte, Timothy Rockall and Iain Jourdan in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
The authors wish to thank the research nurses at RSFT and in particular – Sinead Donlon, Celia Harris, Suzanna Tluk and Humyraa Aziz for their regular contribution in assistance for recruitment and sample delivery.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sentinel Diagnostics provided sampling devices and all reagents for the FOB Gold Wide®. The company was not involved in the design of study, the statistical analyses or the interpretation of data; GUTSfbc is a registered charity (1026791) that awarded a £500 grant towards the study. This money was subsequently not required and returned to the charity.
Ethical approval
Ethics for the project was approved by the London - South East Research Ethics Committee on 28th May 2019 (REC reference: 19/LO/0889, IRAS ID: 260384).
Guarantor
IJ.
Contributorship
SCB, IJ and WM conceived the study. WM was the Principal investigator for the study and primary recruiter. WM wrote drafts of the manuscript. All authors reviewed and edited the manuscript and approved the final version.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
