Abstract
A number of different types of agents have been employed to aid in the visualization of lymph nodes, particularly the sentinel lymph node, and to decrease the tissue destruction associated with the diagnosis of nodal metastases. The current study was performed to see if a novel macromolecular near-infrared fluorescent (NIRF) probe could be used to visualize lymph nodes after intravenous administration (pan-node visualization) or subcutaneous administration (sentinel node visualization), and serve as method for guiding dissection with interventional radiologic and surgical procedures. Cy5.5-PGC, the near-infrared dye Cy5.5 coupled to a protected graft copolymer (PGC), was injected (iv or sc) into nude mice. Twenty-four hours later white light and NIRF images were obtained on (i) the live animal, (ii) a partially dissected animal, and (iii) tissue specimens. With Cy5.5-PGC administered intravenously, axillary nodes were visualized from outside a living mouse. With partial dissection, iliac and aortic nodes were visible as concentrated foci of high-intensity NIRF signals. With subcutaneous injection in the front extremity, axillary and brachial nodes draining the injection site were easily visualized. NIRF imaging provides a nonradioactive method of visualizing lymph nodes through layers of tissue that can be employed with intravenous or subcutaneous injection.
Introduction
Determination of the metastatic status of lymph nodes by removal or biopsy is a critical element in planning therapy and helps provide a more accurate prognosis for cancer patients. However lymph node sampling can result in considerable side effects, such as lymphedema and surgical complications. A wide variety of agents and approaches have been investigated for visualizing lymph nodes, often with the objective of determining the first node on the lymphatic drainage channel from the tumor (sentinel node). In an effort to minimize nodal destruction in breast cancer, it is now common to remove the sentinel node through the localization of methylene blue dye injected intra- or peritumorally and radioactive Tc99m sulfur colloid injected subcutaneously [1–4]. Although reasonably accurate in identifying the sentinel node [5], the method requires the use of two substances, one of which is radioactive, thereby introducing radioactivity into the operating room. While these reagents can be employed to visualize nodes that drain the area of administration, nodes of importance in some cancers cannot be targeted by local administration techniques.
A wide range of diagnostic and therapeutic agents accumulate in lymph nodes after local administration by virtue of the flow of fluid from the interstitium to lymph nodes (for a review, see [6]). Hence a wide variety of approaches have been used to visualize lymph nodes. A much more limited number of substances undergo useful pan-lymphatic collection after intravenous injection. Macromolecules exhibiting a long blood half-life, such as dextran-coated iron oxide nanoparticles [7], a graft copolymer of polylysine and reduced dextran [8], or negatively charged albumins [9], show useful levels of lymphatic accumulation. We therefore hypothesized that the macromolecular probe Cy5.5-PGC [the near-infrared dye Cy5.5 coupled to a protected graft copolymer (PGC)], which has a long blood half-life, would collect in lymph nodes after intravenous administration and would enable the global visualization of lymph nodes by near-infrared fluorescence (NIRF). Since NIRF can penetrate tissues up to several centimeters thick [10,11], we further hypothesized the NIRF from nodes could be visualized through overlaying tissues after intravenous administration, a capability which might help guide minimally invasive sampling. The purpose of the current study was to understand the potential of Cy5.5-PGC as a nodal visualization agent by intravenous and subcutaneous administration.
Materials and Methods
NIRF Probe
Cy5.5-PGC is a graft copolymer consisting of poly-

White light (top) and NIRF (bottom) images of a nude mouse after intravenous injection of Cy5.5-PGC.
Animal and Tissue Imaging
Nude mice (nu/nu) were anesthetized by intraperitoneal injection of ketamine (90 mg/kg) and xylazine (10 mg/kg). Cy5.5-PGC was injected either subcutaneously in the front extremity to evaluate for sentential node imaging, or intravenously (tail vein) for pan lymph node delineation. Two nanomoles of dye were used with subcutaneous administration (n = 5) and 10 nmol with intravenous administration (n = 6). With both routes of administration, animals were anesthetized 24 hr after injection and whole body white light and NIRF images were obtained. For uninjected controls, n = 2. No toxic effects were noted. A previously described small animal whole body imaging system was used [13]. For excitation, the system uses a halogen lamp with a bandpass filter (610–650 nm), an imaging chamber for the animals, a bandpass emission filter (680–720 nm), a 12-bit monochrome CCD camera (Kodak, Rochester, NY) equipped with a 12.5–75 mm zoom lens for recording emission photons, and a computer for image acquisition. Image acquisition time was 60 sec per animal. Image analysis was done using commercially available software (Kodak Digital Science 1D software).
Immediately post in vivo imaging, animals were sacrificed (pentobarbital, 200 mg/kg ip), and were submitted to dissection. Whole body white light and NIRF images were obtained as dissection proceeded. Finally, tissues were taken for the determination of NIRF fluorescence as described [12,13]. Dissected tissues from injected and noninjected animals (about 200 mg) were placed in 96-well microtiter plate wells, and regions of interest were placed manually over the fluorescent image of excised tissues. The mean and standard deviation (SD) of signal intensity of pixel values was recorded. Mouse lymph nodes are between 1 and 1.5 mm in size [14]. The animal protocol was approved by the Institutional Review Committee on Animal Care and was conducted in accordance with National Animal Welfare guidelines.
Biodistribution Studies
Nodal and organ accumulation was assessed 24 hr after tail vein injection of 111In labeled Cy5.5-PGC prepared as described (n = 4) [12]. Animals were sacrificed by pentobarbital as above, and liver, spleen, and lymph nodes were taken. Para-aortic and left and right axillary nodes were pooled to obtain an average nodal concentration of probe. Activity was counted in a well counter (Wallace, Turku, Finland). Concentration of Cy5.5-PGC in tissues was expressed as percentage of injected dose per gram of tissue (% ID/g).

White light (top) and NIRF (bottom) images of a nude mouse from Figure 1 after dissection. Outlines of aortic and iliac nodes are plainly visible through overlaying tissue layers.
Results
Figure 1 shows white light and transcutaneous NIRF images 24 hr after an intravenous injection of Cy5.5-PGC, as might be used for pan-nodal delineation. Tissue penetration properties of near-infrared light are evident by the visualization of the axillary nodes and spleen, through the skin and overlying fat and muscle. As shown in Figure 2, with dissection iliac and aortic nodes were also plainly visible as concentrated foci of high-intensity NIRF signals compared to low-intensity signal from adjacent tissue.
To explore whether Cy5.5-PGC could also be used by subcutaneous administration, Cy5.5-PGC was injected in the left front extremity of a nude mouse. Figure 3 shows the white light and NIRF images of the brachial and axillary nodes. Note that the outline of axillary and brachial lymph nodes (along expected drainage route of injection site) is apparent, even though nodes are surrounded by connective tissue and fat. No signal is seen in contralateral nodes.
The NIRF fluorescence intensities of selected tissues were obtained after intravenous or subcutaneous administration of Cy5.5-PGC. After intravenous injection (10 nmol dye, Figure 4A), NIRF levels were 4 to 10 times higher in liver (1200 ± 126) and spleen (428 ± 245) than in muscle (66 ± 8) or blood (45 ± 14). A similar NIRF biodistribution study, but lacking data on lymph nodes, has been reported [12]. Fluorescence in the axillary nodes (right 740 ± 370; left 666 ± 228) and para-aortic nodes (412 ± 243) was similar to that found in the spleen (428 ± 245). With axillary nodes, the NIRF was sufficient to permit visualization from outside the living animal (Figure 1). With left front extremity subcutaneous injection of 2 nmol Cy5.5-PGC (Figure 4B), NIRF was concentrated in the left axillary node (4616 ± 666) to far higher levels than that obtained with intravenous injection (10 nmol Cy5.5-PGC, Figure 4A). The subcutaneous administration of Cy5.5-PGC provides an efficient technique for obtaining a very high NIRF intensity in nodes draining the site of administration, which can be used to view considerable nodal detail after partial dissection (Figure 3).

White light (top) and NIRF images (bottom) of a mouse after front extremity subcutaneous injection.

Lymph node NIRF after intravenous injection (A) or subcutaneous injection (B). Y-axis is fluorescence in arbitrary units (AU). Also shown are values for tissues from uninjected animals (uninj). Errors are evident only where they were sufficiently large to be seen graphically. (A) and (B) show the same data for uninjected animals.
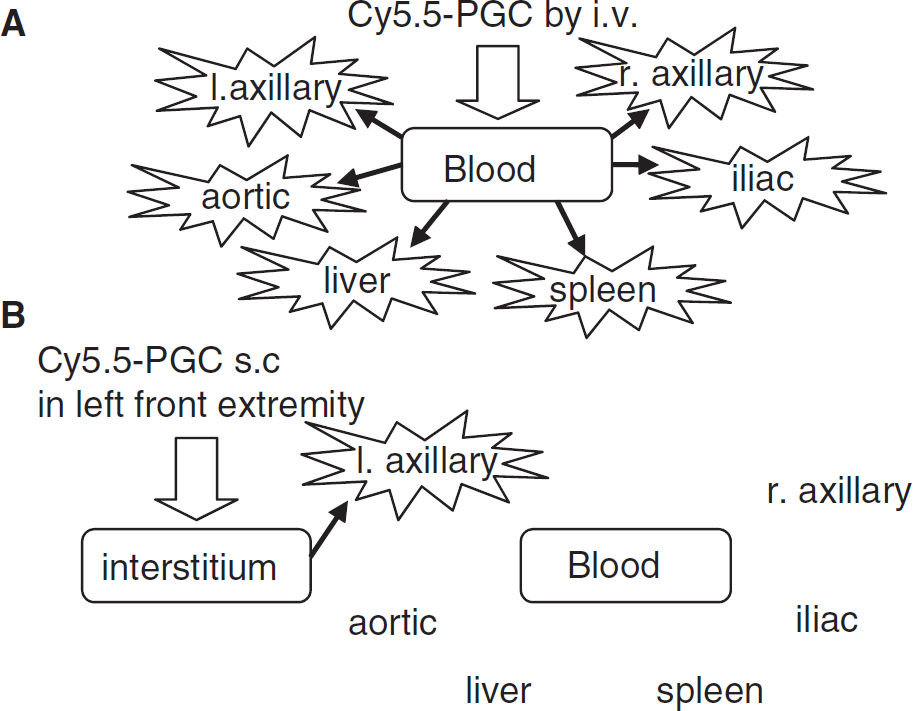
Visualization of lymph nodes after intravenous injection (A) or subcutaneous injection (B).
Using 111In labeled Cy5.5-PGC, we determined that 24 hr after intravenous injection, the concentration of PGC was highest in the liver (4.8 ± 1.3% ID/g), followed by lymph nodes (3.4 ± 0.8% ID/g) and spleen (2.2 ± 0.5% ID/g). Cy5.5-PGC is lymphotropic, achieving concentrations in lymph nodes similar to the major sites of accumulation, the liver and the spleen.
Discussion
Visualizing lymph nodes with the NIRF from Cy5.5-PGC has several major advantages over earlier methods and reagents for visualizing lymph nodes. First, Cy5.5-PGC can be administered either locally or systemically as shown in Figure 5. When injected intravenously, a similar level of NIRF signal was obtained from nodes in a variety of anatomical locations and in the liver and spleen (Figure 4A). However, this selective visualization of the axillary nodes is not due to selective accumulation of the agent in those nodes, based on the NIRF signals from dissected tissues, but rather, was due to the surface weighting of the NIRF imaging method we employed. The development of newer and less surface weighted types of NIRF instrumentation, or minimally invasive signal techniques (see below) may make possible a global delineation/visualization of lymph nodes after intravenous administration of Cy5.5-PGC. A drawback of intravenous administration is that a higher dose was needed to visualize specific nodes because of higher uptake by the liver, spleen, and nodes generally. We used 10 nmol of dye for intravenous injection, compared with 2 nmol for subcutaneous injection. On the other hand, subcutaneous administration efficiently targets Cy5.5-PGC to nodes draining the site of injection (Figure 4B and Figure 5). However, with local administration only nodes draining the site of injection can be observed. The choice between local and systemic administration might be based on the number, location, and physiology of nodes to be sampled and patterns of tissue drainage after local administration.
Another benefit of using Cy5.5-PGC for nodal visualization is the of lack of exposure of patients and operating room personnel to radiation and the elimination of the need for frequent manufacture of the pharmaceutical due to radiochemical decay. Finally, Cy5.5-PGC utilizes fluorescence in the near-infrared range (600–800 nm), which penetrates skin and fat surrounding lymph nodes. This allows visualization of nodal architecture through millimeters of overlaying fat or connective tissue that might be encountered intraoperatively.
With the NIRF reflectance viewing system used here [13], tumors implanted in subcutaneous fat less than 10 mm from the surface have been visualized from outside the animal in several systems [12,15,16]. However, NIRF is attenuated by passage through tissues, thus the signal from organs or tissues closer to the surface appears stronger than those from deep within the animal. Hence, the liver could not be visualized even though it had a high NIRF after dissection (Figure 4A). Two promising approaches for imaging NIRF signals from deep within an animal or patient are the use of fluorescent endoscopes [17,18], and fluorescence-mediated tomographic (FMT) imagers. FMT imagers have demonstrated the capability to image small foci of NIRF signals deep within mice [19]. In addition, FMT imagers has considerable potential to image NIRF signals emanating from the large animals like humans [10]. Using these approaches, the global visualization of lymph nodes after the intravenous injection of Cy5.5-PGC might be possible even with humans.
The NIRF signal obtained from lymph nodes after Cy5.5-PGC intravenous administration reflects in large measure the lymphotrophic nature of the probe, which results from its long blood half-life. Earlier biodistribution studies with intravenously injected 111In labeled Cy5.5-PGC found 31% of the agent in blood 24 hr after injection, indicating the long blood half-life of the agent (see Table 1 of Ref. [12], 15.5% ID/g in blood, assuming a blood volume of 2 ml/mouse). Cy5.5-PGC has a long blood half-life, due to its molecular weight (480 kDa), which prevents renal elimination, and its hydrophilic methoxypolyethelene glycol chains, which protect the molecule from rapid clearance by the reticuloendothelial system. In the current study, we showed that Cy5.5-PGC attained a concentration in lymph nodes (3.4 ± 0.8% ID/g), similar to that in the major organs of the reticuloendothelial system (liver, 4.8 ± 1.3% ID/g; spleen, 2.2 ± 0.5% ID/g). Magnetic nanoparticles, negatively charged albumins and a dextran-based macrocomplex, are two additional examples of materials that are lymphotrophic after intravenous injection [7,9,14]. After subcutaneous administration, the high molecular weight of Cy5.5-PGC (480 kDa) prevents diffusion into the capillary bed, so the agent undergoes a highly efficient collection by the lymphatic system.
It is possible that an additional mechanism, activation of fluorescence due to cleavage of the probe by proteases, contributes to the NIRF from lymph nodes. Cy5.5-PGC is activated by cathepsin B, and its ability to be enzyme activated has been used in the imaging of subcutaneously implanted tumors and polyps [12,20]. Cathepsin B is present in macrophages [21] and normal lymphoid tissues [22]. Future experiments may determine the relative roles of probe accumulation and probe activation in determining the NIRF signal emanating from lymph nodes.
Another diagnostic agent for lymph nodes consists of magnetic nanoparticles which, like Cy5.5-PGC, accumulate in nodes throughout the lymphatic system in animals [7] and humans [23] after intravenous administration. Magnetic nanoparticles produce high-resolution, contrast-enhanced MR images and are used to determine the presence of intranodal metastasis, information that aids in cancer staging and surgical planning. We propose to use Cy5.5-PGC not as a contrast agent for intranodal tumor detection, but as a visualization agent for determining the position of lymph nodes.
Our data suggest that Cy5.5-PGC could be injected either intravenously or locally, as is the current practice with radiocolloids or blue dyes in breast cancer surgery, and would permit visualization by NIRF. In this clinical situation, the nodal NIRF might be determined intraoperatively with relatively simple surface weighted instrumentation consisting of a source of near-infrared light, a camera with a filter for wavelengths that have been Stokes shifted, and a monitor to display the NIRF for the surgeon during the operation. With the development of a variety of new NIRF imaging techniques, Cy5.5-PGC might be administered intravenously and permit the visualization of any lymph node in a wide range of diagnostic procedures. With either form of administration, patients and hospital personnel may benefit from an NIRF imaging method that permits the visualization of lymph nodes through layers of overlaying tissue without the use of radioactivity.
