Abstract
We studied the prevalence and short-term natural course of chronic frequent headache (CFH) in the general population and identified risk factors. In the Netherlands everyone is registered at a single general practice. We sent questionnaires to all persons (n = 21 440) aged 25-55 years, registered at 16 general practices. We compared the characteristics of 177 participants with CFH (>14 headache days/month for >3 months) with 141 participants with infrequent headache (1-4 days/month) and 526 without headache (<1 day/month). The prevalence of CFH was 3.7% [95% confidence interval (CI) 3.4, 4.0]. In 5 months, 12% showed a clinically relevant decrease to <7 days/month. In both headache groups 70% were women vs. 41% in the group without headache. Compared with the group with infrequent headache, the CFH group had more subjects with low educational level [35% vs. 11%; odds ratio (OR) 4.3, 95% CI 2.3, 7.8], medication overuse (62% vs. 3%; OR 38.4, 95% CI 13.8, 106.9), sleeping problems (44% vs. 8%; OR 8.1, 95% CI 3.6, 18.1), a history of head/neck trauma (36% vs. 14%; OR 4.0, 95% CI 2.2, 7.1), high scores on the General Health Questionnaire (62% vs. 34%; OR 2.7, 95% CI 1.3, 3.6) and more smokers (45% vs. 19%; OR 3.1, 95% CI 1.9, 5.3). We conclude that headache frequency fluctuates. CFH is common and associated with overuse of analgesics, psychopathology, smoking, sleeping problems, a history of head/neck trauma and low educational level. Female sex is a risk factor for headache, not for chronification of headache.
Introduction
Chronic frequent headache (CFH), also known as chronic daily headache, is a collective term for primary headaches occurring on >14 days per month for at least 3 months. The prevalence of CFH in the general population is around 4% worldwide (1–4). Many patients start with an infrequent episodic headache type (migraine or tension-type) that gradually becomes more frequent over time until their headaches are almost daily. The cause of this chronification process is unknown.
Clinical experience suggests a causal relationship with overuse of acute headache medication because withdrawal of medication often results in a dramatic improvement of headache frequency (5). Several cross-sectional studies have reported an association between overuse and chronic headache. Two population-based studies in Spain and Taiwan reported that 25% and 34% of subjects with CFH, respectively, overused acute headache medication (1, 4). However, these percentages were not compared with control groups. In a community-based study conducted among elderly Chinese (>64 years old) CFH was associated with analgesic overuse [odds ratio (OR) 79, 95% confidence interval (CI) 19, 321] and overuse was a predictor of persistent CFH at follow-up 4 years later (6). Other factors that have been associated with CFH in the general population include female sex, low educational level, previously married status, arthritis, habitual snoring and a history of migraine (6–8). Because the control groups in these studies included subjects who rarely had headaches (only two headaches a year), these factors could be associated with having headaches regularly, rather than with chronic headache in particular.
There are limited data on the incidence and natural course of CFH. It is estimated that in a specialized headache centre 14% of patients with episodic migraine develop chronic headache during 1 year of follow-up (9). In a general population sample in the USA with a headache frequency of 2–104 days/year, the 1-year cumulative incidence of CFH was 3% (8).
We studied the prevalence and short-term natural course of CFH in the Dutch general population. To identify risk factors for chronification of headache we compared subjects with CFH with subjects with infrequent headaches. Details on clinical features, comorbidity, personality profile and impact on quality of life will be reported separately.
Methods
We studied the prevalence of CFH in the Dutch general population by sending a postal questionnaire (Q1) between January 2002 and September 2003 to all persons aged 25–55 years registered at 16 general practitioners (GPs) located in the regions of Leiden and The Hague. In the Netherlands almost everyone is registered at a single GP, which makes GPs' registers suitable for population-based studies. Leiden and The Hague are cities of 117 000 and 457 000 inhabitants, respectively, located in the province of South-Holland, a mixed area with both urbanization and agriculture. To minimize selective response, the primary objective (assessment of headache) was not explained, but a more general objective, namely evaluation of common health problems and self-treatment, was stated in a standard letter, signed by the GP. The questionnaire contained a number of headache-unrelated questions for masking reasons. We assessed headache frequency and medication use by the following questions: ‘On how many days per month on average did you suffer from headache in the past three months?’ and ‘On how many days per month on average did you take medication to treat your headache?’ We sent two reminders. Answers were given on a five-point frequency scale: on >14 days/month (chronic frequent), on 8–14 days/month (very frequent), on 5–7 days/month (frequent), on 1–4 days/month (infrequent) and on <1 day/month (none). Respondents were allocated into five groups according to headache frequency: chronic frequent headache (CFH), very frequent headache (VFH), frequent headache (FH), infrequent headache (IH) and no headache (NH).
To identify factors associated with chronification of headache, we compared subjects with CFH (headache on >14 days/month) with subjects with infrequent headaches (1–4 days/month). We also compared the CFH group with the NH group (<1 day/month) to discriminate chronification factors from factors associated with headache in general. After about 5 months (range 3–7 months), all individuals who reported CFH and two random samples of the IH and NH groups (each twice as large as the case group), received a second, more detailed questionnaire (Q2) containing questions on demographics, lifestyle factors and headache characteristics. We re-assessed headache frequency with the following question: ‘On how many days per month on average did you suffer from headache in the past six months?’ For further analyses we selected subjects who had the same headache frequency in both Q1 and Q2 (i.e. the stable frequency group). The following additional risk factors were recorded: age of onset of headache, a family history (first-degree relatives) positive for headache, a history of head or neck trauma prior to the onset of headache, sleeping problems, tranquillizer use, use of acute headache medication and caffeine intake. Overuse was defined as: use of analgesics on ≥3 days/week, use of triptans on ≥2 days/week, use of ergots on ≥1 day/week, use of narcotics on ≥10 days/month and use of >5 caffeine units a day. A caffeine unit is one cup of tea, coffee or caffeine-containing soda. We also asked subjects whether they had consulted their GP for headache in the past 6 months.
The General Health Questionnaire (GHQ-28) was used to screen for psychopathology (10). It includes four subscales: somatic physical illness and distress, anxiety/insomnia, social dysfunction, and severe depression, each consisting of seven items. Answers are given on a 4-point Likert scale, ranging from 0 ‘better than normally’ to 3 ‘much worse than normally’, with scores ranging from 0 to 21 for each subscale. Scores can be recoded into (0, 0, 1, 1) with a total scoring range of 0–28 (the GHQ scoring method). We used a cut-off score of 4/5 to define a GHQ case (10). The GHQ-28 has a sensitivity of 0.84 and a specificity of 0.82 in detecting psychopathology (10).
Q2 also contains other questions on clinical features of headache, comorbidity, quality of life, coping strategies and personality profile. These results will be published separately. Subjects in the CFH group received one reminder. Non-respondent CFH subjects received a short questionnaire to assess possible selection bias and included main items such as demographic variables, headache frequency and medication and caffeine use. The VFH (8–14 days/month) group will be followed over time to study the incidence of and risk factors for CFH. The FH group did not receive Q2 and was not analysed further.
Statistical analysis was performed with SPSS, version 11.0 (SPSS Inc., Chicago, IL, USA). Prevalences and differences between groups are presented with 95% CIs. We evaluated factors associated with chronification by comparing the CFH group with the IH group. ORs are given for putative risk factors. We used the Mantel–Heanszel procedure to adjust for potential confounders.
The Medical Ethics Committee of Leiden University Medical Centre approved the study.
Results
Sixteen GP practices participated in the study, seven located in the cities of Leiden and The Hague, five in urban areas and four in villages in rural areas. All GPs estimated the percentage of immigrants in their practice to be <10%, except for one practice, where 50% of patients were non-Western immigrants, mainly from Turkey, Morocco, the Dutch Antilles and Suriname. In total 21 440 subjects received Q1, 16 232 (76%) completed Q1 and 1160 (5%) refused to participate or had moved (Fig. 1). The response per practice varied between 69% and 84%, except for the practice with the high number of immigrants, where only 53% of subjects completed Q1.
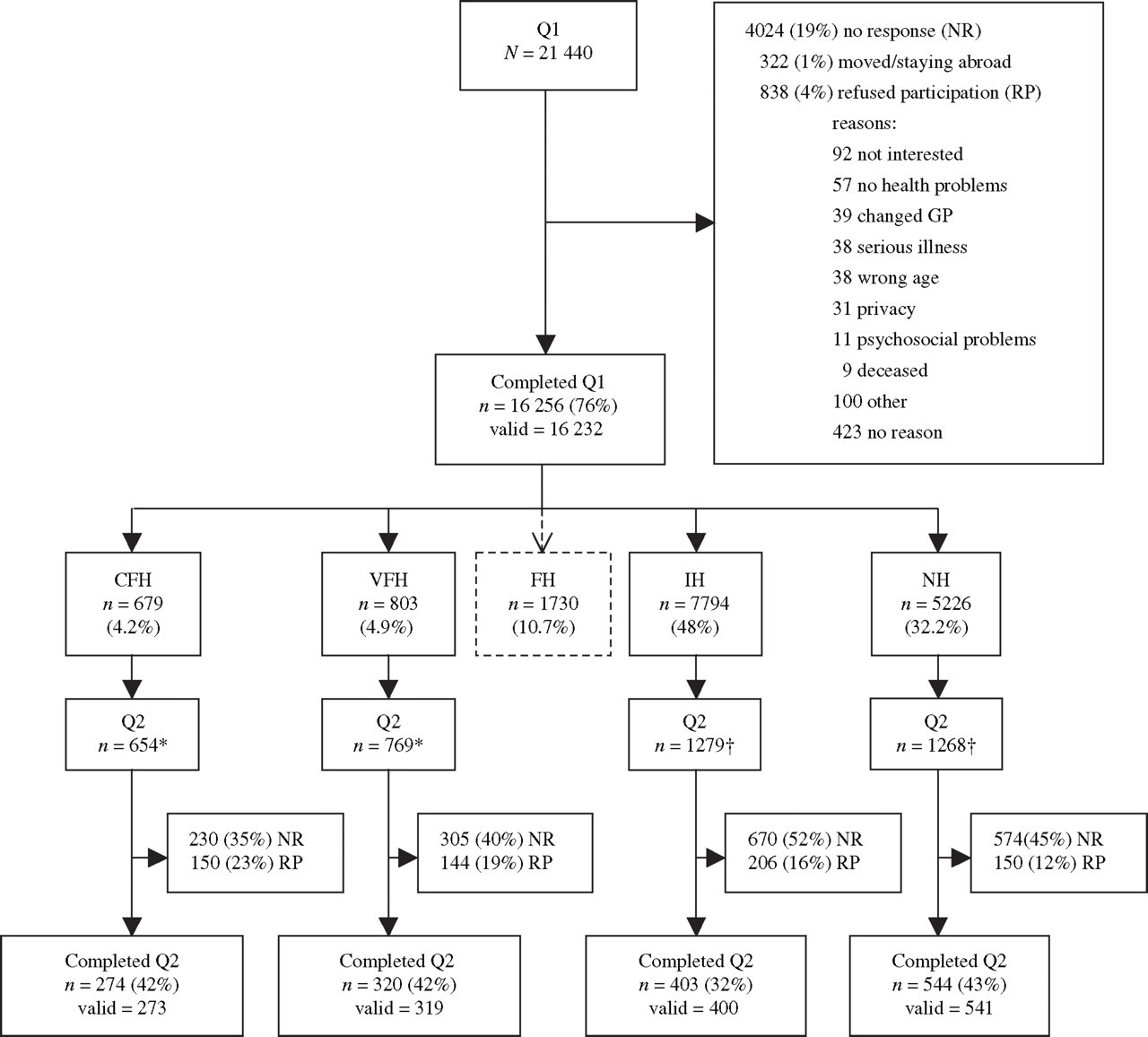
Flowchart of response. ∗Some subjects did not receive Q2 because they returned Q1 after the Q2 mailing date. †Random sample (twice as large as the CFH group0. Q1, General health survey; Q2, second detailed questionnaire; CFH, chronic frequent headache (>14 days/month); VFH, very frequent headache (8–14 days/month); FH, frequent headache (5–7 days/month); IH, infrequent headache (1–4 days/month); NH, no headache (<1 day/month); NR, no response; RP, refused to participate.
Prevalence
Of all 16 232 participants, 679 reported to have CFH (4.2%, 95% CI 3.9, 4.5). In the practice with the high number of immigrants the prevalence of CFH was 12.3% (95% CI 10.1, 14.5). Without this practice, the prevalence of CFH was 3.7% (95% CI 3.4, 4.0). Prevalences of the other headache frequency groups are shown in Fig. 1. Of 679 CFH subjects, 430 (63%) used headache medication on >14 days/month, compared with 32 (4%) in the VFH group, 33 (2%) in the FH group, 15 (0%) in the IH group and 1 (0%) in the NH group.
Follow-up
Q2 was sent to 3970 subjects. The time between Q1 and Q2 was 5 months on average (range 3–7 months). A total of 1541 subjects (39%) completed Q2, 650 (16%) refused to participate and 1779 (45%) did not respond. We excluded five subjects from analysis because they proved to be older than their registered age. Three subjects did not complete Q2 properly and were excluded. Figure 1 shows the response per headache frequency group. Participants who had reported infrequent headache in the first survey (Q1) completed Q2 less often than the others.
Non-respondents analysis
In the CFH group 230 (35%) did not respond to Q2. Mean age of the non-respondents was 40 (SD 8) and 140 (61%) were female. Ninety-two (40%) non-respondents had a non-Dutch name indicating foreign nationality. Sixty-eight (30%) non-respondents completed the short non-response questionnaire, of whom 24 (35%) had a low educational level and 37 (54%) no longer had CFH.
Frequency changes
Re-assessment of headache frequency in Q2 showed that of 273 subjects with CFH in Q1, 177 (65%) had a stable headache frequency of >14 days/month, 62 (23%) had changed to VFH (8–14 days/month) and 34 (12%) now reported a headache frequency of <7 days/month (Table 1). Overuse at baseline (Q1) was not a predictor for persistent CFH at Q2 (OR 1.5, 95% CI 0.9, 2.5). Vice versa, 65 (20%) subjects who had very frequent headache (8–14 days/month) in Q1 changed to CFH over 5 months. Overuse at baseline (Q1) in this group was not a predictor for CFH in Q2 either (OR 2.4, 95% CI 0.9, 6.5). In Q2, 109 (62%) of the stable CFH group still reported overuse of acute headache medication, compared with 26 (27%) of those who had changed to lower headache frequencies, a difference of 35% (95% CI 23, 46). So, in the group who changed to lower frequencies, the percentage of overusing subjects decreased from 51% at baseline to 27% in Q2, while in the stable CFH group there was no change. To assess whether the subjects who changed to lower frequencies had received specific headache treatment we looked at GP consultation and prophylactic use. Remission was not attributable to treatment; only 20 (22%) subjects had consulted their GP for headache in the past 6 months compared with 56 (33%) in the stable CFH group (difference −11%, 95% CI −23, 0) and there was only a 3% difference in the use of prophylactic medication between the two groups (95% CI −10, 3).
Changes in headache frequency between Q1 and Q2

Q1, General health survey; Q2, second detailed questionnaire; CFH, chronic frequent headache (>14 days/month); VFH, very frequent headache (8–14 days/month); FH, frequent headache (5–7 days/month); IH, infrequent headache (1–4 days/month); NH, no headache (<1 day/month).
Numbers in bold are stable headache frequency groups (Q1 = Q2), in total 959 of 1533 subjects (63%). Time between Q1 and Q2 ranged from 3 to 7 months.
Demographics
Further analyses were limited to the groups in which the reported headache frequency did not change over the two surveys (i.e. the stable frequency groups). Table 2 shows the differences in demographic variables between subjects with CFH, infrequent headache and no headache. In both headache groups the majority were women, in contrast to the NH group, where the majority were men.
Demographic variables in stable CFH group vs. stable IH and NH groups
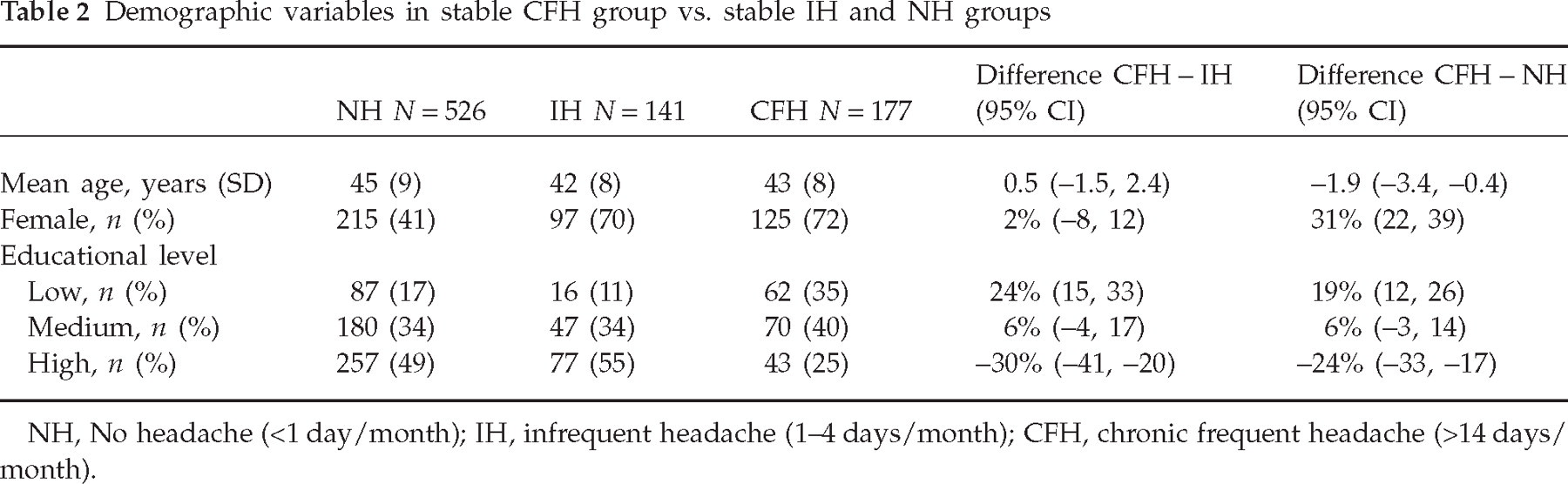
NH, No headache (<1 day/month); IH, infrequent headache (1–4 days/month); CFH, chronic frequent headache (>14 days/month).
Risk factors
Mean age at onset of headache was 19 (SD 11) for the CFH group and 18 (SD 9) for the IH group. In both headache groups 62% of subjects had a family history positive for headache. Table 3 summarizes the prevalence and ORs for putative risk factors for chronification of headache.
Prevalence and odds ratios of putative risk factors for chronification of headache

Values are number of subjects (%) unless otherwise stated.
Adjusted for educational level.
Adjusted for educational level and smoking.
Subjects used medication which could have been prescribed for either headache or a comorbid disorder (e.g. propanolol for migraine or hypertension).
Adjusted for educational level, smoking and medication overuse.
Adjusted for sleeping problems.
NH, No headache (<1 day/month); IH, infrequent headache (1–4 days/month); CFH, chronic frequent headache (>14 days/month); NSAIDs, non-steroidal anti-inflammatory drugs; GHQ, General Health Questionnaire.
Overuse of acute headache medication was strongly associated with CFH. Of the 109 subjects in the CFH group overusing acute headache medication, 90 (83%) overused one class of medication, 16 (15%) and three (3%) subjects overused two and three different classes, respectively. The percentage of smokers was similar in medication overusers (43%) and non-overusers (46%), mean difference 3% (95% CI −19, 12). Caffeine overuse was not associated with CFH. The average intake of caffeine in each group was seven units a day, including coffee, tea, ice-tea and cola.
CFH subjects reported sleeping problems more frequently than subjects with infrequent headache. Sleeping problems were not related to caffeine use. In the CFH group 74 of 170 (44%) reported sleeping problems on >3 nights/week; 66 (39%) had problems falling asleep and 26 (15%) awoke at night with headache. In contrast, 11 of 139 (8%) subjects with infrequent headache had sleeping problems on >3 nights/week; all had problems falling asleep, none awoke at night with headache. Tranquillizer use was higher in the CFH group than in both control groups but was no longer associated with CFH after adjusting for frequent sleeping problems.
In the CFH group, 62% of subjects screened positive for psychopathology. This percentage was the same for the new CFH group (those who changed to CFH). With a sensitivity and specificity of 0.84 and 0.82, respectively, the true prevalence of psychiatric comorbidity in CFH is estimated to be 66%. Results of the GHQ-28 are presented in Table 4.
General Health Questionnaire-28 scores

Values are means (SD) unless stated otherwise.
NH, No headache (<1 day/month); IH, infrequent headache (1–4 days/month); CFH, chronic frequent headache (>14 days/month); GHQ, General Health Questionnaire.
Discussion
We found a prevalence of CFH in the Dutch general population of 3.7%. This is in accordance with previous population-based studies (1–4). Although the prevalence worldwide is around 4%, we found a much higher prevalence in the practice with a high number of non-Western immigrants. Even if we consider all non-respondents in this particular practice to have a low headache frequency, the prevalence would still be higher. In the Netherlands, prevalence of poor reported health is highest among Turks and Moroccans (11). An adverse social and economic position may contribute to the poor health status of these ethnic minorities.
Our prevalence number is a reliable estimate due to the high response to the first questionnaire. The response to the second detailed and extensive questionnaire was low, but yielded high enough numbers to compare risk factors. Demographic characteristics were similar in the respondent and non-respondent CFH subjects, except for the higher percentage of non-Dutch names in the latter. As this is an indication of foreign ethnic origin, the language of the questionnaire might have been too difficult. The question is whether non-response introduced bias in the associations. If non-respondents are healthier than respondents, prevalence estimates of risk factors based on respondents could be overestimated. However, non-response does not necessarily cause bias in associations. In a large population-based study on risk factors for chronic disease conducted in the Netherlands (MORGEN-project) the response rate was 45%. Associations between lifestyle factors and health did not vary according to response status (12).
In many subjects headache frequency changed over time without specific headache treatment. Twelve percent had a clinically relevant decrease from >14 days to <7 days/month. This could be an underestimate, because the time between questionnaires ranged from 3 to 7 months, meaning that some participants were asked about overlapping time periods. This spontaneous change in headache frequency can be seen as regression towards the mean (13) and underscores the need for control groups when assessing efficacy of treatments for CFH. A decrease in headache frequency was associated with a decrease in headache medication overuse. However, medication use at baseline could not predict outcome in Q2. Our data correspond to two population-based follow-up studies in the USA and Taiwan, where after 1 and 2 years, respectively, only 44% and 35% still had CFH (4, 8). Whether subjects had received treatment or whether this was a spontaneous remission was not described.
We found that the majority of CFH subjects overused analgesics. The cross-sectional design of this study makes it impossible to determine the direction of causality. Improvement after withdrawal would make a causal relationship between overuse and chronification of headache likely. Since analgesics are mostly over-the-counter products, the GP may not be informed about the overuse. In fact, the majority had not consulted their GP for headache in the past 6 months. Many CFH subjects, however, frequently also suffer from sleeping problems and a substantial percentage use tranquillizers. Sleeping problems could be a possible cue for GPs to ask about headaches and analgesic use. Only 9% of CFH subjects used prophylactic medication to reduce headache frequency. To prevent overuse, physicians should inform the headache patient about restricting use of acute headache medication and the possibility of prophylactic therapy.
Smoking is associated with CFH. We assumed that medication overusers would show an overall tendency towards substance use; however, tobacco use did not differ between overusers and non-overusers. Nicotine induces dopamine release in the ventral striatum causing positive mood changes, which may relieve negative consequences of pain. Since we have no information on the age of onset of smoking, we do not know whether smoking could be more than a secondary phenomenon.
As found in other studies, CFH subjects had a lower educational level than subjects without CFH (2, 14). A low educational level is an indication of low socio-economic status, which is associated with poor health status in general. We do not think that headache interfered with scholarly achievement, because the mean age at onset of headache was 19 years.
About one-third of CFH subjects reported a history of head or neck trauma prior to the onset of headache. This may be partly due to recall bias. On the other hand, tissue injury might have triggered central sensitization, a pathological change in central pain processing observed in models of chronic pain (15).
In headache clinics, the majority of patients with CFH have comorbid psychiatric disorders (16–18). The most commonly reported disorders are major depression and generalized anxiety disorder, panic disorder and phobias. In our population-based study, 62% of subjects with CFH screened positive for psychopathology, twice as many as in the IH group. Breslau et al. found a bidirectional relationship between migraine and major depression, suggesting shared aetiological factors (19). Alternatively, pain may exacerbate a pre-existing vulnerability to psychopathology, which in turn intensifies the pain (20). This would imply that neither condition can be treated independently of the other.
The strength of our study is the large number of participants and the identification of associated factors by comparing the CFH group with control groups with infrequent headache and no headache. In both headache groups the majority were women, in contrast to the NH group. Female sex seems to be a risk factor for headache, not for the chronification of headache. A limitation of our study is that prevalence of risk factors is based on self-report, which is not as accurate as studies based on interviews by specialists and headache diaries.
We conclude that headache frequency fluctuates spontaneously and chronification is common. In the Netherlands the prevalence of CFH in the general population, aged 25–55 years, is 3.7%. We identified several risk factors to be associated with CFH, including overuse of analgesics, psychiatric comorbidity, smoking, sleeping problems, a history of head/neck trauma and a low educational level.
Competing interests
None declared.
Footnotes
Acknowledgements
This study was supported by an unrestricted grant of the Netherlands Organization for Scientific Research (NWO 940-31-049) and by the ‘Doelmatigheidsfonds van het LUMC’ (LUMC fund to increase efficiency in healthcare). We thank the following GPs for their participation: K. F. van de Bent, A. Boels, J. J. F. M. Brinkmann, T. Buiter-van der Kooi, H. A. Dirkse, L. van Duijn, F. M. M. van Exter, F. G. W. M. Haase, H. van Klei, J. Klein Haneveld, W. Libeton, C. H. Luirink, E. H. J. Smids, M. H. C. van der Velden, R. M. Vermeer, R. Voskuil, P. G. Willemse and B. W. de Zeeuw.
