Abstract
Glyceryl trinitrate (GTN) is a pro-drug dissociating nitric oxide throughout the body. It dilates cephalic arteries without increasing cerebral blood flow (CBF). GTN induces headache in healthy volunteers and migraine attacks in migraineurs. Acetazolamide (Az) increases CBF but does not dilate cerebral arteries. The hypothesis tested here was that Az, by dilating cerebral arterioles but not arteries and thereby decreasing pulsatile stretching of the wall of the large arteries and their perivascular sensory nerves, would reduce or prevent the GTN-induced headache We tested this hypothesis in 14 healthy volunteers. In a randomized, double-blind, cross-over study, they were pretreated with Az or placebo followed on both study days by a GTN infusion of 0.5 μg kg-1 min-1 for 20 min. Headache was scored on a verbal rating scale and a headache diary was kept for 12 h. Mean blood velocity of the middle cerebral artery was measured (transcranial Doppler). Our hypothesis was disproved, as Az did not decrease GTN-induced headache. Unexpectedly but interestingly, GTN combined with Az induced more delayed headache than GTN alone. Furthermore, a migraine-like headache was observed in three volunteers, who did not develop migraine after GTN alone. The fact that a suitable pharmacological intervention may trigger migraine in individuals with no prior migraine may suggest that the ability to develop migraine without aura is a quantitative genetic trait.
Introduction
Our experimental human headache model using intravenous infusion of glyceryl trinitrate (GTN) for headache induction has proved a useful tool in studies of headache mechanisms (1–3). Dilation of large intracranial arteries has been reported both during GTN-induced headache and, variably, in spontaneous migraine attacks (4–7). The intracranial arteries are innervated by trigeminal sensory nerve fibres (8). Stretching of these nerve endings, similar to a strain gauge, might increase sensory afferent activity and headache (9).
Although GTN dilates intracranial arteries, it does not increase regional cerebral blood flow (rCBF) (4, 10). This suggests that the arterioles remain unaffected. In contrast, acetazolamide (Az) almost selectively dilates cerebral arterioles causing considerable rCBF increase (11).
The hypothesis tested here was that Az, by dilating cerebral arterioles but not arteries and thereby decreasing the pulsatile stretching of the wall of the large arteries and their perivascular nerves, would reduce or prevent GTN-induced immediate headache.
This was not confirmed but, surprisingly, Az increased delayed GTN-induced headache considerably and in some caused a migraine attack without aura.
Material and methods
Subjects
The study followed a randomized, placebo-controlled, double-blind, cross-over design. Fourteen healthy subjects, mean age 25.7 years (range 20–31 years), male : female ratio 6:8, were recruited from the Copenhagen Schools of Medicine and Dentistry. The participants had no prior history of migraine and did not have tension-type headache more than once a month. We retrospectively interviewed participants about their family history of migraine, but only half could be contacted. They had no family history of migraine. The participants were examined on two separate occasions, at least 7 days apart. They were challenged with 0.25 mg of GTN sublingually and only those who experienced headache were included. The study was approved by The Danish National Board of Health and The Ethics Committee of Copenhagen County. Participants gave their informed consent, in which the right to withdraw at any time during the study was emphasized. The study was conducted in accordance with the Helsinki Declaration of 1964, as revised in Edinburgh in 2000.
Equipment
The GTN infusion was given using a volume directed pump (Braun Perfusor; Braun, Frederiksberg, Denmark). Blood pressure and heart rate were measured with an automatic, inflatable arm cuff (Tonoprint; Speidel und Keller, Jungingen, Germany). Transscan (2 MHz; EME, Uberlingen, Germany) was used for the transcranial Doppler (TCD) measurements. Handheld probes were used, as fixed probes are unpleasant when subjects develop headache. However, great care was taken to ensure identical placement of the probes. As seen in Results, the variability of the measurements was small. Mean maximal blood velocity in the middle cerebral arteries was calculated as mean of the maximal velocities of four heartbeats. End-tidal CO2 was measured with a capnograph (POET; Criticare Systems, Waukesha, WI, USA). The CO2 in the exhaled air was collected through a line fastened to a facial mask with a large opening, through which the subjects breathed air from the room.
Procedure
No alcohol, smoking, caffeine-containing beverages or medication were allowed for the 12 h preceding the study, and subjects had to be headache free at study start. The syringe containing Az/placebo (saline 0.9%) was prepared by a physician not involved in the study. Baseline values were recorded after insertion of a cannula (Viggo, Venflon; Viggo AB, Halsingborg, Sweden) in the right cubital vein, and 30 min of rest (Fig. 1). One gram of Az or placebo was injected intravenously over 5 min, and after 30 min an infusion of GTN (0.5 µg kg−1 min−1) was given for 20 min. Headache was scored on a 0–10 verbal numerical rating scale, where 0 represents no pain, 5 moderate headache and 10 the worst possible headache. At pain scores > 0, standardized questions were asked in order to characterize the headache quality and accompanying symptoms according to the diagnostic criteria of the International Headache Society (IHS) for migraine without aura (12). Blood pressure and heart rate were measured every 2 min, TCD every 5 min, and CO2 was recorded continuously (Fig. 1).

Schedule of the study.
The participants left the laboratory 1 h after termination of the GTN infusion, and completed a headache diary at home in the following 11 h.
Statistics
Randomization, multicomparison test for Friedman's test, and Wilcoxon–Pratt were made using MEDSTAT. Excel and Statgraphics were used for all other statistical analyses. P < 0.05 was chosen as the level of statistical significance.
The peak headache scores and the summed headache scores obtained in the laboratory, Period I (0–115 min), and recorded headache at home, Period II (2–13 h), were analysed separately, with Wilcoxon (sign rank sum test, headache in Period I) and Wilcoxon–Pratt (Period II). Differences in headache scores over time were analysed with Friedman's test, and the significant differences located with a multicomparison test. One subject reported headache after Az and before GTN. The statistical analyses were done with and without the data from this subject, but as no difference was found, all data were included in the analyses.
Data on the mean maximal blood velocity, systolic and diastolic blood pressure, heart rate and end-tidal pCO2 were calculated as percent of baseline. Four events were chosen for analysis: baseline (t = 0), maximum mean blood velocity (t = 25 min), end of GTN infusion (t = 55 min) and end of study (t = 115 min). The time effect of each study day was analysed with a multifactor analysis of variance (
Side-to-side differences in mean blood velocity were analysed with a paired t-test, at baseline (t = 0 min) and at the maximum mean blood velocity (t = 25 min). No significant difference was found, and further analyses were done using the mean of the two sides.
Results
All participants were headache free at study start and all completed the study. Az and GTN were well tolerated. Most participants described sudden dizziness for a few minutes after infusion of Az. Other well-known adverse events were reported: tingling or numbness in the extremities (n = 4), nausea (n = 1), dizziness (after the initial dizziness) (n = 3), altered taste for carbonated drinks (n = 1), buzzing (n = 2) and headache (n = 1).
Headache
Thirteen of 14 experienced headache on both study days in Period I. Eleven subjects reported headache following Az/GTN vs. eight following placebo/GTN in Period II.
Pretreatment with Az did not influence the peak headache scores in Period I (0–115 min), 2.1 (Az) vs. 2.1 (placebo) (P = 0.68), or in Period II (2–12 h), 3.4 (Az) vs. 1.1 (placebo) (P = 0.14). In Period I, the summed headache score showed no difference between Az and placebo, 12.1 vs. 11.0 (P = 0.86), whereas the summed headache score in Period II increased significantly after Az, 16.5 vs. 2.9 after placebo (P = 0.04) (Fig. 2).

Mean headache scores following acetazolamide or placebo and glyceryl trinitrate (GTN) in Period I (0–115 min) and Period II (2–13 h).
In three subjects the headache fulfilled the HIS criteria for migraine without aura following Az at 3, 3.5 and 6.5 h after the GTN infusion, but none after placebo. Of these, one was adopted, one gave no family information and one had no migraine in first degree relatives. The headache in Period II and its characteristics are listed in Tables 1 and 2.
Headache characteristics, Period II, 2–13 h after intravenous infusion of 1 g of Diamox/placebo
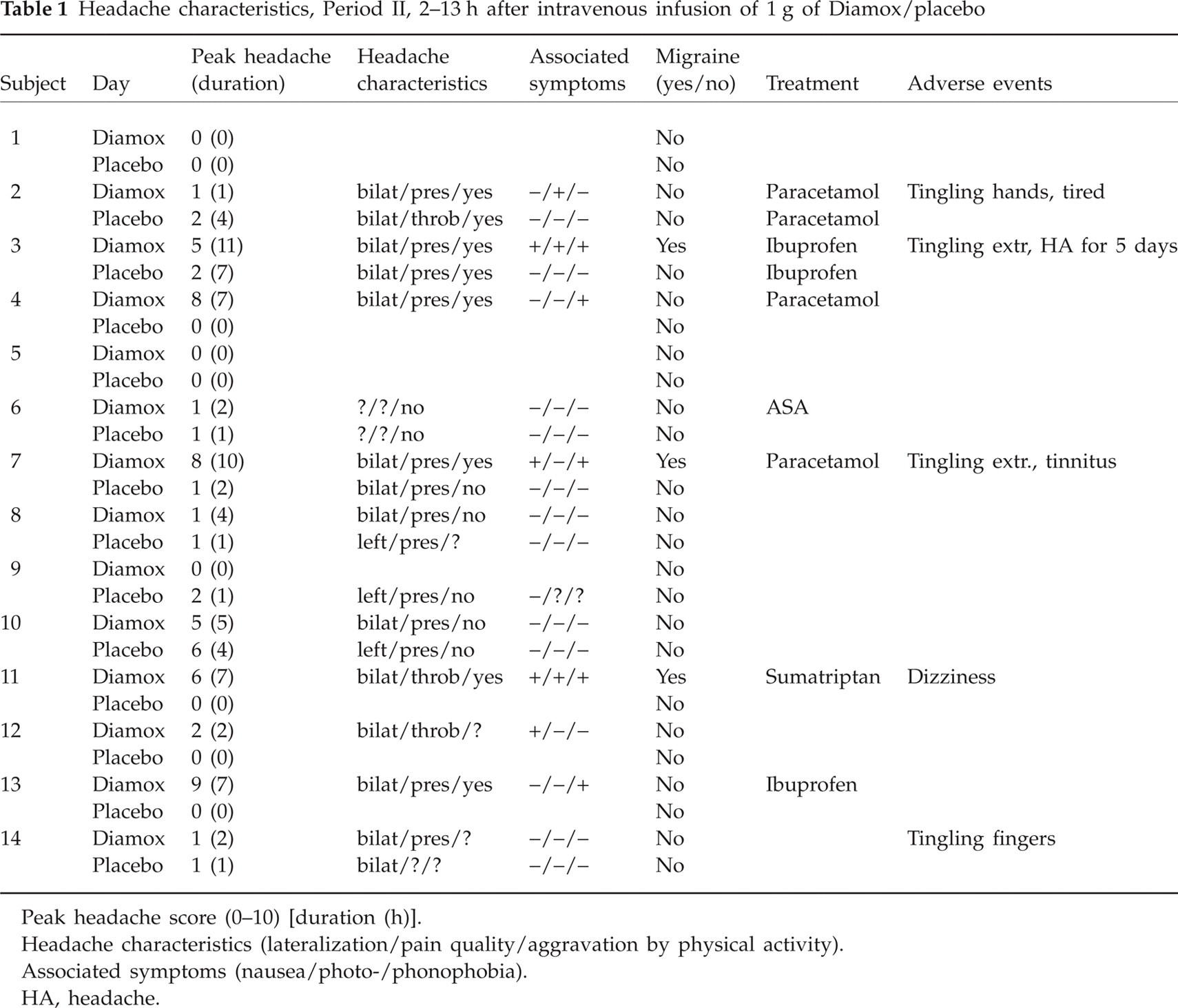
Peak headache score (0–10) [duration (h)]. Headache characteristics (lateralization/pain quality/aggravation by physical activity). Associated symptoms (nausea/photo-/phonophobia). HA, headache.
Headache characteristics, Period II (2–13 h), n = 14
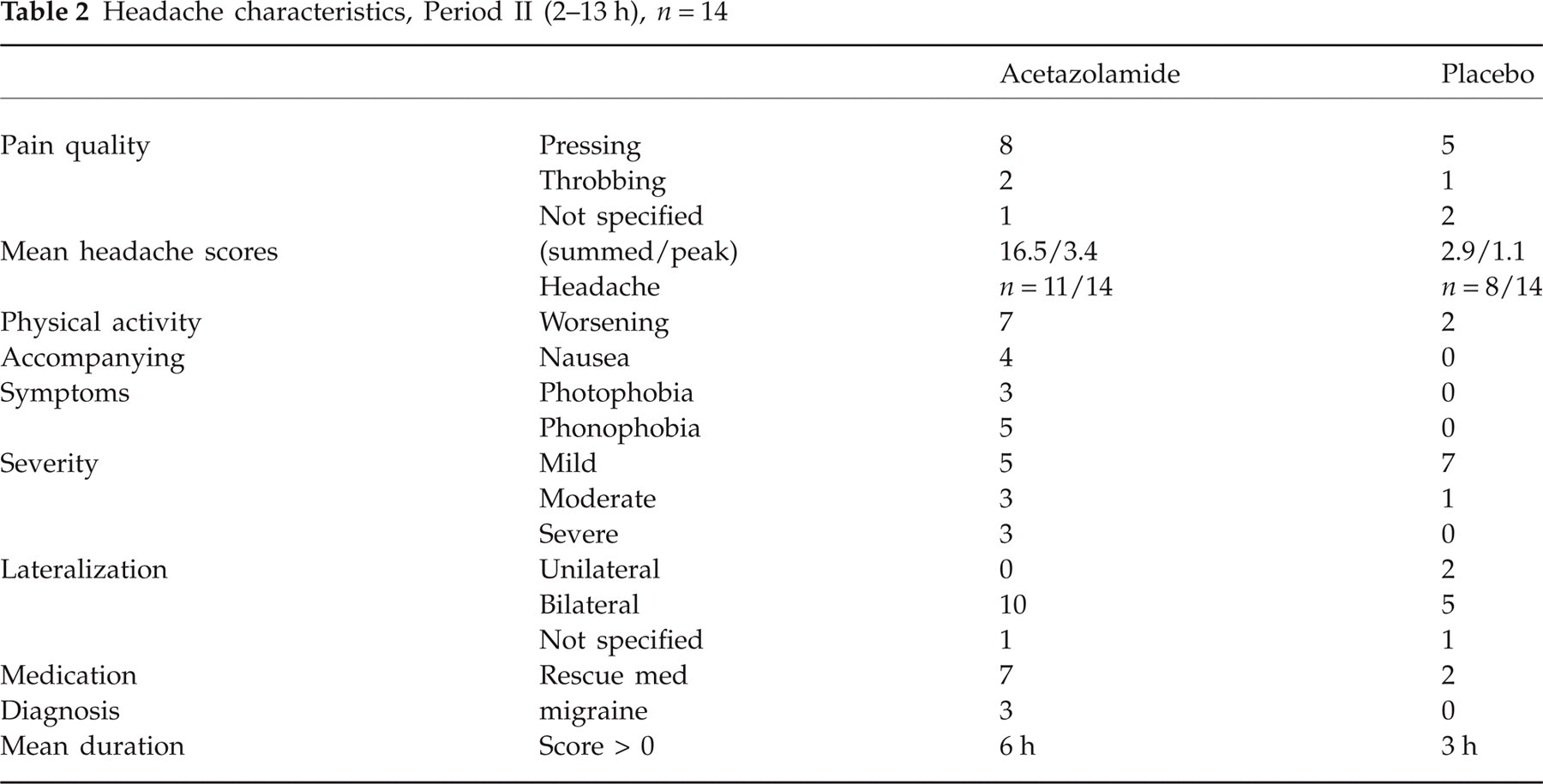
Seven of 11 subjects with delayed headache (including the three with the migraine-like headache) took headache-relieving medication after Az, compared with two of eight with delayed headache after placebo.
Mean blood velocity and end-tidal CO2
Mean blood velocity in the middle cerebral arteries increased following Az to 138% (± 3.4) (P < 0.001) above baseline (= 100%), compared with 102% (± 1.3) (P < 0.001) after placebo (Fig. 3). The increase in velocity was immediate, with a mean increase above the baseline of 17% 5 min after initiation of the Az infusion. The GTN infusion, on the other hand, caused an equal 20% decrease in mean blood velocity within 5 min on both study days.

Maximum mean blood velocities in the middle cerebral arteries, V mean, n = 14. Absolute values, pCO2-corrected data.
End-tidal pCO2 decreased 5% (± 1.8) 15 min after Az (P = 0.05), and increased by 5% (± 1.1) 30 min after placebo (P = 0.02). GTN caused a 2% decrease in pCO2 on the Az day and a 3% decrease on the placebo day.
Blood pressure and heart rate
Systolic blood pressure did not change significantly, neither on the Az day (P = 0.2) nor on the placebo day (P = 0.6). Diastolic blood pressure showed a similar increase from baseline on both days, with a peak of 114% (± 2.3, Az, P < 0.001) and118% (±2.6, placebo, P < 0.001) during the GTN infusion. GTN increased heart rate on both study days to 120% of baseline (± 3.6, Az, P < 0.001) and 113% (± 3.0, placebo, P < 0.01).
Discussion
The main result of the present study was that Az, despite probably decreasing arterial pulsation, did not diminish GTN-induced headache. Thus, our primary hypothesis was falsified. An unexpected but important finding was that the combination of GTN plus Az increased delayed headache after GTN and induced migraine-like headache in healthy volunteers.
The immediate headache
GTN followed by placebo induced an immediate headache during the infusion, which rapidly decreased and essentially was gone in 30 min. Pretreating with Az did not alter the immediate GTN-induced headache in the present study. Thus, the study did not support our initial hypothesis that Az, by dilating arterioles and increasing tissue blood flow, would decrease arterial pulsation and reduce GTN-induced headache.
Theoretically, decreased vascular resistance in the arterioles will decrease pulsation in conductance arteries, but this may not have happened due to counterregulation in conscious human beings. Another possibility is that headache does not arise from or in relation to cerebral arteries, but rather from arteries in the dura mater or extracranial arteries, where blood flow and pulsations are not influenced by Az (11, 13). A third possibility is that direct dilation by Az may involve not only the intraparenchymal arterioles but also small cortical arteries that receive sensory innervation from the trigeminal nerve (8, 11). Perivascular input from pial arteries or arterioles might then offset any beneficial action on the bigger pial arteries. It should also be considered that the increased CBF results in higher blood velocity in the arteries. This would cause increased shear stress leading to endothelial nitric oxide (NO) production (14). That could be the reason why Az has been shown by itself to have some headache-inducing properties (15). One possible conclusion from the present study is that the transarterial pressure in cerebral arteries is not as important for migraine pain as previously thought. However, considering the uncertainties about the mechanisms involved, such a conclusion is probably premature.
The delayed headache
It is interesting that in the present study Az plus GTN caused significantly more delayed headache than GTN alone in normal volunteers. In our previous studies of normal volunteers, GTN caused little delayed headache and no migraine in normal volunteers (16). It is therefore of interest that Az/GTN but not placebo/GTN was able to induce a delayed migraine in normal volunteers who had never had migraine. This delayed headache occurred at variable times in different patients beginning around 4 h after GTN infusion, and it lasted for several hours or until effective migraine medication was taken. In other words, pretreatment with Az in normal volunteers changed their response to GTN from the pattern of normal volunteers into a pattern similar to that observed in migraine patients. The delayed headache in these normal volunteers had many of the characteristics of migraine (Table 1). In three out of 14 individuals it fulfilled the operational diagnostic criteria for an attack of migraine without aura (12). This conversion of a normal response to a response similar to migraine patients has not been observed before and is of paramount importance, because it suggests that many phenotypically normal individuals may develop a migraine attack if challenged with sufficiently strong triggers. It is possible that our subjects to some extent had migraine in their family since we did not initially interview about this, and several could not later be contacted. In a future study it would be interesting to group individuals according to their family history, but it would be necessary to interview all family members directly. Subjects with one or more affected first-degree relatives might be more prone to develop delayed headache because of a higher genetic load. Along this line of thinking, the present results support that all individuals have a genetically and environmentally determined migraine threshold that decides the likelihood of having a migraine attack.
The time at which delayed headache developed in our normal volunteers after Az was so late that all direct haemodynamic effects of GTN and also of Az had disappeared (3, 4, 11). Again, this response is similar to that seen in migraine patients after GTN alone. A biochemical cascade must be triggered that, once triggered, runs its own course and in a few hours results in migraine or migraine-like headache.
In migraine sufferers Shirai et al. (15) found that Az by itself may induce headache. Although without formal diagnostic criteria, it was reported that a migraine attack occurred 1–8 h after administration of oral Az in many patients. It is possible that it is the increased shear stress and endothelial NO formation caused by Az that augments the direct GTN effect in the present study. Shear stress may also explain how Az by itself induces headache or migraine in migraine sufferers.
Haemodynamic results
Previous observations have shown that GTN has no effect on brain arterioles and cranial blood flow, but markedly dilates brain conductance arteries in man (4). Az has the opposite effect, namely a marked dilation of brain arterioles and a marked increase in CBF, but virtually no effect on brain arteries (11).
The response to GTN in the present study was a decrease in velocity, which was seen from the normal level in the placebo pretreated group and from a very high velocity level in the Az-treated group. Despite these different levels, the magnitude of velocity reduction by GTN was almost identical. Our results also demonstrate, as discussed above, that the duration of the haemodynamic effect of Az and GTN is relatively short.
Acknowledgements
The study was supported by The Migraine Trust, United Kingdom and by the Lundbeck Foundation as part of the Lundbeck Foundation Centre for Neurovascular Signalling. We are grateful for the skilful assistance of laboratory technician Kirsten Enghave.
