Abstract
Provoking delayed migraine with nitroglycerin in migraine sufferers is a cumbersome model. Patients are difficult to recruit, migraine comes on late and variably and only 50–80% of patients develop an attack. A model using normal volunteers would be much more useful, but it should be validated by testing the response to drugs of known efficacy in acute migraine. Furthermore, treatment during headache rather than pretreatment is the most naturalist method. To fulfil these requirements we used continuous long-lasting infusion of glyceryl trinitrate (GTN) 0.25 µg kg−1 min−1 for 140 min and gave aspirin 1000 mg, zolmitriptan 5 mg or placebo to normal healthy volunteers. The design was double-blind, placebo-controlled three-way crossover. Our hypothesis was that these drugs would be effective in the treatment of the mild constant headache induced by long-lasting GTN infusion. The headaches did not fulfil the International Headache Society diagnostic criteria for migraine without aura. Moreover, there was no effect on headache of either zolmitriptan or aspirin. Thus our hypothesis was disproved and we conclude that our model is not valid for the testing of new acute antimigraine drugs. Our experiment suggests that headache caused by direct nitric oxide (NO) action in the continued presence of NO is very resistance to analgesics and to specific acute migraine treatments. This suggests that NO works very deep in the cascade of events associated with vascular headache, whereas tested drugs work higher in the cascade. The model suggested here should therefore be tested with other headache/migraine-provoking agents that supposedly work higher in the cascade of events. The need for human models persists, but the solution to this problem is still pending.
Introduction
Human models of migraine have recently received increasing interest. Studies with nitroglycerin, glyceryl trinitrate (GTN), calcitonin gene-related peptide (CGRP) and other molecules have elucidated aspects of migraine mechanisms. Although these substances cause headache that has some migrainous features during infusion and shortly after in normal volunteers, as well as in migraine patients, only migraine patients develop a delayed headache fulfilling diagnostic criteria for a migraine attack (1).
Given the limited predictability of existing animal models of migraine, it is tempting to suggest that human models as described could be useful in the testing of new antimigraine compounds. The most reliable and strongest provoking agent is GTN. This model can be applied in many different ways with a different dose, different lengths of infusion and in normal volunteers as well as in migraine patients. The most obvious human model would therefore be to give GTN in the already validated dose of 0.5 µg kg−1 min−1 for 20 min (1,2) to migraine patients and test new drugs during the provoked migraine headache. However, such a model is highly impractical. First, it is difficult to recruit migraine patients for such experiments. Second, migraine attacks after GTN occur at highly variable time points ranging between 2 and 12 h after the infusion. Third, only 50–80% of migraine subjects developed an attack after GTN, which considerably weakens the statistical strengths of the experiments and necessitates fairly large numbers of subjects. Rather than using such a model, a drug would be tested in spontaneous migraine attacks.
In this study we used normal subjects, because they are easy to recruit, because the immediate GTN headache has some migrainous features, and because nitric oxide (NO) is crucially involved in migraine mechanisms. We gave GTN in a protracted fashion in order to allow treatment during on-going headache rather than pretreatment. Thus, the treatment situation is similar to that in spontaneous headache attacks. Finally, we used a modest dose of GTN that could be expected to be easier to treat than headache after a higher standard dose. We used a general analgesic aspirin and a triptan, zolmitriptan, as validating drugs and tested the hypothesis that these drugs are effective in the described model.
Subjects and methods
Subjects
Prior to study inclusion, it was ascertained that the subjects experienced headache after GTN 0.5 mg sublingually. Sixteen healthy medical student subjects without migraine and with no more then 2 days with tension-type headache per month were included. An additional nine subjects were screened but did not experience headache after a test dose of GTN 0.5 mg sublingually and were not included in the study. The subjects were recruited through advertising among medical students. Good general health was determined by medical history, physical examination, vital signs, electrocardiogram (ECG) and body mass index within the range 19–29 kg/m2. Women of child-bearing potential could participate provided they had a negative pregnancy test and used reliable contraceptive methods. Subjects with known allergy to the drugs used or with regular use of prescribed or over-the-counter medication except oral contraceptive pills were excluded.
Study procedures
The study had a double-blind, randomized, placebo-controlled, three-way crossover design.
Subjects were all scheduled to have three study days separated by at least 7 days. On all three study days the subjects received an intravenous infusion of GTN 0.125 µg kg−1 min−1 for 140 min in the cubital vein by a volume directed pump (IVAC P2000; Basingstoke, UK). GTN (SAD 5 mg/ml) was diluted with saline to a 50-ml 20-µg/ml solution. Supine position was assumed 30 min prior to GTN infusion and until at least 60 min after termination of the infusion. The start of GTN infusion was designated time zero. Subjects were randomized to receive either oral zolmitriptan 5 mg or effervescent acetylsalicylic acid 1000 mg or matched placebo 20 min into the GTN infusion. Randomization was performed by Astra Zeneca and the code was not known to the investigators until after the database was closed. With double dummy technique, zolmitriptan or matching placebo tablets were given with 100 ml of water and effervescent acetylsalicylic acid 1000 mg (2 × 500 mg Bamyl®; Hässle, Mölndal, Sweden) or matching placebo (Samarin, effervescent sodium bicarbonate) as a 200-ml oral solution, prepared by a person not otherwise involved in the study.
Subjects were required to refrain from smoking while in the unit. Caffeine and alcohol intake was restricted in relation to the study days.
Headache parameters
Headache was scored on a numerical verbal rating scale from 0 to 10, where 0 represents no headache, 1 represents a very mild headache, including a feeling of pressing or pulsation (pre-pain), 5 represents a moderate headache and 10 represents the worst possible headache (2). Headache characteristics (unilateral/bilateral, quality and aggravation by physical activity) and accompanying symptoms (nausea/vomiting, phono- and photophobia) were recorded in order to compare with the International Headache Society (HIS) diagnostic criteria for migraine without aura (3). Headache parameters and accompanying symptoms were recorded in the clinic before study start and every 10 min until 60 min after termination of the infusion by a standardized questionnaire. Afterwards, the volunteers used a diary card at home to register headache until 12 h after the start of infusion. In case of severe headache, the subjects were offered rescue medication as paracetamol or non-steroidal anti-inflammatory drugs (NSAIDs).
The ECG was monitored continuously and vital signs were obtained prior to start of study procedures and followed until 3 h after start of the GTN infusion. A follow-up clinical examination, including vital signs, ECG and evaluation of adverse events, was scheduled 10–14 days after the last study day.
This study was conducted from October 2000 to February 2001.
Approvals were obtained from The Danish Medicines Agency and The Scientific Ethical Committee for the County of Copenhagen. Signed informed consent was obtained by each subject. The trial was conducted in accordance with the Declaration of Helsinki amended in Somerset West, South Africa 1996. and conformed to rules for Good Clinical Practice.
Statistics
The primary efficacy parameters were peak headache (0–10) from 110 to 200 min (1.5–3 h after intake of study medication) and area under the curve (AUC) for headache from 80 to 200 (1–3 h after intake of study medication). Results for headache were analysed with non-parametric ANOVA, Friedman's test followed by Dunn's test. The design was powered to observe a 30% difference in headache scores with a type 1 error of 5% and risk of a type 2 error of 10%. For this 15 subjects were needed.
Results
Eight men and eight women participated. The mean age was 24 years (range 19–27 years). GTN 0.125 µg kg−1 min−1 for 140 min caused a mild headache that was usual constant, bilateral and with few accompanying symptoms. In each treatment group the headache was pulsating in four to seven subjects, was accompanied by nausea in 0 to two subjects, and by photophobia in two subjects. The headaches did not fulfil the IHS diagnostic criteria for migraine without aura (3). The one primary efficacy measure, peak of headache from 110 to 200 min, was 2.4 after zolmitriptan (range 0–5), 2.0 after aspirin (range 1–5) and 1.7 after placebo (range 0–9) (NS).
The mean headache values for the three treatment arms are illustrated in Figure 1.
Mean headache score during glyceryl trinitrate intravenous infusion (0.125 µg kg−1 min−1) for 140 min after oral placebo [placebo for aspirin (effervescent sodium carbonate) + placebo for zolmitriptan], aspirin 1000 mg (+ placebo for zolmitriptan) and zolmitriptan 5 mg (+ placebo for aspirin). The vertical dotted line indicates the time point when the drugs were given. Note that there is absolutely no difference for aspirin and zolmitriptan vs. placebo.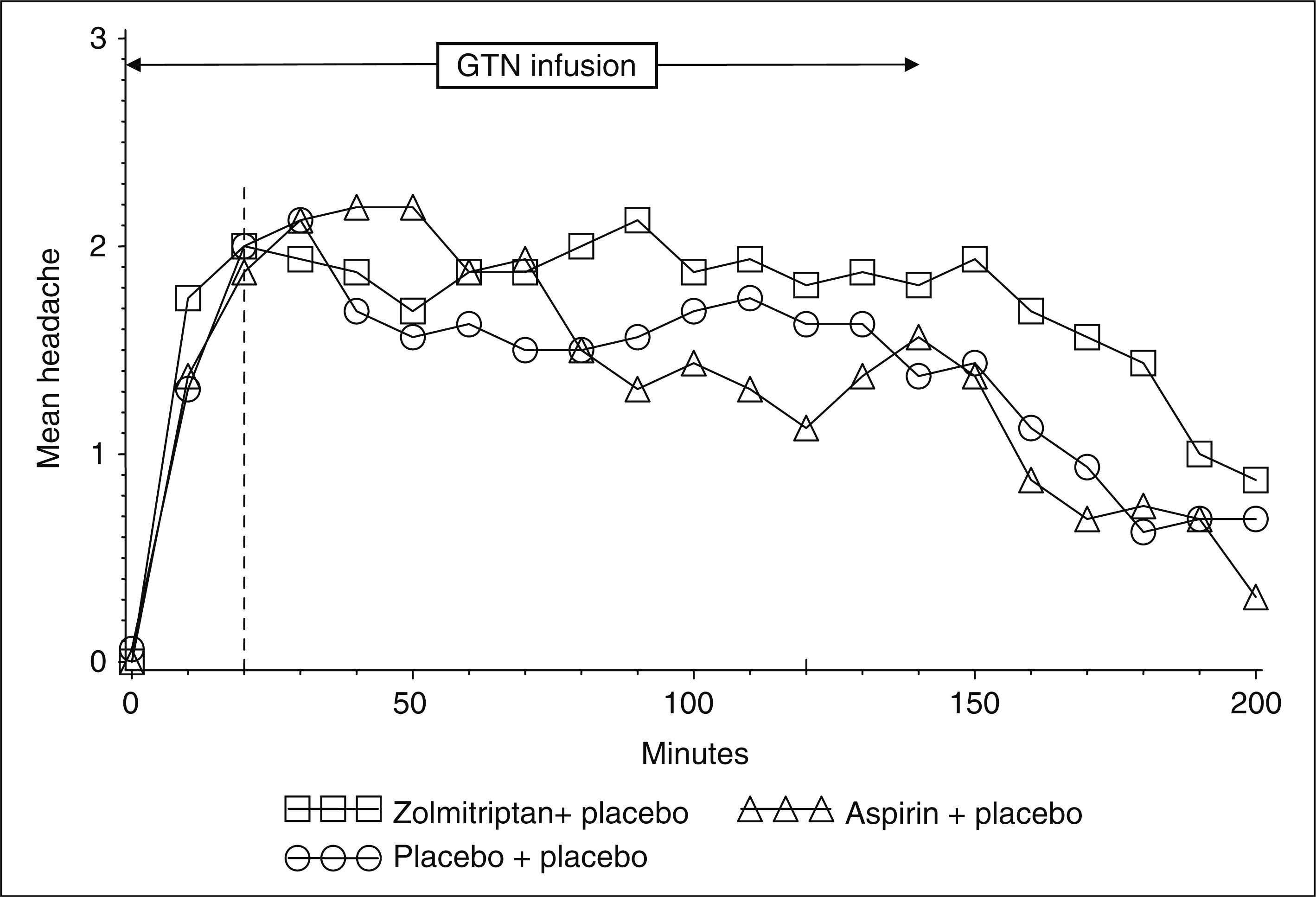
For the other primary efficacy parameter, AUC for headache severity, the median value was 170 severity × min (range 20–480) after zolmitriptan, 130 severity × min (range 0–375) after aspirin and 120 severity × min (range 0–860) after placebo (NS).
The mean baseline mean blood pressure was 84 mmHg and mean heart rate was 63 beats/min. After 20 min GTN infusion, before administration of test drugs, these parameters were largely unchanged with a mean blood pressure of 80 mmHg and a heart rate of 65 beats/min. After 140 min the mean blood pressure was 80 mmHg and the heart rate was 62 beats/min.
Discussion
The main result of the present study was that zolmitriptan and aspirin had no effect on continuing headache caused by a long-lasting infusion of a relatively low dose of GTN. Thus the experiment did not confirm our hypothesis that this design would be a useful surrogate marker of efficacy in spontaneous migraine attacks. This result is surprising and below we discuss the choice of model and of drugs as well as the possible reasons for their failure.
Choice of model
An experimental model must have significant advantages over the natural disease. Ease of recruitment of subjects, more standardized circumstances given better power, less requirement for toxicological studies and repeatability as well as being able to test in a hospital environment are examples of possible advantages of experimental human models. Regarding migraine, the GTN provocation model seems the most promising and it is certainly the best studied.
It can be done in many varieties and the paradox is that the version most closely resembling spontaneous migraine attacks, the delayed migraine after GTN, is so difficult to use that, for the testing of new drugs, it is actually easier to test them in spontaneous attacks.
For a model to be practically useful to the pharmaceutical industry, is should use normal subjects for ease of recruitment immediately on headache infusion in order to test the drug in a stable long-lasting headache. We designed such a model, but, as discussed above, it did not respond to standard treatments.
Choice of validation drugs
Aspirin was included in our study because it is the most used drug for migraine worldwide. Only 10–15% of migraineurs use a triptan in Denmark and the UK (4), while the rest probably use aspirin or another NSAID. Aspirin is a general analgesic with effect on almost all kinds of pain including episodic tension-type headache (5,6), and it is effective in human experimental pain models (7). Highly soluble aspirin 1000 mg is effective in migraine (8) but its mode of action in migraine is uncertain. Animal studies indicate that NSAIDs, including aspirin, may inhibit central trigeminal neurons in nucleus caudalis (9,10). This mechanism of action should work for all kinds of pain in the head, but aspirin was without any effect in the present vascular headache model.
Zolmitriptan 5 mg, double the recommended dose of 2.5 mg (11), was expected to work in the GTN headache model in normal volunteers because sumatriptan given as 6 mg subcutaneously followed by GTN or placebo had effect in normal volunteers (12). Both headache and arterial dilation because of GTN were significantly decreased after sumatriptan from after placebo. Oral zolmitriptan has a significant effect in spontaneous migraine attacks detectable 30 min after administration and is probably maximal after 2 h (13). It also has vasoconstrictor effects in man. Thus, oral zolmitriptan 2.5 mg investigated in 16 migraine patients outside of attack constricted both brachial, carotid and superficial temporal artery (STA) 1.5 h after intake (14). Sumatriptan was in the previous study (12) given as pretreatment, whereas zolmitriptan in the present study was given during a continued infusion of GTN. Thus, sumatriptan was already bound to the vascular receptor as demonstrated by constriction of the STA (12) before the GTN infusion was started, whereas zolmitriptan in the present study was administered during an ongoing GTN infusion.
Possible mechanisms of action of NO, aspirin and zolmitriptan
The GTN in the low dose used here caused only a mild headache in normal volunteers, and aspirin usually cures mild spontaneous headache. However, it has been suggested that aspirin works via inhibition of cyclooxygenase, thereby inhibiting the formation of prostanoids, some of which induce the formation of NO (15). If the mechanism of aspirin in mild to moderate spontaneous headache is to reduce prostanoid formation and the consequent reduction of NO formation, then this mechanism would be bypassed by exogenous NO delivered by GTN in the present study.
How do we then explain the lack of effect of zolmitriptan? The most widely known mechanism of action of the triptans is cerebral vasoconstriction. Sumatriptan 6 mg given by subcutaneous (s.c.) injection constricts the middle cerebral, the middle meningeal artery and the STA (12,16). Given as a tablet, zolmitriptan had a smaller but significant constrictor effect on STA (14). It is possible that the vasoconstrictor effect of zolmitriptan in the present study was too small to influence GTN-induced headache, whereas the faster and greater effect of s.c. sumatriptan reduced nociceptive input. The other main mechanism of action of triptans is a binding to and stabilization of prejunctional nerve terminals to inhibit leakage of neurotransmitters into the perivascular space, or binding to the central terminal inhibiting impulse transduction at the first synapse (17,18). The neurotransmitters act on receptors in cellular membranes and subsequently on intracellular pathways. NO acts downstream of classical neurotransmitters by stimulating intracellular soluble guanylyl cyclase. It is therefore not surprising that stabilizing nerve terminals will have no effect on headache caused by NO.
Implications for use of the GTN model in drug development
With the present negative results at hand, how should a useful human model be designed? There are at least two possibilities. GTN may still be useful, but the active drug must then be given as pretreatment before GTN is administered. This was effective in our previous study (12), but probably because of a marked vasoconstrictor effect of s.c. sumatriptan. Thus, we are not optimistic about this model. The other possibility is to use other provoking substances such as CGRP or other migraine-inducing substances. They may work higher in the cascade of migraine mechanisms and hence may be more responsive to migraine drugs. If that is so, it might be possible with such substances to use a design similar to that in the present study, i.e. giving the experimental treatment during continuing infusion of the migraine-inducing substance. The infusion would have to be continued for at least 2 h and preferably 3 h in order to give enough time for the drug to work.
Conclusions
A naturalistic human migraine model in non-migraine healthy volunteers cannot be developed with GTN, because: (i) GTN did not induce migraine headaches in our population; and (ii) neither aspirin nor zolmitriptan had any effect in such a model.
Footnotes
Acknowledgements
The study was supported by AstraZeneca and by the Lundbeck Foundation via the Lundbeck Foundation Centre for Neurovascular Signalling (LUCENS).
