Abstract
Small-animal fluorescence-enhanced imaging involves the detection of weak fluorescent signals emanating from nanomolar to picomolar concentrations of exogenous or endogenously produced fluorophore concurrent with the rejection of an overwhelmingly large component of backscattered excitation light. The elimination of the back-reflected excitation light of the collected signal remains a major and often unrecognized challenge for further reducing the noise floor and increasing sensitivity of small-animal fluorescence imaging. Herein, we show that the combination of three-cavity interference and holographic super notch filters with appropriate imaging lenses to collimate light improves rejection of excitation light, enabling more accurate imaging. To assess excitation leakage, the “out-of-band (S(Λ x ))” to “in-band (S(Λ m )–S(Λ x ))” signal ratio from phantom studies and the target-to-background ratio (TBR) from in vivo animal imaging was acquired with and without collimating optics. The addition of collimating optics resulted in a 51% to 75% reduction in the ratio of (S(Λ x ))/(S(Λ m )–S(Λ x )) for the phantom studies and an improvement of TBR from 11% to 31% and of signal-to-noise ratio from 11% to 142% for an integrin-targeting conjugate in human glioma xenografts.
Introduction
Fluorescence-enhanced tomography and planar imaging studies for in vivo molecular imaging require deep tissue penetration of light for activation of ultra-low concentrations of fluorophore. However, the collection of accurate fluorescent light measurements requires the separation of weak fluorescent signals from an over-whelmingly large component of scattered excitation light. Consequently, fluorescence-enhanced imaging is limited by the high noise floor that arises due to the excitation light leakage through the band-pass and band-rejection interference filters that have been employed extensively in near-infrared (NIR) fluorescent imaging studies [1–20]. Yet the rejection of excitation light for fluorescence-enhanced imaging in small animals easily rivals the stringent requirements for removal of excitation light in Raman spectroscopy, which employs holographic filters for removal of collimated excitation light. Table 1 provides a listing of reported efforts for in vivo fluorescence imaging in animals [1–4,6–8, 10,12,16,17,19,20] and tissue phantoms [5,9,11,13–15, 18], which shows that few investigators, inclusive of our own group, employ collimation and appropriate filters with sufficient optical densities for collection of weak fluorescent signals. Despite the nonoptimal excitation light rejection, studies nonetheless report fluorescent target depths ranging from 1 to 4 cm with fluorophore concentrations ranging from 100 fmol to 2.5 μmol in phantom studies and 1 pmol to 5.4 μmol of dye per animal within in vivo small-animal imaging studies. The fluorescent imaging of deeper tissues at lower concentrations of fluorophore requires improved effective blockage of excitation light.
In a first attempt to address the problem of excitation light leakage for fluorescence imaging in tissues, Houston et al. [11] reported the efficiency of excitation light rejection using band-pass and holographic notch filters, which retained high fluorescence signals arising from 100 fmol located as deep as 4 cm in tissue mimicking phantoms using time-dependent, frequency-domain photon migration (FDPM) measurements. FDPM measurements refer to the propagation of intensity-modulated light through tissue-like scattering media and the measurements of the re-emitted amplitude, IAC, phase-delay relative to the incident light, δ, and the average intensity, IDC, which is analogous to continuous wave or conventional intensity-based imaging. Although increased depth penetration and lower fluorophore concentrations depend upon improved excitation light rejection in both time-dependent and time-invariant measurements, few investigators recognize that interference filter performance deteriorates as the angle of incident light deviates from zero degrees [21,22]. As detailed in Table 1, most fluorescence imaging and tomography studies do not collimate light for the normal incidence that is required for efficient excitation light rejection. Recently, Chen et al. [15] employed a convex lens to collimate the detected light delivered via a fiber optic prior to its passing through an interference in order to overcome the excitation light leakage. Unfortunately, the assessment of excitation light leakage was not addressed. Herein, we report a study to illustrate the impact of collimation and excitation light leakage upon fluorescence imaging.
Review of Literature for Efficient Collection of Fluorescence Signal in Phantom and Animal Studies

Not given.
In this study, we employ collimating lenses between interference filters in order to align the collected excitation and fluorescent light normal to the filter surfaces for efficient rejection of excitation light. To quantitate filter performance, we compute the ratio of “out-of-band (S(Λ x ))” to “in-band (S(Λ m )–S(Λ x ))” signals registered on a gain-modulated intensified charge-coupled device (ICCD) used to image a tissue-mimicking phantom in the presence of and in the absence of 10 nmol concentration of indocyanine green (ICG). To quantitate image improvement owing to improved excitation light rejection, we imaged the molecular targeting of the integrin αvβ3 receptor with an arginine–glycine–aspartic acid (RGD) peptide–dye conjugate in xenografts of human glioma [23] and assessed improvement in target-to-background ratio (TBR) and signal-to-noise ratio (SNR). Using both phantom and animal imaging, we demonstrate the importance of proper optical design when employing interference filters to reject excitation light.
Materials and Methods
Phantom Studies
The phantom consisted of a 1-L black cubic box filled with 1% Liposyn, a fat emulsion whose scattering properties mimic those of tissue. The 1% solution was prepared by volumetric dilution of 20% stock solution (Abbott Laboratories, North Chicago, IL) using deionized, ultra-filtered water. ICG (Sigma-Aldrich, St. Louis, MO) was initially dissolved in deionized, ultra-filtered water and then added to 1 L of 1% Liposyn to formulate a 0.01 μM solution. Sodium polyaspartate (PASP) (Sigma-Aldrich) was added at 5.2 molar excess of ICG to stabilize the dye from noncovalent interactions [24]. The surface of the phantom was illuminated with excitation light at 785 nm and emission from ICG was collected at 830 nm.
Animal Studies
For the in vivo imaging studies, 6- to 8-week-old female athymic nude mice (nu/nu: 18–22 g) were purchased from Harlan Sprague Dawley (Indianapolis, IN). Animals were maintained in a specific pathogen-free mouse colony in the Department of Veterinary Medicine (The University of Texas M.D. Anderson Cancer Center). The animal protocol was approved by the American Association for Laboratory Animal Care and all experiments were conducted in accordance with guidelines of the Institutional Animal Care and Use Committee.
1 × 106 cells of human glioma U87 were implanted subcutaneously into the thigh region of mice. Tumor mass was allowed to grow approximately 1–5 mm diameter in size. IRDye™800 (LI-COR, Biotechnology, NE, USA) bound to RGD peptide [23] was injected into the tail vein of an anesthetized mouse at a dose of 20 nmol per animal. The RGD peptide is known to target the integrin αvβ3 receptor, which is overexpressed in the U87 tumor model. IRDye™800 has similar excitation and emission spectra as ICG and was excited at 785 nm and its fluorescence was collected at 830 nm. Twenty-four hours following the intravenous administration of peptide-conjugated dye, the mice were anesthetized by 1.5–2.0 vol.% of isoflurane (Isoflo, Abbott Laboratories) using mechanical ventilation and imaged using an ICCD camera.
Instrumentation
FDPM measurements of the phantom and tumor-bearing mice were conducted using the homodyned ICCD camera system, which has been described elsewhere [13,25]. The CCD camera (series AT200, model SI512B, Photometrics, Tucson, AZ) is comprised of a 512 × 512–array of potential wells (pixels) that convert incident photons into electrons. The size of a pixel is 24 × 24 μm and the active imaging area of a CCD chip is 12.3 × 12.3 mm. FDPM consists of employing an incident, intensity-modulated light excitation source that creates a “photon density wave” that propagates continuously throughout the tissue to produce a fluorescent “photon density wave” that propagates to the surface for collection by a gain-modulated ICCD imaging system. Figure 1 illustrates the detected intensity (solid line) at one point in the image field in response to source intensity (dotted line). The measurable quantities in typical frequency-domain data are the phase shift, δ, the intensity amplitude, IAC, and average value of intensity, IDC, which is analogous to conventional intensity-based imaging. The modulated excitation light was expanded over the region of interest (ROI) using an optical diffuser [26] and a convex lens to illuminate the phantom surface uniformly. The area of illumination was 64 cm2 and field of view (FOV) was 16 cm2 for phantom studies. In animal studies, the FOV was 30.25 cm2.
Analysis of FDPM Measurements
FDPM images of IAC, δ, and IDC were gathered using homodyning procedures and computer acquisition programs (PMIS Image processing Software, Photometries for phantom studies and V++ Digital Imaging System Software, Digital Optics, Auckland, New Zealand for animal studies) as previously described [14]. Five binned images (128 × 128 pixels) were acquired each with 0.8 sec of integration time as a function of phase delay between the image intensifier and the laser diode operated in homodyne mode. From the average of the phase-sensitive images acquired by the CCD camera, both IAC and δ were computed using Fast Fourier Transform (FFT) analysis of zero-averaged data. For IDC measurements, a dark noise obtained without laser excitation was subtracted from the images.
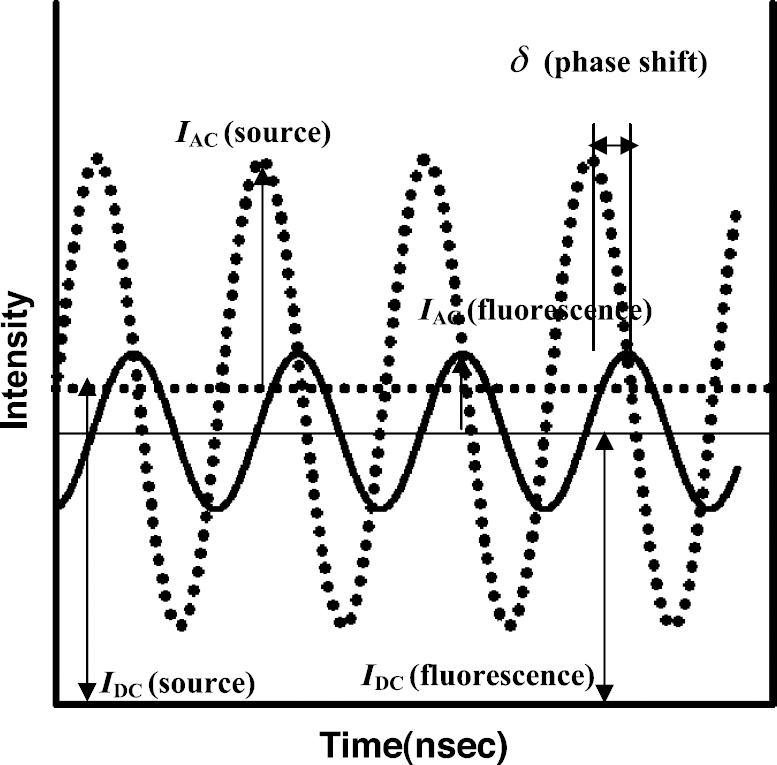
Schematic of FDPM measurements used in NIR optical spectroscopy and tomography. (From Hawrysz DJ, Sevick-Muraca EM, Neoplasia, 2(5), 388, 2000; with permission)
Selection of Optical Filters and Collimating Optics
The schematics of the optical measurements employed for investigating the improvement of excitation light rejection by collimation are illustrated in Figure 2. In order to reject off-axis light with angle of incidence greater than 27° on to the interference filters, 1-in. lens tubes (Thorlabs, Newton, NJ) housed the combination of lenses and filters between the Nikon focusing lens and the photocathode of the image intensifier. A three-cavity 830-nm band-pass filter with 10.33 nm full width at half maximum (830FS10-25, Andover, Salem, NH: optical density [OD] = 5.5 at 785 nm) and a holographic super notch filter (HSPF785.0–1.0, Kaiser Optical System, Ann Arbor, MI: OD > 6.0 at 785 nm) were employed to provide direct rejection of the 785–nm excitation light and to selectively pass emission light. As shown in Figure 3, the performance of interference filters deteriorates as the angle of incidence deviates from zero. In order to direct collimated, normal incident light on the interference filters, two collimating lenses (NIR Achromatic doublets; focal length, f′ = 40 mm; 1 in. of diameter; Thorlabs) were positioned based upon a ray tracing calculation (Optics Lab Optical Ray Tracing Software, Science Lab software, Carlsbad, CA) which modeled the doublet lens as thin lens. Because the diameter of the doublet lenses was greater than their thickness, this is an appropriate assumption [27]. The inserts to Figure 2(A) and (B) provide ray tracings from a single point on the image plane formed from the Nikon lens in order to illustrate the impact of collimation. Typically, as denoted in the insert in Figure 2(A), there are three classifications of light directed into the first lens (7): (a) incident parallel light rays; (b) light rays passing through the center of lens; and (c) light rays passing through the focal point of first lens. All light incident upon the lens can be expected to be bounded by the light ray tracings (a), (b), and (c) as illustrated in the insert to Figure 2(A). Initially consider the incident parallel ray (a), which arrives at zero incident angle on the holographic filter (8). Because it is at zero incident angle, excitation light within this ray will be blocked and will not travel further for detection. Fluorescent light will pass. Excitation light in ray (b) may pass through holographic filter (8) and interference filter (6) owing to its nonzero angle of incidence, but will be blocked from detection owing to collimation by lenses (7) and (5) and its zero angle of incidence on holographic filter (4). Finally, excitation light in ray (c) may pass through holographic filter (6) but will be collimated by lens (7) to have zero angle incidence on interference filter (6). Emission light, but no excitation light, in ray (c) will be detected. The distance between object and image plane of Nikon focusing lens is based upon Equation 1:

The insert to Figure 2(B) shows uncollimated image delivery to the interference filters in which only excitation light in parallel ray (a) is blocked from detection and excitation light in all other rays may pass through the filters in varying amounts, dependent upon their incident angle.
Experimental Design and Analysis
The image of the surface of the tissue phantom in the presence and absence of uniformly distributed ICG provided a method to assess excitation light leakage. Excitation light leakage is defined as the signal, S(Λ
x
), or average pixel intensity values associated with the image of the scattering surface taken in the absence of ICG in the solution while using band-pass and holographic filters. The fluorescence signal, S(Λ
m
), is likewise averaged from the pixel intensity values associated with the image taken in the presence of ICG in the solution. Measurement parameters such as the intensifier gain were held constant for S(Λ
x
) and S(Λ
m
). The S(Λ
x
) signal represents “out-of-band” transmission signals, whereas the difference, S(Λ
m
)–S(Λ
x
), represents the “in-band” transmission signal. The transmission ratio of the filter and lens combination, R, is given by:


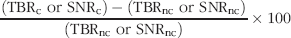

Experimental setup employed for the improvement of excitation light leakage (A) with collimating optics and (B) without collimating optics. Numbered components include: (1) CCD camera, (2) laser diode, (3) image intensifier, (4) holographic filter, (5) collimating lens, (6) band-pass filter, (7) collimating lens, and (8) holographic filter. θ represents angle of incidence on the filter plane.

(A) Optical density of 830 nm interference filter as a function of incident angle (from Andover with permission). (B) Optical density of Kaiser holographic filter as a function of incident angle (reproduced from Handbook of Vibrational Spectroscopy [22] with permission).
Results
Phantom Studies
The transmission ratios of average IDC and IAC images acquired with (solid bars) and without (nonshaded bars) collimating optics are shown in Figure 4(A) and (B) as a function of distance, r, from the center of the 18-mm diameter photocathode. In all cases, a reduction of 51–75% of transmission ratio, R(IDC) and R(IAC), occurred when collimating optics were employed. Additionally, for uncollimated images, a departure of the incident angle from normal increases the transmission ratios, R(IDC) and R(IAC), with increasing distance from the center of the image intensifier as shown in the insert of Figure 2(B). Without collimation, the largest off-axis incident angle of light occurred at the edge of image intensifier which corresponds to the radius 7.73 mm on the photocathode. Significant differences at a confidence level of .05 occurred between the mean transmission ratio computed from collimated and uncollimated light as predicted by the hypothesis t test [28]. Consistent with our previous reports [11], measurements of IAC have a reduced out-of-band transmission in comparison to IDC, owing to the natural frequency filtering of ambient, out-of-band light.
Images Collected from Molecularly Targeted Agents in Animals
To translate the implication of our phantom results for in vivo imaging, we extended experiments of excitation light leakage to animal imaging. Figure 5(A–D) shows the posterior view of a xenograft bearing a 5-mm diameter glioma (U87) provided by IAC and IDC collected with (A and C) and without (B and D) collimation optics, respectively. Images in Figure 5(A–D) are displayed as the raw data with individual color scales. The incident excitation illumination remained unchanged between collimated and uncollimated imaging measurements.

Transmission ratio of DC intensity (R(IDC)) (A) and AC amplitude (R(IAC)) (B) computed from filter combination (holographic–band-pass–holographic filters) as a function of r, the distance from the center of photocathode Black and white bars indicate out-of-band to in-band ratio of imaging system with and without collimating optics, respectively. Error bars represent standard deviations of intensity with ROI.
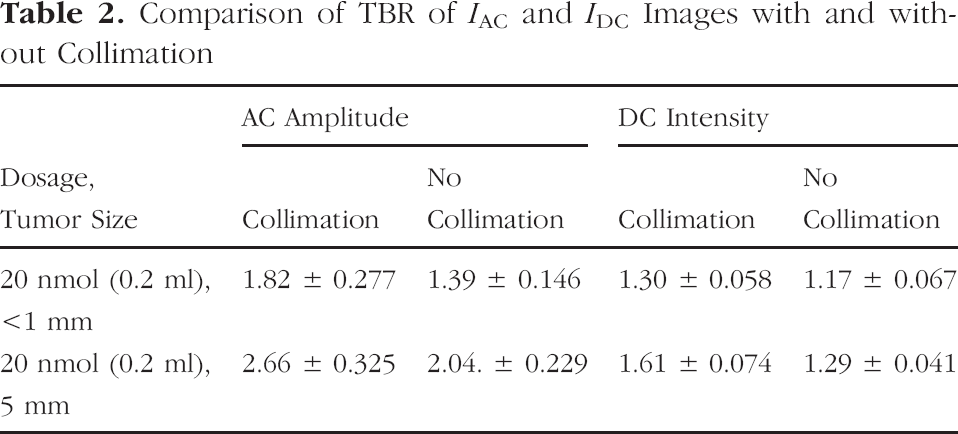
Tables 2 and 3 list the TBR and SNR for two animals bearing a <1 mm and 5 mm diameter glioma imaged with both IAC and IDC 24 hr after administration of 20 nmol of the RGD–IRDye800. TBR obtained from the images acquired with a collimating lens improved from 11% to 31% over that of uncollimated images. The SNR of IAC and IDC from collimated images increased from 11% to 142% over that derived from the corresponding uncollimated images.
Discussion
Molecular imaging using conjugated fluorescent agents requires registration of a weak fluorescent signal within the efficient rejection of an overwhelming large excitation light signal. In small-animal fluorescence optical imaging, the requirements for excitation light rejection exceed or rival that of Raman spectroscopy. Improved imaging sensitivity depends upon better rejection of excitation light in order to reduce the noise floor for evaluating deeper and smaller amounts of dye. Although molecular imaging using fluorescent agents is currently restricted to small animals and shallow penetration depths, when translated into the clinic, the issues of depth of penetration and amount of agent injected will crucially depend upon improved excitation light rejection. Reduction of the noise floor should be accomplished both through (i) the judicious choice of a fluorophore with a large Stoke's shift (as done herein with the choice of IRDye800 or ICG), (ii) selective illumination of the tissue with narrow spectral sources close to the excitation of the fluorophore, (iii) proper selection of combinational optical filter sets with OD > 6 at Λ x , and (iv) by collimating the image incident upon interference filters. OD at the excitation wavelength describes the attenuation characteristics (i.e., a filter with OD = 3 transmits 0.001 times the amount of the normally incident excitation light). Few investigators (including in the past, ourselves) focus upon the importance of collimated excitation light rejection in their studies and few employ filters with OD > 6. Although we have demonstrated the improvement of filter sets and light collimation using FDPM measurements through measurement of out-of-band to in-band transmission, TBR and SNR, the retrofitting of conventional optical cameras with collimating lenses before the placement of interference filters can have similar impact on intensity-based images.
We have presented our TBR and SNR measurements primarily for IAC which requires no background subtraction because only the modulated light is registered in the measurement. Because both the excitation and generated emission light is modulated, excitation light leakage is as problematic for FDPM as it is for conventional intensity based imaging. Although one could conceivably create “difference” images prior to and following administration of molecularly targeting fluorescent agents, the problems of co-registration of images especially after long times after administration (such as imaging 24 hr following administration of agent as shown herein) and the propagation of measurement error limits the sensitivity of a “differencing” approach. Thus, excitation light rejection is paramount for molecular imaging with fluorescence. For example, consider the signal generated from a femtomolar dose of fluorescent agent whose signal is collected with an interference filter of OD = 3. The collected signal levels at the emission wavelength can be 6 orders of magnitude less than the reflected and scattered excitation light. In this case, the signal acquired before administration (comprised mainly of nonrejected excitation light) and after the administration of the agent (whose signal is may still be predominantly comprised of nonrejected excitation light but with a small fluorescent component) differs by a small signal level, which can be within standard error of the measured intensity values.

IAC image of an animal bearing 5-mm diameter glioma lesion (A) with and (B) without collimating optics. IDC image of an animal bearing 5-mm diameter glioma lesion (C) with and (D) without collimating optics. Color bars represent raw signals for each image.
Comparison of TBR of IAC and IDC Images with and without Collimation
Intensity measurements or IDC images have the added complication of ambient and dark noise counts added into the measurements which require backsubtraction, whereas FDPM measurements eliminate the need for backsubtraction as only the modulated signal is measured [11]. Consistent with our past work, Figure 4 shows that transmission ratio is higher for IDC than for IAC images. Nonetheless, upon proper filtering and through the use of collimation optics, improvement occurs in both IDC and IAC images of the phantom surface.
In addition to assessing “out-of-band” signal in a controlled phantom study, we translated these results into improvements in in vivo small-animal planar imaging. Although the performance of emerging, molecularly targeting optical agents is typically assessed using CCD-based imaging of the values of TBR and SNR, Tables 2 and 3, and Figure 5 show that these figures of merit may be a function of excitation light rejection capabilities as well as the performance of the targeting agent. From visual comparison of Figure 5(A) with Figure 5(B) and Figure 5(C) with Figure 5(D), one can see that collimation reduces the background in the tissue contralateral to the tumor region. In small-animal fluorescent imaging, excitation light leakage through interference filters can therefore reduce TBRs which are used as the figure of merit for reporting the specificity of targeting fluorescent conjugates. As shown in the study by Kwon et al. [20], the dose-dependent response of integrin-targeting agent reported in terms of TBR did not correlate with the dose-dependent rate of conjugate uptake obtained from dynamical imaging measurements, which are less impacted by excitation light leakage than TBR. Table 2 also shows that collimation increases the TBR of IAC and IDC images and TBR of IAC images are greater than those of IDC images regardless of collimation. These in vivo imaging results confirm the conclusions for target phantom work which show that IAC images may be more sensitive than IDC images owing to reduced ambient photon noise [11].
Comparison of SNR of Averaged IAC and IDC Images with and without Collimation

Table 3 shows that the SNR of collimated IAC and IDC images were greater than those of uncollimated IAC and IDC images for all animals due to reduced background signal. The larger background signal in the uncollimated image causes high out-of-band to in-band ratio in phantom studies and a low TBR in animal studies when compared with the images made with collimated light. The contrast (defined as TBR-1) for the collimated images was approximately 1.6 to 2.1 times that demonstrated in the uncollimated images, suggesting that serious consideration of excitation light rejection capabilities be taken into account when assessing the specificity of fluorescent, targeting agents.
Footnotes
Acknowledgments
This work was supported in part by the National Institutes of Health (R01 CA67176 [EMS] and R01 EB000174 [CL]).
