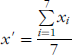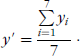Abstract
Micro-computed tomographic (micro-CT) imaging provides a unique opportunity to capture 3-D architectural information in bone samples. In this study of pathological joint changes in a rat model of adjuvant-induced arthritis (AA), quantitative analysis of bone volume and roughness were performed by micro-CT imaging and compared with histopathology methods and paw swelling measurement. Micro-CT imaging of excised rat hind paws (n = 10) stored in formalin consisted of approximately 600 30-μm slices acquired on a 512 × 512 image matrix with isotropic resolution. Following imaging, the joints were scored from H&E stained sections for cartilage/bone erosion, pannus development, inflammation, and synovial hyperplasia. From micro-CT images, quantitative analysis of absolute bone volumes and bone roughness was performed. Bone erosion in the rat AA model is substantial, leading to a significant decline in tarsal volume (27%). The result of the custom bone roughness measurement indicated a 55% increase in surface roughness. Histological and paw volume analyses also demonstrated severe arthritic disease as compared to controls. Statistical analyses indicate correlations among bone volume, roughness, histology, and paw volume. These data demonstrate that the destructive progression of disease in a rat AA model can be quantified using 3-D micro-CT image analysis, which allows assessment of arthritic disease status and efficacy of experimental therapeutic agents.
Introduction
Rheumatoid arthritis (RA) is a debilitating, chronic inflammatory disease characterized by synovitis and formation of destructive pannus tissue, causing progressive cartilage erosion, bone remodeling and destruction, pain, and loss of joint function [1,2]. Although the etiology of the disease remains unclear, the clinical presentation and disease progression are known. In short, the synovial tissue in the peripheral joints becomes inflamed and hypercellular. This hyperplastic synovial tissue or pannus becomes locally invasive, resulting in cartilage and bone destruction, which leads to severe pain and loss of joint function and integrity [1,2]. Following the diagnosis of RA, the current American College of Rheumatology (ACR) guidelines of disease management recommend radiography of involved joints to establish a baseline for monitoring disease progression and treatment response [3]. Radiographs, although inexpensive and established, are ultimately limited by the two-dimensional (2-D) representation and the inability to image synovitis [4]. A recent prospective study demonstrated the limited ability of conventional radiography to detect erosive bone lesions in finger and wrist joints in arthritic patients [5]. Additionally, a recent review by Ory [6] highlighted plain film radiography deficiencies that include feature obscurity or enhancement due to improper positioning and scoring variations among radiologists. Such limitations would also plague preclinical animal research, within which drug treatments are being developed to treat RA.
Advances in preclinical medical imaging aim to provide detailed, noninvasive anatomical and pathophysiological metrics that accurately monitor disease progression and treatment efficacy [7]. In this context, it was deemed important to perform micro-computed tomography (micro-CT) to evaluate its potential in monitoring bone changes in small rodent models of RA and develop semi-automated algorithms to quantify the disease status. In that regard, advanced imaging methods and analysis may provide additional preclinical data to assist the advancement of an experimental drug. Ultimately, imaging methods may contribute to the goal of providing a more personalized standard of clinical care based on the disease presentation of the individual patient.
In this work, quantitative approaches are presented to objectively analyze bone degradation. These methods supplement the collection of other imaging markers that have been developed to monitor arthritic (and osteoporotic) disease status using medical imaging. These markers include a range of architectural parameters [8–12] as well as joint space distance and bone mineral density [13–15]. Presented in this article are volumetric and roughness analyses from micro-CT images that were used to characterize the pathology of adjuvant-induced arthritis (AA) in rats. The AA rat model presents bone and cartilage changes similar to RA, within a shorter disease progression time frame compatible with pharmaceutical research [16,17]. From high-resolution micro-CT images of excised rat ankles, bone volumes were calculated from the bones of the ankle tarsus (talus, cuneiforms, navicular, and cuboideum) excluding the calcaneous. Volumetric analysis was compared to histopathological evaluation and to paw volume measurement, which is a common preclinical method of staging disease progression. A new approach to quantify the increase in bone surface roughness is included, and its value to micro-CT imaging of arthritic bone changes is demonstrated.
Materials and Methods
Animal Preparation
Two-month-old, female Lewis rats (Charles River, Kingston, NY) were housed according to Millennium Pharmaceuticals' IACUC standards. The animals (n = 10) were randomized into two groups of five animals each. In the AA group, arthritis was induced on Day 0 by an intradermal injection in the pad of right hind foot with 0.1 mL Complete Freund's Adjuvant containing 3 mg/mL Mycobacterium tuberculosis (Difco Laboratories, Lee Labs, Grayson, GA). Control rats were injected with an equal amount of mineral oil only. Paw volume, a marker of swelling, was measured using a plethysmometer (Stoelting, Physiology Research Instruments Division, Wood Dale, IL), and the onset of arthritis was indicated by increased paw volume, which appeared approximately 10–12 days postinjection. Both paw volume and body weight were measured throughout the study. Twenty-one days after adjuvant injection, the rats were euthanized with CO2, and the right hind paw was amputated and fixed in 10% neutral-buffered formalin. Micro-CT imaging was performed prior to histology.
Micro-CT Imaging
Micro-CT imaging was performed on a Scanco μCT-20 (Scanco Medical, Zurich, Switzerland). The excised hind paws were secured in 15-mm imaging tubes and immersed in formalin. Approximately 600 30-μm-thick slices were acquired on a 512 × 512 image matrix with isotropic, nominal (digital) resolution (30 × 30 × 30 μm) using 110 msec exposure time (50 kVp). Other scanner details include: Microfocus fan-beam X-ray with 7-μm spot size; 1024 element, 25 μm pitch detector; 8 W (160 μA); 580 projections; back projection reconstruction algorithm with convolution and correction for fan beam. Imaging time was approximately 11 hr per sample.
Histopathology
Following micro-CT imaging, the hind paws were decalcified in Immunocal (American MasterTech Scientific, Lodi, CA) for 3 days (solution change each day). After decalcification, paws were embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E). Histopathological evaluation of the severity of joint inflammation was scored using four parameters: cartilage/bone erosion, pannus development, inflammation, and synovial hyperplasia. Each histopathological parameter was scored from 0 to 4, yielding a maximal value of 16 for each joint, with higher scores indicating more severe disease.
Data Analysis
Bone volume analysis. Reconstructed micro-CT data were imported into the software package Analyze 4.0 (BIR, Mayo Clinic; AnalyzeDirect). Background and soft tissue were segmented from the bone in the images using histogram analysis. The threshold was set as the midpoint between background and bone regions of the histogram. Volumetric analysis was performed on the tarsus bones: talus, lateral cuneiform, medial cuneiform, intermediate cuneiform, navicular, and cuboideum (excluding the calcaneous). The bones were segmented as individual objects using a region-growing algorithm with six-point, 3-D connectivity. The number of voxels in each object (i.e., bone) was counted and translated into a volume, as described in Equation 1.
where the cubed constant is the 30-μm isotropic resolution factor, Vi is a voxel in the object of interest, and N is the total number of voxels in the object.
Bone roughness analysis, the Boundary Index (BI). For this analysis, the talus bone was segmented from the surrounding ankle bones and exported as a separate image file. The bone–background interface was determined using fixed threshold analysis, identical to the method described for the volumetric measurements. Bone objects with an area less than 1 mm were removed. The BI is then calculated, on a slice-by-slice basis, as the ratio of a “smooth” estimate of the object (slice) perimeter to the actual (true) perimeter. A sample object is shown diagrammatically in Figure 1 to illustrate the meaning of the BI and the difference between the actual and the smooth object perimeter. The edge of the object was defined as pixels with an adjacent background neighbor. The perimeter, P, was quantified (slice-by-slice) as the sum of the edge pixel distances around the boundary of the object as represented in Equation 2:
when i = N, xi+1 = x1 and yi+1 = y1,
where pixel (x1, y1) is a random edge point (i.e., the seed point) and pixel (xi+1, yi+1) is the clockwise, adjacent edge pixel, and N is the number of edge pixels in the object.
To determine the smooth perimeter (slice-by-slice), the perimeter was redefined as the average pixel locations in seven-pixel increments. Briefly, the average position of seven adjacent perimeter pixels was calculated, and the pixel coordinates of position were stored—(x′,y′) in Equations 3a and 3b:

The boundary index, BI, is the coefficient of variation (CV) of the ratios of the smooth perimeter to the actual perimeter from all slices in the image. This diagram illustrates the parameters obtained from a single slice. The perimeter of the object is shown in the dark gray boxes. Starting at a seed point (denoted by), the program moves clockwise to determine the average perimeter location of a seven-pixel segment. The average location in this example object is shown by the dark-bordered box. The smooth perimeter, shown by the line, is calculated from the distance separating the average perimeter pixels.
The program then moved (clockwise about the object) to the next segment of seven-perimeter pixels, and again the average position, (x′,y′), was recorded. This process was repeated until the entire perimeter was traversed. In most cases, the perimeter was not an integer number of seven-pixel increments; therefore, the last average perimeter coordinate was calculated as the average position of the final segment, regardless of the size. The array of average edge pixel coordinates was then analyzed to quantify the smooth perimeter, SP, which was defined using the Euclidean distance measure shown above:
where pixel (x′1, y′1) is an average edge pixel used as the seed point for the calculation, pixel (x′oi+1, y′i+1) is the next average edge pixel (clockwise), and M is the number of average edge pixels in the object.
Finally, the ratio of the smooth perimeter to the perimeter was calculated for each object. A smooth surface (e.g., a sphere) has a ratio near 1, due to the similarity between the actual perimeter and the seven-pixel smoothed perimeter. This was confirmed with imaging phantoms (data not shown). Also with phantoms, we verified that rough surfaces have a ratio less than 1 due to the deviation of the smooth perimeter from the actual perimeter. We hypothesized that the smooth surface of normal bone may have a ratio somewhat less than 1 due to the complexity of the surface. In comparison, a diseased, eroded bone would have a significant decrease in the ratio. On a slice-by-slice basis, the ratio of the smooth perimeter to the actual perimeter was computed. The BI was defined as follows:
as the coefficient of variation (CV) of the set defined as the ratio of the smooth perimeter (SP, Equation 4) to perimeter (P, Equation 2) of all n slices. Given the complexity of bone shape, the BI parameter was measured independently from three orthogonal directions (i.e., x, y, and z), and the final index is the average of the BI measured from these orientations. This will minimize any directional anisotropy resulting from bone objects not being digitally oriented in the same position.
Statistical Tests
All data are presented as mean and standard error of the mean (σM). Statistical significance testing was performed on all analysis parameters using t tests, except histology scores which used a Mann–Whitney test, and a p value #x003C; .05 was considered significant. The micro-CT analysis was performed blindly and independent of histopathology, and comparisons were made at the end of the experiment.
Results
Figure 2 shows representative 3-D-rendered micro-CT images from the perspective of the dorsal aspect of the foot of control and AA rat ankles. The bone erosion is obvious in the image of the diseased ankle as compared to the image of the normal ankle. Micro-CT images taken from representative control and arthritic animals are shown in Figure 3B and Figure 3D, respectively. Corresponding (maximally overlapping) representative histological sections of control and arthritic animals are shown in Figure 3A and Figure 3C, respectively. Both histology and micro-CT slices are in the sagittal plane, and the micro-CT images were chosen to show the talonavicular joint shown in the histology, albeit at different scales. The arrows indicate the relative orientations of the images. The normal histology section (Figure 3A) shows intact joint spaces, normal cartilage and bone morphology, and normal cellularity. In contrast, the section from the arthritic animal (Figure 3C) illustrates the severe joint damage seen in this disease model, characterized by synovial hyperplasia and inflammation, as well as significant pannus development and cartilage/bone destruction. In Figure 3D, the integrity of the bones is compromised by significant erosion and pannus infiltration. Although micro-CT is insensitive to the pannus (and other soft tissue), the bone destruction resulting from the pannus infiltration can easily be visualized. The micro-CT signatures show void spaces and surface roughness which translate into a lower bone volume index and a higher bone roughness (BI) index.

3-D-rendered micro-CT images from the perspective of the dorsal aspect of the foot of normal (left) and AA (right) rat joint. The normal joint has smooth bone surface with intact joint architecture, whereas the joint from the AA rat presents with significant bone erosion and compromised joint integrity.
The panels in Figure 4 profile the quantitative results of the bone volume measurement (Figure 4A), histopathological evaluation (Figure 4B), BI (Figure 4C), and paw volume (Figure 4D). Figure 4A plots the absolute change tarsal bone volume between control and arthritic animal groups. The arthritic bone significantly declined by 27% (p = .004) as compared to sham. All histopathology signatures, shown in Figure 4B, were significantly different between the control and AA rat groups. Specifically, the total histopathology scores changed from 1 (±0) and 14 (±1) for control and arthritic joints, respectively (p = .008). In Figure 4C, the BI is graphed for the control and arthritic talus samples. The roughness of the bone, defined by the BI (arbitrary units), was 0.040 (±0.002) for normal talus bones and 0.062 (±0.006) for arthritic talus bones—an increase of 55% in the arthritic samples (p = .01). Finally, Figure 4D shows the plethysmometer-measured volume of the control and arthritic rat paws were 1.4 (±0.05) and 3.2 (±0.3), respectively—a 129% increase (p #x003C; .001).
Discussion
This study demonstrates the success of region-specific bone volume and roughness analysis in an animal study of an adjuvant-induced RA model. Existing analysis methods and biomarkers include a range of structural parameters that probe bone and tissue [8–12]. Compared to other micro-CT analysis techniques—such as trabecular thickness and connectivity measurements—the bone volume measurement may be performed at lower resolution, allowing for faster scans and higher throughput—optimal for therapeutic evaluation. Specifically, even digitally interpolating data from 30 to 90 μm isotropic resolution resulted in comparable absolute bone volumes (data not shown). Recently, another preclinical application demonstrated the use of volumetric measurements of ossified regions of the menisci in a guinea pig model of osteoarthritis, suggesting that volumetric analysis is unambiguous and relevant for the evaluation of therapeutics [18].

Representative H&E stained, histological sections (A and C) and micro-CT image slices (B and D) in the tarsal of the animals shown in Figure 2. The green arrows indicate the relative orientation of the images. (A) An H&E stained section from a control animal in this study. The presentation of the joint space, synovial lining/fluid, cartilage, and bone is normal with no signs of inflammation, hyperplasia, or erosion. (B) A representative 30-μm-thick micro-CT image slice from the normal animal shown in panel (A). This image shows the intact architecture of a normal joint and smooth bone surfaces. (C) An H&E stained section from a diseased animal in this study. The section from the AA rat demonstrates the severe presentation of established RA. The joint space is significantly compromised by inflammation and hyperplasia of the synovium; further, the cartilage and bone are severely eroded and infiltrated by the pannus. (D) A representative 30-μm-thick micro-CT image slice from the diseased animal shown in panel (C). This image shows drastic bone erosion, which results in the significant reduction in bone volume and increase in roughness.
The tarsal bones were chosen for analysis in this study because they were undisturbed in the joint removal process and demonstrate maximal sensitivity to disease-related changes. It is interesting to note that the talus bone presented with less, albeit significant, bone erosion as compared to the overall volume loss (14% vs. 27%, respectively). Bone erosion was more pronounced in the mid-foot region, as indicated by the 31% reduction in bone volume. Ultimately, the combined volume of the tarsal and talus bones may be best for pharmaceutical research given the high sensitivity and low overall variability. Further, despite the high (30 μm) resolution of the scan, the analysis is macroscopic in comparison to the histopathology—providing a view of the entire sample and the simultaneous option of region-specific measurements unavailable in whole sample global metrics such as paw volume.
In this particular study, one sample in the disease group did not demonstrate significant bone loss in the micro-CT scans. The histological analysis supported this finding with positive (abnormal) inflammation and hyperplasia scores despite the absence of pannus development or cartilage/bone erosion. This animal may have had reduced or delayed disease development, which is supported not only by histology and CT, but also by the paw swelling data for this animal. In general, the histopathology scores and paw volume (swelling) measurements were consistent; although, to establish good quantitative correlations, interrogation over a variety of disease states and larger group sizes would be needed.

(A) Absolute changes in tarsal bone volumes in control and arthritic animal groups. (B) Histopathological scoring for cartilage/bone erosion, pannus development, inflammation, synovial hyperplasia, and total score. (C) The roughness estimate from the Boundary Index, BI, for the control and arthritic talus bones. (D) The plethysmometer-measured volume of the control and arthritic rat paws.
The rough, eroded bone surface, which conventional radiography has limited ability to quantify [4,5], is a hallmark of arthritic disease [1,2]. The boundary index, BI, was devised to quantify this issue. The BI method may be more sensitive to subtle changes in the bone surface than the global bone volume analysis. On the talus, chosen for its consistent disease presentation, the change in bone surface roughness was larger than the change observed for talus bone volume measurements (55% vs. 14%, respectively). In our current analysis, the BI parameter is a global index of an isolated bone; however, this method can be adapted to include additional anatomical regions. Further, development is ongoing for a second-generation 3-D roughness method that will map focal regions of erosion.
The intrinsic 2-D nature of BI computation confers some orientational sensitivity to the index. In our calculations, we have seen the BI to change by relatively large amounts (55% reported here to 34% on the same data analyzed from a single direction), although statistical significance was unchanged. It is important to note that the 55% difference observed here is an average from three orthogonal planes, thus minimizing the directional anisotropy of the index.
Computation of surface roughness is complicated by the need to separate topography (form) from local roughness. The BI captures this aspect by using the ratio of true to smoothed perimeter and using an optimized ruler size that maximizes the difference between the BI measurement of normal and arthritic talus bones. In this context, it should be recognized that small ruler sizes (in this case, <5 pixels) would oversample the perimeter, thus minimizing the definition of rough; while large ruler sizes (#x003E;13 pixels), would undersample the perimeter and would misclassify a normal change in form as roughness. In this study, the choice of 7 is optimal; however, it is not the only justifiable one. In our testing, BI analysis with rulers in the range of 5–11 pixels yields similar results. Other studies may require another selection of ruler size because it will depend on anatomy, resolution, object size, type of damage, and so forth. For instance, a change in the resolution would require two important considerations. First, it is likely that a change in ruler size would be required due to a change in relative voxel size. Second, it also is important to note that the resolution change could reduce the sensitivity of roughness due to the larger spatial dimensions. However, insofar as rat talus bones are concerned, one set of optimal parameters (as presented here) is sufficient to use as a platform for rapid analysis if all other experimental conditions are kept uniform across studies. Hence, once defined for particular bone(s) and imaging paradigm, the BI estimate of roughness is expected to be stable and robust across studies.
For rapid therapeutic evaluation, quantitative indices are necessary for expression of efficacy in terms of response rates. The quantification of bone erosion seen in these models can be used for analytical studies in drug discovery, providing a rapid and semi-automated environment for optimization of lead compounds. Absence of such indices requires semi-quantitative, subjective scoring paradigms, which are sometimes variable and inevitably slower to implement. The volumetric measures shown here are relatively easy to obtain and demonstrate the bony destruction that would be expected in the advanced stages of erosive RA. Although the scan times appear prohibitive to high-throughput or in vivo research, this issue is solved by later generation scanners capable of achieving images of comparable resolution and quality in a fraction of the time. Bone volumetric or roughness measurements can be further coupled with other pathological estimates, such as bone mass, mineralization, and porosity measurements, which have already shown value in preclinical pharmaceutical research [7,13–15,18–20]. The addition of any complementary information regarding the therapeutic effects of a test compound could dramatically improve the laborious drug discovery process [21,22]. Capabilities to extend such visualization to in vivo studies would provide spatial and temporal correlates of specific functional benefits obtained from therapeutic interventions or target modulation experiments.
Footnotes
Acknowledgments
We thank the valued contributions of Dr. Steven Carr and Craig Muir.