Abstract
Carbachol induces headache in healthy subjects, but the migraine eliciting effect of carbachol has not previously been studied. We hypothesized that the cholinomimetic agonist carbachol would induce headache and migraine-like attacks in migraineurs. Carbachol (3 µg/kg) or placebo was randomly infused into 18 patients with migraine without aura in a double-blind crossover study. Headache was scored on a verbal rating scale from 0 to 10. Velocity in the middle cerebral artery (VMCA) and diameter of the superficial temporal artery (STA) were recorded. Fifteen patients experienced headache after carbachol compared with eight after placebo (P = 0.039). There was no difference in incidence of migraine-like attacks after carbachol (n = 8) compared with placebo (n = 6) (P = 0.687). Carbachol caused a decrease in VMCA (P = 0.044), but no change in STA (P = 0.089) compared with placebo. The study demonstrated that carbachol provocation is not a good model for experimental migraine.
Keywords
Introduction
Acetylcholine is a parasympathetic transmitter found in nerve fibres surrounding cerebral vessels (1–3). Acetylcholine interacts with endothelial muscarinic receptors (4–8), increases the production of endothelial nitric oxide (NO) via NO synthase (NOS) and dilates human cephalic vessels (9). The acetylcholine analogue, carbachol, degranulates mast cells and induces plasma protein extravasation (PPE) in the rat dura mater (10,11). Animal studies have shown that acetylcholine activates nociceptive fibres in the cornea (12) and carbachol in the skin (13). Furthermore, acetylcholine causes localized pain in man when applied to a blister base (14) or given as intra-arterial injection (15).
Recently, we showed that carbachol infusion induced mild headache and dilated cephalic vessels in healthy subjects (16). Carbachol, like acetylcholine, induces vasodilatation via endothelial NO production (17). It does not pass the blood-brain barrier (BBB) (18). These data indicate that carbachol-induced headache and vasodilatation are likely to be caused by endothelial production of NO. In contrast, the well-known migraine trigger and NO donor, glyceryl trinitrate (GTN), is highly lipid soluble and easily penetrates membranes, including the BBB. NO from GTN can therefore act both peripherally and centrally. The carbachol model of headache provides an opportunity to investigate whether an intravasally delivered drug acting outside the BBB may activate endothelial NOS and thereby induce migraine-like attacks.
In the present study, we hypothesized that intravenous infusion of carbachol would cause delayed migraine-like attacks in migraine patients without aura. We therefore performed a double-blind, placebo-controlled, crossover study in patients with migraine without aura.
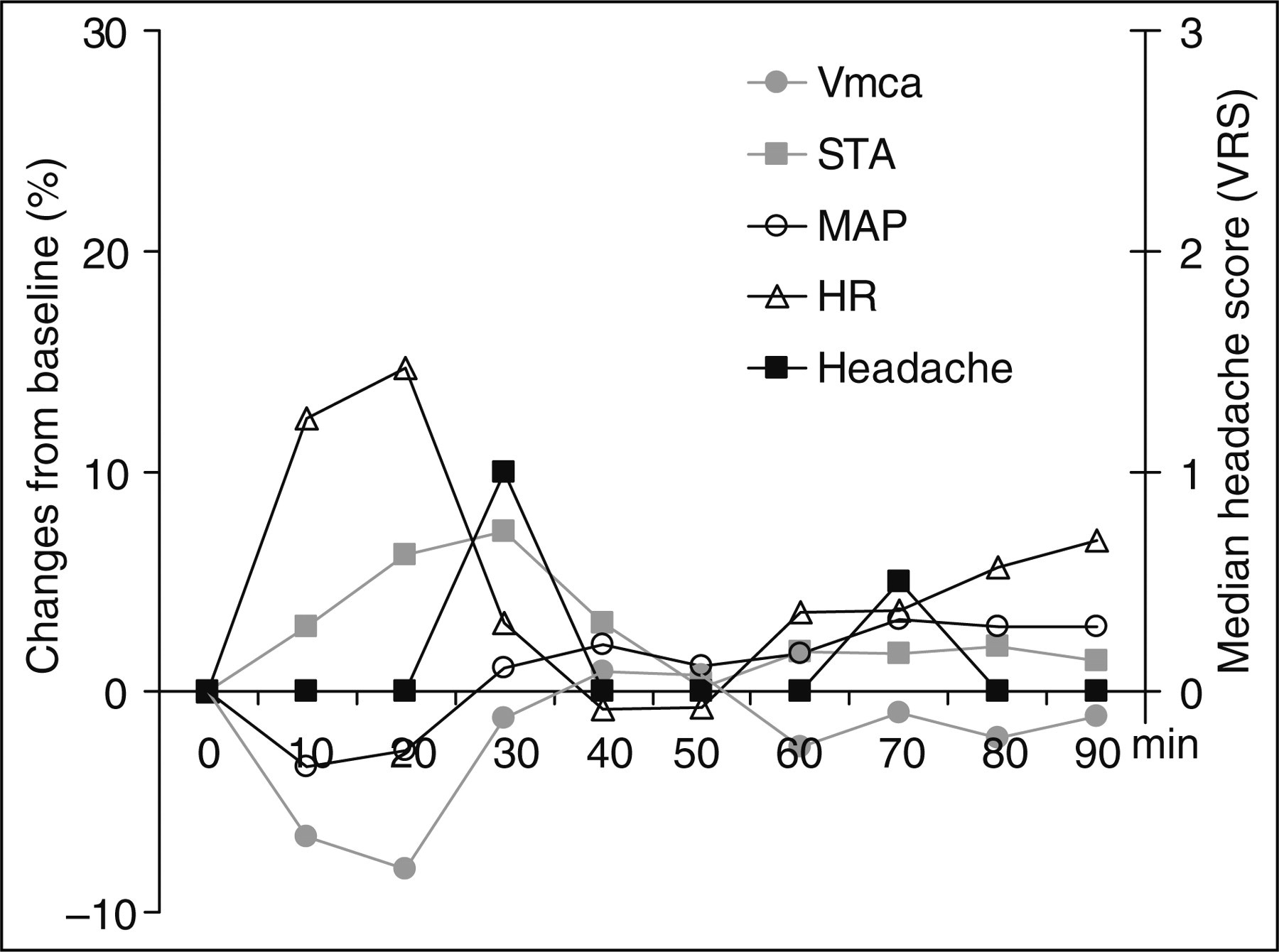
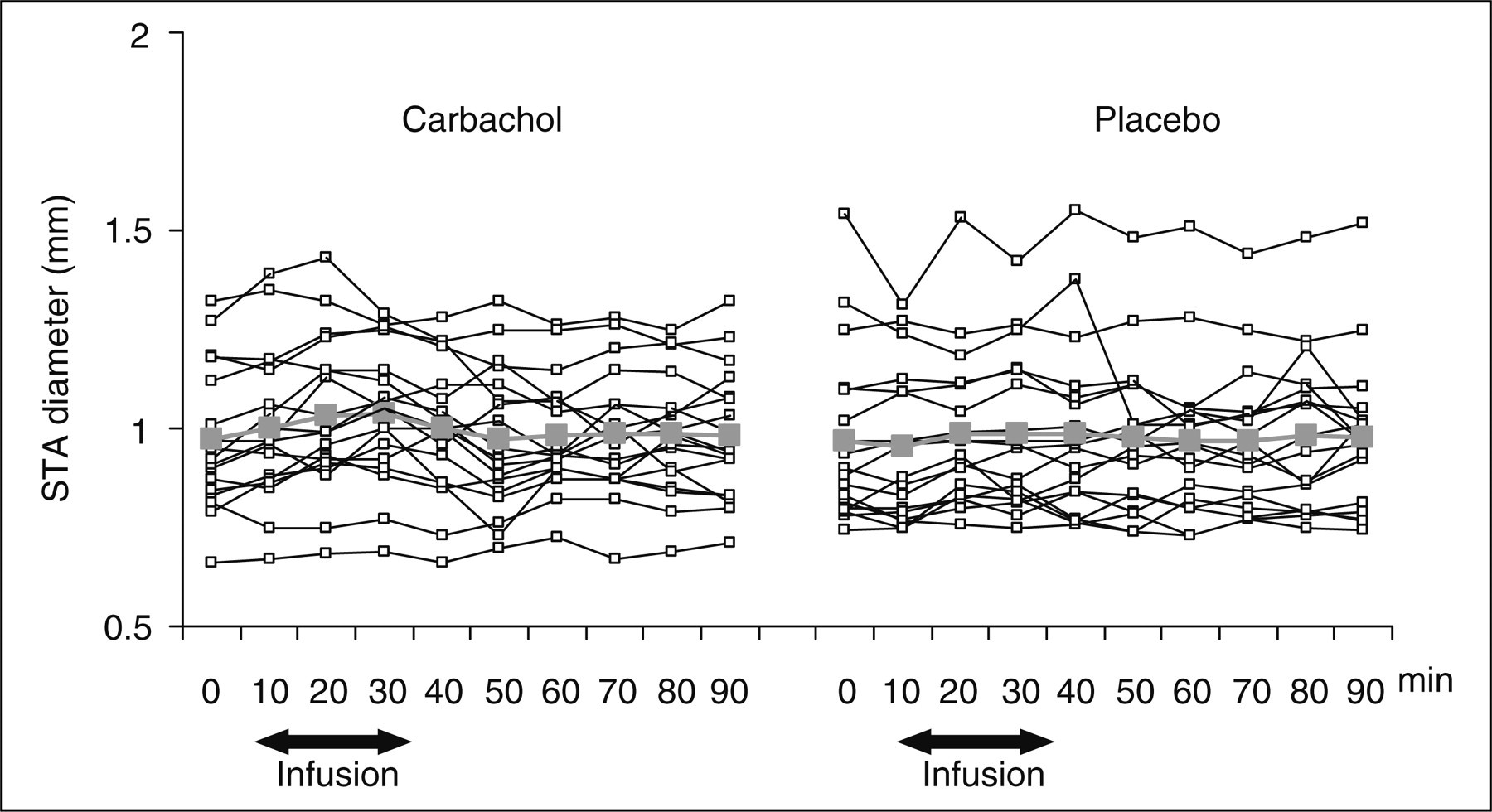
Individual and median headache scores on a verbal rating scale (VRS) from time 0–12 h after carbachol (▪) and placebo (□) infusion in migraine patients. Thick grey lines are median headache scores. The incidence of headache was higher after carbachol (n = 15) compared with placebo (n = 8) (P = 0.039). There was no difference in incidence of migraine-like attacks after carbachol (n = 8) compared with placebo (6) (P = 0.687). Haemodynamic changes from baseline in migraine patients after carbachol in comparison with median headache score on a verbal rating scale (VRS). Individual and mean blood flow velocities in the middle cerebral arteries (VMCA) assessed by transcranial Doppler ultrasonography for carbachol (▪) and placebo (□) day. Thick grey lines show mean values. Infusion of carbachol in migraine patients resulted in a decrease in VMCA during the infusion phase (0–30 min, P = 0.044) but not the post-infusion phase (30–90 min, P = 0.533) compared with placebo. Individual and mean diameters of the superficial temporal artery (STA) assessed by high-resolution ultrasonography for carbachol (▪) and placebo (□) day. Thick grey lines show mean values. Carbachol did not change STA diameter compared with placebo during the infusion (P = 0.089) or post-infusion phases (P = 0.779).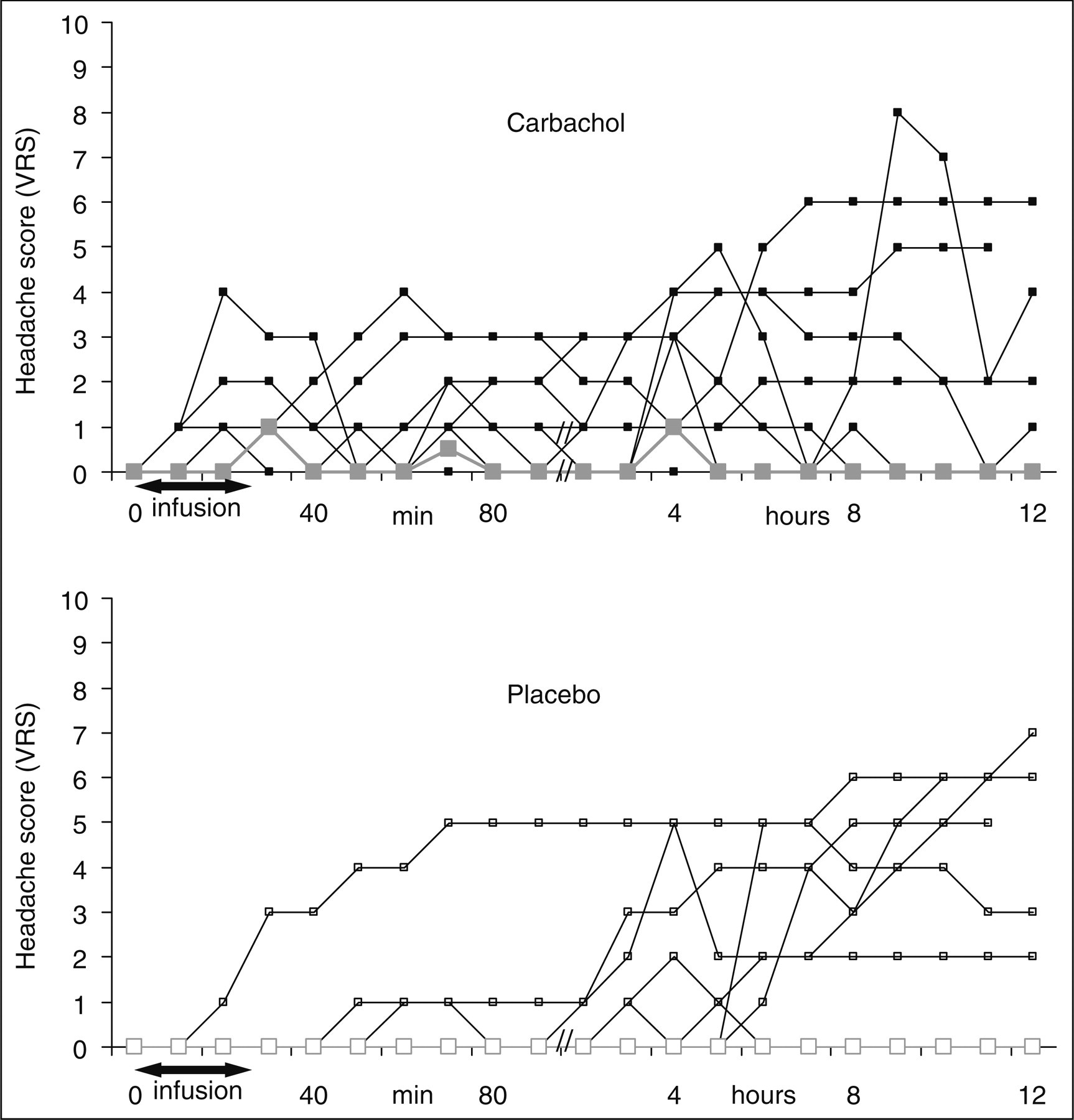

Design and methods
We recruited 18 patients with migraine without aura (MO) according to the International Headache Society (IHS) (19). Exclusion criteria were: a history of any other type of headache (except episodic tension-type headache less than 5 days per month); any daily medication apart from oral contraceptives; and serious somatic or psychiatric disease.
The Committees on Biomedical Research Ethics of the Capital Region of Denmark approved the study (HC20070049). All subjects gave informed consent to participate in the study, which was undertaken in accordance with the Helsinki Declaration of 1964, as revised in Edinburgh in 2000.
Experimental design
All subjects were randomly allocated in a balanced order to receive 3 µg/kg carbachol or placebo (isotonic saline) over 25 min on 2 days separated by a least 1 week. Before the experiment each subject underwent a general physical examination. All subjects reported to the laboratory, headache free, at 08.30. The experiment was postponed if the subject had had migraine attack within 5 days before the start of the study. The intake of coffee, tea, cocoa or other methylxanthine-containing foods or beverages was not allowed for the last 8 h prior to the study. All procedures were performed in a quiet room at a temperature of 25°C. The subjects were placed in the supine position and a venous catheter was inserted into the right antecubital vein for drug infusion. The subject then rested for at least 30 min before baseline measurements of blood pressure, heart rate (HR) and ECG were performed, and the infusion started using a time and volume controlled infusion pump. Headache intensity, mean velocity of the middle cerebral artery (VMCA), superficial temporal artery (STA) diameter, end-tidal partial pressure of CO2 (PetCO2), adverse events and vital signs were recorded before and then every 10 min until 90 min after the beginning of infusion. The subjects were discharged from the hospital after finishing the measurements and were asked to complete a headache diary every hour until 12 h after start of the infusion. The diary included headache characteristics and accompanying symptoms, any rescue medication, adverse events and whether headache was believed to mimic their usual migraine attack. Subjects were allowed to treat headache with over the counter rescue medication or their usual migraine treatment.
Headache and migraine-like attack criteria
Headache intensity was recorded repeatedly on a verbal rating scale (VRS) from 0 to 10 (0, no headache; 1, a very mild headache, including a feeling of pressing or throbbing—pre-pain; 10, worst imaginable headache) (20). Headache characteristics and associated symptoms were also recorded to determine the quality and type of the headache.
Experimental headache induced by infusion of a neurotransmitter cannot fulfil strict IHS criteria for migraine without aura (19). First, the migraine-like attacks reported are induced by pharmacological substances and can therefore not be spontaneous, though they phenotypically mimic spontaneous migraine attacks in the majority of patients (21,22). Second, most spontaneous migraine attacks develop in a matter of hours and often go through a phase where they phenomenologically fulfil the criteria for tension-type headache before the headache gets worse, becomes unilateral and has the associated symptoms required for migraine. For this reason attacks aborted by migraine-specific treatment before fulfilling all criteria for migraine were accepted in the new criteria for chronic migraine (23). Patients in experimental provocation studies cannot be denied treatment of the induced attacks and often treat before all migraine criteria are fulfilled.
Based on these considerations we have used the following two criteria for a migraine-like attack induced 0–12 h after infusion of an experimental drug.
Migraine-like attack attacks fulfilling either 1 or 2:
Headache fulfilling criteria C* and D for migraine without aura (19).
Headache described as mimicking usual migraine attack and treated with a triptan.
Moderate to severe pain intensity is considered ≥4 on the VRS.
Middle cerebral artery blood flow velocity
VMCA was recorded bilaterally with transcranial Doppler (TCD) with hand-held 2 MHZ probes (Multidop X; DWL, Sipplingen, Germany). Fixed probes were avoided, because they may cause discomfort and even headache (24). Four recordings were taken and averaged at each time point. One recording is a time-averaged mean over 4 s or approximately four cardiac cycles. Identification of the MCA was done as previously described (25). Every TCD recording was performed by the same trained physician (HWS). PetCO2 was recorded simultaneously with TCD recordings using an open mask that caused no respiratory resistance (ProPaq Encore®; Welch Allyn Protocol, Beaverton, OR, USA). According to Dahl et al. (26), the changes in diameter (Δd) of MCA can be calculated if CBF is unchanged, which we showed in healthy subjects. Thus, after correcting VMCA with e0.034 for each mmHg change in PetCO2(27) the change in MCA diameter can be estimated as:

Diameter of the superficial temporal artery
Diameter of the frontal branch of the STA was measured by a high resolution ultrasonography unit (Dermascan C; Cortex Technology, Hadsund, Denmark: 20 MHz, bandwidth 15 MHz) as previously described (28,29).
Vital signs
Heart rate and blood pressure were measured every 10 min using an auto-inflatable cuff (ProPac Encore®; Welch Allyn Protocol). ECG (Cardiofax V; Nihon-Koden, Shinju-ku, Tokyo, Japan) was monitored on a LCD screen and recorded on paper every 10 min.
Data analysis and statistics
All values are presented as mean values (±
The half life of carbachol is not known, but infusion of carbachol in healthy subjects has previously shown vascular effects occurring primarily during infusion (16). We therefore defined two phases during the hospital phase (0–90 min): an infusion phase as 0–30 min and a post-infusion phase from 30 to 90 min after start of infusion. The hospital phase was followed by a post-hospital phase 1.5–12 h after start of infusion.
We calculated area under the curve (AUC) according to the trapezium rule (30) to obtain a summary measure and to analyse the differences in response between carbachol and placebo. Baseline was subtracted before calculating AUC to reduce variation between sessions within subject.
The primary end-points were: the difference in incidence of headache or migraine-like attacks between carbachol and placebo; and the difference in AUC for headache score during infusion (0–30 min), post-infusion (30–90 min) and post-hospital phase (1.5–12 h) between carbachol and placebo. The secondary end-points were difference in AUC for VMCA, diameter of STA, heart rate, MAP, systolic blood pressure, diastolic blood pressure and PetCO2 between carbachol and placebo day.
Binary categorical data were analysed with McNemar test. Analysis of AUC values was performed with a paired two-way t-test, except headache scores where data were tested with the Wilcoxon signed rank test. We tested for period and carry-over effects for all baseline variables with the Mann–Whitney test and independent t-test.
All analysis was performed with
Results
Eighteen migraine patients (18 F, mean age 31, range 19–44 years) were screened and completed the study on both study days. The migraine patients had a mean attack frequency of 25 attacks per year, ranging from 6 to 54 attacks per year.
We found no differences in vascular variables between the two experimental days at baseline, except for VMCA, which was higher on the carbachol day compared with the placebo day (P = 0.038). There was no carry-over or period effect for baseline values of VMCA, STA, MAP, HR or PetCO2.
Headache and migraine-like attacks
Headache characteristics after carbachol in 15 migraine patients
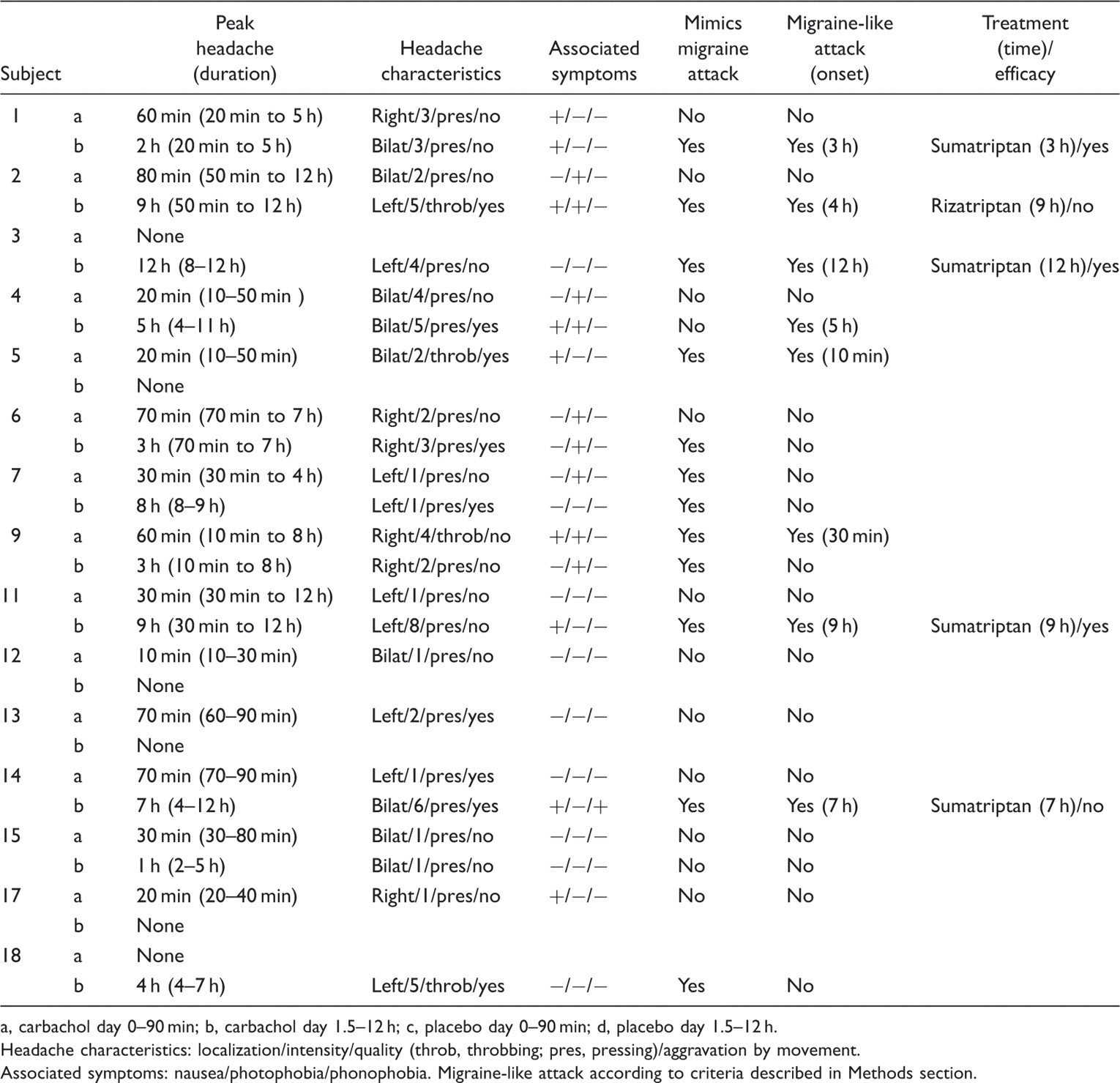
a, carbachol day 0–90 min; b, carbachol day 1.5–12 h; c, placebo day 0–90 min; d, placebo day 1.5–12 h.
Headache characteristics: localization/intensity/quality (throb, throbbing; pres, pressing)/aggravation by movement.
Associated symptoms: nausea/photophobia/phonophobia. Migraine-like attack according to criteria described in Methods section.
There was no difference in incidence of migraine-like attacks after carbachol (n = 8) compared with placebo (n = 6) (P = 0.687). The median peak headache score was 2 (range 0–8) after carbachol. The median time to peak headache after carbachol was 1.0 h (range 0–12). After carbachol five subjects took triptans compared with four subjects after placebo.
During the hospital phase AUC for headache was larger after carbachol compared with placebo during the infusion (AUC0–30 min, P = 0.012) and the post-infusion phases (AUC30–90 min, P = 0.028). There was no difference in AUC between carbachol and placebo (AUC1.5–12 h, P = 0.972) in the post-hospital phase.
Middle cerebral artery
Mean peak haemodynamic variables 0–90 min after carbachol infusion in 12 healthy subjects and 18 migraine patients

Data from healthy subjects have previously been published elsewhere (16).
MCA, middle cerebral artery; STA, superficial temporal artery; MAP, mean arterial blood pressure.
Carbachol did not change PetCO2 compared with placebo during infusion (AUC0–30 min, P = 0.054) or post-infusion (AUC30–90 min, P = 0.668).
Superficial temporal artery
There was no difference in STA diameter for carbachol compared with placebo during infusion (AUC0–30 min, P = 0.089) or post-infusion phases (AUC30–90 min, P = 0.779) (Figs 2 and 4 and Table 2).
Heart rate and mean arterial blood pressure
We found an increase in heart rate for carbachol compared with placebo during infusion (AUC0–30 min, P = 0.001), but not during post-infusion (AUC30–90 min, P = 0.589) (Fig. 2 and Table 2).
The MAP decreased for carbachol compared with placebo during infusion (AUC0–30 min, P = 0.012), but not during post-infusion (AUC30–90 min, P = 0.201). Diastolic blood pressure decreased during infusion (AUC0–30 min, P = 0.005), but not during post-infusion (AUC30–90 min, P = 0.529). Systolic blood pressure did not differ between carbachol and placebo during infusion (AUC0–30 min, P = 0.514), but it increased post-infusion after carbachol (AUC30–90 min, P = 0.034).
Adverse events
Adverse events were recorded and reported during both the infusion and post-infusion phases. Increased saliva (P < 0.001), sweating (P = 0.001), urge to void (P = 0.039), lacrimation (P = 0.016), heat sensation (P < 0.001) and headache (P = 0.002) were more often reported for carbachol than placebo.
Discussion
The major finding of the study was that there was no difference in incidence of migraine-like attacks after carbachol compared with placebo in migraine patients without aura. Furthermore, carbachol induced more headaches than placebo, but only of mild intensity.
Carbachol vs. GTN in migraine
Given that carbachol dilates cerebral vessels through activation of endothelial NO, we expected that carbachol infusion, similar to GTN infusion, would result in a high percentage of migraine-like attacks in migraine patients without aura. Thus, migraineurs reported migraine-like attacks in 80% of cases after GTN (0.5 µg/kg) and in only 10% of cases after placebo (21). In the present study 44% of patients reported migraine-like attacks after carbachol vs. 33% after placebo. Furthermore, the GTN study reported median peak headache intensity of 5.5, a decrease in VMCA of 30.1% (21) and using a lower dose of GTN (0.25 µg/kg) an increase in STA diameter of 37% in migraineurs (31). In comparison, we observed a median peak headache intensity of 2 after carbachol, a decrease in VMCA of 8.1% and an increase in STA diameter of 7.2%. Taken together these data suggest that carbachol in the dose given is weaker than GTN in induction of migraine-like attacks and vasodilatation.
Why did carbachol fail to induce migraine-like attacks?
GTN-induced headache is dose dependent (20). Therefore it is possible that the dose of carbachol applied in this study was insufficient to cause migraine-like attacks. In the pilot study we found that 3 µg/kg carbachol is the maximal dose tolerable, because larger doses induced intolerable systemic side effects in healthy controls (16). Thus, a higher dose of carbachol could not be applied. It is likely that carbachol did not induce sufficient endothelial NO production to trigger migraine-like attacks. This is also suggested by the low response in haemodynamic variables in the present study, which is comparable with the response to carbachol in healthy subjects (Table 2). In addition, it has been shown that the effects of carbachol can be less NO dependent (32). Acetylcholine would be preferable to carbachol, as the natural agonist. It is, however, practically impossible to administer acetylcholine in a human headache model, and carbachol is the best agonist to mimic the effects of acetylcholine.
We found that after carbachol 83% of migraine patients developed headache with a mean peak headache intensity of 2 (range 0–8) vs. 75% of healthy subjects with a mean peak headache intensity of 1 (range 0–3) (16). Thus, the current dosage of carbachol seems to elicit almost the same headache response in migraineurs and healthy subjects. It could be argued that a difference in incidence of migraine-like attacks could be confounded by a difference in intake of rescue medication on the two trial days. However, on the carbachol day five subjects took oral triptans compared with four subjects on placebo day. The high placebo response of migraine-like attacks could influence the results of the present study. In contrast to previous studies, where usually 0–17% of subjects experience a migraine-like attack after placebo (21,28,33), a high number of patients (33%) experienced migraine-like attacks after placebo. We cannot find any good explanation for this unusual placebo response. We have previously performed a study with similar design, where PACAP38 or placebo was infused and none had a migraine-like attack on placebo (34). However, the six subjects reporting migraine-like attack on the placebo day might be very sensitive to the stress of the experiment. Thus, the current study exemplifies the importance of conducting human experimental headache studies in a balanced randomized placebo-controlled double-blind crossover design.
Carbachol did induce more headaches in migraine patients compared with placebo, which could be attributed to production of endothelial NO. Other mechanisms involved could be activation of dural nociceptors (12,13), degranulation of dural mast cells (10) and release of endothelial prostacyclin (PGI2) (35,36). These mechanisms should be investigated in future human and animal models.
Conclusion
The present study showed that carbachol provocation is not a good model for experimental migraine. Carbachol in the maximal tolerated dose induced mild headache, but no more migraine-like attacks than placebo and a low degree of cephalic dilatation. The lack of migraine induction can be due to the dose limitation posed by systemic side effects.
Footnotes
Acknowledgements
The authors wish to thank laboratory technicians Winnie Grønning Nielsen and Lene Elkjær for their excellent and dedicated assistance. The study was supported by the Mauritzen La Fontaine Foundation and the Lundbeck Foundation via the Lundbeck Foundation Center for Neurovascular Signaling.
