Abstract
Background
Meningiomas are generally slowly growing intracranial tumors. They are often incidentally diagnosed, given that symptoms may be absent even in cases of an enormous tumor size. Headache is a frequent but not consistent symptom. Therefore, we examined the association between structural, biochemical and histochemical tumor parameters with preoperative as well as postoperative occurrence of headache.
Methods
In our study, we prospectively investigated 69 consecutive patients enrolled for meningioma neurosurgery. Anatomical, histological and biochemical parameters were acquired, and headache parameters were registered from the clinical report and from a questionnaire filled by the patients before neurosurgery. The headache was re-evaluated one year after neurosurgery. The study was designed to exploratively investigate whether there is an association of acquired clinical and biological parameters with the occurrence of preoperative and postoperative headache.
Results
Edema diameter and the proliferation marker MIB-1 were negatively associated with the incidence and intensity of preoperative headache, while the content of prostaglandin E2 in the tumor tissue was positively associated with preoperative headache intensity. Headache was more prevalent when the meningioma was located in the area supplied by the ophthalmic trigeminal branch. Compared to preoperative headache levels, an overall reduction was observed one year postoperative, and patients with a larger tumor had a higher headache remission. In parietal and occipital meningiomas and in those with a larger edema, the percentage of the headache remission rate was higher compared to other locations or smaller edema. Multivariable analyses showed an involvement of substance P and prostaglandin E2 in preoperative headache.
Conclusions
The study demonstrates new associations between meningiomas and headache. The postoperative headache outcome in the presented patient sample is encouraging for the performed neurosurgical intervention. These results should be tested in a prospective study that incorporates all patients with meningiomas.
Abbreviations
calcitonin gene-related peptide
nitric oxide
prostaglandin E2
Introduction
Meningiomas are the most frequent type of all primary intracranial tumors. In more than 90% of cases they are benign (1) and are clinically only recognized when they are accompanied by neurological deficits or abnormal functions. Meningiomas generally grow slowly and remain frequently untreated until serious symptoms occur. The aim of treatment is the complete surgical removal of the tumor, which may be difficult or impossible due to the anatomical location (2). Headache is the most frequent symptom and occurs in about two thirds of the patients (3,4). Whether a meningioma causes a headache may depend on compression of specific structures or an increase in intracranial pressure. Other reasons may be endogenously formed substances that are assumed to interact with intracranial nociception. Among such substances, which may be elevated in patients’ plasma during headaches or which can induce headaches, are neuropeptides, in particular calcitonin gene-related peptide (CGRP), nitric oxide (NO) and its metabolites, and prostaglandin E2 (PGE2). In a histochemical investigation of pituitary tumors, substance P or CGRP immunoreactivity was found in about one quarter of specimens; however, this was not correlated with the occurrence of headache (5,6). Also, no correlation was found between gene expression of the meningioma subtypes and particular features such as vasodilation, associated headache or key substances like neuropeptides or NO synthases (7). In highly malignant glioblastomas, the neuronal NO synthase isoform was increased but this was not the case in lower-grade glioblastomas or meningiomas (8). The prostaglandin PGE2 is a common inflammatory mediator, a metabolite of arachidonic acid formed through cyclooxygenases 1 and 2. In addition to its vasodilatory effect, PGE2 sensitizes nociceptors (9) and also has central nociceptive effects (10). Cyclooxygenase inhibitors are among the most frequently used drugs for unspecific headaches. The inducible cyclooxygenase 2 was demonstrated immunohistochemically in many meningiomas and, as for the NO synthase, the degree of cyclooxygenase 2 expression was associated with the malignancy of the tumors (11). However, meningioma-associated headaches did not correlate with cyclooxygenase 2 expression (7). The growth of meningiomas seems to be promoted in an autocrine manner (12), but also by the peptide cholecystokinin. A possible link between the production of cholecystokinin and headache has not been studied so far. The first aim of our study was to identify biological meningioma parameters that are associated with preoperative occurrence and intensity of headache. Prior studies looked at postoperative outcome other than headache in patients with meningioma, including investigation of the meningioma location, the meningioma cell type and grading, coexisting symptoms, an occurrence of particular postoperative symptoms, the operating techniques, age subpopulations, the recurrence rate and the outcome due to additional procedures or diagnostics. (7,13–24). Thus, the second aim of our study was to identify parameters in an operated patient sample that are predictive for postoperative headache, which has not been focused on before.
Materials and methods
Ethical approval and patient recruitment
The study was approved by the ethical review board of the Medical Faculty of the University of Erlangen-Nürnberg. In total, all 69 patients diagnosed with meningioma and operated on at the Department of Neurosurgery of the University of Erlangen-Nürnberg in the period 2010–2012 were included in the study, no patients were excluded. A written informed consent was provided by all patients or their legal representatives. Patient charts containing clinical data, laboratory measurements and radiological results of the patients were acquired for analysis.
Questionnaire and data acquisition
Within the 69 included operated patients, 59 completed a headache-focused questionnaire developed by Dr. Michael Küster (KSFB-Bonn, devised by the Pain Center of the German Pain Society in Bonn, Germany). The original German questionnaire is available with permission as a supplement, in addition there is also an English translation of the questionnaire. The patients answered the questionnaire at least until the day before the surgery. In all cases, the questionnaire was completed before an optional medication with dexamethasone. Some patients had help from relatives to complete the questionnaire. From this questionnaire with 39 items, only the following items were selected to investigate a possible association with headache and biological parameters of the meningioma. Patients with more than infrequent headache (once per month, according to ICHD 3-beta) were classified as headache-positive and asked to rate the typical intensity of their headache. The headache intensity was rated on a numerical rating scale ranging from 0 to 51. The numerical rating scale was additionally graded by a verbal pain rating, the latter not being considered in our analysis. None of the patients with meningioma undergoing neurosurgery in the study period were excluded; three of these patients were considered to have meningiomatosis. Within the patient cohort, the prevalence of other diseases was recorded. Three patients had an intracranial pathology, that is, one patient had a cerebral infarction, one had a thalamus infarction, and one had a prolactinoma. In addition, one patient suffered from epilepsy, two had a psychoorganic syndrome, one had leukemia, one had Cushing’s disease, one had multiple sclerosis, and one had a spinal osteoblastoma. One patient suffered from migraine, which persisted after the surgery. In addition, the localization of the headache and the intake of medication for pain relief was recorded.
In patients with preoperative headache, this was classified as “7.4.1 Headache attributed to intracranial neoplasm” according to ICHD 3-beta (25). This is due to the presence of headache, an intracranial neoplasia, the temporal relation of headache to the intracranial neoplasia in most of the patients and headache relapse in relation to the neurosurgery. For those patients in which 7.4.1 C2b “headache has significantly improved in temporal relation” to the operation does not apply, the best classification might be 7.4.1D, that is patients “not better accounted for by another ICHD-3 diagnosis”.
The occurrence of postoperative headaches one year after surgery was assessed during a follow up visit or via phone contact by the neurosurgeon. The postoperative evaluation determined headache with the same criteria and on the same 0–51 pain scale as used by the preoperative questionnaire. The meningioma size was determined in preoperative magnetic resonance imaging. The diameter of the preoperative edema was determined by the neurosurgeon F.S. using preoperative magnetic resonance imaging.
Biological tumor analysis
Samples of 61 meningiomas were processed for immunohistochemistry and their neuropeptide content. The resected tissue was placed in isotonic electrolyte solution and immediately frozen in liquid nitrogen until further processing. For immunohistochemistry, one part was fixed in 4% paraformaldehyde with 0.01% picric acid. The fixation period was adjusted for the size of the tissue, assuming a diffusion of about 0.5 mm/h. The samples were incubated in phosphate buffered saline overnight and immersed in 10% sucrose for 24 hours before freezing. Finally, they were embedded in TissueTek, frozen in methylbutane and stored at −20℃. The second part of the tumor sample was stored at −20℃ without fixation to determine the total content of substances addressed below (26). Briefly, the cryoconserved samples were thawed and washed in synthetic interstitial fluid containing, in mM: 107.8 NaCl, 26.2 NaCO3, 9.64 Na-gluconate, 7.6 sucrose, 5.55 glucose, 3.48 KCl, 1.67 NaH2PO4, 1.53 CaCl2 and 0.69 MgSO4 (27). After weighting, samples were immersed in 2 M acetic acid, heated to 95℃ and boiled for 10 minutes. Afterwards, the tissues were homogenized with a custom-made homogenizer. The samples were boiled a second time for 10 minutes at 95℃ in 2 M acetic acid and centrifuged for 30 minutes at 10,000 g. The supernatant was removed and 3 M NaOH was added to adjust to a pH of 5.2–7.4. In these samples, the content of PGE2, substance P and CGRP was determined by specific enzyme-linked immune assays as indicated by the manufacturer (Bertin, France) and as previously described (28). Whole protein content was determined by a kit (ThermoFischer, Waltham, MA). The protein concentration and the neuropeptide content of the samples was detected photometrically by a microplate reader (Opsys MR-Dynex Technologies, Germany). The tumor proliferation index MIB-1 of most tumors has been described (29). This uses a monoclonal antibody as a cell proliferation marker on routine histologic material to determine the proliferative activity of Ki-67 protein (30). The latter is present during all active phases of the cell cycle and is therefore a useful tool for detecting the clinical course of the disease.
Statistical procedures
The study was prospective and designed to investigate potential associations of acquired parameters with occurrence of preoperative and postoperative headache in an exploratory manner. Further, associations between acquired parameters had an exploratory character and therefore no endpoints were predefined. First, independent parameters were pairwise tested for correlation. This was calculated with a product-momentum correlation for two continuous variables and by a Spearman correlation when at least one non-parametric variable was included. Due to the exploratory nature of the present study, in this phase no correction for multiple testing was employed. Chi-square tests were used to test 2 × 2 and 2 × 3 case contingency tables with a Yates’s correction where required. Two groups were compared by t-tests for independent samples. Bonferroni-Holm correction was used to adjust p-levels in case of linked tests. Multiple groups were evaluated by analysis of variance (ANOVA), followed by Tukey’s HSD post-hoc test for pairwise comparisons.
To specifically address a possible involvement of the neuropeptides CGRP and substance P and the lipid mediator PGE2 in preoperative headache, two hierarchical, multiple regression models were applied: To predict the occurrence (yes and no) of preoperative headache in all patients, binary logistic regression was used. Firth logistic regression was applied due to the low number of patients without preoperative headache taking pain medication (31,32). For patients with preoperative headache, its intensity (1–51) was modeled using multiple linear regression. In both logistic and linear regression, the following combination of predictors was used: Model 1 contained pain medication (yes and no) and maximal tumor diameter; in Model 2, concentrations of CGRP, Substance P and PGE2, all log-transformed to base 2, were entered in addition. The likelihood ratio Chi-square statistic was used to test i) if Model 1 predicts headache occurrence and intensity; ii) if Model 2 predicts headache occurrence and intensity; iii) if Model 2 predicts headache occurrence and intensity better than Model 1; iv) to what extent the individual variables contribute to the models; and v) if the individual variables predict headache occurrence and its intensity.
All statistical tests were two-sided, p < 0.05 was considered statistically significant. Data are presented as mean ± SEM, in case of skewed distributions with a Shapiro-Wilk test for normality of p < 0.05, median and interquartile range is given. Analyses were performed using Statistica (StatSoft, Tulsa, USA) and IBM SPSS Statistics 24.
Results
Patient characteristics and sample size in absolute numbers and percentage.

Preoperative headache: Univariate analyses
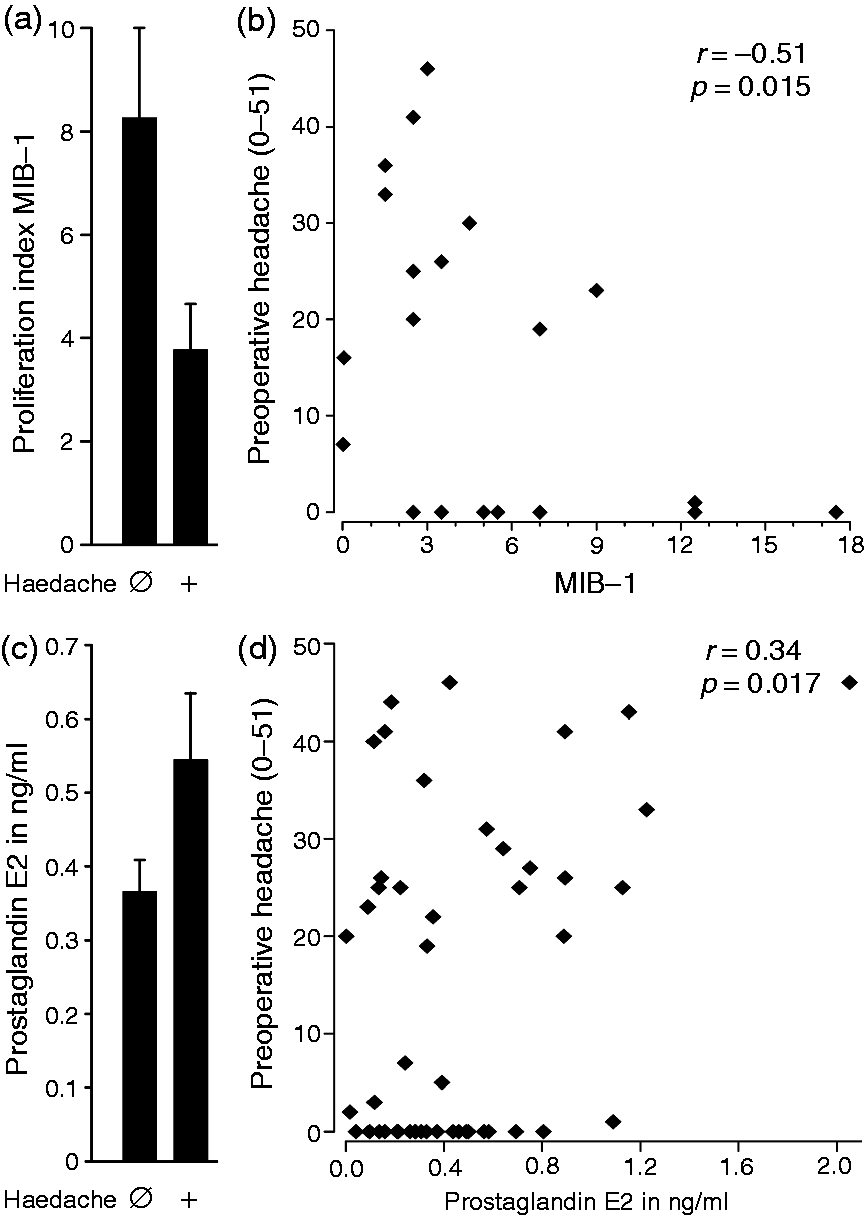
Preoperative headache exceeding an infrequent rate (once per month, according to ICHD 3-beta) was reported by 60% (38/63) of patients. The headaches did not conform to characteristic primary headaches such as migraine or cluster headache, no characteristic pattern of the headache was recorded, as it was reported in previous studies (33). The preoperative incidence of headache was 71% (27/38) in patients with meningiomas innervated by the first trigeminal branch, which is higher than 44% (11/25) for the second and third trigeminal branch (p = 0.032, Chi-square test, Figure 1). Compared to the functional classification, in a simpler anatomical classification to frontal, temporal, parietal and occipital, no significant differences were observed. The preoperative incidence or intensity of the headache was not associated with the maximum tumor diameter (Rs = 0.004 and −0.076, p = 0.98 and 0.57, Spearman correlations). The location of the sensation of headaches was not associated with the location of meningiomas. This is in agreement with a previous intraoperative study, where stimulation of different sites of the exposed dura mater caused widespread referral of pain, which did not coincide with the expected trigeminal areas (34). For the 12 patients, who had both a unilateral headache and a unilateral meningioma, there was no positive or negative association between the side of the headache and the side of the meningioma (p = 0.61, Chi-square test). Patients with an edema above 10 mm had a preoperative headache rating of 9 ± 3, which is lower than the 19 ± 3 reported by patients with smaller edema (p = 0.044, n = 24 and 28, U-test, Figure 1(b)). The edema around the tumors was negatively associated with the preoperative headache intensity (R = −0.28, p = 0.048, product-momentum correlation, Figure 1(c)). The tumor proliferation index MIB-1 was higher in patients without than in patients with preoperative headache (p = 0.020, U-test, determined in 24 patients, Figure 2(a)). The MIB-1 was negatively associated with the intensity of preoperative headache (R = −0.51, p = 0.015, product-momentum correlation, Figure 2(b)). Median PGE2 levels in meningioma tissue were 354 pg/ml (interquartile range 158–581) and were positively correlated with the intensity of preoperative headache (R = 0.34, p = 0.019, n = 48, product-momentum correlation, Figure 2(c),(d)). Substance P levels failed to reach significance (R = 0.37, p = 0.071, n = 44, product-momentum correlation).
(a) Preoperative headache is associated with innervation. Meningiomas were classified according to the innervation territory of the three trigeminal branches and spinal nerves C1–C3. Meningiomas in the territory of the ophthalmic branch were more frequently associated with headache compared to the other cases. (b) Preoperative edema diameter, here thresholded at 10 mm, is negatively correlated with the preoperative headache rating. (c) Scatterplot of the edema around the tumors and the preoperative headache intensity. Biological parameters associated with preoperative headache. (a) Patients without preoperative headache had a higher tumor proliferation index MIB-1 compared to patients without preoperative headache. (b) The MIB-1 was negatively correlated with the intensity of preoperative headache. (c) Prostaglandin E2 concentration in meningiomas of patients with and without preoperative headache. (d) The levels of prostaglandin E2 in the meningioma were correlated with the intensity of preoperative headache.
Preoperative headache: Multivariable analyses
Hierarchical multivariable regression models testing the effects of CGRP, substance P and PGE2 concentrations within meningiomas on preoperative headache occurrence and intensity.

Note. Bold numbers denote significant associations with preoperative headache.
In Firth logistic regression, Model 1 including the variables pain medication and maximal tumor diameter predicted the occurrence of headache significantly better than a basic model without predictors (Model 1: χ2 (2) = 13.0, p = 0.002), whereby only the variable pain medication contributed significantly (χ2 (1) = 12.8, p < 0.001). To test the predictive value of CGRP, substance P and PGE2, their concentration was added in addition (Model 2). Although Model 2 also significantly predicted headache occurrence in general (Model 2: χ2 (5) = 12.4, p = 0.029), it was not better than Model 1 (Model 2 vs. Model 1, p = 0.91).
In multiple linear regression, Model 1 did not significantly predict headache intensity (R2 = 0.19, χ2 (2) = 4.28, p = 0.09). Addition of CGRP, substance P and PGE2 concentration significantly improved the model, increasing the coefficient of determination by 0.36 to 0.55, which was statistically significant (Model 2 vs. Model 1: χ2 (3) = 11.2, p = 0.010). While the effect of CGRP was not significant, substance P and PGE2 significantly predicted headache intensity (p = 0.006 and 0.014). The partial correlation coefficients for substance P and PGE2 concentration with preoperative headache were 0.512 and 0.509, respectively.
Calculation of overall likelihood ratio statistics with respective p-values showed that pain medication, maximal tumor diameter, substance P and PGE2 concentration significantly predicted preoperative headache, whereas no such effect was observed for CGRP concentration.
Post-operative headache
The percentage of all patients without postoperative headache increased with the T-staging (Rs = 0.25, p = 0.043, Spearman correlation, Figure 3(a)). Also, within patients with preoperative headache, the percentage of patients who became headache-free after the surgery increased with the T-staging (Rs = 0.34, p = 0.042, Spearman correlation, Figure S4). These results are similar when an at least ≥50% reduction in the postoperative headache rating is considered instead of complete absence of headache. Further of interest is the predictive value of the anatomical location. In patients with frontal and temporal meningioma, the postoperative incidence of headache was about half of the preoperative incidence; for the parietal and occipital cases, all patients were headache-free after the operation (Figure 3(b)). Anatomical locations of particular interest did not comprise enough patients to allow meaningful statistical analysis. In 57 of 69 patients, the meningioma could be fully removed. Complete vs. incomplete removal was not predictive for occurrence of postoperative headache (p = 0.86, Chi-square test). Patients with a preoperative edema above 10 mm reported a postoperative headache in 8% (2/26), which is lower than the 31% (10/32) of patients reporting headache with smaller preoperative edema (p = 0.028, Chi-square test, Figure 3(c)).
Postoperative headache. (a) Patients with high tumor progression had a large reduction in headache due to neurosurgical intervention. (b) A complete postoperative absence of headache was observed for parietal and occipital localized meningiomas, compared to a reduction in frontal and temporal cases. (c) Percentage of patients with and without postoperative headache separated by preoperative edema. (d) Within the subgroup of patients with preoperative headache, the percentage of patients with and without postoperative headache separated by preoperative edema. The percentage of patients is given within the bars of the stacked histograms.
According to the study protocol, the three patients with intracranial pathology were not excluded. The supplementary statistics tables provide all results presented above also as sub-analysis for the 66 patients without intracranial pathology, which did not show substantial differences.
Discussion
We investigated patients’ headache together with structural, biochemical and histochemical tumor parameters before and after meningioma surgery. Edema diameter and malignancy index MIB-1 were negatively correlated with preoperative headache occurrence, and a large tumor size was the most important parameter for a high chance of a postoperative headache reduction. In our patient sample, a high level of tumor progression favors intervention and in these patients the biggest improvements in tumor-associated headache are predicted.
It should be noted that the study investigated a preoperative patient cohort seen and selected for neurosurgery at the Department of Neurosurgery of the University of Erlangen-Nürnberg. This might implicate a selection bias compared to a cohort of patients with meningioma at the stage of first diagnosis. For example, the percentage of patients with symptoms due to the meningioma is likely to be enriched in a preoperative cohort referred to neurosurgery when compared to all subjects with a meningioma, of which many are asymptomatic or not diagnosed. On the other hand, only one migraine patient was in our sample, substantially below an expected 6.2 of 69 patients with migraine, calculated on the basis of age-matched prevalence (35). We cannot exclude undiagnosed primary headaches in the present study. However, most primary headaches occur earlier in life and are therefore typically known already at the time of meningioma incidence. In primary headache patients, referral to structural diagnostics might have a higher threshold. Further, even for those who received structural diagnostics, there might be a bias against referral to neurosurgery. Together both reasons might explain why primary headaches did occur less frequently in our patients than expected from an unbiased sample.
There was a substantial positive correlation between the tumor size and the edema diameter, which is not surprising. However, tumor size but not edema diameter was a good predictor for postoperative outcome. Although there are many studies about brain tumor headache and possible involvement of diverse substances which might be released, there are still open questions that have to be answered. There is seminal work regarding the pain-sensitive structures in the meninges, which are perceived as localized headache according to the site of stimulation (36,37). They showed that the dura mater is sensitive to stimulation, and pain is in particular observed when stimulation was performed close to arterial vessels, where the branches of the trigeminal nerve travel alongside.
Pre-existing primary headache is a risk factor for developing headache from a brain tumor, which suggests a predisposition for secondary headache (4). In more than 80% of the patients, the headache changed with the occurrence of the tumor. Stimulation of the trigeminal ganglion led to an increase in regional cerebral blood flow in the frontal and parietal cortex of cats (38). The particular importance of the neuropeptide CGRP in meningeal nociception was the topic of several investigations (39–41). Headaches associated with brain tumors were commonly assumed to be the result of intracranial pressure and mechanical traction on pain-sensitive structures. In addition to the mechanical activation suggested by the results of Ray and Wolff (37), there are likely also chemical pronociceptive effects of tumors affecting the nociceptive afferents. For patients with brain tumors of all etiologies, the incidence of headache was about 50% (42).
For our analysis, meningiomas were subdivided into different innervation territories of the three trigeminal branches and the cervical spinal nerves C1-C3. Meningiomas in the area of the ophthalmic branch were more frequently associated with preoperative headache compared to locations associated with other branches of the trigeminal nerve. The innervation of the ophthalmic branch includes the supraorbital region. It was described that periorbital allodynia occurs due to an injury in the somatosensory cortex (43). There is convergent evidence that chemical sensitization or activation of nociceptive afferents innervating the dura mater can cause facial allodynia (44,45). The most likely explanation is convergent input to central nociceptive structures, because the ophthalmic branch of the trigeminal nerve innervates most of the facial areas and the dura mater (46). Furthermore, the involvement of the cavernous sinus has been considered as a source of headache, since several structures involved in headache generation such as the internal carotid artery and the ophthalmic branch of the trigeminal nerve pass here, but this remained rather speculative (47). Dural stretch was a traditional explanation for the occurrence of headache, which has not been substantiated.
Our findings show a negative correlation between preoperative edema diameter and preoperative headache rating of the patients. This appears counterintuitive at first glance, as a larger edema diameter might cause more intracranial pressure and headache. It should be considered that pressure at nerves could cause a conduction block and thereby suppress or silence neurons. An important aspect is the tumor growth rate. Fast growing tumors are more rapidly detected than slowly growing ones. The faster growing tumors, also associated with more edema, might dislocate other structures more effectively compared to the slower growing tumors, which may invade structures without displacing them. Furthermore, the tumor subtype might be important, and there might be pronociceptive paraneoplastic effects of particular tumor types.
Patients without preoperative headache had a higher tumor proliferation index (MIB-1). As mentioned above, faster growing tumors seem to be detected by other symptoms than pain. The concentration of PGE2 in the meningioma was weakly positively associated with preoperative headache. PGE2 is a well-known sensitizer of nociceptive molecular targets such as TRPV1 (49), which could further promote pronociceptive cascades, including neurogenic inflammation and activation of non-neuronal structures. Inflammatory reactions caused by the tumor or brain injuries can induce plasma extravasation and extracellular accumulation of fluid and proteins. A dysfunction of tight-junction proteins may also play a role in the occurrence of edema. Inflammation can also be associated with an increased permeability of the blood brain barrier, which might increase edema, and which might expose nerve fibers to additional excitatory substances.
Fortunately, a large fraction of patients is headache-free after resection. This occurs more frequently in patients with larger tumors compared to smaller tumors. The relief of headache in particular in patients with larger tumors could be explained by the mechanical effects of the larger volume removed, but other parameters associated with size such as growth rate and malignancy should also be considered. Associated with the latter factors are also biochemical changes, particularly paracrine (paraneoplastic) effects of the tumor on its surroundings, which could contribute to secondary headache.
Considering the tumor location, patients with preoperative headache and tumors with parietal or occipital location were completely headache-free after surgery. An explanation might be the available space, which might be more tightly limited in areas where expansion might affect the brainstem. Still, more than half of the patients with temporal and frontal tumors became headache-free after surgery. By trend, patients with a large preoperative edema had more relief of headache after resection of their tumor, which again could be explained by the mechanical alteration as well as biochemical changes associated with the edema. In males, the WHO grade was higher, and tumor diameter and edema larger compared to females. This could be explained by more aggressive tumors in males, but more likely by a well-described higher level of indolence in males, seeking medical attention in a later stage. A final point to be considered is whether neurons grow into tumors due to chemo-attractive stimuli or are changed in their peripheral expression. Larger tumors had a higher substance P concentration. Substance P levels were most substantial in tumors located in the frontal lobes, but this might at least partially be explained by the meningeal innervation pattern.
To specifically address a possible involvement of CGRP, substance P and PGE2 in preoperative headache with adjustment for confounders, we used multivariable regression models. The intake of pain medication and tumor size were regarded as potential confounders; thus, they were added in a first regression model (Model 1). Adding CGRP, substance P and PGE2 in addition (Model 2) allowed testing for if and which of these variables have additional predictive value. First, Firth logistic regression was used to model the occurrence (rating 0 vs. > 0) of preoperative headache. The fact that pain medication significantly predicted the presence of preoperative headache is not surprising. The addition of CGRP, substance P and PGE2 concentration did not improve the predictive value of the model, indicating that there is no evidence that these molecules are involved in the occurrence of preoperative headache. Importantly, due to the limited statistical power of this exploratory trial, this cannot be interpreted as no involvement, it merely indicates the absence of evidence for an involvement.
Regarding pain intensity (rating 1–51) in patients with preoperative headache, however, multiple linear regression supports an involvement of substance P and PGE2. Together with CGRP concentration, the concentration of these two molecules within meningiomas explained considerably more variance of preoperative headache intensity than pain medication and tumor size alone. The results suggest that doubling of substance P and PGE2 concentration increase headache intensity by 4.4 and 2.5 units. The non-significant effect of CGRP concentration means that there could be an association with preoperative headache, but our data provide no evidence for it. Significant overall likelihood ratio chi-square statistics suggest that pain medication, maximal tumor diameter, substance P and PGE2 predict preoperative headache generally.
Limitations of the study: The chosen questionnaire has some disadvantages. This includes the pain rating scale, which uses a verbal grouping that might affect the linearity of the scale, a non-standard number range, and an untypical upper end of the scale. Further, the questionnaire was not specifically designed for the present study, and therefore an unnecessary effort was required to answer questions that were not of interest for this study. Patients were included in the neurosurgery department, therefore at a stage when a surgical option was at least seriously considered. This limits generalization to other patient cohorts, for example at an earlier stage in the clinical decision tree. Based on the late inclusion, the study lacks presurgical data such as headache frequency and intensity based on a headache diary. It should be considered that dexamethasone application before surgery might have had an influence on the levels of the biomarkers. Postoperative headaches were investigated once, one year after surgery; no time course of headache incidence was acquired. The limited sample size did not allow for statistically significant numbers in a finer categorization and might have overlooked existing associations due to the generalization required to get sufficient numbers per group. A placebo rate due to surgical procedures should be considered when evaluating the results (50), especially taking into account that more invasive procedures have higher placebo rates.
With special focus on associated headache, several new associations between clinical and experimental measures have been found in operated patients with meningioma. However, it has to be noted that due to the exploratory character of this study, these results need to be interpreted with caution and considered to be new hypotheses. Furthermore, due to the observational study design, no causal relationships can be established. In this regard, a causal involvement of substance P might be probed in a clinical study testing the efficacy of the approved NK1-receptor antagonist aprepitant as a symptomatic therapy against tumor-induced headache.
Key findings
Preoperative headache intensity was associated with the innervating trigeminal branch, the surrounding edema, tumor proliferation index MIB-1 and with prostaglandin E2 content. After adjusting for pain medication intake and maximal tumor diameter, an increase of substance P and of PGE2 concentration within tumors was associated with higher preoperative pain intensity. A high maximal tumor diameter was a predictor for a low probability of postoperative headache
Supplemental Material
Supplemental material for Pre- and postoperative headache in patients with meningioma
Supplemental material for Pre- and postoperative headache in patients with meningioma by Franziska Guenther, Ferdinand Swozil, Stefan Heber, Michael Buchfelder, Karl Messlinger and Michael JM Fischer in Cephalalgia
Footnotes
Acknowledgements
We like to thank Birgit Vogler, Annette Kuhn and Susanne Haux-Örtel for excellent technical assistance. We thank Rok Blagus and Georg Heinze (Medical University of Vienna, Institute of Clinical Biometrics) for statistical advice with regards to multivariable analyses. Author contributions: FG designed the study, acquired patient data, performed experiments and drafted the manuscript. FS designed the study, acquired material and interpreted patient data. SH performed multivariable analyses and wrote respective parts of the manuscript. MB contributed to the manuscript. KM coordinated the study and wrote the manuscript. MJMF supported experiments, performed statistical analysis, compiled the graphical presentation and wrote the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
