Abstract
Infusing glyceryl trinitrate as a donor molecule, we have used electrophysiological and c-fos immunostaining techniques to study the effects of nitric oxide on neurones in the nucleus trigeminalis caudalis. Following infusion of glyceryl trinitrate, responses of neurones to electrical stimulation of periorbital cutaneous afferents were potentiated and threshold for activation of neurones by stimulation of dural afferents was reduced. Expression of c-fos was unchanged by glyceryl trinitrate compared to saline controls. Intradermal injection of capsaicin in the periorbital area increased c-fos expression in nucleus trigeminalis caudalis; this was significantly potentiated by glyceryl trinitrate. These results suggest that, in the anaesthetized rat, glyceryl trinitrate alone may not acutely activate the trigeminovascular system to a significant degree at doses that cause headache and later trigger migraine headache in migraineurs. Nevertheless, it is susceptible to exogenous nitric oxide in that activation of trigeminal neurones through cutaneous or dural pathways is potentiated. This may in some measure underlie the pathogenesis of migraine headache.
Introduction
The search for the pathophysiological basis of migraine has recently begun to highlight the putative role of nitric oxide (NO) in the generation of spontaneous migraine attacks. The main unpleasant side-effect of NO-donors such as glyceryl trinitrate (GTN), used therapeutically as coronary vasodilators, is throbbing headache; indeed GTN has been used for many years as an agent provocateur in the study of headache (1). Consequently, migraineurs are known to be supersensitive to the rapid-onset headache-inducing effects of GTN (2) and, by inference, to NO itself. In normal subjects GTN infusion induces a rapid-onset headache which usually disappears within minutes of stopping infusion; the antimigraine drug sumatriptan attenuates this immediate headache (3). Migraineurs experience a more severe initial headache that persists at a moderate level for several hours before developing into a full-scale migraine headache (4–6). Furthermore, infusion of a nitric oxide synthase inhibitor into migraineurs has been shown to abort ongoing attacks (7).
It is now accepted that the pain of migraine almost certainly arises from nociceptors in intracranial vessels and sinuses (8, 9). Sensory innervation of these structures is predominantly by small diameter peptidergic afferents, many of which project via the trigeminal nerve to second order neurones in the nucleus trigeminalis caudalis (NTC) and superficial dorsal horn of the upper cervical spinal cord (10–14). Electrical stimulation of the trigeminal ganglion in man is known to evoke intense pain, as is direct electrical or mechanical stimulation of the superior sagittal sinus, the pain referred usually to an area within the ipsilateral trigeminal territory (15, 16). Moskowitz and colleagues (17) devised a model, subsequently much used and modified, to visualize plasma extravasation in the dura following activation of the trigeminovascular system either electrically by stimulating the trigeminal ganglion, or chemically by infusing inflammatory mediators into the sagittal sinus. Variations of this paradigm have since been employed to probe the effects of stimulating the trigeminovascular system and to characterize the response properties of afferents innervating the cerebral vasculature (18–21).
Stimulation of pain-producing vascular structures such as the sagittal sinus activates second order neurones in the NTC and cervical dorsal horn. This has been shown electrophysiologically and by mapping the appearance in second order neurones of fos protein (FOS) (14, 22, 23), considered an index of post-synaptic activity correlating with nociceptor activation (24–26). Large systemic doses of GTN promote FOS expression in the trigeminal nucleus (27, 28), in a pattern similar to that seen following noxious stimulation of facial afferents (29). Indeed the latter study, together with many others, clearly demonstrates the convergence of intracranial meningeal and vascular afferents with facial cutaneous fibres upon second order neurones in the NTC (e.g. 22, 23, 30, 31).
In view of this convergence, and the idea that GTN acts upon the intracranial vasculature (6), we have probed the effect of GTN on activation of NTC neurones. To avoid traumatic sensitization of intracranial trigeminovascular or meningeal afferents by craniotomy, we looked indirectly for an effect of GTN on this pathway by measuring the magnitude of NTC neurone responses to electrical stimulation of facial periorbital skin afferents. In a separate study, we measured GTN-induced changes in electrical threshold of dural sensory fibres, stimulating through thinned skull and again avoiding major craniotomy. We also looked for induction of FOS in the brainstem by GTN and quantified the effect of GTN on FOS expression induced by noxious stimulation of facial skin using infraorbital intradermal injection of capsaicin.
Methods
Animals and preparative surgery
Male Wistar rats (200–250 g) were anaesthetized with an intraperitoneal injection of urethane (1.5 g/kg). For electrophysiological experiments, polythene cannulae were introduced into the trachea, left femoral artery and left femoral vein to facilitate artificial ventilation, monitoring of blood pressure and intravenous administration of saline or drugs, respectively. Core temperature was maintained at 37 ± 1°C by means of a rectal probe connected to a homoeothermic heating blanket controller. Animals were secured in a stereotaxic frame and the atlanto-occipital membrane removed together with the dorsal aspect of the C1 vertebra. In most experiments, animals were allowed to breathe spontaneously. However, if breathing became uneven or laboured, animals were paralysed and artificially ventilated; end-tidal CO2 was monitored and maintained at 4–5% by adjusting tidal volume. For FOS experiments, the left femoral vein alone was cannulated and animals were kept warm as above.
Electrophysiology
With reference to a stereotaxic atlas of the rat brain and using the obex as a landmark, glass-covered tungsten microelectrodes (tip diameter, approximately 2 μm; impedance, approximately 1 MΩ) were inserted into the NTC. Recordings were made from neurones receiving robust input from either periorbital afferents (response magnitude study) or both periorbital and dural afferents (activation threshold study). Although measurements of dural receptive field and mechanical threshold yield useful information about response properties of central neurones, we decided against risking sensitization through the surgical trauma involved in exposing the dura. Electrical stimulation of the dura was made through a thinned window in the skull (see below). In all experiments we strove to keep both electrical and mechanical stimulation to a minimum to avoid inducing sensitization.
To achieve an optimum signal from a single neurone, the electrode was driven vertically into the brainstem whilst electrically stimulating either periorbital skin or dura and stroking the periorbital area with a paintbrush. Neurones were selected for study if they responded to afferent stimulation by firing spikes of at least twice the amplitude of background noise (Fig. 1).
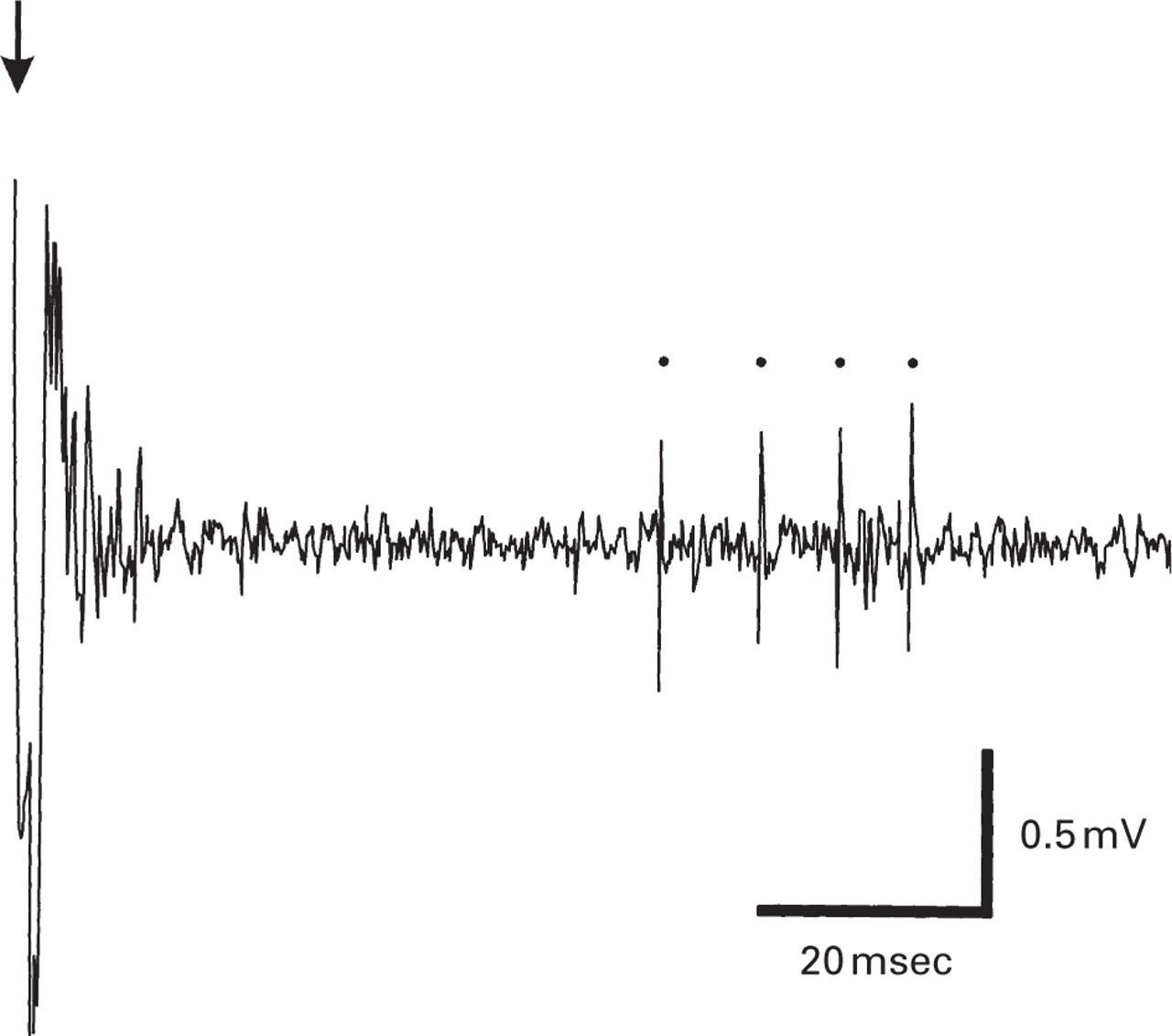
Activation of an NTC neurone by electrical stimulation of ipsilateral periorbital receptive field. Stimulating pin electrodes were inserted into the skin just below the anterior (positive) and posterior (negative) corners of the eye. The arrow indicates the time of stimulus delivery and the latencies of the spikes evoked in this example are 55.9, 64.6, 71.3 and 77.4 ms. Based on a conduction distance of approximately 25 mm, and allowing 1 ms for synaptic delay, the latency to the first spike corresponds with a conduction velocity of 0.46 m/s, indicating that the fibre(s) giving rise to the response in this neurone are most likely C type. Within its periorbital receptive field, the cell responded very strongly to firm pinch but only weakly to light touch.
A Neurolog NL800 stimulus isolator (Digitimer, Welwyn Garden City, UK) was used to deliver single constant current pulses (1–10 mA, 0.2 or 2 ms, 0.1 Hz), either percutaneously through stainless steel pin electrodes to the ipsilateral periorbital skin, or through gold-plated electrodes (convex tip diameter 0.5 mm) to the ipsilateral dura. Signals were amplified (× 5k) and filtered (0.3–5.0 kHz) using Neurolog equipment and passed through a Humbug noise eliminator (Quest Scientific, North Vancouver, Canada). Neuronal activity was monitored on several oscilloscopes; the signal was also digitized (VR10B Data Recorder, Instrutech Corp., Great Neck, USA) and saved on videotape. Sections of interest were played back later and stored on computer for analysis with Spike 2 software (Cambridge Electronic Design, Cambridge, UK).
Response magnitude experiments
Long-duration recordings were made from neurones with a stable response to a constant, single shock, submaximal stimulation (0.1 Hz) of the periorbital skin. After 10–20 min of recording, either saline or GTN was infused for a period of 30 min. Responses were then followed for up to 30 min post-infusion. Only one neurone was studied from each animal.
Neurones chosen for study in these experiments were either completely quiescent or firing very infrequent single spikes between stimuli. Nevertheless, between and within cells there was considerable variation in the magnitude of responses, i.e. number of spikes evoked by any given stimulus. For analysis therefore the response of each cell was calculated by summing the evoked spikes for each six stimuli per minute of recording; these minute totals were then normalized to the mean spikes-per-minute value for the 10 min immediately prior to infusion. The ‘percentage of pre-infusion response’ values thus obtained for each cell for each minute throughout each experiment (saline infusion and GTN infusion) were subjected to two-way repeated measures analysis of variance (
Activation threshold experiments
A small electric drill with a dental burr was used to thin the skull at appropriate points overlying the vasculature. Small holes were drilled until only a thin wafer of bone remained over the dura. The bone wafer was then gently fractured using a 23G needle point. A fragment of saline-soaked Sterispon gelfoam was then placed in each hole to ensure good contact for the stimulating electrodes and the entire area was then covered in a film of mineral oil to prevent drying and minimize the spread of electrical stimuli to surrounding tissue.
Single shocks were thereby delivered to the dura overlying or adjacent to the superior sagittal sinus and middle meningeal artery to locate neurones in the NTC receiving input from these vascular structures. Threshold stimulus required to elicit firing of each trigeminal neurone with dural afferent input was determined. In each experiment the protocol was to continuously evaluate as many single units as possible during 30 min prior to, 30 min during and 30 min after infusion of GTN or saline. In the animals receiving GTN infusion, six neurones evaluated immediately prior to the start of infusion were held and tested again within 15 min after the start of infusion. The mean threshold±standard error of the mean (SEM) was calculated for the cells recorded prior to infusion and for the combined group recorded during infusion and post-infusion. Student's t-test was then used to compare pre-infusion threshold with during/post-infusion threshold within and between saline and GTN experimental groups. The aim of this protocol was to provide large population samples and to include a subset of neurones recorded both prior to and during GTN infusion.
C-FOS induction
In animals prepared as above, GTN was infused for 30 min. Saline was infused in place of GTN in controls. One-and-a-half hours after the end of infusion, animals were perfused transcardially with 200 ml of saline, followed by 500 ml of 4% paraformaldehyde in 0.1
Capsaicin-treated animals
Capsaicin causes pain by activating C fibres when injected intradermally. Infraorbital injection of capsaicin was therefore employed as a positive control to elicit neuronal activation and FOS expression in the NTC (29). Immediately following the infusion period, a 50 μl bolus of 0.02% capsaicin was injected intradermally into the infraorbital region on the right side of the face, using a Hamilton syringe with 30-gauge needle.
FOS staining
Cryostat sections (20 μm) were cut coronally from the obex to a distance approximately 5.52 mm caudal to this level. Every sixth section cut was mounted on Superfrost slides and air-dried. Slides from control and experimental animals were processed simultaneously for FOS immunocytochemistry. Phosphate-buffered saline (0.2
Drugs and other chemicals
Glyceryl trinitrate (Nitronal, Lipha Pharmaceuticals, West Drayton, UK) was diluted from 1 mg/ml in water to 10 μg/ml with 0.9% saline. An infusion rate of 3 ml/hour was used to give a standard infusion volume of 1.5 ml in 30 min to all animals, thereby delivering a dose of 2–2.5 μg/kg/min GTN in rats weighing 200–250 g. This dose was considered supramaximal for the headache-inducing effect of GTN (see Iversen et al. 1989). Where neuromuscular blockade was required for artificial ventilation, gallamine (80 mg/kg) was injected intravenously. Capsaicin (Sigma U.K.) was dissolved at a concentration of 0.02% in 10% ethyl alcohol, Tween 80 and saline.
Results
Electrophysiology
Response magnitude
In the first set of experiments, five single units were monitored before, during and after GTN infusion and four during infusion of an equivalent volume of 0.9% saline. All of the neurones studied were located in an area 2.0–3.5 mm caudal to the obex and 1.5–1.8 mm lateral to the midline. The range of recording depths, measured from the surface of the brainstem was 50–310 μm. The majority of cells encountered with periorbital receptive fields, including all those chosen for study, were responsive to both light touch (air-jet or paintbrush deflection of hair) and noxious stimulus (pinch of skin). Electrical stimulation within the receptive field of each cell at an intensity sufficient to activate C fibres (1–10 mA, 2 msec) elicited a response typically consisting of action potentials clustered in early (latency < 15 ms) and late (latency > 30 ms) phases. Due to the very short latency of some of the early phase spikes it was impossible to discriminate them clearly from the stimulus artefact. Therefore, in all cases the number of spikes in the late phase was counted for analysis (Fig. 1).
Of the cells recorded during GTN infusion, the mean latency to the first spike of the late phase was 43 ± 5 ms. With an estimated conduction distance of 25 mm, this corresponds to a conduction velocity of 0.57 m/s, within the range for C fibres. In Fig. 2, post-stimulus-time histograms (PSTH) have been constructed from the responses of two cells recorded before, during and after infusion of GTN and 0.9% saline, respectively. These examples clearly illustrate potentiation of the response to periorbital stimulation following infusion of GTN (2 μg/kg/min, 30 min). By contrast, the magnitude of the other unit's response does not increase following saline infusion.

Cumulative PSTHs constructed from periorbital stimulus-evoked responses of two trigeminal neurones. Each histogram is constructed from 60 sweeps recorded over a period of 10 min.. a, baseline prior to infusion of GTN; b, end/post GTN infusion: recorded over 5 min before and 5 min after end of 30-min GTN infusion. The response is seen to have increased substantially in magnitude. Compared with baseline (c), 30-min infusion of 0.9% saline produces no increase in the magnitude of the response of this neurone to periorbital stimulation (d).
The mean change in response magnitude of four out of five units studied during infusion of GTN is shown in Fig. 3. Four cells were studied as controls where saline was infused in place of GTN. When normalized to pre-infusion baseline, the magnitude of responses over the last 5 min of GTN infusion had increased to 156 ± 11% (n = 4). When GTN- and saline-treated cells were assessed over a 50-min period (10 min pre-infusion to 10 min post-infusion) the magnitude of the post-GTN response was significantly increased in comparison to both the pre-GTN response (P < 0.001) and the control cell response at the end of saline infusion (P < 0.02; two-way repeated measures
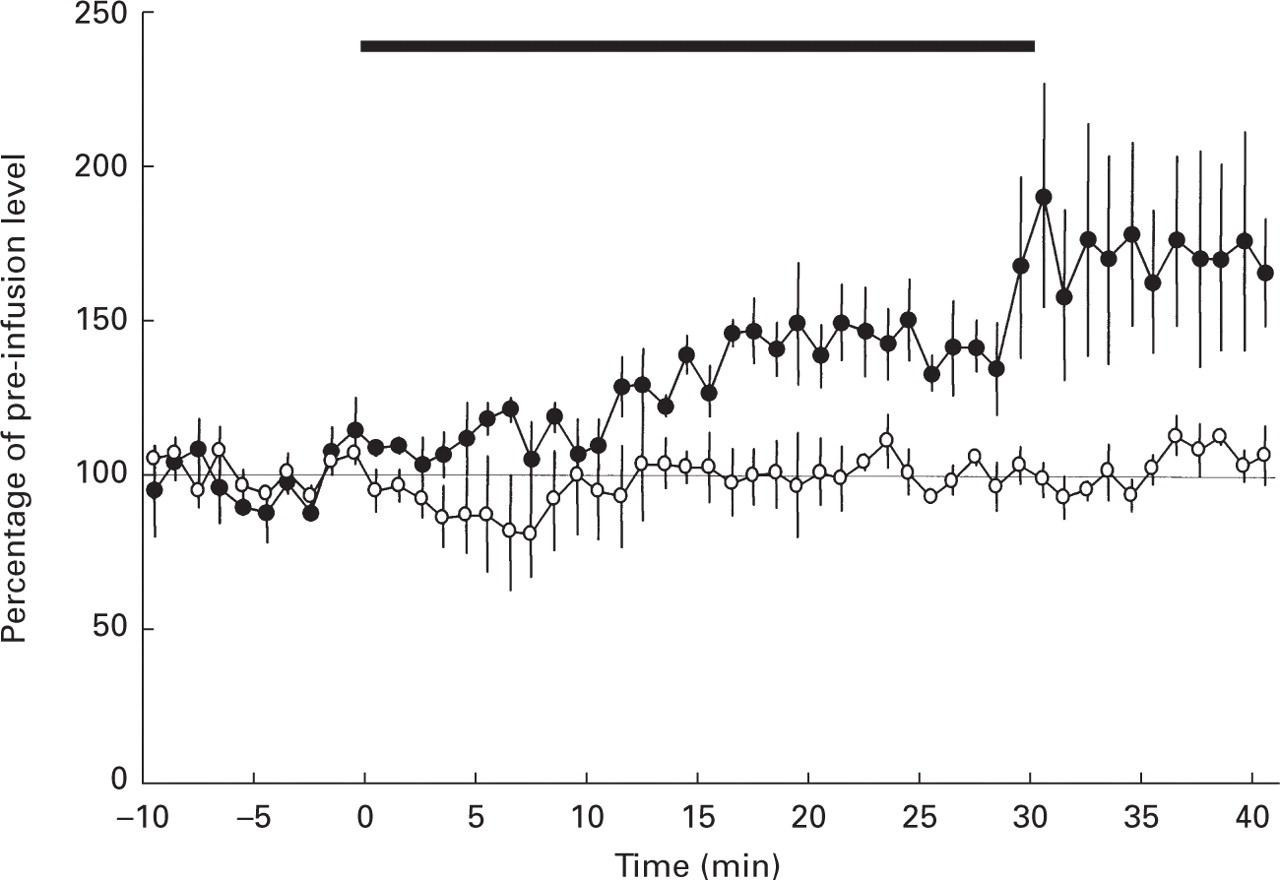
Increase in evoked response of trigeminal neurones during infusion of GTN, 2–2.5 μg/kg/min (•). Saline (○). The solid bar over the record indicates the 30-min infusion period. Each point represents the mean± SEM of four cells; values are normalized to pre-infusion baseline levels. At the end of infusion, in GTN-treated animals, response magnitude was significantly increased over pre-infusion baseline (P < 0.001) and over responses of control cells (P < 0.02).
The response characteristics of neurones studied during saline infusion were similar to those described above, i.e. wide dynamic range, early and late phases of firing, mean latency to first spike of late phase, 45 ± 5 ms. There was no significant change in response magnitude of these cells during or after the infusion period (see Figs 2 and 3).
Activation threshold
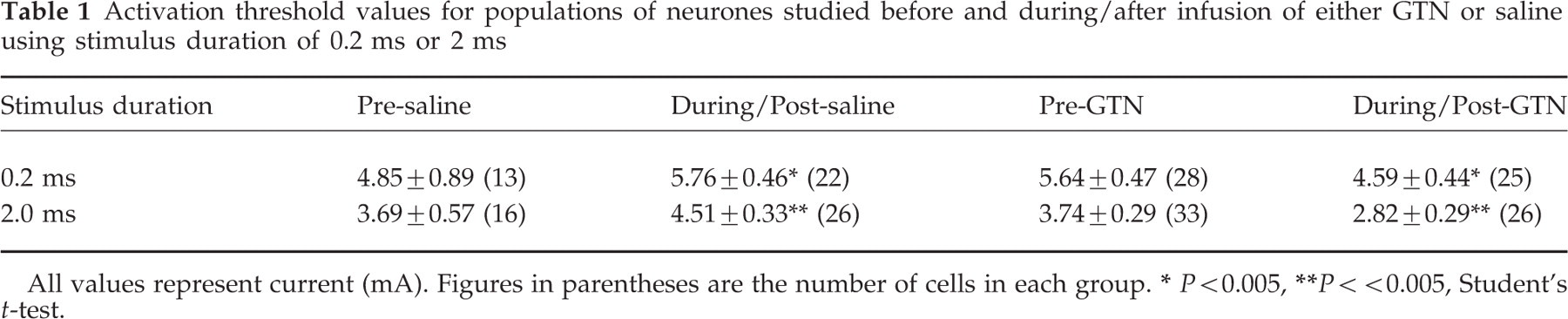
Dural afferent stimulus intensity required to activate NTC neurones was measured in 35 units before GTN infusion and 29 units during or within 30 min of stopping GTN infusion. Six of these cells were evaluated both before and during GTN infusion. Current intensity required to elicit a minimum response (usually a single spike) was first measured using a 2-ms pulse. Subsequently, where possible, to ensure that a short latency spike had not been obscured by the lengthy stimulus artefact, threshold was also measured with a 0.2-ms pulse. The threshold values obtained in saline and GTN infusion experiments are given in Table 1 and compared in the histogram in Fig. 4.

Effect of GTN on dural stimulus activation threshold of NTC neurones. □ Pre-GTN or saline infusion; ▪ Post-infusion. During or up to 30 min after a 30-min infusion of GTN, the current intensity required to activate NTC neurones was reduced in comparison to activation current measured during/after infusion of saline (∗P ≪ 0.005, Student's t-test). Stimulus duration, 2 ms; figures in parentheses denote numbers of cells.
Activation threshold values for populations of neurones studied before and during/after infusion of either GTN or saline using stimulus duration of 0.2 ms or 2 ms
All values represent current (mA). Figures in parentheses are the number of cells in each group.
P < 0.005,
P < < 0.005, Student's t-test.
Compared with the mean pre-infusion levels, activation thresholds measured during and after GTN infusion were reduced though this difference was not significant. In the case of the six neurones evaluated both before and after the start of GTN infusion, threshold was reduced, but again, not significantly. This subset of data has therefore not been presented separately.
In control experiments infusing 0.9% saline, a small, consistent but not significant increase in threshold was observed during/post-infusion compared with pre-infusion. However, the difference between mean threshold of cells recorded during/post-GTN and of those recorded during/post-saline, was highly significant (P < 0.005, Student's t-test) for both 0.2-ms and 2-ms stimuli.
The magnitude of the response at threshold (i.e. the minimum response) remained, in almost all cases, a single spike. There were no differences in spatial distribution between any of the groups studied. The range of recording depths of cells studied was: pre-GTN, 110–850 μm (398 ± 33 μm mean± SEM); during/post-GTN, 50–750 μm (368 ± 36 μm); pre-saline, 100–670 μm (343 ± 38 μm); during/post-saline, 100–970 μm (415 ± 42 μm).
FOS immunocytochemistry
Incubation of tissue with the primary anti-FOS antibody, resulted in specific staining of FOS-IR cells (see Figs 5 and 6). Negative control sections treated with normal rabbit serum in place of the primary antibody, produced no positive staining. Positively staining cells were mostly located in the NTC, the nucleus reticularis lateralis and the nucleus tractus solitarius. Staining in these areas occurred reproducibly in all animals and was used as an internal positive control for FOS-IR.
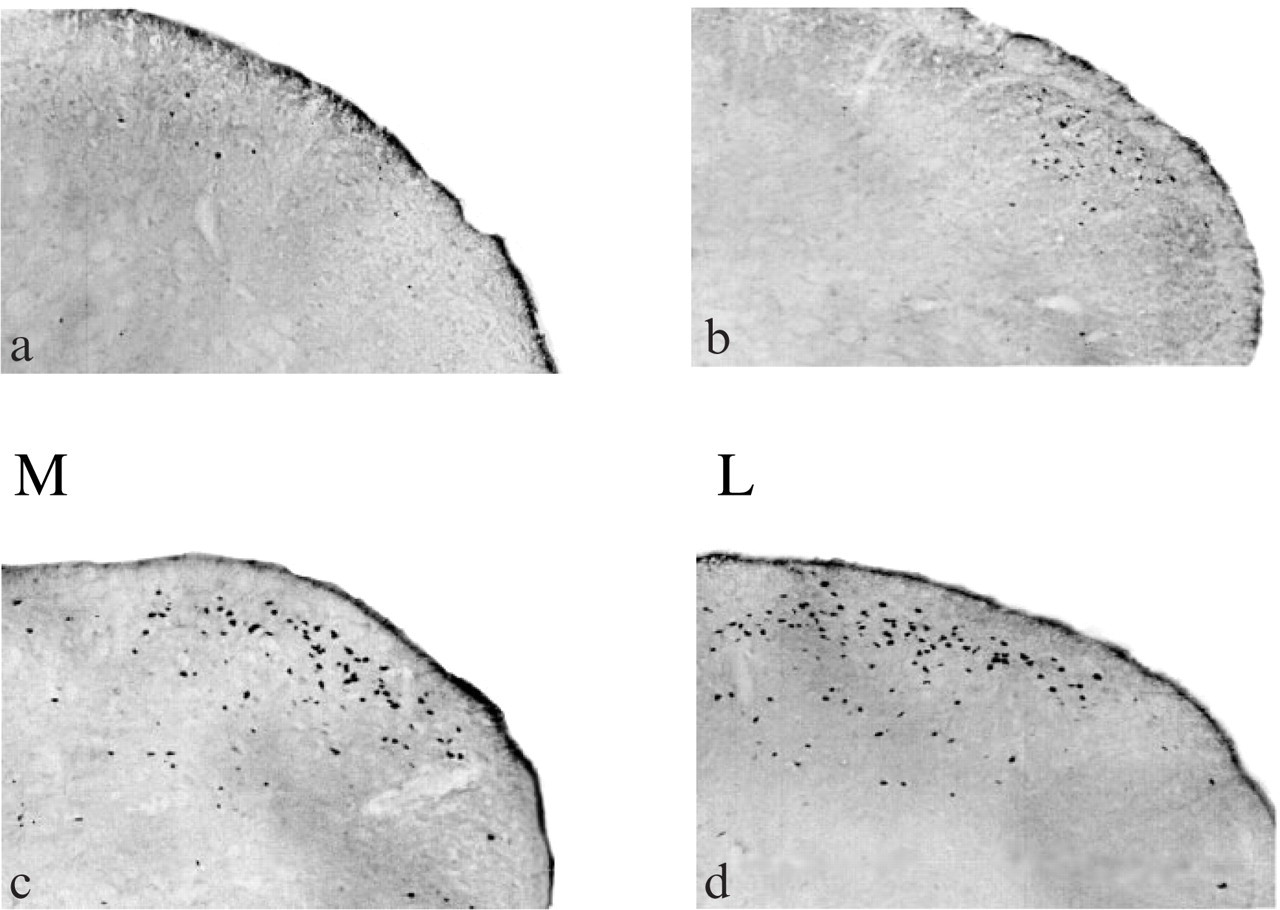
Photomicrographs showing neurones with FOS immunoreactivity within superficial laminae of the NTC at a distance of 3–3.5 mm caudal to the obex. Sections were taken from the right side of the animal, ipsilateral to capsaicin injection. ‘M’ and ‘L’ denote, respectively, medial and lateral aspects of the sections. A, saline infusion; B, GTN infusion; C, saline followed by periorbital intradermal capsaicin; D, GTN followed by capsaicin.
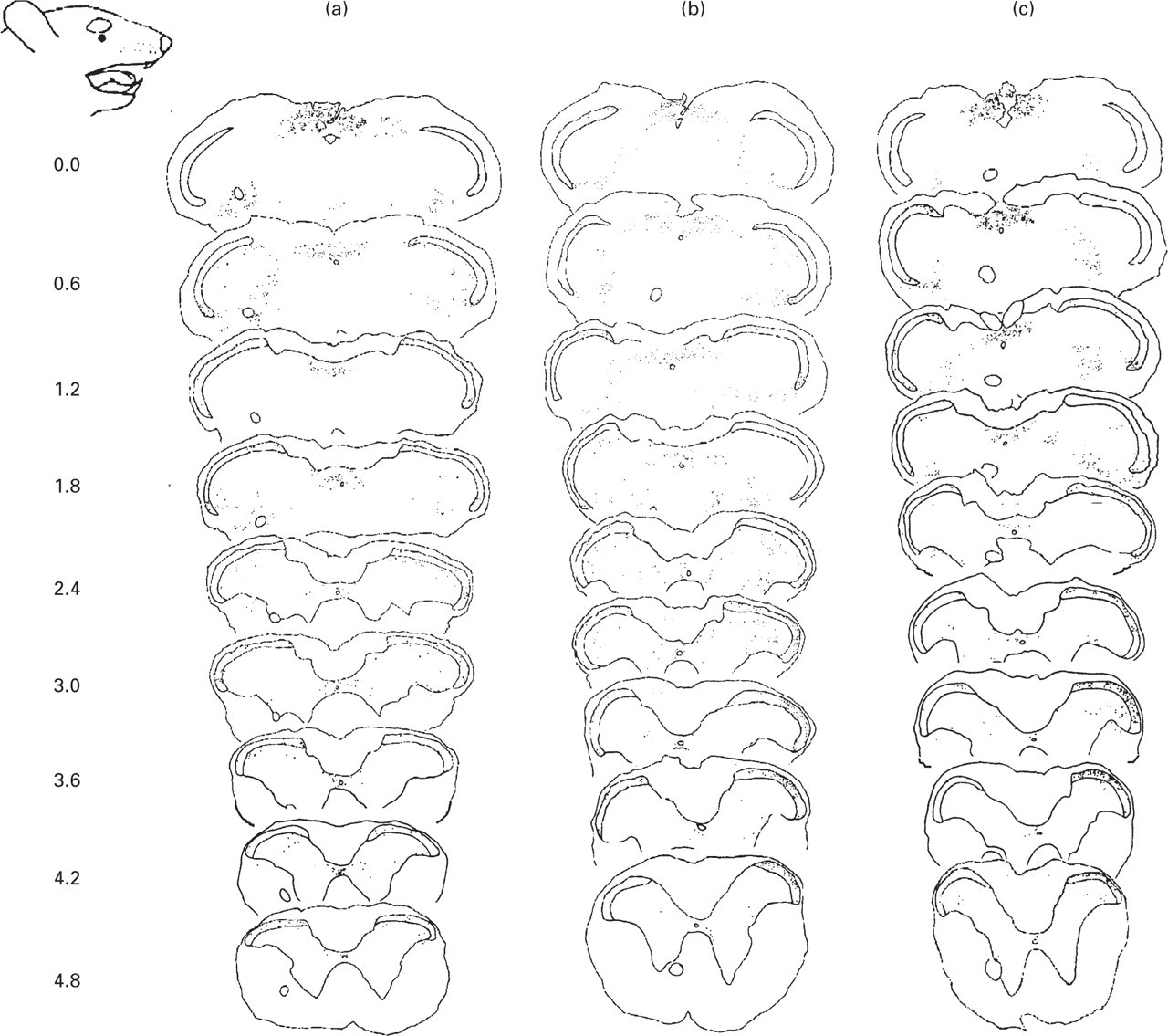
Camera lucida drawings of coronal sections from rat brainstem and upper cervical spinal cord, at distances extending caudally from the obex (marked to the left of the sections). The drawings show the distribution of FOS-IR following: a, GTN infusion; b, saline infusion followed by intradermal injection of capsaicin to the right periorbital area; c, GTN infusion followed by capsaicin. The approximate site of the capsaicin injection is indicated on the rat face profile. Pinholes in sections were used to mark the contralateral side to the capsaicin injection.
GTN vs. saline and immediate perfusion
A small peak in labelling (occurring at approximately 0.72–1.32 mm caudal to the obex) is present in all experimental groups (Fig. 7). Of roughly equal magnitude in all groups, it suggests that FOS-IR at this level is unrelated to the experimental protocol. No other peaks in labelling were discernible in tissue from animals either infused with GTN (n = 3), with saline (n = 3) or perfused immediately following anaesthetization (n = 3). In the GTN-infused animals, the overall mean cell count/section of 10 ± 1 was not significantly different from that of either the saline-infused (9 ± 1) or the immediately perfused (8 ± 1) animals (Fig. 8). Clearly, basal levels of FOS expression in NTC were affected neither by GTN infusion nor by animal preparation (handling, anaesthesia and preparative surgery).

Changes in mean FOS-IR cell counts at different rostrocaudal levels of the NTC from three groups of animals: A. GTN alone evokes little FOS expression. The counts and distribution were not significantly different from those of saline-infused animals (not shown). B. In animals undergoing sham-operations or infused with saline, periorbital capsaicin injection produces a peak in FOS expression at 3–4 mm from obex; at all points between the upward arrow-heads counts are significantly increased over those from animals infused with saline or GTN alone (P < 0.05,
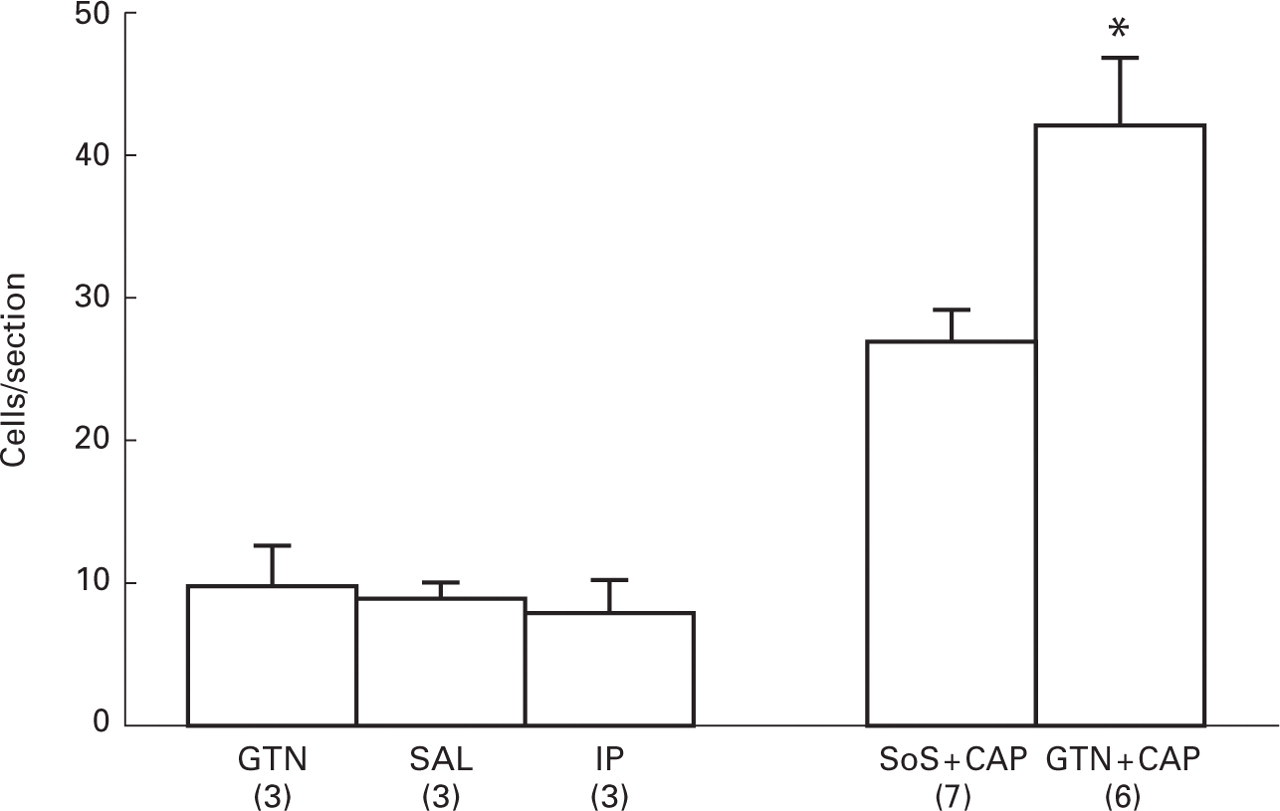
Levels of FOS-IR recorded from different experimental groups. No significant difference was observed when comparing counts from animals infused with either GTN or saline (SAL) or immediately perfused (IP) after anaesthetization. In saline-infused or sham-operated animals, periorbital capsaicin injection (SoS + CAP) produced a large increase in FOS-IR which was significantly potentiated by GTN (GTN + CAP; two-way
Capsaicin-treated animals
In these experiments, infraorbital capsaicin injection was used as a positive control to evoke neuronal activation and expression of FOS-IR in the NTC. In sham-operated (n = 3) and saline-infused (n = 4) animals periorbital capsaicin injections produced a large increase in FOS-IR ipsilaterally within the superficial layers of the NTC. The increase in FOS-IR was significant with respect to both non-capsaicin-treated controls and GTN-infused animals at between 2.76 and 4.44 mm caudal to obex (two-way
When capsaicin was injected immediately after GTN infusion, FOS labelling in the NTC increased significantly above the level attributed to capsaicin treatment alone (Figs 7 and 8). The number of FOS-IR cells/section rose overall by 56% from 27 ± 2, recorded for control capsaicin-treated animals, to 42 ± 2 for GTN and capsaicin-treated animals.
Discussion
Nitric oxide-donating compounds precipitate migraine headache attacks (6) and the severity of migraine during spontaneous attacks is significantly reduced by inhibition of NO synthase (7). In this study we have demonstrated that exogenous NO increases the response of trigeminal neurones to stimulation of facial afferents and reduces the intensity of dural afferent stimulation required to activate these cells. Additionally we have shown that c-FOS induction in NTC by chemical stimulation of facial afferents is amplified by exogenous NO. We will discuss the likely mechanism and locus or loci of these actions in relation to current knowledge of the putative role of NO in the pathogenesis of migraine.
Peripheral action?
Laser Doppler flow measurements have been used to show that doses of GTN which evoke cranial pain also cause significant dilation of cerebral arteries with a time course similar to that of the pain (6). Moskowitz's group had earlier shown that unilateral topical application of GTN to feline cerebral arterioles caused dilation, which was attenuated by an antagonist of calcitonin gene-related peptide and abolished by ipsilateral trigeminal ganglionectomy (33). Sumatriptan, which seems to exert its anti-migraine effect by inhibiting afferent peripheral terminals (34–36), reduces GTN-evoked headache (3) and blocks GTN-induced cerebral dilation (37). All of this suggests that GTN-evoked headache arises at least in part through an action on perivascular afferent terminals. In our experiments therefore NO derived from GTN may be acting to excite perivascular nociceptor terminals. Concomitant release of CGRP may then dilate vessels leading, in turn, to further excitation of the sensory terminals.
Before GTN infusion there is unlikely to be a significant excitatory input to the NTC from the small-calibre dural perivascular afferents (18); therefore, responses of neurones to stimulation of facial skin is probably generated by signals from these afferents alone. During GTN infusion, in addition to being driven by facial afferent stimulation NTC neurones may now receive convergent traffic from dural afferents, resulting in an enhanced response (see 21). Similarly, if NO derived from GTN acts upon meningeal terminals, an electrical stimulus of a given strength delivered to the dura overlying the vasculature may recruit more C fibres. If so, the result would be transmission into the NTC of a more powerful afferent volley, which in turn would be more likely to result in firing of neurones.
Central action?
In addition to the likely sensitization of peripheral terminals, the possibility of a central action of NO in our experiments should be considered. NO-producing cells are abundant in the superficial laminae of the spinal cord (38–40) and production of NO is implicated in the development and maintenance of persistent hyperalgesia through central sensitization (41–45). NO donors delivered intrathecally evoke long-lasting hyperalgesia (46) and systemically administered GTN, which readily crosses the blood–brain barrier (47), rapidly reverses depression of central neurone firing rate by the NO synthase inhibitor NG-nitro-
These findings suggest a highly labile system in which the physiological effects of exogenous NO become apparent almost immediately, such that a 30-min infusion period would be time enough for exogenous NO to increase the gain of NTC neurones responding to either dural or facial afferent signals, or both. If this were the case then it might be expected that dorsal horn nociceptive transmission could be ubiquitously facilitated by systemic GTN. We have not attempted to investigate this. However, in their early investigations, Iversen et al. (5) reported that volunteers experiencing GTN-induced headache reported no pain from any other part of the body; nor does there appear to be any evidence to suggest that GTN reduces nociceptive thresholds anywhere other than within the myofascial tissue of the head (49). Therefore the notion of a central component to the action of NO in this model remains speculative.
Induction of FOS
Previous reports of GTN-induced trigeminal FOS expression describe studies in which GTN was administered subcutaneously as a single dose of 10 mg/kg (27, 28, 50). These reports present a picture of FOS expression in which multiple factors, including baroreceptorial activity and direct activation of central neurones, are likely to contribute to the widespread expression in many brainstem nuclei and higher centres.
Our study differs from those of Tassorelli et al. in two important respects. First, we examined FOS expression at a shorter latency (1.5 h) after GTN administration than the time point (4–5 h) chosen by Tassorelli et al. Whilst we saw no expression of FOS above basal levels in superficial NTC 1.5 h after GTN infusion, we cannot rule out the possibility that FOS expression may have occurred later. Second, the dose of GTN which we used, whilst supramaximal to the dose producing maximum headache in volunteers (5), was still much lower than that used by Tassorelli et al. (27, 28, 50). Nevertheless, were intracranial perivascular afferents to be sensitized by GTN, leading to activity in second order neurones in the same manner in the rat as is apparently the case in human volunteers, the expectation is that this would be revealed in elevated FOS levels at the appropriate trigeminal locus. Substantial FOS expression is induced in NTC by stimulation of the intracranial vasculature (14, 32) or meninges (51). This expression is attenuated by the anti-migraine drugs dihydroergotamine (52) or sumatriptan (35, 53), in the latter case presumably by an action on peripheral terminals as sumatriptan is reported to be active centrally only after disruption of the blood–brain barrier (54).
Notwithstanding the absence of a FOS signal in response to GTN alone, the FOS response to infraorbital injection of capsaicin was clearly potentiated by GTN. Therein lies the rationale for the use of capsaicin as the generator of a signal in the NTC. Whilst GTN at this dose apparently does not, in the anaesthetized rat, excite meningeal afferents sufficiently to evoke activity in trigeminal 2nd order neurones, it nevertheless modulates the system. This may occur via direct action of NO upon NTC neurones, though, as discussed earlier, the lack of evidence to support reduction of thresholds to nociceptive stimuli anywhere other than the head weighs against this idea. It seems more likely that by generating input from meningeal afferents to produce sub-threshold depolarization of NTC neurones, GTN may facilitate transmission at these synapses, in our experiments thereby amplifying a signal generated in cutaneous afferents by capsaicin.
In our electrophysiological studies, we purposely selected neurones that were quiescent between stimuli. That none of these cells began to fire spontaneously during or after GTN infusion is in line with the results of our FOS experiments, suggesting that GTN, at this dose, does not acutely evoke frank excitation of NTC neurones. To further explore the nature of these effects, one approach would be to record directly from dural perivascular afferents, either from teased fibres (see 19) or in the trigeminal ganglion (18), to determine whether normally quiescent afferents begin to fire during GTN infusion. Alternatively, one could repeat the experiments described here and seek to block the increase in evoked responses and FOS induction with sumatriptan, which has been shown to reverse GTN-induced elevation of cortical NO (55). Depression of these effects would suggest a peripheral site of action; preliminary studies in our laboratory support this latter hypothesis (56).
In conclusion, we have demonstrated that the NO donor GTN, at doses that cause immediate headache and later precipitate migraine headache in human volunteers, facilitates transmission of afferent signals in the caudal trigeminal nucleus in anaesthetized animals. Work is under way in our laboratory to ascertain the site or sites of this effect and the mechanisms involved.
Footnotes
Acknowledgements
The authors wish to acknowledge Miss Vivien Cheah for excellent technical support. Thanks also to SmithKline Beecham who funded Martyn Jones and Isobel Lever. Support was also provided by the Medical Research Council. These data were presented in part at the British Pharmacological Society meeting, January 2000.
