Abstract
Background: Adenoviral gene transfer has been shown to be effective in cardiac myocytes in vitro and in vivo. A major limitation of myocardial gene therapy is the extracardiac transgene expression. Methods: To minimize extracardiac gene expression, we have constructed a tissue-specific promoter for cardiac gene transfer, namely, the 250-bp fragment of the myosin light chain-2v (MLC-2v) gene, which is known to be expressed in a tissue-specific manner in ventricular myocardium followed by a luciferase (luc) reporter gene (Ad.4 × MLC250.Luc). Rat cardiomyocytes, liver and kidney cells were infected with Ad.4 × MLC.Luc or control vectors. For in vivo testing, Ad.4 × MLC250.Luc was injected into the myocardium or in the liver of rats. Kinetics of promoter activity were monitored over 8 days using a cooled CCD camera. Results: In vitro: By infecting hepatic versus cardiomyocyte cells, we found that the promoter specificity ratio (luc activity in cardiomyocytes per liver cells) was 20.4 versus 0.9 (Ad.4 × MLC250.Luc vs. Ad.CMV). In vivo: Ad.4 × MLC250.Luc significantly reduced luc activity in liver (38.4-fold), lung (16.1-fold), and kidney (21.8-fold) versus Ad.CMV (
Introduction
Myocardial gene transfer is a promising tool to target specific abnormalities of the failing heart. Using adenoviral vectors, we recently demonstrated that over-expression of sarcoplasmic reticulum Ca(2+)-ATPase (SERCA2a) not only improves contractility [1,2] but also survival [3] in animals with heart failure. However, a major limitation of adenoviral in vivo gene transfer into the myocardium is the expression of transgenes in other organs due to the systemic distribution of virus. Adenoviral constructs show a native tropism for hepatic tissue, but are also found in the lung, thymus, brain, and kidney, regardless of the viral delivery technique [4,5]. This undesired transgene expression may exhibit potential side effects such as inflammation and cell apoptosis in noncardiac tissues, raising important safety concerns [6,7]. A variety of strategies has evolved to overcome these obstacles. Modifying the viral construct itself turned out to be crucial for increasing organ specificity. Once the virus is in the cell nucleus, the actual expression of the transgene is maintained by its promoter, since these viral regulatory elements are responsible for starting transcription of the delivered transgene. Controlling gene expression with a promoter specific to cardiac tissue could avoid transgene activation in other tissues than the heart. Several different cardiospecific promoters have been investigated so far, including α-myosin heavy chain (α-MHC), atrial natriuretic peptide (ANP), and myosin light chain-2v (MLC-2v) promoters [8,9]. In tissue culture experiments, the myosin light chain (MLC-2v) promoter promises high tissue specificity [10]. We hypothesize that alterations in the 250 base pair (250-bp) fragment of the promoter construct may improve the tissue specificity to a point that in vivo cardioselective gene transfer becomes feasible. The present study investigates whether an adenoviral vector driven by an enhanced cardiospecific promoter construct can result in selective transgene expression in rat hearts without significant expression in other organs. To test whether different gene delivery techniques affect promoter activity and specificity, viral constructs are evaluated using localized and systemic myocardial delivery techniques.
Studies of cardiac gene transfer usually rely on postmortem analysis, thus making an assessment of the temporal dynamics of transgene distribution extremely difficult. Optical imaging of myocardial transgene expression in live animals is a relatively new technique, and its application to evaluate temporal and spatial characteristics of cardiac gene expression in the same animal offers significant advantages [11,12]. In this experiment, we use a cooled charged couple detector (CCD) camera to detect and quantify the dynamic course of gene expression rate efficacy and organ specificity of a new cardiospecific promoter fused with a firefly luciferase reporter gene.
Materials and Methods
Promoter Construction
Generation of Adenovirus
Adenoviruses have been generated as previously described [13]. Briefly, the different plasmids were recombined in
Gene Transfer In Vitro
LLC-PK1 cells (kindly provided by Dr. Joseph Bonventre, Department of Medicine, Massachusetts General Hospital) were maintained in culture at 95% air, 5% CO2 at 37°C in Dulbecco's modified Eagle's medium containing 10% fetal bovine serum. HepG2 cells and COS cells were cultured according to standard protocols. Cardiomyocytes were prepared from 1- to 2-day-old rats by use of the Neonatal Cardiomyocyte Isolation System (Worthington Biochemical, New Jersey), plated in six-well plates at 5 × 105 cells/well, and cultured in RPMI 1640/5% FCS/10% horse serum (HS). After 72 hr, cells were incubated with or without adenoviral vector for 48 hr at a multiplicity of infection (MOI) of 10 pfu/cell. Cells were harvested, washed with PBS, and were processed for luciferase assay according to the protocol (Promega, Wisconsin).
Gene Transfer In Vivo
All animals received humane care in compliance with the “Guide for the Care and Use of Laboratory Animals” published by the National Institutes of Health (NIH Publication No. 85-23, revised 1985).
Optical Imaging of Transgene Expression
The Xenogen In Vivo Imaging System (IVIS) consists of a cooled CCD camera mounted on a light-tight specimen chamber, a cryogenic refrigeration unit, a camera controller, and a computer system for data analysis. Both the IVIS and its imaging analysis software are commercially available (Xenogen, New Jersey). Transgene bioluminescence was imaged in vivo as previously described [11,14]. After intraperitoneal injection of reporter substrate
Luciferase Assay and Evaluation of Gene Expression Rate Efficiency
After sacrifice, organs (heart, liver, lung, kidney) were dissected and immediately frozen in liquid nitrogen. Samples were weighed, homogenized in lysis buffer (1% (v/v) Triton X-100, 10% glycerol, 25 mM Trisphosphate pH 7.8, 2 mM DTT) and centrifuged. The supernatant was used to perform luciferase assays as reported elsewhere [15].
Luciferase Activity Given in RLU/mg

Five groups of 105 cells were used for each experiment.
Statistics
Comparisons between groups were carried out using a two-tailed Student's
Results
Gene Transfer In Vitro
To evaluate cell-type specific expression and relative promoter-strength in vitro, rat neonatal cardiomyocytes, HepG2 cells, kidney cell line LLC-PK1, and COS cells were infected with the viral constructs Ad.4 × MLC250.Luc, Ad.CMV.Luc, Ad.MLC2v.Luc, and Ad.Luc, respectively, at a multiplicity of infection of 10 (10 pfu/cell). In rat neonatal cardiomyocytes, infection with Ad.4 × MLC. Luc resulted in a 4.8-fold lower luciferase expression than with the CMV promoter. However, in noncardiac cells, luciferase expression was significantly reduced under the control of the 4 × MLC promoter compared with the CMV promoter as shown in Table 1. In HepG2 cells Ad.4 × MLC250.Luc was found to have reduced luciferase activity by 103-fold compared with the CMV promoter. Similar results were found in the LLC-PK1 kidney cells and COS cells (53-fold and 44-fold reduction with 4 × MLC250, respectively). Cells infected with the promoter-less construct Ad.Luc showed only background activity. We also found that the full-length promoter MLC-2v had lower activity than the 4 × MLC250 promoter with also a higher specificity when compared with HepG2 and LLC-PK1 cells.
Gene Transfer In Vivo
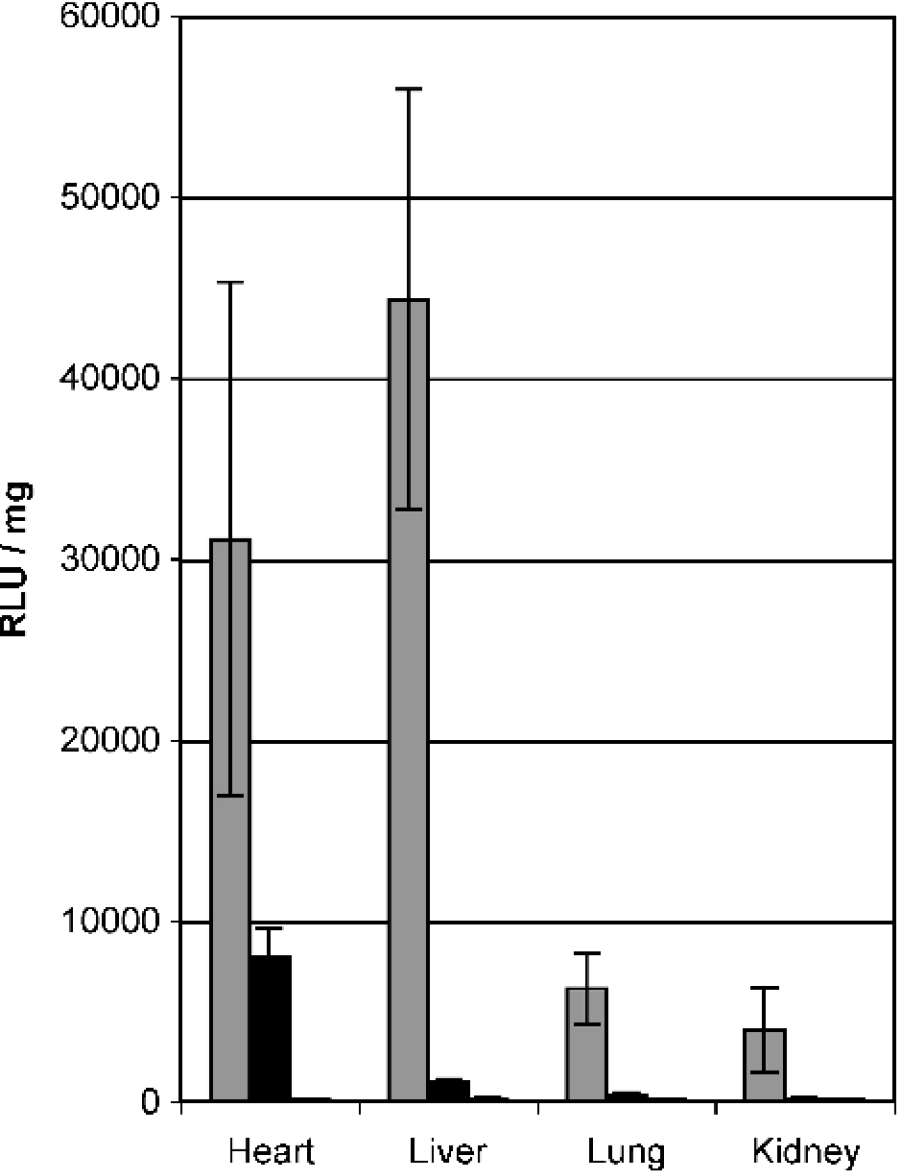
Gene expression after coronary perfusion in vivo after direct myocardial injection. Luciferase activity in four different organs harvested 3 days after coronary perfusion with Ad.CMV.Luc (gray bars) or Ad.4 × MLC250.Luc (black bars).
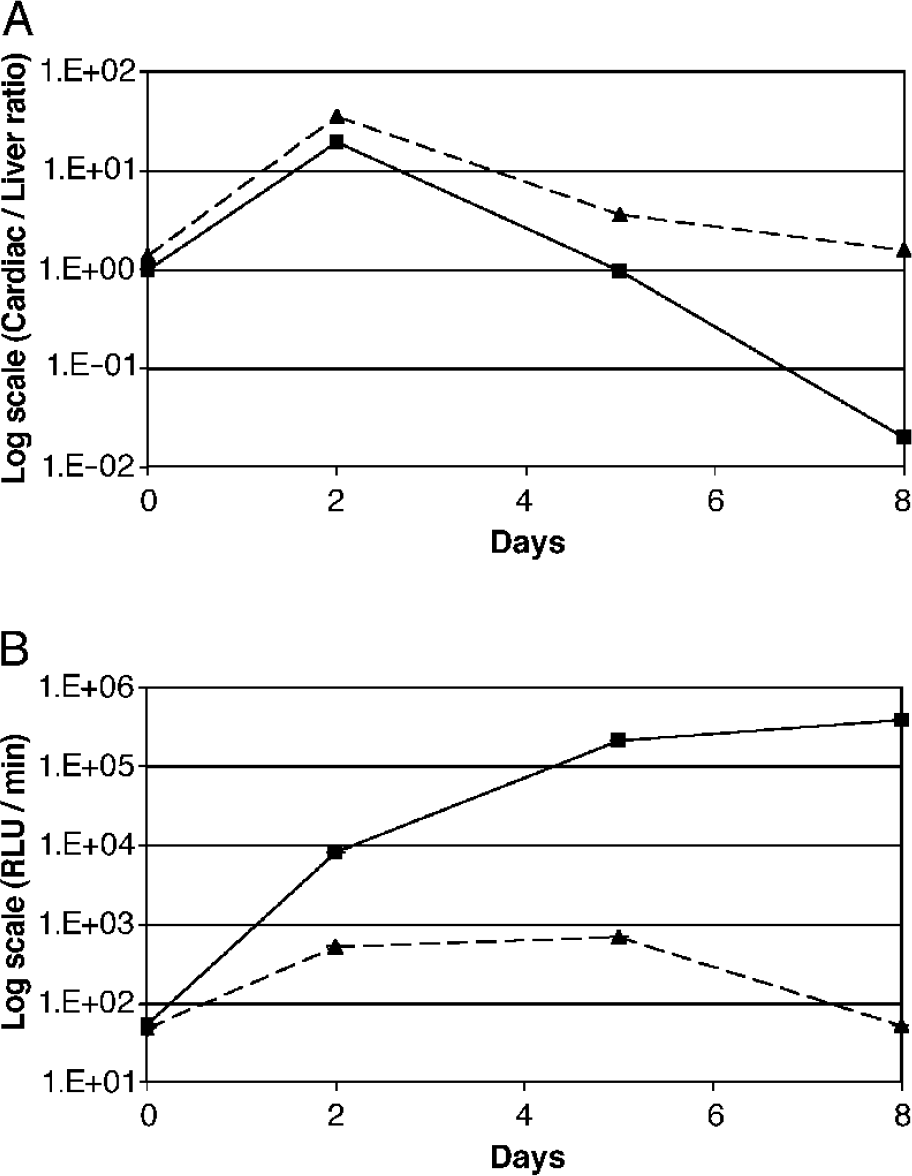
(A) Time variation of myocardial specificity ratio. Both groups show a loss in myocardial specificity over time. Animals transduced with Ad.CMV.Luc show a 50-fold decrease in myocardial specificity between Day 5 and Day 8 (solid line). In contrast, myocardial specificity of animals transduced with Ad.4 × MLC250−2v.Luc decreased only 2-fold after Day 5 (dashed line). (B) Optical imaging of hepatic luciferace activity. Promoter activity in the liver shows a continuous increase in rats transduced with Ad.CMV.Luc (solid line). Animals receiving Ad.4 × MLC-2v.Luc showed a peak hepatic activity at Day 5 with a subsequent decrease of promoter activity to baseline levels (dashed line).
Discussion
Adenoviruses have become one of the main vectors for gene transfer, mainly due to their high gene expression rate efficiency [16]. Unlike commonly used drugs, viral vectors not only reach the heart but also other major organs including the liver, kidney, and lung, exhibiting potential serious side effects.
A variety of cardiospecific promoters have been investigated to address this problem, including atrial natriuretic factor (ANF) [9], cardiac troponin T (TnT) [17], alpha-myosin heavy chain (MHC) [8], and myosin light chain-2 (MLC-2v) [8,10,18]. We concentrated our efforts on the MLC-2v promoter because in vitro studies have indicated that this promoter sequence shows a higher restriction to cardiac tissue than other promoters tested [8]. A conserved 28-bp element (HF-1-3) within the rat cardiac MLC-2v gene has been shown to confer cardiac muscle-specific gene expression [19]. We therefore constructed a promoter hybrid containing four copies of the MLC-2v promoter element HF-1-3. (4 × MLC250) to amplify this cardiospecific expression.
Myocardial Promoter Activity
Evaluation of gene expression rate of a new promoter construct is fundamental for proper promoter assessment. Although many investigators demonstrate promoter characteristics regarding its restriction to myocardial cells, only very few studies show a quantitative assessment of promoter activity in myocardium and other tissues both in vitro and in vivo [8]. A study by Franz et al. [8] observed a 10-fold reduction of myocardial gene expression rate by using an unaltered full-length MLC-2v promoter versus the RSV promoter. Our results show that the newly constructed 4 × MLC250 promoter causes a 4–6-fold lower myocardial gene expression rate when compared with the CMV promoter after coronary perfusion or direct myocardial virus delivery, however, it shows a higher

Optical imaging of reporter gene expression. Animals imaged on Day 2 (top) and Day 5 (bottom) after cardiac gene transfer. Both rats emit significant myocardial luciferase activity at Day 2 and Day 5. Animal transfected with Ad.CMV.Luc shows prominent hepatic luciferase activity starting at Day 5. Control rat shows background signal. Note that the bioluminescent scales are different for control rat, Ad. CMV.Luc and Ad. 4 × MLC250−2v.Luc to account for the wide range of cardiac luciferase activity observed.
Myocardial Specificity
Although exogenous viral promoters such as CMV are active in a wide range of tissues, it has been reported that CMV promoters show a particular tropism to hepatic tissue [4,5]. This effect has been attributed to significant side effects of adenoviral gene transfer [6,7]. We here introduce a myocardial specificity ratio (myocardial promoter activity/hepatic promoter activity) to approximate the effects and side effects of different promoters in vivo and in vitro. A myocardial specificity ratio > 1 indicates a higher myocardial than hepatic promoter activity. Not surprisingly, coronary perfusion of virus caused the highest amount of virus distribution in the systemic circulation, resulting in a myocardial specificity ratio of 7 versus 0.7 (4 × MLC250 vs. CMV). Although 4 × MLC shows a 10-fold reduction in hepatic tropism versus CMV, this result indicates that the use of coronary perfusion techniques even with cardiospecific promoters may involve more risks than other more localized gene delivery methods.
Temporal Characteristics of Promoter Activity
Recently, Wu et al. [11] could show that optical imaging of cardiac reporter genes is feasible in living rats. In this experiment, we extended the use of optical imaging in rats to monitor and compare temporal changes of myocardial promoter specificity. In accordance with the common dynamics of adenoviral mediated transgene expression [20,21], both promoters showed a loss of myocardial activity over time, with its greatest activity at Day 5. Although this dynamics could also be observed in hepatic activity of the 4 × MLC250 promoter, the CMV promoter showed a constant increase in hepatic transgene activity until Day 8, causing a dramatic loss of its myocardial specificity ratio (50-fold vs. 2-fold; CMV vs. 4 × MLC250). This late increase in hepatic transgene activity can be explained this way: (1) a cumulative leakage of adenovirus from the myocardium after direct myocardial injection, leading to a predominantly hepatic gene expression rate [11]; (2) adenoviral constructs show a native tropism for hepatic tissue, irrespective of the promoter used [22,23]; and (3) transgene expression was finally facilitated by a nonspecific promoter (CMV). In contrast, the newly established 4 × MLC250 promoter was able to mainly preserve its myocardial specificity ratio from Day 5 to Day 8, counteracting the abovementioned obstacles.
In this study, we evaluated a new cardiospecific promoter in vivo and vitro. 4 × MLC250 is highly selective for myocardial tissue and shows a significantly higher myocardial specificity ratio in vivo and in vitro than a nonspecific CMV promoter. Although the 4 × MLC250 promoter was not exclusively active in myocardial tissue, we could demonstrate that 4 × MLC250 greatly reduces late hepatic transgene activation, thus lessening an important safety concern after intracardiac gene transfer. Our results provide evidence that tissue-specific promoters may be successfully enhanced, and will play an important role in the quest for safer viral gene transfer.
Footnotes
Acknowledgments
This work was supported in part by grants from the National Institutes of Health: HL 57623, HL 71763, HL73375, (R. J. H.); HL069842 (F. dM), HL 697782 (U. S.) and the Older Americans Independence Center (U. S.). O. Y. B. is supported by a grant from the Max Kade Foundation, New York. R. J. H. is a Paul Beeson scholar of the American Federation of Aging Research.
