Abstract
Micro-computed tomography (micro-CT) is becoming an accepted research tool for the noninvasive examination of laboratory animals such as mice and rats, but to date, in vivo scanning has largely been limited to the evaluation of skeletal tissues. We use a commercially available micro-CT device to perform respiratory gated in vivo acquisitions suitable for thoracic imaging. The instrument is described, along with the scan protocol and animal preparation techniques. Preliminary results confirm that lung tumors as small as 1 mm in diameter are visible in vivo with these methods. Radiation dose was evaluated using several approaches, and was found to be approximately 0.15 Gy for this respiratory-gated micro-CT imaging protocol. The combination of high-resolution CT imaging and respiratory-gated acquisitions appears well-suited to serial in vivo scanning.
Introduction
Micro-computed tomography (micro-CT) refers to computed tomography that is conducted on a nearly microscopic level comparable to that achievable on a clinical CT system in human subjects [1,2]. Micro-CT is currently widely used to evaluate bone specimens noninvasively [3–10], and has been applied to static vascular and lung imaging [11–15]. In vivo lung tumor studies using micro-CT have produced images in which lung nodules are detectable [16]. However, image quality is generally greatly improved by scanning the mice after sacrifice [2,14].
Cone-beam micro-CT is the acquisition of a complete volume of CT data in a single revolution of the X-ray tube—detector assembly, and is different from the fan-beam systems currently used in clinical CT scanners. Kinney et al. [4] utilized micro-CT to examine trabecular bone architecture in a rat osteoporosis model in vivo. This study used a high-resolution CT instrument called an X-ray tomographic microscope, which demonstrated a limiting resolution of 8 μm [17]. The measured histomorphometry values of the rat trabecular bone based on the X-ray tomographic microscope were in good agreement with data from conventional methods obtained on the same specimens. This system rotates the object of interest, and would complicate the evaluation of ventilated live animals. Another recently published in vivo cone-beam micro-CT study monitored cancellous bone alterations in hindlimb-unloaded female rats [8]. Images using this micro-CT prototype system had voxel dimensions of 20 × 20 × 26 μm and successfully detected 3-D trabecular microarchitecture changes in vivo. This prototype system does not rotate the animal subject, and was specifically designed for imaging small laboratory animals in vivo.
Micro-CT scanners typically provide excellent images of tissues with high natural contrast, such as bone and lung. High detail in vivo images of the thoracic region of animal models could be useful in many research fields, and particularly in studies of lung cancer treatment effects. Respiratory-gated micro-CT could potentially reduce breathing motion and motion-related artifacts. More importantly, respiratory gating would also provide the means to image the lung during full inflation, resulting in an increase in lung tissue image contrast, and thus a marked improvement in image quality.
Radiation exposure is a potential concern for in vivo micro-CT scanning, particularly if serial scanning is desired. We used several approaches to evaluate radiation dose for in vivo micro-CT acquisitions. Animal preparation and handling techniques are also critical to the success of respiratory-gated micro-CT scanning. Typical respiratory gated micro-CT images of normal laboratory animals (mice and rats) are presented as well as preliminary correlation of gated micro-CT images and lung histology.
Materials and Methods
Micro-CT Scanner
The micro-CT system described in this article (Figure 1) is manufactured by General Electric Medical Systems (model RS-9, London, Ontario). The micro-CT scanner X-ray tube has a fixed tungsten anode with a nominal focal spot size of 50 × 30 μm. This X-ray source operates at 35–80 kVp and 100–500 μA, and includes 1.8 mm of aluminum equivalent filtration. The detector is a charge coupled device with an active imaging area of 112 × 56 mm and an element size of 27 μm. Isotropic voxels are produced using a modified Feldkamp [18] reconstruction process and are 91 × 91 × 91 μm in the in vivo scan mode. The reconstructed 3-D image matrix can be viewed in any plane (axial, sagittal, coronal, oblique) without loss of detail (Figure 2). The standard scan acquisition size is typically 6 cm × 6 cm × 4 cm.

General Electric Medical Systems micro-CT that was utilized in all studies.
The scan parameters that are consistent for both gated and nongated in vivo scan acquisitions include: 80 kVp, 450 μA, 100 msec per frame, and calibration images (bright and dark fields). The differences in the protocol parameters are listed in Table 1. The dark-field correction is obtained in order to imitate maximum mass attenuation, and no X-rays are produced. The bright-field correction measures the maximum absorption on the detector and uses 450 μA with 80 kVp in order to imitate minimal mass attenuation where the X-rays are fully detected. These two calibration images are used to normalize the detector element readings for any drift that may occur, or to identify an element not functioning properly, and scales the acquired detector data accordingly. The bright- and dark-field images are obtained after scanning of the object is complete and require approximately 2 min.
Laboratory animals that have been deeply anesthetized typically develop a specific pattern of respiration in which gasping breaths are separated by relatively long time periods. This generally results in a nearly motion artifact free resulting image (Figure 3A). However, because image collection occurs at near full expiration, lung tissue contrast is quite compromised and the lungs appear gray due to the relative lack of air presence. When the same mouse was ventilated to control breathing, motion artifacts are more apparent, but the improvement in lung tissue contrast more than compensates for the motion blurring which is generally limited to the diaphragm region (Figure 3B).
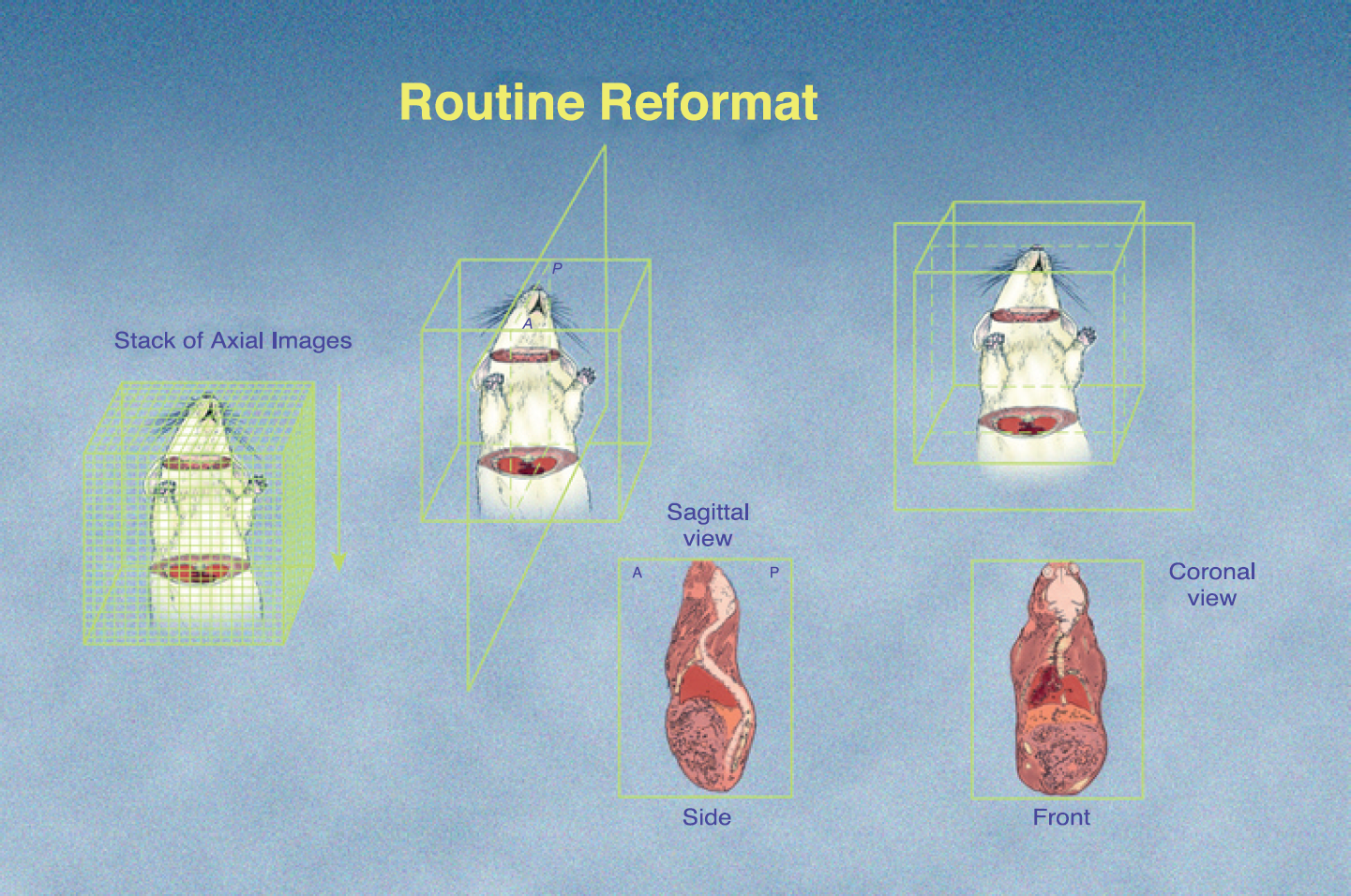
The cone-beam micro-CT reconstruction process produces a 3-D matrix of isotropic voxels. This 3-D dataset can be sampled from any orientation without loss of detail. Coronal views in particular appear well-suited to the presentation of thoracic images.
The Gated Protocol is Used for Thoracic Scanning In Vivo
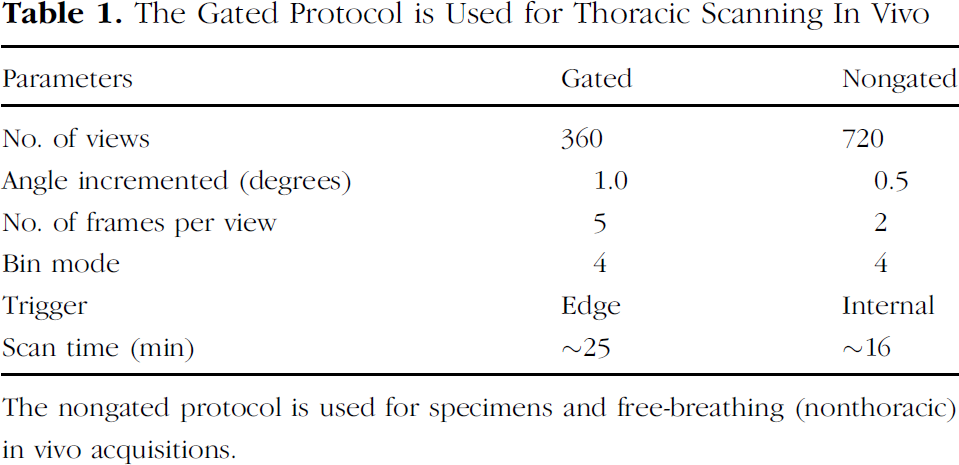
The nongated protocol is used for specimens and free-breathing (nonthoracic) in vivo acquisitions.
Radiation Dose
We assessed radiation dose associated with this micro-CT scanner to determine radiation risk. We positioned a calibrated CT ion chamber (Capintec, Ramsey, NJ) in air at the isocenter of the scanner bore opening, and the radiation signal detected was integrated over the entire scan time in the low-resolution in vivo nongated scanning mode. We used these results to predict the radiation dose for the respiratory-gated acquisition case, because they were different only for X-ray flux, expressed in mA sec [mA sec = X-ray tube current × Beam on time in seconds] and can be easily scaled. We also used skin dose monitors (SDMs) (McMahon Medical SDM, San Diego, CA) to estimate the entrance exposure both in air at isocenter, and on the external surface of an expired mouse.

(A) Axial micro-CT view of a free-breathing 129SV mouse. (B) Axial micro-CT view of the same mouse as in part A, at the same anatomic location with the same gray level settings, obtained with respiratory gating Note that although lung tissue contrast is improved with a gated acquisition, motion artifact is not completely eliminated (blur at liver edge). Acquiring the CT image data at full inspiration improves the lung tissue contrast and the visualization of blood vessels.
Animals
Initial in vivo micro-CT scan protocols were developed using untreated mice (C57Bl/6) and rats (Fischer 344). Properly preparing the animals for the micro-CT scan is essential to reducing image artifacts. We initially anesthetized each animal with 4–5% isoflurane gas followed by an injectable anesthetic. The subcutaneous injection consisted of a combination of: 10% Dormitor (Pfizer Animal Health, New York, NY), and 10% atropine sulfate (American Pharmaceutical Partners, Los Angeles, California). The injectable anesthetic further relaxed the animal and facilitated intubation. We then intubated the animal using an intravenous catheter as an endotracheal tube over a fiber-optic laser attached to an illuminator that allows direct visualization of the oropharyngeal opening for intubation (BioTex, Houston, TX). We use a standard 20-gauge, 2.5-cm-long intravenous catheter as an endotracheal tube for a standard 25–30 g mouse (Figure 4A), and a standard 14-gauge intravenous catheter for a typical 180–200 g rat. After endotracheal intubation, we placed the animal in an acrylic half pipe and immobilized it in a supine position. We connected the endotracheal tube to a SAR-830/P small animal ventilator (CWE, Ardmore, PA) and maintained breathing on 1–2% isoflurane gas (Figure 4B). We typically set the breathing rate at 100 breaths per minute for mice, and 75–80 breaths per minute for rats. We scan live animals with the radiation scatter guard in the open position (Figure 4B), and chest motion was monitored using a flashlight during the scan session by trained veterinary technicians. To date, over 500 mice have been imaged utilizing this in vivo gated micro-CT protocol.
Gated Micro-CT Acquisition—Normal Animals
For respiratory-gated in vivo scanning, the micro-CT X-ray shutter motion is initiated by the ventilator. This ventilator produces an electronic trigger to initiate micro-CT data collection just after the maximum inhalation phase of the respiration cycle (Figure 5). Imaging at full inspiration takes advantage of the natural contrast available in the lung by expanding the lung volume. The X-ray tube collimator open time is 100 msec (one frame of signal data) during maximum inhalation of the mouse's breathing cycle. Because signal acquisition occurs during the entire range of lung displacement, breathing motion is not eliminated.

(A) Mouse endotracheal intubation for respiratory-gated in vivo micro-CT scanning. (B) Intubated mouse that has been connected to the ventilator and positioned in the micro-CT scanner for respiratory-gated in vivo micro-CT image acquisition.

Graph depicting the breathing cycle of the mouse and the triggering of the micro-CT acquisition. The micro-CT acquires signals for 100 msec and the average breath cycle time is 600 msec.
An initial scan session was conducted to determine the optimal ventilation pressure to use for the mouse C57Bl/6 strain in vivo. The ventilator measures air pressure applied to the lungs in units of centimeters of water pressure; 12, 14, 16, and 18 cm of water pressure were used during micro-CT scanning of four different mice. Each image set was evaluated independently by two chest radiologists to determine optimal image quality.
Two normal F-344 rats weighing around 185 g were scanned with respiratory-gated micro-CT to establish that our animal preparation and scan procedures resulted in acceptable image quality, and to begin to collect information regarding normal patterns of micro-CT rodent respiratory structure.
Mouse Lung Tumor Image Correlation
Dr. Tyler Jacks (Massachusetts Institute of Technology) developed a mouse model (129SV) carrying targeted mutations in the k-ras gene [19]. The k-ras gene is an oncogene, and this mutation causes the development of multifocal lung adenocarcinomas in 100% of these mice. Lung tumors are generally first detected microscopically at 2 weeks of age, and tumor number and size increase continuously until they essentially fill the thoracic cavity (mean survival time is 300 days). The growth rates and total number of tumors are variable in this mouse model, which makes noninvasive micro-CT imaging advantageous to studies that employ this model. After micro-CT imaging, each mouse was kept in the positioning sled and euthanized with CO2, then flash frozen with liquid nitrogen. Each mouse was placed into a −20°C freezer for a minimum of 24 hr to ensure complete freezing. The mouse was removed from the sled and placed into a wooden block filled with colored water and returned to the freezer. Once the water was frozen, the entire block was sliced using a wood planer. Slices of ice/tissue (125–500 μm) through the entire lung field were removed and the block face was imaged with a digital camera. Preliminary validation studies were carried out in which the block face images of the lungs were visually inspected for lung tumors and then directly compared to the corresponding micro-CT images in the same plane to confirm the presence of tumors observed in the micro-CT images of the lungs.

Coronal views from respiratory-gated in vivo micro-CT acquisitions from four different untreated C57Bl/6 mice. The number in the upper left-hand corner of each image indicates the airway pressure used during the image acquisition. The images obtained with 12, 14, and 16 cm of water pressure, respectively, revealed minimal distention of the bronchi. The image set obtained with 18 cm of water pressure showed good contrast and lung volume, but also revealed distended bronchi. The image set obtained at 16 cm of water pressure had the best tissue contrast, without distention of the bronchi.

Respiratory-gated in vivo micro-CT coronal images of two normal rats highlight the detail that is visible. (A) This image illustrates damage in the right apex of the lung. This damage is believed to be a result of local hemorrhage that was caused by the intubation process. (B) This illustrates a coronal slice exhibiting normal anatomy.
Results
Radiation Exposure Measurements
The results of several repeated measurements were consistent and indicated that the radiation dose delivered to the ion chamber in air is about 0.12 Gy, 0.137 Gy for the SDM in air and 0.104 Gy for the SDM on the skin of an expired mouse when used in the in vivo (nongated) mode. The average radiation dose for the nongated micro-CT acquisition for the three approaches used was 0.12 Gy. For a respiratory-gated acquisition, the dose would increase by approximately 25% due to a slight increase in overall mA·sec, which would result in a radiation dose of 0.15 Gy for a single respiratory-gated micro-CT acquisition. The dose threshold in mice for radiation-induced lung damage is about 10–15 Gy [20–22], depending upon the mouse strain irradiated. Histological studies have found that for a single radiation dose of 20 Gy or less to the thorax, little change in the lungs occurs within 3 months after irradiation [23]. To produce a single whole-body dose of 20 Gy would require 133 sequential respiratory-gated micro-CT scans. Paulus et al. [2] also measured micro-CT radiation dose and found that it was equivalent to only 5% of the LD50/30 dose.
Gated Micro-CT Acquisition—Normal Mice
The resulting images from the ventilation pressure respiratory-gated micro-CT study are shown in Figure 6. Eighteen centimeters of water pressure caused distention of the air passages in this C57Bl/6 mouse. Twelve centimeters of water pressure eliminated airway distention but did not provide sufficient lung expansion or contrast. Sixteen centimeters of water pressure was selected for general use after visual assessment by two radiologists because it resulted in a compromise between lung volume and airway expansion.
Gated Micro-CT Acquisition—Normal Rat
The image in Figure 7A illustrates damage in the right apex of the lung. This damage is believed to be a result of local hemorrhage that was caused by the intubation process. In Figure 7B, a coronal slice exhibiting normal anatomy demonstrates that excellent image quality can be obtained by respiratory-gated micro-CT of rats.

Registered axial histopathologic block-face section and respiratory-gated in vivo micro-CT image of a normal (control) mouse. No grossly visible or radiographic evidence of lung tumors. H = heart, T = trachea, V = thoracic vertebra, and S = spinal cord. The scale on the left is a 1-cm scale divided into 1-mm increments.
Mouse Lung Tumor Image Correlation
Normal mouse organs and structures were readily observed for micro-CT images and corresponding gross histology sections (Figure 8). Adenocarcinoma lesions visible in micro-CT images were also successfully correlated to gross histology sections as shown in Figure 9. Lung tumors as small as 1 mm in diameter can be appreciated in these images. Although tumor boundaries were not always well defined, they tended to be visually discernible from nearby vascular structures when additional views (axial/coronal/sagittal) were displayed.
Discussion
Undertaking in vivo respiratory-gated micro-CT imaging requires the cooperation of a skilled multidiscipliary team that includes imaging physics expertise, sophisticated veterinary support, and experienced animal researchers. The animals must be properly anesthetized and immobilized to ensure no movement during image acquisition. Respiratory gating of the micro-CT acquisition also requires endotracheal intubation and ventilation of the animals. The position of the animals in the micro-CT must allow visual access to the chest to confirm proper ventilation during the imaging procedure.

Registered axial block-face section image and axial respiratory-gated in vivo micro-CT image of mouse. Lung tumors are outlined in blue and the lung field is outlined in green. Tumor location and size are well matched for this mouse between these different images. G = gall bladder, E = esophagus, L = liver, and S = spinal cord.
Gated micro-CT acquisition does not eliminate respiratory motion artifact. By keeping integration time to a minimum and acquiring image data at the same point in the respiratory cycle, blurring due to the mouse's breathing cycle is minimized. Respiratory motion causes some image blur especially near the diaphragm. If the exposure time (currently 100 msec) was further compressed, respiratory motion could be reduced. This approach would require a longer scan time to maintain current image quality. Other acquisition parameters (number of frames and views) could potentially be adjusted to balance a shorter trigger period. A different scanner design capable of faster data acquisition would be an alternative approach to reducing respiratory motion blur.
We found that a ventilator setting of 16 cm of water pressure was optimal for the respiratory gated for in vivo thoracic imaging for mice. This level of air pressure provided a good compromise between lung volume, lung contrast, and airway expansion. Further studies are currently being conducted to compare the advantages of using a constant peak volume of air versus the constant pressure protocol for respiratory-gated scanning of mice in vivo.
Images acquired of normal rats in vivo with respiratory-gated micro-CT revealed detailed lung anatomy. Many blood vessels and lung structures were readily visualized. Future studies involving micro-CT scanning of rat models are being investigated.
The correlation of respiratory-gated micro-CT images and histopathology for a k-ras mouse model of lung tumors is proceeding. Our frozen block-faced imaging method appears well suited to this task. As in all sectioning methods, however, this technique is limited to acquiring one view of the block-faced images (axial/coronal/sagittal) per animal even though any arbitrary view can be generated from each animal from the 3-D micro-CT dataset. It also destroys all tissues in the process, which does not allow for cell typing of tumors.
We evaluated radiation exposure delivered by this system when operated in nongating (free-breathing) mode, and predicted the exposure that would result from applying the respiratory-gated acquisition mode. These preliminary estimates indicate that radiation damage is unlikely to occur due to in vivo micro-CT imaging unless very frequent scan sessions are desired, and for quite an extended period of time. We currently recommend that repeated in vivo gated micro-CT scans be performed no more frequently than at weekly intervals unless a very short-term study is desired.
Image contrast in respiratory-gated micro-CT images could be further improved if intravenous contrast could be used, in a manner similar to a clinical CT study. Unfortunately, current contrast materials are cleared from the bloodstream of laboratory animals well before the 20-min acquisition is complete. Longer-lasting contrast materials are currently under development for this application. The relatively small total blood volume of a typical laboratory mouse (5–6% of a mouse's body weight) renders the use of intravenous contrast somewhat risky for in vivo scanning if animal subject recovery is important.
Soft tissue detail with this imaging device has been unimpressive to date (note the lack of liver structure in Figure 9). The utilization of interperitoneal iodinated contrast material could potentially improve soft tissue imaging in micro-CT by coating the surfaces of the abdominal organs and allowing their margins to be more readily visualized. Optimal doses and timing of bolus injections would have to be carefully examined prior to implementation of this approach.
Respiratory-gated micro-CT acquisition appears to be a promising method for the noninvasive observation of certain kinds of lung pathology in vivo with acceptable radiation risk.
Footnotes
Acknowledgments
The authors thank Belinda Rivera, BS, RVT, for her skilled efforts and invaluable assistance in the development of this procedure. The authors also would like to acknowledge the technical assistance provided by Brooke Walters, BS.
This work was supported in part by grants from the National Cancer Institute (CA-17772, CA-64193, and CA-98013).
