Abstract
We included 58 patients with meningioma in a prospective study to analyse the prevalence of and risk factors for different types of meningioma-associated headache. Twenty-three patients (40%) had meningioma-associated headache. Of these, the pain was migraine-like in five (22%) and tension-type headache (TTH)-like in 13 (57%). Sixteen of 21 (76%) experienced relief of pain intensity of at least 50% after 18–24 months. Univariate analysis revealed bone-invasive growth pattern (P = 0.007) as a risk factor for headache and intake of antiepileptic drugs (P = 0.04) or large surrounding oedema (P = 0.04) as possible protective parameters. For migraine-like headache, risk factors were a positive history of migraine (P = 0.009) and bone-invasive growth pattern (P = 0.046) and, for TTH-like headache, only bone-invasive growth pattern (P = 0.009). Binary logistic regression analysis added to assess predictability and interaction effects could not identify a single factor predicting the occurrence of headache in the presence of a meningioma (correct prediction in 74% by a model consisting of bone-invasive growth pattern, history of head surgery, intake of antiepileptic drugs, temporal tumour location and moderate and large surrounding oedema). Analysis of 38 tumour specimens could not confirm the hypothesis that the occurrence of headache correlates with the expression magnitude of signal substances known to be present in meningiomas [stroma cell-derived factor 1, interleukin (IL)-1β, IL-6, vascular endothelial growth factor A] or thought to be relevant to headache/pain pathophysiology [prostaglandin-endoperoxide synthase 2, calcitonin-related polypeptide alpha, nitric oxide synthase (NOS) 1, NOS2A, NOS3, transforming growth factor-alpha, tumour necrosis factor, tachykinin, vasoactive intestinal peptide]. The affection of bone integrity and the expression of molecules thought to be relevant to headache pathophysiology might be important for meningioma-associated headache in predisposed individuals.
Introduction
Meningiomas are the second most frequent primary brain tumours (1) with a prevalence of 1–2% in routine autopsies (2). They can present with a progressive neurological deficit, epileptic seizures, and headache (3). Headache is more prevalent in patients with meningioma (77%) than in patients with brain tumours in general [31–71% (4–8)]. Brain tumour-associated headache in general is similar to tension-type headache (TTH) in 40%. A pulsating migraine-like quality tends to be more common in patients with meningioma (8). To date, there is no definite evidence for an association between pain and gender, age, and the mechanical factors tumour size, tumour location and a surrounding oedema (8–10). Pre-existing headache is the main risk factor for the occurrence of headache in brain tumour patients. This raises the question of whether the mechanisms of headache in brain tumour patients are related to the mechanisms in primary headache. It remains to be determined whether the local production of signal substances by meningiomas is associated with the occurrence of headache.
In this prospective study we analysed 58 patients with meningioma for clinical (intrapersonal, tumour-associated) risk factors for headache. As a second step, we tested the hypothesis that the expression of various signal substances correlates with the occurrence of headache. Candidate substances were those expressed by meningiomas [stroma cell-derived factor-1 (CXCL12) (11), interleukin (IL)-1β (12), IL-6 (12,13), vascular endothelial growth factor A (VEGF-A) (14,15)] or those thought to be relevant to headache pathophysiology [prostaglandin-endoperoxide synthase 2 (PTGS2; COX2) (16), calcitonin-related polypeptide alpha (CALCA; CGRP) (17), nitric oxide synthase (NOS) 1 (nNOS), NOS2A (iNOS), NOS3 (eNOS) (18), transforming growth factor-alpha (TGF-α), tumour necrosis factor-alpha (TNF; TNFSF2) (19), tachykinin precursor 1 (TAC1; substance P) and vasoactive intestinal peptide (VIP) (20,21)].
Patients and methods
Study population
We prospectively recruited patients undergoing meningioma surgery at the Neurosurgery Department of the University of Munich between June 2007 and February 2008. The study was approved by the Ethics Committee at the University of Munich (project no. 233-07). A total of 60 consecutive patients with the clinical and radiological diagnosis of meningioma agreed to participate and gave their written informed consent. Two of these were excluded after surgery because of a histopathological diagnosis of follicular lymphoma and malignant glioma, resulting in finally 58 patients. All parameters for data collection were predefined at the onset of the study to enable identification of meningioma-associated headache and its risk factors by statistical analysis. Patients were interviewed about personal details (age, sex, family history of headache or cardio-/cerebrovascular events) and asked to fill in a standardized questionnaire, which included information on pre- and postoperative symptoms with a focus on headache. We were interested in the chief complaint that led to the diagnosis (by chance, neurological or neuropsychological deficit, epileptic seizure, headache), facial and back pain, past medical history, general medication, cardiovascular risk factors and alcohol abuse. The presence and classification of pre-existing headache were recorded using a self-administered headache questionnaire validated for migraine, TTH and trigeminal autonomic cephalalgia (22,23). In addition, we asked for subjective alterations of pain quality, intensity, frequency and location. A new headache was distinguished from a pre-existing headache by a change of the headache syndrome according to the self-administered headache questionnaire (see below) or by a marked subjective alteration of pain quality, intensity, frequency or location. Patients were additionally asked about their pain treatment habits (pain medication and prophylaxis: substance, frequency). The entity meningioma-associated headache was defined as a new headache if a medication overuse headache (MOH) could be excluded [intake of pain medication on >15 (non-triptan analgesics) or 10 days (triptans, opioids, combination analgesics) per month for >3 months]. Meningioma-associated headache was furthermore categorized as migraine-like or tension-type-like using the self-administered headache questionnaire. To describe secondary headaches not classified by this questionnaire, further characteristics were noted: frequency (constant, daily, >15/month, more than weekly, less than weekly), duration (< 1 h, 1–4 h, 4–72 h, 3–7 days, >7 days), intensity on the nominal analogue scale (ranging from 1 for little pain to 10 for maximum pain), headache character (dull, pulsating, stinging), location (frontal, temporal, parietal, occipital, hemispheric), side (left, right, one side alternating, bilateral), associated symptoms (nausea, visual deficit, lacrimation, phono- or photophobia, none) and aggravating factors (posture, early morning worse than evening, exercise, none). Eighteen to 24 months after surgery, every patient with tumour-associated headache was contacted to evaluate an alteration of the headache (categorized as no alteration, relief of at least 50% or worsening). Each patient underwent a standardized neurological examination on the day of admission.
Tumour characteristics
Information on the neoplasm was obtained from the histopathological characterization and from neuroradiological imaging [magnetic resonance imaging (MRI) or contrast-enhanced computed tomography (CT)] as regards tumour size, location, surrounding oedema and growth pattern (bone invasion, non-invasive). The main location of the tumour was chosen from the following: left, right, bilateral or medial, and frontal, temporal, parietal, occipital, hemispheric, medial or infratentorial. Tumour size was defined as the product of the two largest dimensions (in cm) and was used as a measure to differentiate between small (< 10 cm2), medium (10–20 cm2) and large tumours (> 20 cm2). Multifocal tumours were assigned to a special category. Bone-invasive growth pattern was defined as local thickening of the bone adjacent to the meningioma reaching >150% in comparison with the respective contralateral calvarium. The surrounding oedema was estimated subjectively on a scale of 0–3, from no oedema (0) to extensive oedema (3).
Quantitative polymerase chain reaction from tumour tissue
During surgery, tumour tissue was removed as indicated by the neurosurgeon: one sample was analysed by neuropathology, the second, if available, was stored immediately at −80 °C until analysis.
Total RNA was isolated using the RNeasy Fibrous Tissue Mini Kit with proteinase K and DNase I digest (all Qiagen, Hilden, Germany). Total RNA (1 µg) was reverse transcribed in a final volume of 20 µl using the RNA-to-cDNA Kit (Applied Biosystems, Darmstadt, Germany). Quantitative polymerase chain reaction was performed in triplicates on TaqMan low-density arrays using Gene Expression Master Mix (all Applied Biosystems) using 500 ng total RNA converted to cDNA per port.
The following primer/probes were selected: glyceraldehyde-3-phosphate dehydrogenase (GAPDH; Applied Biosystems assay number Hs99999905_m1), CXCL12 (Hs00930455_m1), IL-1β (Hs00174097), IL-6 (Hs00985639_m1), VEGF-A (Hs00900054_m1), PTGS2 (COX2, Hs00153133_m1), CALCA (CGRP, Hs00266142_m1), NOS1 (nNOS), Hs00167223_m1), NOS2A (iNOS, Hs00167257_m1), NOS3 (eNOS, Hs00167166_m1), TGF-α (Hs00608187_m1), TNF (TNFSF2, Hs00174128_m1), TAC1 (substance P, Hs00243225_m1), VIP (Hs00175021_m1), actin-β (Hs99999903_m1) and PPIA (cyclophilin A, Hs99999904_m1).
Data were analysed using the ΔCT-method independently with three housekeeping genes (GAPDH, actin-β, and PPIA).
Statistical analysis
Statistical analysis of clinical data was performed using SPSS 16.0 for Windows (SPSS Inc., Chicago, IL, USA). In a first step, categorical and continuous variables were analysed using χ2 or Fisher's exact test (in case of expected values < 5) and two-sample t-test to identify significant correlations. All probably relevant variables from the first step (predefined with P < 0.15) were analysed further by binary logistic regression to assess interaction effects and to determine the set of variables most likely to predict the occurrence of headache in association with a meningioma. The probability of correct prediction with these variables was assessed with the Hosmer–Lemeshow test. Data are presented as means ± standard deviation [
Results
Study population
Characteristics of the 58 study patients in respect of age, gender and medical history

NA, not available; TTH, tension-type headache.
Tumour characteristics
Tumour characteristics of the 58 study patients, including histopathology, size, location, size of the surrounding oedema and growth pattern

n.s.d., no specific diagnosis; m, meningioma.
Meningioma-associated headache
Significant (P < 0.05).
Pre-operatively, one patient had a change from migraine to TTH-like headache and six had a marked alteration of headache characteristics = new headache. One had MOH, three had an unchanged headache and seven had lost their headache = no new headache.
Daily dosage of antiepileptics of the individual patient: levetiracetam 500 mg, 1000 mg, carbamazepine 600 mg, 800 mg, pregabalin 300 mg, phenytoin 300 mg, lamotrigine 50 mg.
Intrapersonal and tumour-associated risk factors for meningioma-associated headache were assessed first by univariate and subsequently by binary logistic regression analysis.
Univar., univariate analysis: χ2, Fisher's exact test or Student's t-test; Bin. log. reg., binary logistic regression; OR, odds ratio; 95% CI, 95% confidence interval; NA, not available; TTH, tension-type headache; n.s.d., no specific diagnosis; m, meningioma; MI, myocardial infarction.
There was an abnormal neurological examination in 13 of all patients with tumour-associated headache (13/23, 57%). There was no difference between TTH-like (8/14, 57%) and migraine-like headache (3/5, 60%).
Characteristics of meningioma-associated headache concerning frequency, duration, location, intensity, trigger factors, character and additional symptoms

NAS, nominal analogue scale; Horiz. pos., worsening in horizontal position; N/V, nausea/vomiting; P/P, phono-/photophobia; Alt. vision, altered vision.
Of all 23 patients with meningioma-associated headache, two could not be reached after 18–24 months despite great effort (at least four telephone calls over 4 weeks and one written letter). The remaining 21 described a marked relief of their headache in 16 cases (76% of 21) and only five realized no change (two) or even a worsening (three).
Tumour characteristics as risk factors for headache
Risk factors for the occurrence of headache in association with meningioma are listed in Table 3. On univariate analysis, significant correlations were found for bone-invasive growth pattern (positive, P = 0.007) and for intake of antiepileptic drugs (protective, P = 0.04; daily dosage of antiepileptics of the individual patient: levetiracetam 500 mg, 1000 mg, carbamazepine 600 mg, 800 mg, pregabalin 300 mg, phenytoin 300 mg, lamotrigine 50 mg) or large surrounding oedema (protective, P = 0.04). No association was found for age, gender, pre-existing headache, cardiovascular risk factors, medication other than antiepileptic drugs, tumour histopathology, location, or size. Binary logistic regression using the parameters bone-invasive growth pattern, history of head surgery, intake of antiepileptic drugs, temporal tumour location as well as moderate and large surrounding oedema could not identify a factor predicting the occurrence of headache in the presence of a meningioma (correct prediction by the model in 74%, sensitivity 57%, specificity 86%, data not shown).
Intrapersonal and tumour-associated risk factors for the subgroups migraine-like and tension-type headache-like meningioma-associated headache in comparison with no meningioma-associated headache at all were assessed first by univariate and subsequently by binary logistic regression analysis

Significant (P < 0.05).
Univar., univariate analysis: χ2, Fisher's exact test or Student's t-test (mean age); Bin. log. reg., binary logistic regression; OR, odds ratio; 95% CI, 95% confidence interval; NA, not available; TTH, tension-type headache; n.s.d, no specific diagnosis; m, meningioma.
In the subgroup TTH-like meningioma-associated headache, univariate analysis identified bone-invasive growth pattern as positively correlated (P = 0.009) with head pain (Table 5). A model consisting of bone-invasive growth pattern, intake of oral contraception, and two kinds of histopathology (meningothelial, no specific diagnosis) was able to predict headache correctly in 85% (sensitivity 46%, specificity 100%, data not shown), but could not identify a single parameter as a significant factor by binary logistic regression.
Yield of tumour tissue
In 20 cases, no tumour specimen could be obtained due to bone invasion or small tumour size
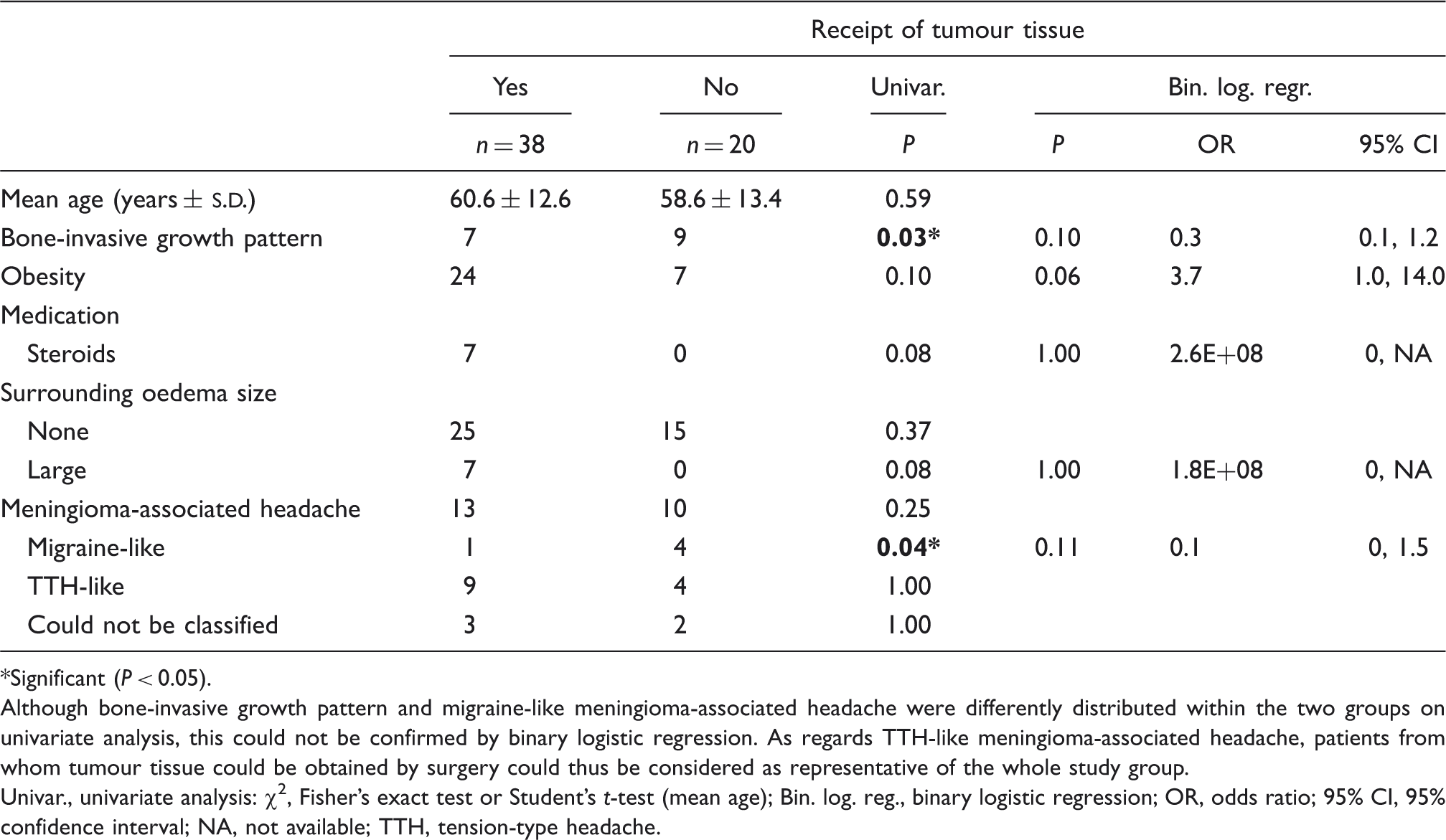
Significant (P < 0.05).
Although bone-invasive growth pattern and migraine-like meningioma-associated headache were differently distributed within the two groups on univariate analysis, this could not be confirmed by binary logistic regression. As regards TTH-like meningioma-associated headache, patients from whom tumour tissue could be obtained by surgery could thus be considered as representative of the whole study group.
Univar., univariate analysis: χ2, Fisher's exact test or Student's t-test (mean age); Bin. log. reg., binary logistic regression; OR, odds ratio; 95% CI, 95% confidence interval; NA, not available; TTH, tension-type headache.
Gene expression profile in meningioma and correlation with headache
Comparison of the expression of 13 target genes in meningioma tissue between patients with tumour-associated TTH-like headache and patients without tumour-associated headache

Values below the detection limit (Det.Lim.) are in italics. P-values were calculated using the Mann–Whitney U-test and were >0.05 for all genes (not significant) even without correction for multiple testing.
GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
IL, interleukin; NOS, nitric oxide synthase; PTGS2, prostaglandin-endoperoxide synthase 2; TAC1, tachykinin precursor 1; TGF-α, transforming growth factor-alph; TNF, tumour necrosis factor-alpha; VEGF-A, vascular endothelial growth factor A; VIP, vasoactive intestinal peptide.

Expression of CALCA, CXCL12, IL-1β, IL-6, NOS1, NOS2A, NOS3, PTGS2, TAC1, TGF-α, TNF, VEGF-A and VIP was measured by TaqMan polymerase chain reaction in 38 meningioma specimen (black triangles) from patients with (n = 13) and without (n = 25) meningioma-associated headache. Horizontal solid lines indicate medians, dashed lines the detection limit. P-values were calculated using the Mann–Whitney U-test and were >0.05 for all genes (not significant) even without correction for multiple testing. Gene expression is displayed relative to the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Essentially similar results were obtained with two other housekeeping genes (ACTB and PPIA, data not shown). IL, interleukin; NOS, nitric oxide synthase; PTGS2, prostaglandin-endoperoxide synthase 2; TAC1, tachykinin precursor 1; TGF-α, transforming growth factor-alph; TNF, tumour necrosis factor-alpha; VEGF-A, vascular endothelial growth factor A; VIP, vasoactive intestinal peptide.
Discussion
We prospectively analysed patients with meningioma for the occurrence and classification of meningioma-associated headache according to the International Classification of Headache Disorders, 2nd edn (24). The prevalence of headache (40%) and its predominant similarity to TTH (57%) obtained using a self-administered headache questionnaire [sensitivity and specificity of 0.85 and 0.85 for migraine and 0.6 and 0.88 for TTH, respectively (23)] is in accordance with the literature describing headache in 31–71% of patients with brain tumours (4–8). According to our definition, five patients were included as having a new headache (5/23, 22%) as they described an explicit alteration of their pain characteristics, although no change in headache syndrome according to the self-administered questionnaire was documented. Therefore, one limitation of this study remains, as is in all studies on secondary headache with pre-existing headache, that there is a final uncertainty in the true frequency of meningioma-associated headache. Another limitation is that multiple comparisons were made, increasing the likelihood of type 1 errors. One major consequence would thus be that additional prospective studies should confirm our data with a predefined focus on the relevant characteristics identified in our exploratory work.
In our study 10 patients with meningioma had tumour-associated headache with normal neurological examination (i.e. 17% of 58). This frequency of headache without abnormal neurological examination is markedly higher than reported in the literature [< 1% in (25) and 2% in (8)]. We conclude that patients at the age of our study group (mean 60 years) should receive an imaging study in a case of new-onset headache even if no focal neurological sign is present.
The clinical picture and especially the biological basis vary largely between the different brain tumours. Malignant glioma and metastases demonstrate different behaviour from meningiomas, which are known to be benign, slow-growing and seldom produce elevated intracranial pressure. Therefore, the mechanisms described in our study cannot be interpolated to the other types of brain tumours, as their pathophysiological principles of producing headache might be different. Previous studies have identified pre-existing headache as a risk factor for brain tumour-associated headache in general in a patient cohort with various tumour entities (9). In our study focusing on meningioma patients, this was not the case for all types of previous and tumour-associated headache. However, specifically a history of migraine was predictive of tumour-associated migraine-like headache, and the intake of antiepileptic drugs was protective against meningioma-associated headache on univariate analysis. This suggests different pathophysiological mechanisms for migraine-like headache in meningioma patients in contrast to TTH-like headache and other tumour entities. In other words, the increased risk for tumour-associated headache might be restricted to patients with a positive history of migraine—and in this case might result in migraine-like headache. The mechanism of reduced headache in patients taking antiepileptic drugs is unclear. Migraine prophylactics such as valproic acid or topiramate are also effective in the treatment of epilepsy, indicating a group-related mechanism in meningioma-associated headache, although none of the antiepileptic drugs used in our study is recommended as first choice in migraine treatment (26). As cross tables demonstrate correlations and not causative relationships, the tendency of frequent use of non-opioid pain medication in the subgroup migraine-like tumour-associated headache might be interpreted as self-treatment of the headache attack. The alternative interpretation that headache is produced by the pain medication is unlikely, as the mechanism of medication overuse was excluded in our definition of meningioma-associated headache. These associations were not confirmed by binary logistic regression analysis. In our speculative interpretation, however, the mechanisms that induce headache with migraine characteristics in patients with meningioma might be related to the mechanisms involved in primary migraine. The report that patients with prior headaches also had a greater risk of post-lumbar puncture headache (27) supports this hypothesis. Although not confirmed on binary logistic regression, univariate analysis identified bone-invasive growth pattern as a risk factor correlating with the occurrence of meningioma-associated headache and with its subgroups migraine-like and TTH-like headache. This supports the old idea of the ‘traction on intracranial pain-sensitive structures’ as the mechanism of pain generation (28–32). It might be of particular importance that the cranial region is innervated by the trigeminal and the greater occipital nerve. Bone invasion might therefore result in irritation of these pain-sensitive structures with subsequent predisposition to headache. Whether especially an invasion of the cavernous sinus as proposed in other studies (33) is correlated with headache would be of particular interest. As a great part of the imaging studies were non-digital axial CT or MRI, such an assessment was not possible with our raw data, which require additional prospective studies. Speculation that the affect on bone and nerve integrity by tumour invasion is involved in the mechanism of pain generation is concordant with the elevated risk of post-craniotomy headache in patients with surgical removal of an acoustic neuroma using the retrosigmoid approach with additional intradural drilling of the posterior aspect of the internal acoustic meatus (34). This post-craniotomy headache might also be the reason for the three patients experiencing a worsening of their headache despite tumour removal.
Besides a pre-existing headache syndrome in the past medical history (8,9), the literature has not identified age, sex and neoplasm location (10) or tumour size (9) as risk factors for headache. Tumour-associated headache is thus more likely in patients with a predisposition to primary headache; the mechanism, however, was independent of tumour size, location and surrounding oedema. The physical impression of neighbouring tissue as a mechanism of headache generation is therefore unlikely. A limitation of our study is that tumour size was assessed by a two-dimensional approach. Using the third dimension in addition would have been more accurate for tumour volume determination and might have given a different result. Further research on the influence of the exact tumour volume on headache generation is therefore necessary. One alternative conclusion from these findings could be that the tumour produces substances that might trigger headache attacks depending on the magnitude of expression. These substances might act regionally or systemically in the sense of neurotransmitters producing headache in the individual. The clinical parameters meningioma-associated headache in general and its subgroup tension-type-like headache were tested for correlation with the expression pattern of the tumours. The subgroup migraine-like headache could not be analysed, since only one sample could be obtained. We were able to confirm the expression of molecules with importance for the regulation of angiogenesis, vasculogenesis and vascular permeability in the case of VEGF-A (14,15,35), stimulation or inhibition of tumour proliferation in the case of IL-1β or IL-6 (12,13), and meningioma cell proliferation in the case of CXCL12 (11), as previously shown in the literature. We could also demonstrate in meningioma the expression of NOS1, NOS2A, NOS3, TAC1 (substance P), and VIP, which are thought to be relevant to the pathophysiology of migraine (18,20,21), and of PTGS2 (COX2), TGF-α and TNF with putative relevance to headache or pain in general (16,19). CALCA (CGRP) (17) RNA above the detection limit was found in only one tumour.
The hypothesis that the occurrence of headache, migraine-like headache or tension-type like headache correlates with the level of expression of one of these molecules in tumour tissue, however, could not be confirmed in our study. This is consistent with Levy et al. (36), who analysed the expression of CALCA (CGRP) and TAC1 (substance P) in pituitary adenoma and could not demonstrate an association with the occurrence of headache.
The relevance of meningioma expressing molecules important for headache pathophysiology [i.e. NOS, PTGS2 (COX2), TGF-α, TNF and CALCA (CGRP)] is unclear. As regards headache, however, we could speculate that, although no correlation of expression with headache was found, these molecules could be relevant in predisposed individuals. A pre-existing headache syndrome is thought to be a risk factor for secondary headache (8,27), as confirmed in our study by univariate analysis in the subgroup migraine-like headache. This predisposition could be important when combined with the expression of these substances, so that predisposed individuals are more likely to develop headache than those without predisposition independent of the magnitude of expression.
Footnotes
Acknowledgements
We thank Ms K. Ogston for her help in preparing the manuscript. The study was supported by a grant from the BMBF (Chronic headache, project D1), and the Förderprogramm für Forschung und Lehre (FöFoLe, reg.-no. 561) of the University of Munich.

