Abstract
The role of nitric oxide (NO) in migraine has been studied in the experimental glyceryl trinitrate (GTN)-infusion headache model. We hypothesized that GTN-induced headache may activate the trigeminovascular system and be associated with increased levels of sensory neuropeptides, including calcitonin gene-related peptide (CGRP). CGRP, vasoactive intestinal peptide (VIP), neuropeptide Y (NPY) and somatostatin plasma levels were measured before and after placebo/sumatriptan injection and during GTN-induced headache. Following a double-blind randomized cross-over design, 10 healthy volunteers received subcutaneous sumatriptan 6 mg or placebo. This was succeeded by 20 min of GTN (0.12 µg kg−1 min−1) infusion. At baseline no subject reported headache (using verbal rating scale from 0 to 10) and the jugular CGRP-like immunoreactivity (-LI) level was 18.6 ± 2.5 pmol/l. After a 20-min intravenous infusion of GTN 0.12 µg kg−1 min−1, median peak headache intensity was 4 (range 2–6) (
Introduction
Nitric oxide (NO) has been shown to be involved in migraine pathophysiology (1,2), hence the role of NO has been studied in an experimental headache model using glyceryl trinitrate (GTN) infusion (3). The exact mechanism by which NO induces headache and migraine is not fully understood. However, GTN induces an immediate headache during the infusion and this is followed by a late phase in migraine patients resembling genuine migraine attacks after several hours (2,4). GTN is a NO donor, which provides the possibility to evaluate if NO acts directly as a vasodilator or on parts of the pain signalling pathways through an increase of second messengers such as cyclic guanosine monophosphate (cGMP). GTN could also work more indirectly through increased production or release of other signalling molecules (5,6). One of several interesting signalling molecules is the neuropeptide, calcitonin gene-related peptide (CGRP) (7). During spontaneous acute migraine, an increase of CGRP in blood from the external jugular vein was found, which declined after pain relief by sumatriptan treatment (8). These findings during acute migraine attacks have been reproduced in several studies (9–11) where CGRP levels correlated with the severity of pain. In one single, but larger study these findings could not be reproduced, perhaps due to lower pain intensity in the subjects (12). There is no doubt that CGRP plays an important role in migraine pathophysiology, and recent studies have shown that the CGRP antagonists olcegepant (13) and telcagepant (14) are effective in treating acute migraine attacks. In relation to the GTN headache model, Wei et al. (15) found that GTN caused CGRP release and suggested that both the dilation and the headache were caused by CGRP; however, this has later been questioned (16). Therefore, we hypothesize that GTN-induced headache occurs in parallel with an increase in CGRP and/or other peptide signalling molecules. This was tested by measuring the immediate effects of sumatriptan/placebo and GTN on CGRP, vasoactive intestinal peptide (VIP), neuropeptide Y (NPY) and somatostatin in blood from the external jugular and cubital veins in healthy subjects. Since only healthy subjects were included, no significant appearance of a delayed headache was expected, thus a time line of 90 min was chosen for the current study.
Methods
Ten healthy volunteers, four women, six men, mean age 30.2 years (range 26–37 years), mean bodyweight 72.6 kg (range 54–84 kg) were included after informed consent. They had no history of migraine and never or less than 1 day of tension-type headache a month. It was a double blind, randomized, placebo controlled cross-over study in which they were pre-treated with sumatriptan (6 mg) or placebo subcutaneously 15 min before the start of a 20-min GTN infusion. Each subject was investigated twice, with at least 6 days apart.
The study was performed in accordance with the Helsinki Declaration of 1981 and was approved by the Danish National Board of Health and Welfare and the ethical committee of the County of Copenhagen.
On the day of the study the subjects attended the laboratory at 08.30 h. Breakfast, but no coffee, tea, alcohol or smoking was allowed for a period of 12 h before medication. A cannula was inserted into the right cubital vein for GTN infusion. Furthermore, cannula were inserted into the left cubital vein and external jugular vein on the dominant side, respectively, for blood sampling.
After 30 min of rest in the supine position, 6 mg sumatriptan (GlaxoWelcome, Uxbridge, UK) or placebo (isotonic saline) was injected subcutaneously in the deltoid region. After 15 min, GTN (glyceryl trinitrate; SAD, Denmark) 0.12 µg kg−1 min−1 was infused for 20 min by a volume directed pump (Braun Perfusor, Melsungen, Germany). Blood samples were collected at baseline, and at 15, 35 and 95 min after placebo or sumatriptan injection. (See Figure 1 for time line of experiment.)
Time line of experiment. Following collection of baseline blood samples, either 6 mg sumatriptan or placebo was injected subcutaneously. After 15 min, glyceryl trinitrate (GTN; SAD, Denmark) 0.12 µg kg−1 min−1 was infused for 20 min. At the end of GTN infusion the third set of blood samples was collected. The fourth and final set of blood samples was collected 95 min after placebo or sumatriptan injection.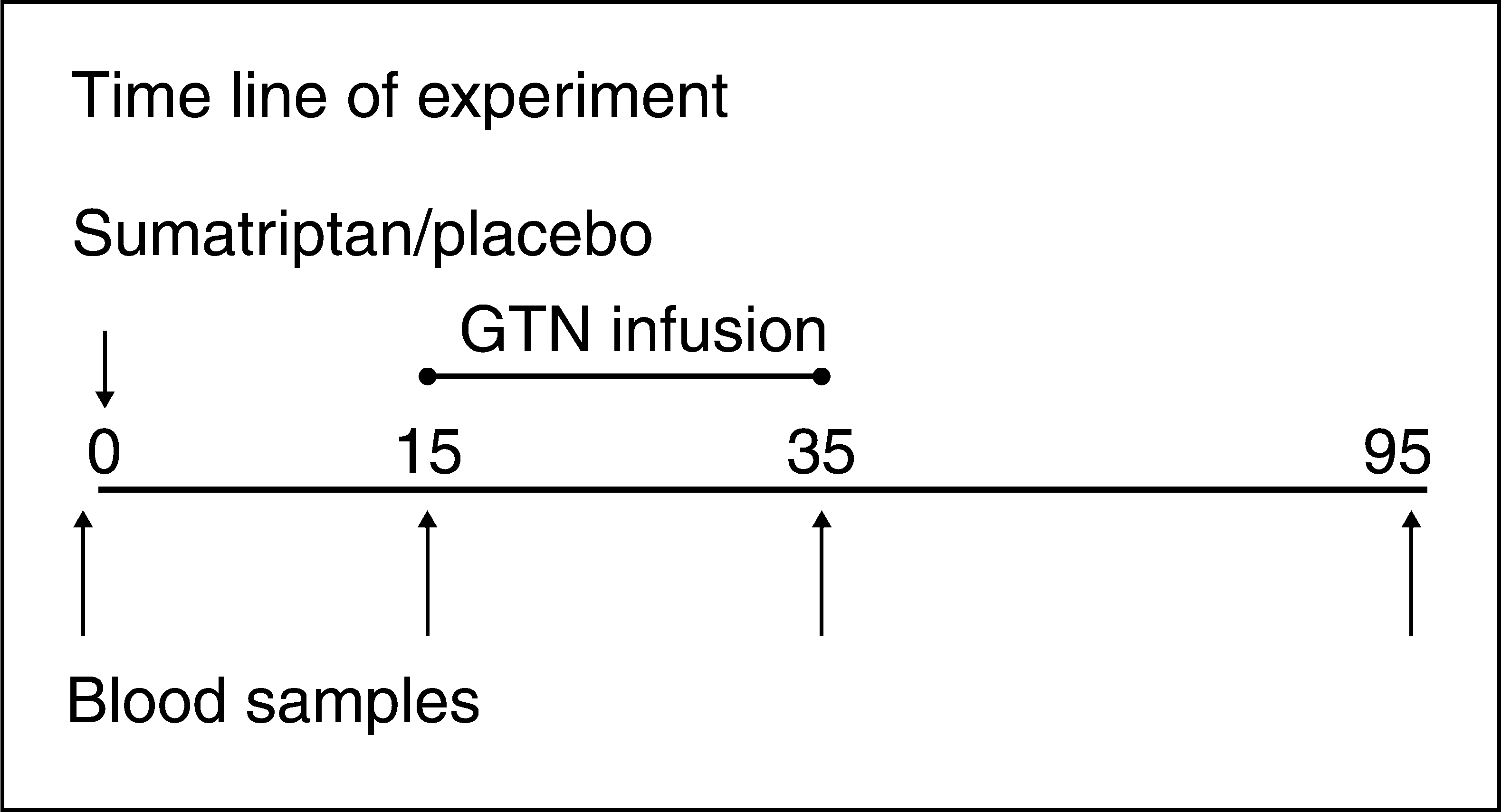
Headache intensity was scored on a numerical verbal rating scale from 0 to 10, 1 representing a very mild headache (including feeling of pressing or pulsation), 5 a headache of medium severity and 10 the worst possible headache (17). Luminal diameters of the radial and temporal arteries were measured with a high-resolution ultrasound unit (Dermascan CR; Cortex Technology, Hadsund, Denmark) (18). Headache scores and artery diameters were recorded at baseline, 10 and 15 min after the subcutaneous injection, every 5 min during the GTN infusion and at 10-min intervals during the following hour. Part of these data have been presented before (19).
Analysis of neuropeptides
Blood was collected through the inserted cannulas in the external jugular vein and the cubital vein of the subjects. Blood was sampled at baseline, 15 min after sumatriptan/placebo injection, at the end of the GTN infusion and 1 h following the end of the GTN infusion. The blood was collected in pre-chilled vials, placed on ice and immediately centrifuged at 4°C. Plasma was removed and immediately frozen and stored at −80°C. After collection of all samples, they were shipped on dry ice to the place of analysis (Göteborg). All neuropeptides, CGRP-, NPY-, VIP- and somatostatin-like immunoreactivity, were analysed according to previously described methods (8,20).
The details of the neurochemistry, the cross-reactivity with small peptide fragments and associated peptides are described previously (21–23). Since antibodies are directed towards a part of the authentic peptide, the antibodies recognizing this sequence also when present in associated peptides or fragments, it is referred to as ‘like immunoreactivity’ (-LI) in this study. All other assays are made in the same way. Plasma levels of CGRP-LI were quantified using a rabbit antiserum (R-8429) in final dilutions of 1 : 37 500. Iodinated CGRP was purified by high-performance liquid chromatography (HPLC) allowing minimum detection levels of 10 pmol/l.
Parallel control investigations showed that when 70 pmol/l human αCGRP (Auspep, Melbourne, Australia) was injected into a separate set of vials containing plasma and assayed with the same CGRP-LI method, a 25 ± 10% recovery of CGRP was detected.
Immunoreactive VIP-LI was determined using rabbit antiserum (code 78752; Milab, Malmö, Sweden) at a dilution of 1 : 40 000. The detection limit was 4 pmol/l.
Somatostatin-LI was measured using rabbit somatostatin antiserum (K18; Milab) at a dilution of 1 : 25 000. It is known to react to cyclic somatostatin (100%), linear somatostatin (50%) and [tyr1]-somatostatin (38%). The detection limit is 5 pmol/l and interassay variation is 8% (24).
NPY-LI was measured by radioimmunoassay with a rabbit antiserum as previously described (20). Antiserum (200 µl, diluted 1 : 40 000) was incubated with 100 µl of standard (synthetic NPY; Peninsula, Belmont, CA, USA) or with plasma samples for 24 h. The samples were then incubated for another 24 h with 200 µl (approximately 2500 c.p.m) of a HPLC-purified 125I-NPY tracer (20). Bound and free 125I-NPY were separated using dextran-coated charcoal. Each sample was assayed in serial dilutions and corrected for non-specific binding. The detection limit was 11.7 pmol/l.
Statistics
The intersubject coefficient of variations was calculated as the
Furthermore, a repeated measurements analysis (
Results
Headache scores and haemodynamic parameters have all been published previously (19). Below a short summary of the main headache and haemodynamic findings is given as a supplement to the results from the neuropeptide analysis.
Headache scores
On the placebo day all subjects reported a mild to moderate headache during the GTN infusion, median headache score being 4 (range 2–6). After sumatriptan pretreatment, only mild headache was reported during the GTN infusion by eight of 10 subjects with a median score of 1.5 (range 0–5), while two had no headache.
Diameter of radial and temporal arteries
Sumatriptan significantly decreased the diameter of the temporal artery (25 ± 3%) and radial artery (14 ± 3%). During GTN infusion the radial and temporal artery diameters both increased significantly, the temporal artery showing a larger response of approximately 40%. However, there was no significant difference in the diameter increase between pretreatments.
Mean blood velocity in middle cerebral artery
Mean blood flow velocity (
Blood pressure and heart rate
Diastolic blood pressure decreased significantly during GTN infusion. Although diastolic blood pressure remained higher after sumatriptan pretreatment, the decrease showed no significant difference between pretreatments. No significant changes were seen for systolic blood pressure or heart rate on either day.
Neuropeptides
Mean plasma values of measured neuropeptides
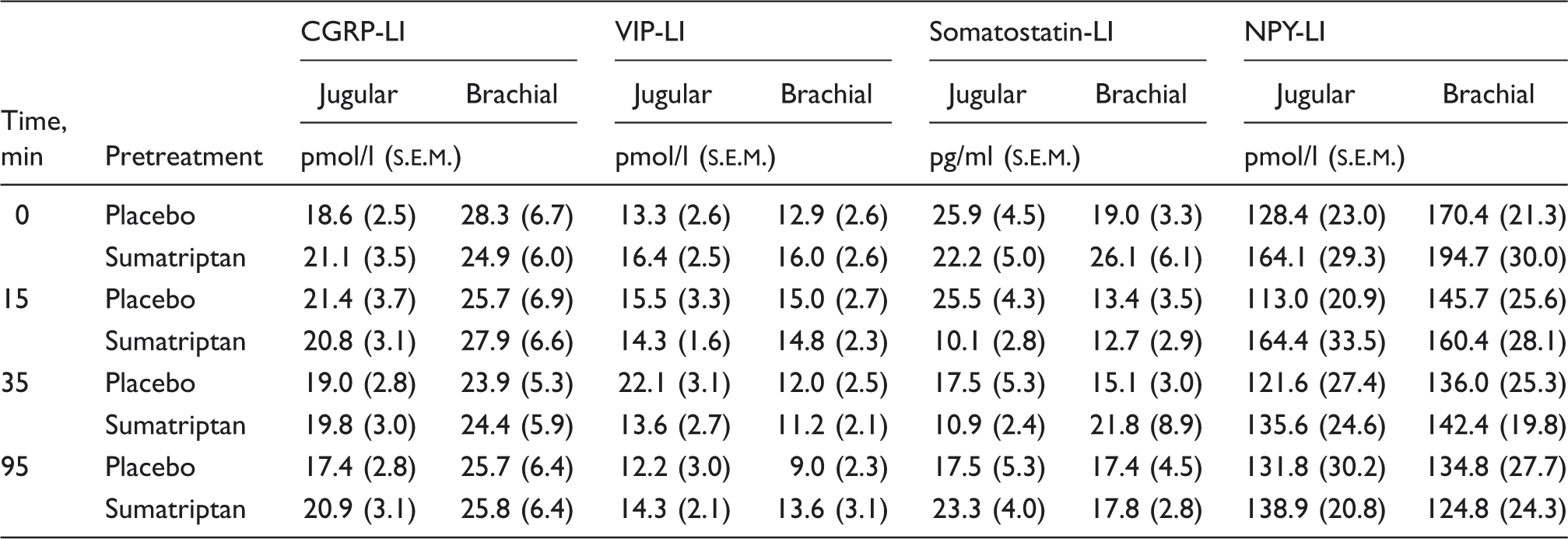
Due to technical reasons, blood was not drawn from the jugular vein in one subject on either day and in one subject on the placebo day.
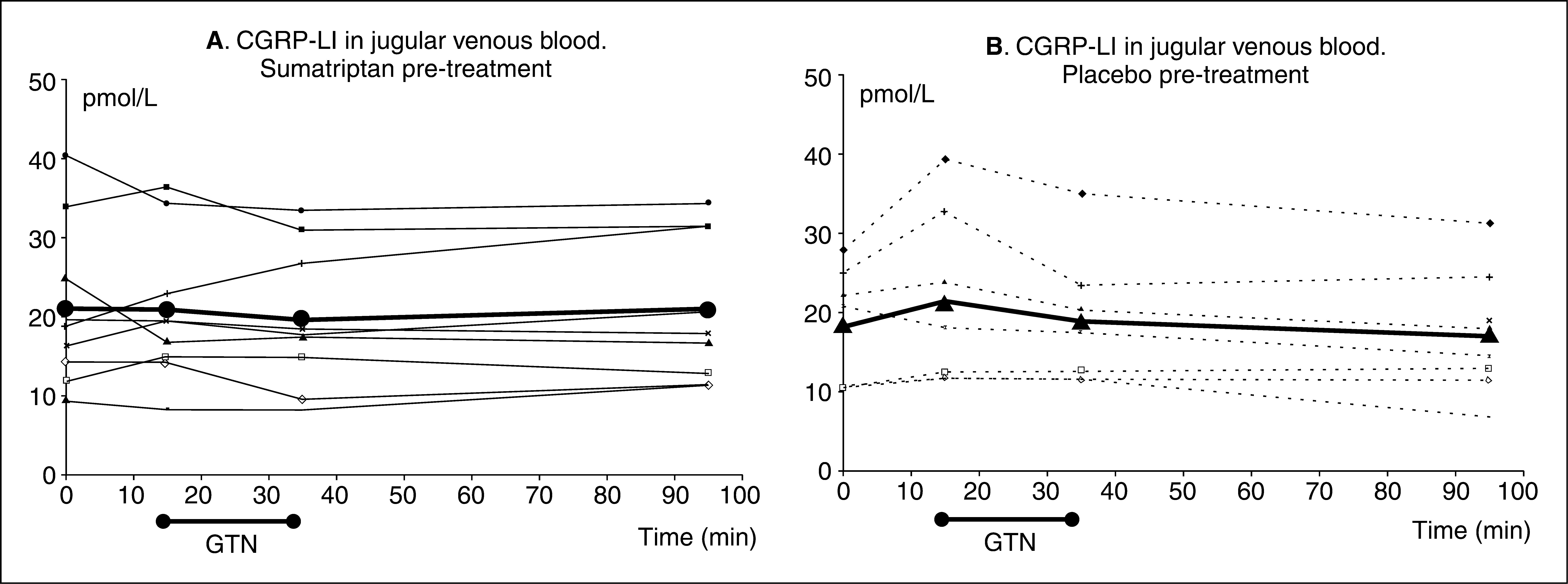
CGRP (Figures 2 and 3)
The coefficient of variation for CGRP-LI in jugular blood was 38% between subjects, 23% over time and 29% between days; for CGRP-LI in cubital blood it was 75% between subjects, 14% over time and 14% between days. The between-subject variation was very high in the cubital blood due especially to one outlier, who had high levels of CGRP-LI on both days (Figure 2). If this subject was excluded the interindividual variation of CGRP-LI in cubital blood was 45%.
Calcitonin gene-related peptide-like immunoreactivity (CGRP-LI) levels in cubital venous blood. CGRP-LI levels in cubital venous blood after sumatriptan pretreatment (A) and placebo pretreatment (B). The mean CGRP-LI plasma level is shown as a bold line, and individual measurements are shown as thin lines. For reasons unknown, one subject displayed very high plasma levels of CGRP-LI after both sumatriptan and placebo pretreatment. There was no overall effect of time on CGRP-LI levels in cubital venous blood; however, a minor effect of sumatriptan pretreatment was detected ( Calcitonin gene-related peptide-like immunoreactivity (CGRP-LI) levels in jugular venous blood after sumatriptan pretreatment (A) and placebo pretreatment (B). The mean CGRP-LI plasma level is shown as a bold line, and individual measurements are shown as thin lines. An overall effect of time was found (
A significant difference was detected between days in baseline cubital blood CGRP-LI levels (
With repeated measurement analysis there was no overall effect of time on CGRP-LI levels in cubital venous blood; however, a minor effect of pretreatment was detected (
There was no significant correlation between CGRP-LI levels and the mild headache score in this study, or between CGRP-LI levels and the blood velocity in the middle cerebral artery (MCA) (
VIP
The coefficient of variation for VIP-LI in jugular blood was 51% between subjects, 34% over time and 60% between days, for VIP-LI in cubital blood it was 60% between subjects, 52% over time and 98% between days. The high variation between days that is also seen for somatostatin was due to generally higher levels on day 1 than day 2, perhaps reflecting a degradation of the signalling molecules over time of storage.
Repeated measurement analysis showed no overall effect of time and pretreatment in VIP-LI levels in either jugular (
There were no significant differences between baseline values of VIP-LI levels in cubital and jugular venous blood between days (
Somatostatin
The coefficient of variation for somatostatin-LI in jugular blood was 46% between subjects, 34% over time and 76% between days, for somatostatin in cubital blood it was 51% between subjects, 51% over time and 58% between days.
No overall effects of time or pretreatment were detected in somatostatin-LI levels in jugular (
Somatostatin-LI levels did not change between days (
NPY
The coefficient of variation for NPY-LI in jugular blood was 51% between subjects, 56% over time and 23% between days, for NPY-LI in cubital blood it was 40% between subjects, 40% over time and 33% between days.
In general, just as with VIP-LI, NPY-LI levels showed no significant differences in any parameter investigated in jugular (
No significant day-to day changes in cubital (
Discussion
The present study has shown (19) that GTN infusion results in a mild immediate headache; the median peak headache score amounted to 4 (range 2–6, of maximum 10) at 15 min after the start of the infusion (total time 20 min). As reported before (19), there was a slight but significant (
In the present study we report on the neurochemical analysis of CGRP-, VIP-, NPY- and somatostatin-LI performed in parallel to the haemodynamic measurements in a controlled study of healthy volunteers, using blood drawn from the jugular and cubital fossa veins. Furthermore, the interindividual coefficient of variation as well as the day-to-day and over time coefficient of variations of the neuropeptides are reported for the first time.
Except for CGRP-LI in the cubital fossa venous blood, none of the peptides showed any significant differences between days.
We observed that sumatriptan (6 mg subcutaneously) in itself increased the cubital CGRP-LI levels, compared with placebo, within the first 15 min. After placebo injection no significant changes were seen in cubital CGRP-LI levels within the first 15 min. Infusion of GTN, on both the placebo and the sumatriptan pretreatment days, induced no significant changes or difference between days. These initial minor changes in CGRP-LI levels were not reflected in the jugular venous blood. In agreement, de Hoon et al. (26) observed that GTN did not elicit any rise in plasma CGRP-LI levels. Similar findings have been observed experimentally upon administration of GTN in the rat (16). Thus, taken together, there are no data to suggest that GTN
In relation to headache and vascular reactivity, sumatriptan pretreatment reduced the immediate GTN-induced headache; median score 1.5 vs. 4 after placebo (
The CGRP-LI levels did not correlate with the small reduction in the immediate GTN-induced headache seen after sumatriptan pretreatment. In previous studies showing CGRP-LI changes, sumatriptan was given to subjects with complete migraine or cluster headache attacks or in animals after activation of the trigeminovascular system (10,11,20,30,31). It is likely that genuine migraine and/or cluster headache attacks involving activation of the trigeminovascular system are required.
In general, depending on the neuropeptide analysed, the coefficient of variations of all neuropeptides were high between subjects, ranging from 14% (cubital CGRP-LI) to 56% (jugular NPY-LI); furthermore, the variation within subjects was between 14% (cubital CGRP-LI) and 98% (cubital NPY-LI) and over time from 14% (cubital CGRP-LI) to 56% (jugular NPY-LI) with the methods of analysis used in the present study, which are similar to the previously used analysis (8,12). The smallest variations between measurements were found with the CGRP-LI analysis from the cubital blood. Thus, when designing future studies measuring neuropeptides, this range of variations might be taken into consideration. In addition, it may be considered that the assays do not measure the entire peptide but only a part of the molecule (usually the C-terminal end), thus other molecules or fragments of the full-length peptide may be measured as well. This has been demonstrated by HPLC analysis and is observed even if optimal sampling conditions are used (21,23). As soon as a vesicle-stored neuropeptide is released, the abundance of peptidases degrade them.
Based on the present data, there is no indication that GTN-induced immediate headache is associated with neuropeptide release either in the local (jugular vein) or in the systemic (cubital vein) circulation. However, the plasma levels may not reflect the central pain signalling processes.
The results are in agreement with the data from Juhasz et al. (30), who found no change in CGRP-LI during immediate GTN-induced headache. Juhasz et al., however, found a decrease in CGRP-LI plasma concentrations in migraine patients after sumatriptan treatment of GTN-induced migraine in migraine patients. Unfortunately, placebo was not included and the plasma levels of CGRP-LI were measured only late in the headache phase of the induced migraine, and thus at a later stage in the cascade of events than presently (11). The caveat of the observations is that the trigeminovascular system must be activated in order to have a positive response of the trigeminovascular system, where the 5-HT1B receptors act pre-synaptically to inhibit the release of CGRP (10). Just as in migraine attacks, sumatriptan does not prevent the acute attack induced by GTN.
In support of the present findings, no immediate change in CGRP-LI was found when inducing headache or migraine by administration of sildenafil, which increases the second messenger of NO cGMP (32).
In conclusion, markers of sympathetic (NPY-LI) and parasympathetic (VIP-LI) neuronal signalling were not affected by short-term GTN infusion, which corresponds to previous findings. Further, the marker for the sensory nerve signalling CGRP showed an early non-specific increase immediately after sumatriptan pretreatment in healthy volunteers. However, it should be noted that in the present study sumatriptan was administered as pretreatment, whereas other studies have administered it after initiation of headache (11,30). Furthermore, within the first hour after GTN infusion, there was no CGRP-LI increase in the local or systemic circulation.
Footnotes
Acknowledgements
The study was supported by The Danish Research Counsel, The Danish Headache Society, Glaxo Research and Development Ltd and The Copenhagen County.
