Abstract
Introduction: Preventive treatment with topiramate is effective for overall reduction of migraine frequency, but there are few data regarding its efficacy on perimenstrual migraines. To determine whether topiramate can prevent perimenstrual migraines, we analyzed data from premenopausal women as a subgroup of the Prolonged Migraine Prevention with Topiramate (PROMPT) study.
Methods: In total, 198 women from the PROMPT study with menstrually related migraine (MRM) were evaluated. After a one-to-two–month prospective baseline period, patients received open-label topiramate (50–200 mg/day) for six months.
Results: During topiramate treatment, mean monthly migraine frequency was reduced from 7.03 at baseline to 4.36 (mean change: −2.66; p < .001, endpoint analysis). Mean percentage reductions were similar for migraines during and outside the perimenstrual period (−45.9% and −46.1%, respectively). In patients with aura, reductions in migraine days with (−48.3%) or without (−43.4%) aura were similar to those in patients without aura (−45.4%). Reductions were also similar whether women were taking combined oral contraceptives (−47.0%) or were not (−46.6%).
Conclusions: Topiramate reduces the frequency, but not severity or duration, of perimenstrual migraines in women with MRM, including migraines with and without aura, and regardless of combined oral contraceptive use.
Introduction
Migraine is a common neurological disorder that runs in families and appears to have a substantial hereditary component (1). It affects around three times as many women as men, with estimated yearly prevalences of 18% and 6%, respectively (2,3). In women, prevalence of migraine is generally highest between puberty and menopause; it has been suggested that hormonal changes during the menstrual cycle are a major trigger in those susceptible to migraine (4–9). Perimenstrual migraines are said to differ from migraines outside this period in that they are more intense, of longer duration, and more difficult to treat with acute medications (10–12).
Several medications are recommended for prevention of migraine, including selected members of classes as varied as beta-blockers, calcium channel antagonists, serotonin antagonists, serotonin reuptake inhibitors, tricyclic antidepressants and anticonvulsants (13–15). Of these, the anticonvulsant topiramate has the highest level of evidence for efficacy and tolerability (16–18). While there is evidence for effectiveness of these agents in the overall reduction of migraine frequency, little is known about their efficacy on perimenstrual migraines in particular. To assess whether topiramate was able to affect the occurrence of perimenstrual migraines, in addition to its well-known capability of reducing migraine frequency overall, we analyzed the results obtained in premenopausal women as a subgroup of one of the largest migraine prevention trials to date: the Prolonged Migraine Prevention with Topiramate (PROMPT) study (19).
Methods
This was a post-hoc analysis of the PROMPT trial (protocol TOPMAT-MIG-303; EudraCT number 2005-000321-29), details of which have been published (19). In brief, adults with history of migraine for at least one year, with a mean of ≥4 migraine days per month during the three months before trial entry, were eligible. Patients were excluded if they had used migraine prophylactic medication in the month before trial entry, or had experienced poor or no efficacy with more than two regimens of migraine prophylactic medication. Women who were pregnant or breastfeeding were excluded, and all women of childbearing age were required to have a negative pregnancy test before enrollment and to confirm that they would use adequate contraception throughout the study. Detailed inclusion and exclusion criteria have been described previously (19). All patients provided written informed consent. The trial was carried out in accordance with guidelines on Good Clinical Practice from the International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH), and was approved by local ethics committees.
Patients started the study with a four-to-eight-week prospective baseline (PB) phase with no preventive study medication. Occurrences of migraines (including start and end times) and auras, acute medication use for migraine headaches, and the days of menstruation (where applicable) were recorded in a diary. Patients then entered a 26-week open-label phase in which they received topiramate (titrated to 100 mg, with further adjustments to 50–200 mg/day permitted). Patients completing the open-label phase entered a 26-week double-blind phase in which they were randomly assigned to continue their final topiramate dose or switch to placebo.
For the subgroup analysis on menstrually related migraine (MRM), women qualified if they were premenopausal, aged <50 years and experienced migraine during the perimenstrual period (PMP) in at least two of the first three cycles in the study. Participants were also required to have diary records with complete menstrual data for the first three cycles of the study and at least the last two cycles of the open-label phase, and complete migraine data in the PB and open-label phases (Figure 1). Women with chronic migraine (≥15 migraine days per four weeks) and women with a cycle length >40 days in any of the initial three menstrual cycles in the study were excluded.
Composition of patient set for post-hoc analysis. MRM = menstrually related migraine.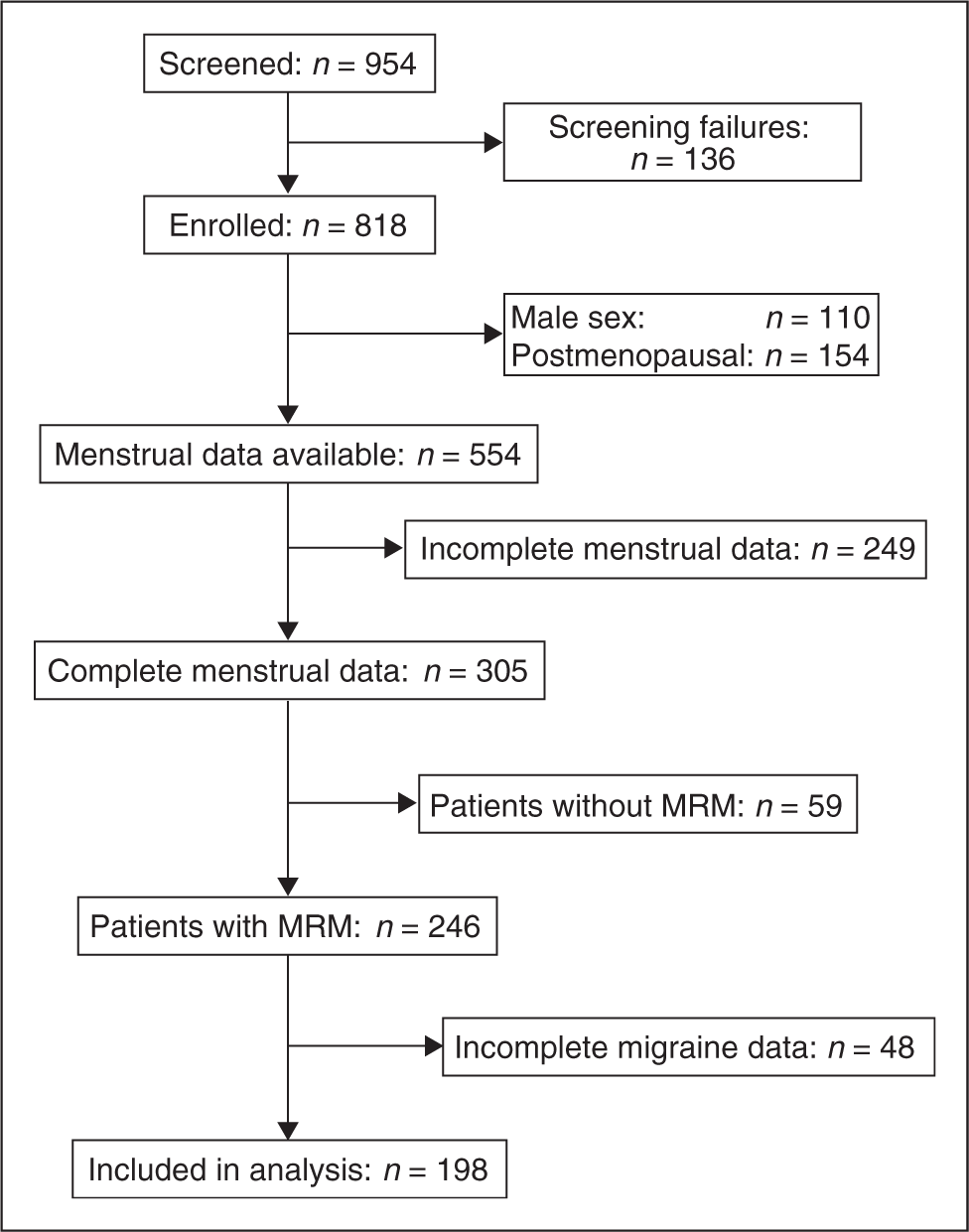
Definitions
A “migraine” was defined as a continuous period of migraine headache, and a “migraine day” was defined as a calendar day during which the patient experienced migraine headache at any time (19).
The PMP was defined as the day of menstruation start, plus the previous and subsequent two days (i.e. a five-day period around the start of menstruation) (12,20). “Menstrually related migraine” was defined as migraine during the PMP in at least two of three menstrual cycles and additionally at other times of the cycle, as per the Appendix of the International Classification of Headache Disorders, second edition (ICHD-II) (20). The initial three menstrual cycles in the study determined whether women were classified with MRM. The start date of each migraine was used to determine whether it belonged to the PMP. All migraines, with or without aura, were considered for the analysis during the PMP and the non-PMP. Qualifying women were additionally categorized either as “patient with aura” (MA) if they experienced aura at least once during the study, or “patient without aura” (MoA), in accordance with previously described methods (21).
Cycle length normalization
In the analysis, menstrual cycles started with a PMP and ended the last day before the next PMP started. To improve inter-individual comparability of the data, menstrual cycles were “normalized” to 28 days: each cycle consisted of a five-day (fixed) PMP and a (variable) non-PMP. To enable full monthly (28-day) migraine frequencies by adding PMP and non-PMP migraine frequencies, non-PMP frequency data were divided by the remaining cycle length (i.e. full cycle length in days, −5) and multiplied by 23. Migraine duration and severity data were based on the original, non-normalized cycles.
For day-by-day presentation of normalized migraine frequencies during baseline and topiramate treatment (Figure 2), the first and last complete cycles of the PB and open-label periods, respectively, were used.
Frequency distribution of migraine days at baseline (first cycle) and at the end of the 6-month topiramate treatment phase (last cycle) in women with menstrually related migraine (N = 198). For each day of the menstrual cycle, normalized to 28 days, the fraction of patients with a migraine day is presented. PMP = perimenstrual period.
Analyses and statistical methods
Migraine severity was recorded using a three-point scale (1 = mild, 2 = moderate, 3 = severe). For each patient in any given period (i.e. PMP, non-PMP or total four-week period), migraine severity was defined as the sum of the severity scores for the migraines that occurred in that period, divided by the number of migraines. Migraine duration (in hours) was defined in a similar manner to migraine severity. Percentage changes from baseline were analyzed as symmetrized percentage change, calculated as 100 × (endpoint − baseline)/(endpoint + baseline), unless endpoint + baseline was equal to zero, in which case percentage change was defined as zero. Values presented in the text are the symmetrized percentage changes transformed back to regular percentage changes.
Frequencies of migraines and migraine days, and means of migraine duration and migraine severity, are presented for the first four weeks of the PB (“baseline”) and the last four weeks of open-label topiramate treatment (“endpoint”); it was not necessary for either period to start with the PMP. Change and percentage change from baseline to endpoint were calculated for each patient, and values were averaged to obtain the means for all patients. Within-group changes were tested for significance using the Wilcoxon signed rank test and using an alpha-level of 5% two-sided. In accordance with the exploratory nature of this post-hoc analysis, no corrections for multiple comparisons were made.
Results
Patients
Baseline demographics and disease characteristics in the subgroup of women with MRM compared with the full study population

MRM = menstrually related migraine. SD = standard deviation.
Cycle lengths
The mean (standard error [SE]) length of patients’ first cycle in the study was 27.4 (0.22) days (range, 19–37 days; mode, 28.0 days) (Supplementary Table 1). On average, cycle length did not change during topiramate treatment; mean length of the last cycle was 27.7 (0.28) days (range, 16–42 days; mode, 28.0 days), and the change in cycle length versus baseline was not statistically significantly different from zero (mean, + 0.26 days; mode, 0.0 days, confidence interval [CI] −0.38, −0.91 days). During the MRM screening period (the first three cycles), all women had migraines during and outside the PMP. In the first cycle, 27% of migraine days occurred during the five-day PMP and 73% during the non-PMP (Figure 2).
Migraine days and migraines
Throughout topiramate treatment, the number of migraine days, and the numbers for the PMP and non-PMP migraine days separately, decreased significantly versus baseline (Figure 3). At the end of the six-month treatment phase, the mean ( ± Frequency of monthly migraine days during the prospective baseline phase and at the end of the 6-month topiramate treatment phase. Data are shown as means ± SE for the perimenstrual period (PMP), the non-PMP and overall, in women with menstrually related migraine (N = 198). ***p < .001 for change vs. baseline.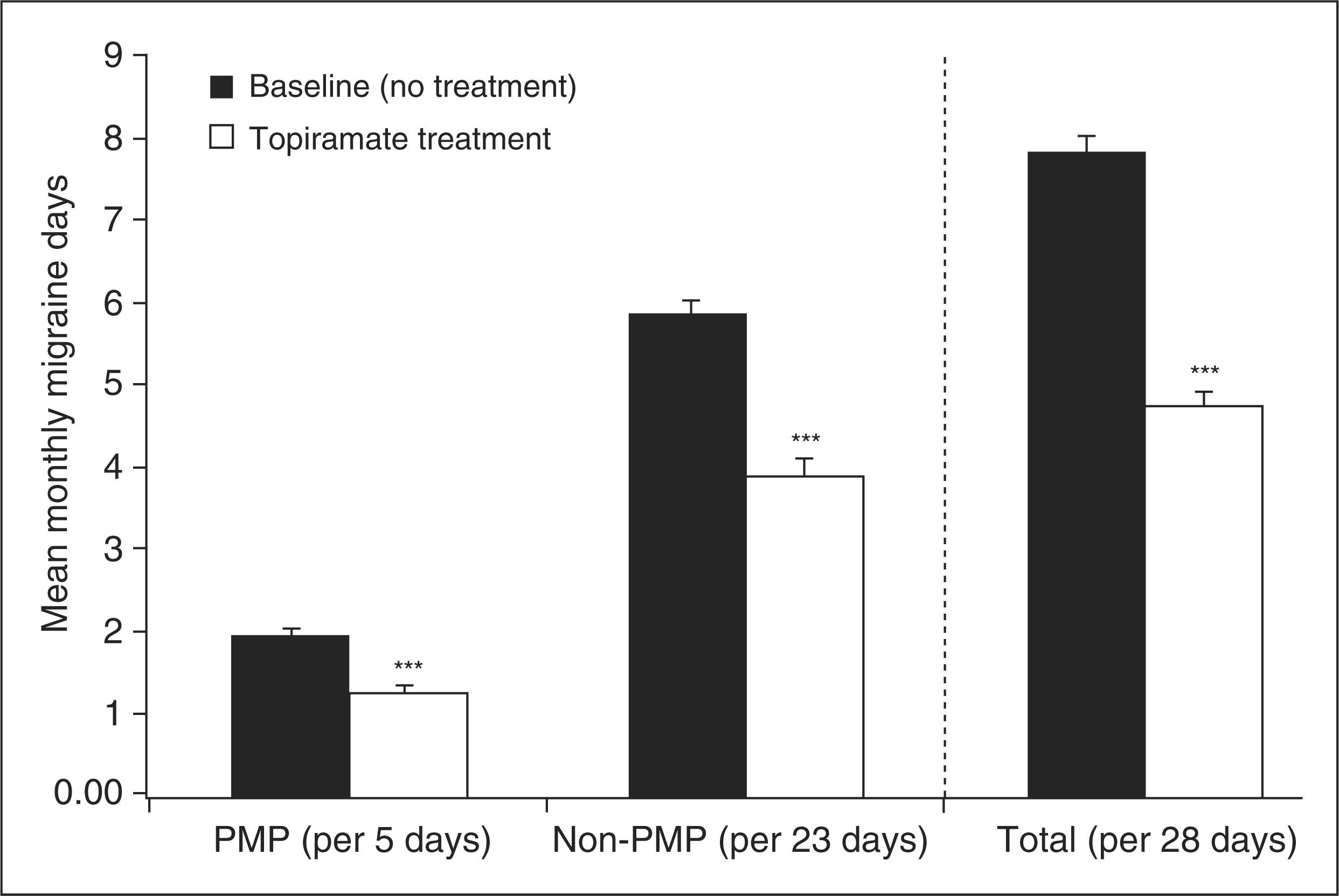
Frequency, severity and duration of migraines during the prospective baseline phase (baseline) and at the end of the 6-month open-label topiramate treatment phase in women with MRM
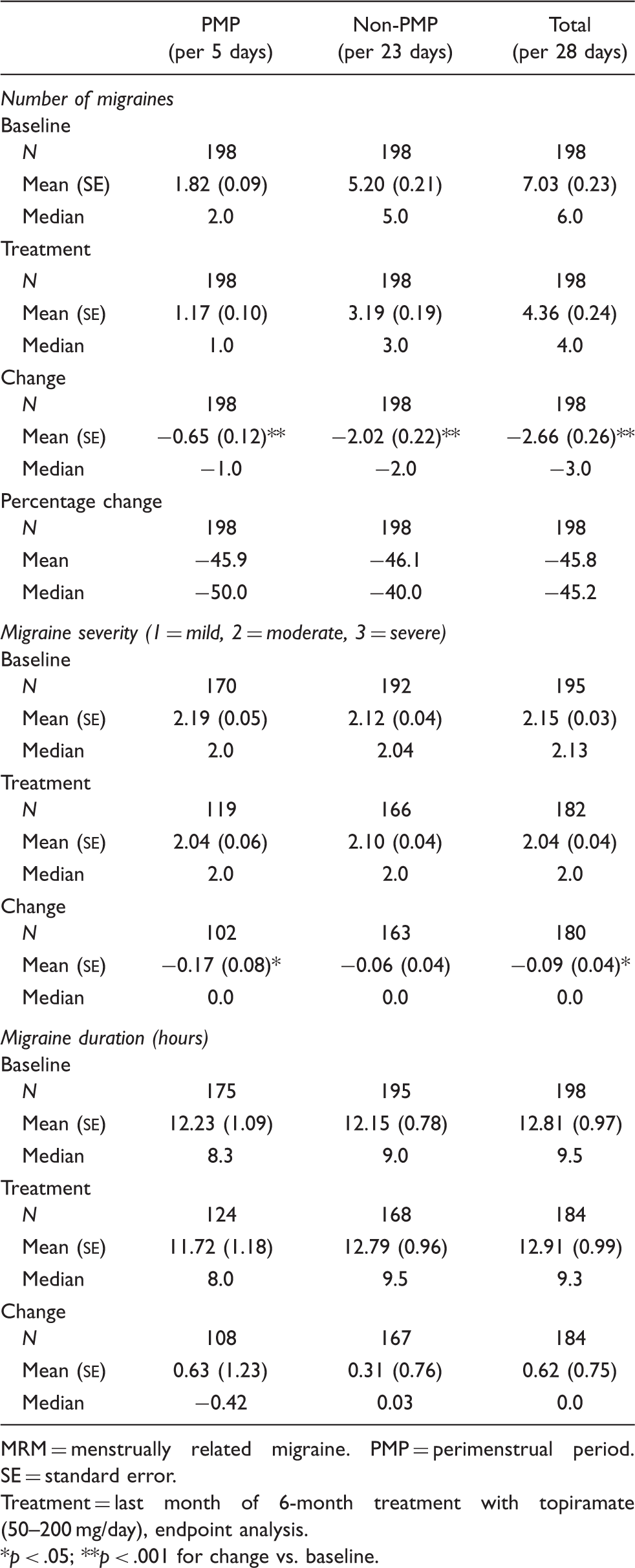
MRM = menstrually related migraine. PMP = perimenstrual period. SE = standard error.
Treatment = last month of 6-month treatment with topiramate (50–200 mg/day), endpoint analysis.
*p < .05; **p < .001 for change vs. baseline.
Auras
Migraine days in the baseline period and at the end of the topiramate treatment period for patients with migraine aura (migraines with and without aura analyzed separately) and without migraine aura
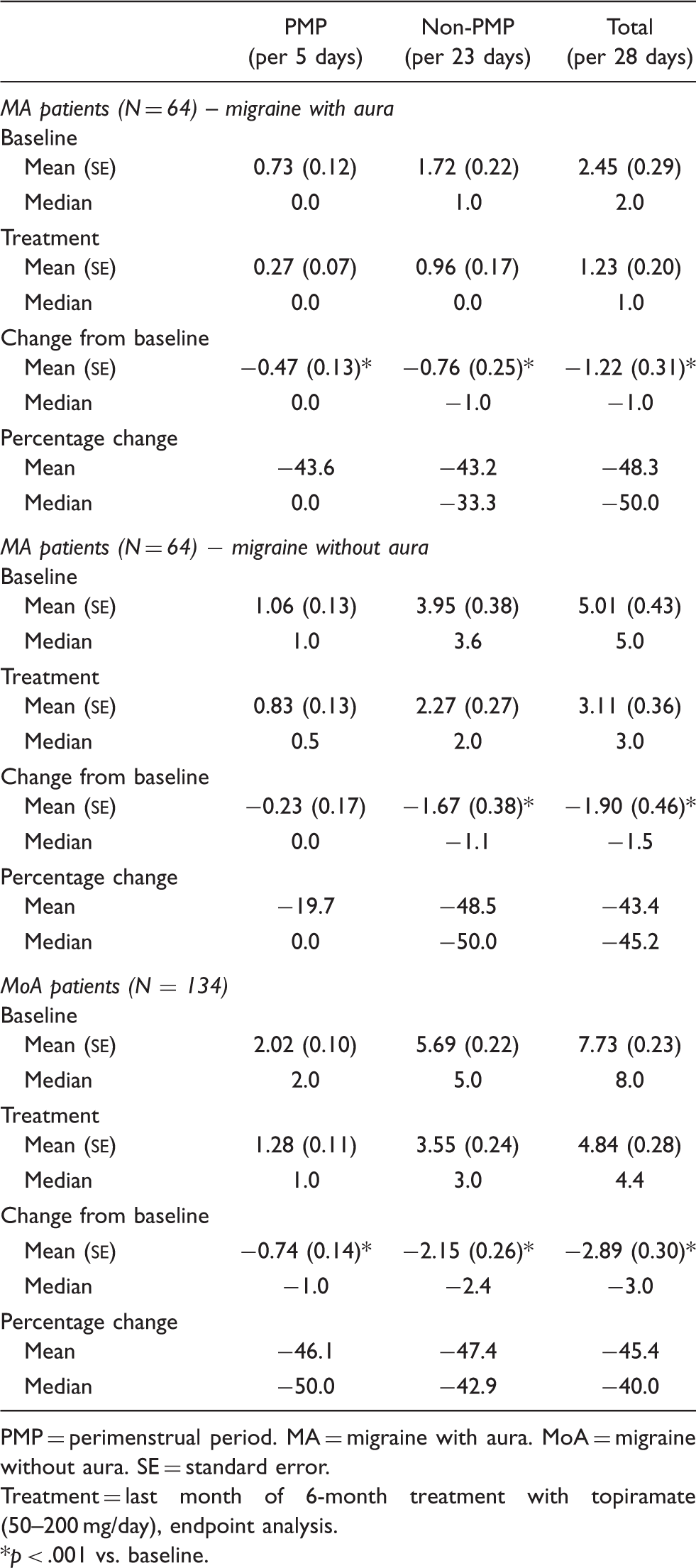
PMP = perimenstrual period. MA = migraine with aura. MoA = migraine without aura. SE = standard error.
Treatment = last month of 6-month treatment with topiramate (50–200 mg/day), endpoint analysis.
p < .001 vs. baseline.
Combined oral contraceptive use
Overall, 58 of the 198 women (29%) used a combined oral contraceptive (COC). In these women, the peak in migraine occurrence around menstruation was more pronounced than in women not taking a COC (an average of 2.24 and 1.86 migraine days per five-day PMP, respectively, at baseline) (Supplementary Table 2). Peak incidence of perimenstrual migraines at baseline was 0.57 (cycle day 2) in women using COCs and 0.42 (cycle day 3) in those not using COCs. The overall reduction in migraine frequency was, however, 47% both in women taking and those not taking COCs. Women taking COCs appeared to experience slightly greater relief of migraines outside the PMP (mean change: −56% vs. −41% in the PMP) (Supplementary Table 2), but this may be due to the variability of these percentages.
Discussion
The present analysis included more than 300 women with complete menstrual data, of whom 81% fulfilled the criteria for MRM (perimenstrual migraines during at least two of three consecutive cycles). Topiramate reduced the number of perimenstrual migraines, which in turn may have reduced the probability of women meeting the MRM criteria. Nevertheless, the very high percentage of MRM in our study group of premenopausal women with migraine supports the view that menstruation is a strong migraine-provoking factor.
Complete migraine and menstrual data were available for almost 200 women with MRM, representing one of the largest MRM trial populations ever analyzed. The results clearly indicate that topiramate treatment reduces the occurrence of perimenstrual migraines, with effectiveness similar to that observed for migraines outside the PMP. Except for frequency, which was clearly higher during PMP than non-PMP, we found no differences in duration or severity between PMP and non-PMP migraines. Topiramate treatment did not affect migraine duration in either period, and reductions in severity were minor, reaching statistical significance for perimenstrual migraines only. These data are consistent with the results for the overall trial population (19). Others have reported a similar lack of effect of preventive treatment on duration and severity for another antiepileptic drug, valproate (22). As the reductions in migraines and migraine days were the same in the PMP and non-PMP (endpoint analysis after six months: 46% for migraines, 46–48% for migraine days), we conclude that PMP and non-PMP migraines respond similarly to preventive treatment with topiramate. Indeed, some authors have previously reported no increased severity of migraines during the PMP compared with the remainder of the cycle (reviewed in Martin and Lipton, 2008 (23)).
Literature reports about longer duration and higher severity for perimenstrual migraines (11,12) may, therefore, be explained by the higher frequency of migraines during this period, leading to the subjective perception of raised duration and severity overall in terms of the percentage of time spent with migraine during the PMP relative to the remainder of the menstrual cycle.
At trial entry, a minimum average migraine frequency of once per week was specified, so no conclusions can be drawn about lower frequencies. Women with pure menstrual migraine (i.e. occurring exclusively during the PMP) were therefore absent from the present trial. It would be interesting to investigate whether topiramate is also effective in women with pure menstrual migraine, although migraine prophylaxis might not be appropriate for many of these women, who have a comparatively low migraine frequency.
The similarity of PMP and non-PMP migraines is further underscored by the lack of difference between migraines with and without aura. The ICHD-II classification introduces tentative definitions for menstrual migraine and MRM, based on the assumption that elevated frequency during the PMP is solely the result of an increase in migraines without aura (20). For theoretical and practical reasons, however, it is difficult, if not impossible, to discriminate between migraines (without aura) originating specifically from hormonal changes around menstruation and migraines (with or without aura) belonging to the regular monthly background. Furthermore, data from the present analysis do not support such a division. During the PMP we saw an increased number of migraines with and without aura compared with the non-PMP, and both migraine types were reduced during treatment. The general perception that the rise in frequency during the PMP above the existing background involves only migraines without aura has, to our knowledge, never been supported with robust data. Results from the present trial, which excluded patients with a low (<4 migraine days per month) or a very high (≥15 migraine days per month) migraine frequency, indicate that migraines with aura were elevated during the PMP, similar to migraines without aura. These results contradict the perception stated in many published reports (9,11,24,25), and therefore questions the validity of excluding migraines with aura in the proposed ICHD-II definition of MRM. The discrepancy may be explained by the fact that, in the present study at least, only one-third of patients had migraine with aura, in whom only one-third of migraines were actually preceded by an aura (21). The high likelihood (89%) that any new migraine will be one without aura, therefore, might have led others to conclude that the extra migraines observed during the PMP were all migraines without aura.
Others investigating menstrual migraine and MRM have focused on changes in hormone levels and patterns, and have concluded that the occurrence of migraines is associated with the drop in serum estrogen levels that occurs at the end of the regular cycle and immediately precedes menstrual bleeding (9,12). Similar, or more pronounced, fluctuations occur during use of COCs, and COC use, particularly withdrawal during the medication-free week of the month, has been associated with an increased propensity to develop perimenstrual migraines (26). Indeed, the drop in estrogen levels and accompanying rise in migraine frequency can be prevented by continuous, uninterrupted COC use, although migraines tend to recur when treatment is lifted (26–28). In our analysis, women receiving COCs showed a somewhat higher migraine prevalence during the PMP than those not receiving COCs, in line with previous reports (26). Firm conclusions cannot be drawn from this comparison, however, as these groups were analyzed retrospectively and COC use was not prospectively assigned in a random manner. Topiramate treatment nevertheless appeared to reduce PMP migraine frequency regardless of COC use.
When considering the results of this analysis, it should be borne in mind that PROMPT was not designed primarily to evaluate MRM; menstrual data were collected in a systematic manner to enable post-hoc subgroup analyses. This may be perceived initially as a weakness, but the absence of focus on this matter might in fact have served to prevent any specific bias when enrolling patients into the study (and subsequently into the MRM subgroup analysis). As this was a post-hoc analysis of open-label data, the findings should be confirmed in a randomized, controlled study.
Based on previous reports and recommendations, the present ICHD-II criteria define the PMP as a five-day period, starting two days before and ending two days after the start day of menstrual bleeding (20). Based on the present results, however, the increase in migraine days above what appears to be the background (i.e. the average frequency during the remainder of the menstrual cycle) extends beyond day + 3 even up to day + 5 (Figure 2). For future updates of the ICHD-II classification, results of this study support extension of the definition of the PMP to a seven-day period (from day −2 until day + 5). This has the additional numeric advantage of a one-week PMP versus a three-week non-PMP, thus completing the standard four-week cycle length. Adapting the definition accordingly might help to increase the versatility of the distinction between these periods, and facilitate communication and interpretation of these numbers among health care professionals and with patients in everyday clinical practice.
In conclusion, this analysis of women with MRM as a subgroup of those enrolled in a large migraine prophylaxis and withdrawal study showed that migraines occurring during the PMP appeared to be no different in nature from those outside the perimenstrual period. This finding applied to women using COCs and those using other methods of contraception, and also applied to the duration and severity of attacks, as well as the occurrence or absence of migraine aura. Migraines during the PMP were, however, more frequent than those in the remainder of the cycle. Percentage reductions in migraine frequency during topiramate treatment were similar with regard to PMP and non-PMP migraines, and were also consistent with the overall study population.
Footnotes
Disclosure
The study was funded by Janssen-Cilag EMEA. Gianni Allais, Margarita Sanchez del Rio and Hans-Christoph Diener have participated in clinical trials and advisory boards for Janssen-Cilag. Chiara Benedetto declares no conflict of interest. Barbara Schäuble and Joop van Oene are employees of Janssen-Cilag EMEA.
Acknowledgements
The authors would like to thank the investigators and all others who participated in PROMPT. The trial and subsequent analyses were sponsored by Janssen-Cilag EMEA. Assistance with the drafting and completion of the paper was provided by Daniel Booth (Bioscript Stirling, Ltd.) and funded by Janssen-Cilag EMEA.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
