Abstract
The objective of this study was to estimate and contrast the occurrence of ictal and interictal cutaneous allodynia (CA) in individuals with migraine with and without temporomandibular disorders (TMD). Both TMD and CA are common in migraine and may be associated with migraine transformation from episodic into a chronic form. Herein we hypothesize that TMD contributes to the development of CA and to more severe headaches. In a clinic-based sample of individuals with episodic migraine, the presence of TMD was assessed using the research diagnostic criteria for myofascial or mixed (myofascial and arthralgic) TMD. Ictal CA was quantified using the validated Allodynia Symptom Checklist (ASC-12). The ASC-12 measures CA over the preceding month by asking 12 questions about the frequency of allodynia symptoms during headaches. Interictal CA was assessed in the domains of heat, cold and mechanical static allodynia using quantitative sensory testing. Our sample consists of 55 individuals; 40 (73%) had TMD (23 with myofascial TMD and 17 with the mixed type). CA of any severity (as assessed by ASC-12) occurred in 40% of those without TMD (reference group), 86.9% of those with myofascial TMD (P = 0.041, RR = 3.2, 95% CI = 1.5–7.0) and in 82.3% of those with mixed TMD (P = 0.02, RR = 2.5, 95% CI = 1.2–5.3). Individuals with TMD were more likely to have moderate or severe CA associated with their headaches. Interictally (quantitative sensory testing), thresholds for heat and mechanical nociception were significantly lower in individuals with TMD. Cold nociceptive thresholds were not significantly different in migraine patients with and without TMD. TMDs were also associated with change in extra-cephalic pain thresholds. In logistical regression, TMD remained associated with CA after adjusting for aura, gender and age. TMD and CA are associated in individuals with migraine.
Introduction
A subgroup of episodic migraine sufferers evolve to a stage where they have headaches on more days than not (chronic migraine, CM) through a process often termed migraine progression or migraine transformation (1,2). Because this process occurs in a sizeable minority of migraine sufferers, identifying factors that predict the change from episodic migraine to CM is of public health importance (3). Identifying these risk factors will provide insights into the mechanisms of disease progression and provide a foundation for the development of interventions intended to modifying the course of the illness (4).
Among the risk factors for CM, comorbidities figure prominently (5,6). There is evidence that migraine is comorbid with a range of other pain disorders, including limited evidence for temporo-mandibular disorders (TMDs) (7–11). In patients with both TMDs and migraine, three sorts of overlapping possibilities are likely. First, TMDs may function as an independent source of pain, adding to the overall health burden (12). Second, TMDs may worsen existing primary headaches and therefore add to the burden of the headache disorders (12). Finally, TMDs may be a risk factor for the development of CM (13,14).
TMDs and migraine may be linked through the phenomenon of central sensitization and cutaneous allodynia (CA) in the distribution of the trigeminal nerve. The prevalence of CA is higher in chronic than in episodic migraine, and is very low in tension-type headache, suggesting that CA is at least a marker of migraine frequency (15). In longitudinal analyses, CA is a predictor of incident CM (15). CA is believed to arise from sensitization of the first and second order neuron in the sensory pathway. Sensitization of neurons in the trigeminal nucleus caudalis may also arise during TMDs (16,17). This would be of importance, because repetitive activation of trigeminovascular neurons may produce long-term changes. For example, repetitive activation of modulatory pain pathways involving the periaqueductal grey area (PAG) may lead to impairment of function or partial neuronal cell damage, through the liberation of free radicals in the PAG or in areas involved with migraine generation. Iron deposits in the PAG demonstrated using MRI methods are consistent with this concept (18).
Because both TMDs and CA have been hypothesized as risk factors for migraine progression from episodic into CM, and TMDs may contribute to the development of central sensitization, herein we measure CA in migraine sufferers with and without TMDs, hypothesizing that those with TMD are more likely to have CA.
Methods
Our sample was enrolled in a tertiary care outpatient headache clinic during the year of 2008. We enrolled consecutive patients meeting the following inclusion and exclusion criteria: age between 18 and 65 years; an established diagnosis of episodic migraine with or without aura according to the criteria of the second edition of the International Classification of Headache Disorders (12); attack frequency ranging from 2–14 headache days per month; and willing and able to participate in the study.
All patients were seen at least 48 h after their last headache attack, when they had no headaches.
Assessment of TMD
Classification of temporomandibular disorders (TMD) according to research diagnostic criteria (RDC/TMD) (19)

Assessment of CA
We assessed CA using two validated methods. To estimate the presence of allodynia over a 3-month period, we used the 12-item Allodynia Symptom Checklist (ASC-12) (17,22,23). To obtain an immediate and objective measure of allodynia, we used quantitative sensory testing (QST). Accordingly, we obtained measures of CA over a period of time (ASC-12) and during the visit to the clinic (QST). Assessments of CA happened in a blinded fashion to the assessment of TMD. Although participants were aware of the hypothesis of the study (because informed consents listed it), they were not given a diagnosis of TMD until the end of the study.
The ASC-12 questionnaire
The ASC-12 included questions about the frequency of various allodynia symptoms in association with headache attacks, as per the validation study (17). For individuals with more than one type of headache (e.g. migraine and also tension-type headaches), questions were directed to the ‘most severe type of headache’, based on the prior evidence indicating that the most severe type was likely to be migraine (24). Instead of using a dichotomous option (yes or no), the response categories were never, rarely, less than half the time, and half the time or more.
Individual ASC items were scored as 0 (i.e. never or rarely, or does not apply to me), 1(less than half the time) and 2 (half the time or more), yielding scores that ranged from 0 to 24. In the development of ASC, alternative scoring strategies were evaluated but did not alter the results. The validation process defined the following categories, based on the ASC- 12 scores: no allodynia (0 to 2), mild (3–5), moderate (6–8) and severe (9 or higher).
Quantitative sensory testing
QST procedures were used to determine pain thresholds for cold, heat and mechanical stimuli using standardized procedures (16). Heat and cold skin stimuli were delivered through a 30 × 3 × 30 mm2 thermode (Thermal Sensory Analyzer-2001, Medoc, Ramat Yishai, Israel) attached to the skin at a constant pressure and their pain thresholds were determined by using the method of limits (25). Thresholds were measured in the following locations in a randomized order: left and right periorbital (LV1 and RV1); left and right masseter (LM and RM); and left and right forearm ventral skin (Lfor and Rfor).
To determine temperature pain thresholds, we allowed the skin to adapt to a temperature of 32°C for 5 min, and then tests were initiated, with temperatures varying at a standardized rate (1°C/s) until pain sensation was perceived (threshold for pain determined). Thermal stimuli were repeated three times each, and the mean of the values was considered as the threshold.
Mechanical pain thresholds were determined by using a set of 20 calibrated von Frey hairs (VFH; Touch-TestTM Sensory Evaluator, North Coast Medical Inc., Morgan Hill, CA). Each VFH monofilament was assigned a scalar number in an ascending order. Because a linear relationship exists between the log force and the ranked number, mechanical pain thresholds are expressed as VFH numbers rather then their forces (g). Each monofilament was applied to the skin three times for 2 s, and the smallest VFH number capable of inducing pain in two of three trials was considered the threshold.
We used summary tables and descriptive statistics to summarize our findings. The Fischer test was used to compare proportions. ANOVA with post-test was used for comparisons among the three groups, after normality was demonstrated. We used Spearman Rank correlation to test correlations between CA and DTM. Tests were two-tailed, and a significant level of 0.05 was defined. We powered our study to detect seven units of difference in the QST with a standard deviation of 10 (sample required for 80% power was 53 participants). As part of the exploratory analyses, we separated patients with mixed TMD into those with and without pain. Because we found no evidence of differences, we elected to maintain the mixed group as defined.
Results
Our sample consists of 55 individuals. Forty subjects met RDC criteria for one or more types of TMD (72.72%). The 15 subjects without TMD were largely female (73.3%) and had a mean age of 45.9 years. There were 23 subjects with myofascial TMD (95% female, mean age = 39.9 years); 17 had mixed TMD (88.2% female, mean age = 43.2 years) and only three of them had no pain in the temporomandibular joint (17.6%). Pooling individuals with any form of TMD together, they were more likely to be female, although differences did not reach statistical significance (P = 0.07). The groups did not differ significantly regarding age.
Migraineurs with no TMD had a mean of 7 days of headache per month; those with myofascial TMD had a mean of 8.11; mixed TMD had a mean of 7.7 (differences are non-significant).
CA during headaches (ASC-12)
Presence and severity of headache-related allodynia, as measured by the Allodynia Symptom Checklist (ASC-12)
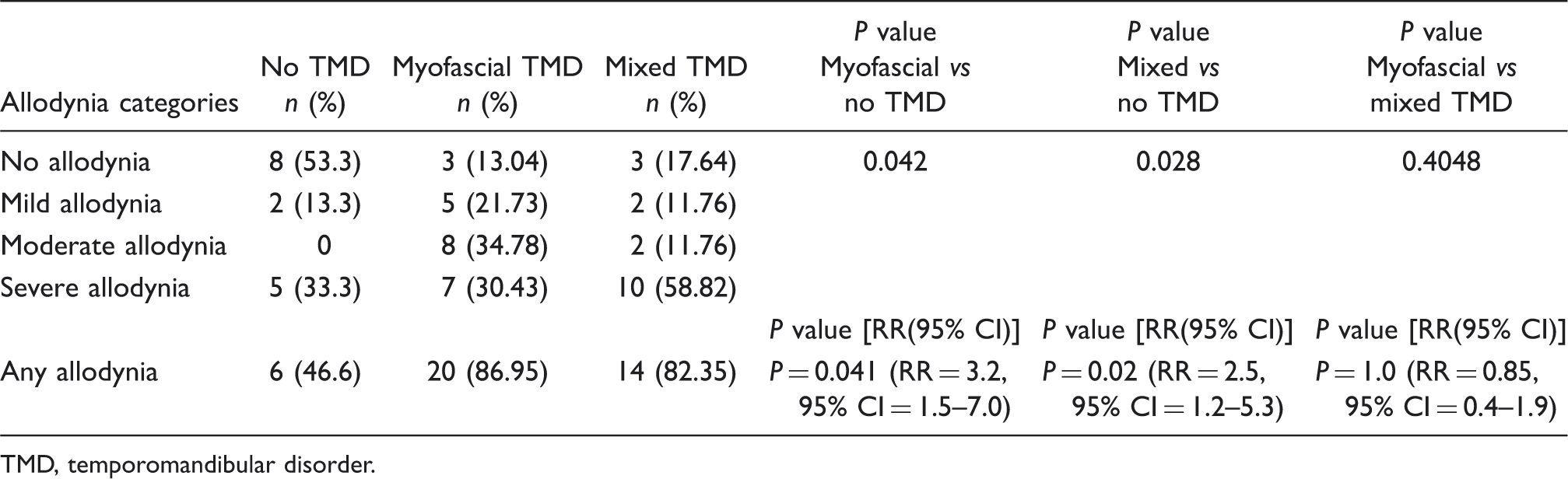
TMD, temporomandibular disorder.
ASC-12 provides a measure of CA overall and separate quantitative indices in each of three domains (Fig. 1). CA scores were higher for each type of allodynia in the myofascial group vs no TMD group, and were highest in the mixed group. The differences were statistically significant (overall and for all domains) in the contrasts between the mixed and no TMD groups.
Allodynia scores, as measured by Allodynia Symptom Checklist-12.
Quantitative sensory testing
Interictal nociceptive thresholds to temperature, as measured by quantitative sensory testing, as a function of presence and type of temporomandibular disorders (TMD)
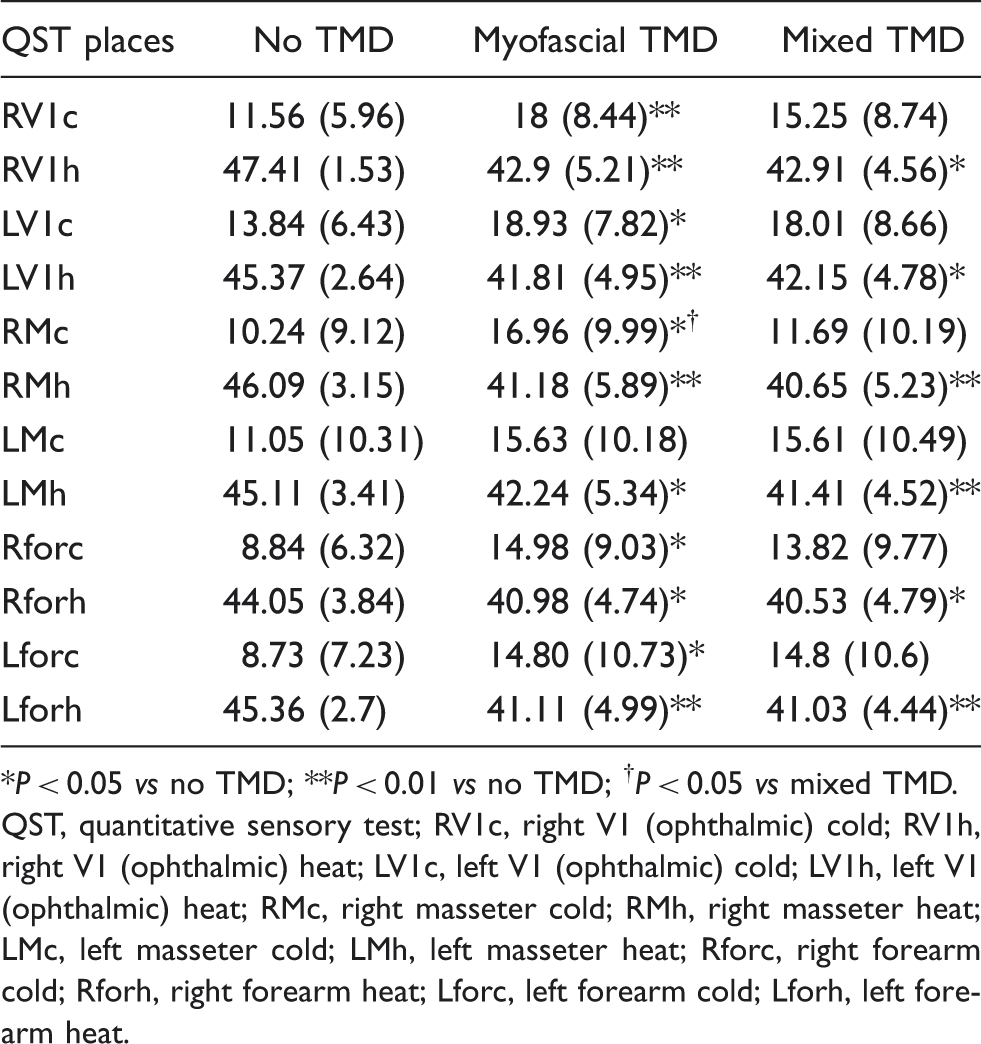
*P < 0.05 vs no TMD; **P < 0.01 vs no TMD; † P < 0.05 vs mixed TMD.
QST, quantitative sensory test; RV1c, right V1 (ophthalmic) cold; RV1h, right V1 (ophthalmic) heat; LV1c, left V1 (ophthalmic) cold; LV1h, left V1 (ophthalmic) heat; RMc, right masseter cold; RMh, right masseter heat; LMc, left masseter cold; LMh, left masseter heat; Rforc, right forearm cold; Rforh, right forearm heat; Lforc, left forearm cold; Lforh, left forearm heat.
Interictal mechanical nociceptive thresholds, as measured by quantitative sensory testing, as a function of presence and type of temporomandibular disorders (TMD)
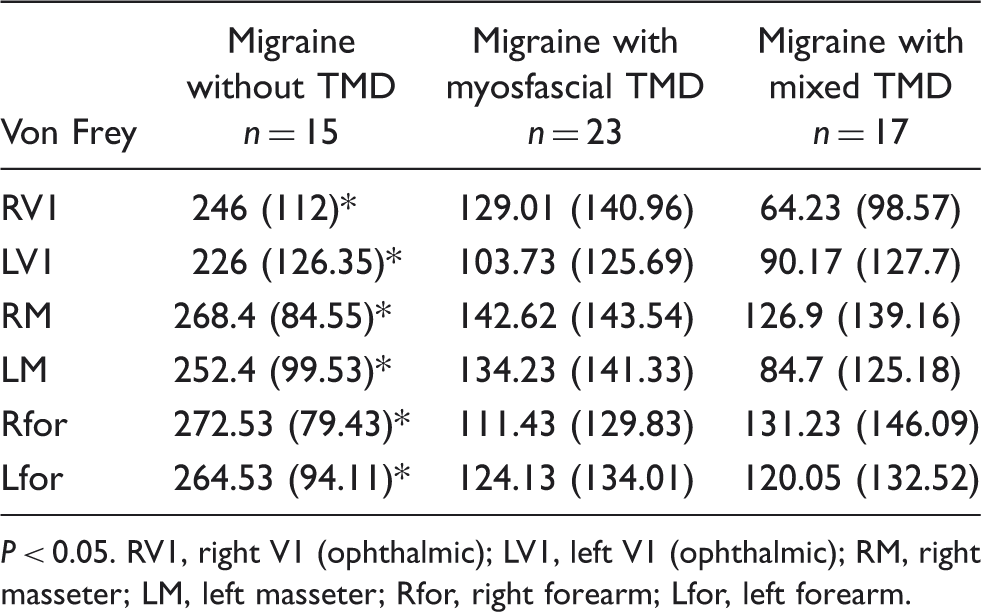
P < 0.05. RV1, right V1 (ophthalmic); LV1, left V1 (ophthalmic); RM, right masseter; LM, left masseter; Rfor, right forearm; Lfor, left forearm.
In logistical regression, we modeled allodynia (moderate or severe vs none or mild) as a dichotomous outcome using TMD, aura, gender and age as predictor variables. TMD was an independent predictor of allodynia in the fully adjusted model (OR = 1.8, 95% CI = 1.3–2.3).
Discussion
In this study, 72.2% of unselected episodic migraine sufferers from a headache specialty practice met criteria for TMD. Because both CA and TMD involve trigeminal nociceptive transmission and because both are associated with migraine we assessed the relationship between TMD and allodynia in migraine (10,15). Our results can be summarized as follows. (i) TMDs are frequent in individuals with migraine seeking care in a headache centre. (ii) During headache attacks, individuals with episodic migraine and TMDs are two to three times more likely to experience CA symptoms, as measured by the ASC-12; furthermore, they are significantly more likely to experience moderate or severe CA symptoms. (iii) Individuals with episodic migraine and TMD have significantly higher ASC-12 scores, as compared with individuals without TMD. (iv) Interictally, as assessed by QST, thresholds for heat and mechanical nociception are significantly lower in individuals with TMD. (v) For cold nociceptive thresholds, mean values were significantly higher for myofascial TMD as compared with controls; differences were not significant for mixed TMD. This is likely to be driven by our sample size. For some parameters both the means and SDs are virtually identical for the myofascial and mixed groups, but the first group reached significance, when compared with the no TMD group, while the latter group did not. (vi) TMDs were associated with change in extra-cephalic pain thresholds. (vii) Myofascial and mixed TMDs are similarly associated with CA and pain thresholds.
The relative frequency of TMDs was higher in this sample than in previous reports from general populations. This issue should be confirmed and the reasons for this potential association should be explored. It is possible that individuals with migraine and TMDs have more severe disease and are more likely to be referred to tertiary centres. As both migraine and TMD predispose to allodynia and sensitization, having one disorder (e.g. migraine) might predispose to the development or diagnosis of the other (e.g. TMD). Shared environmental exposures or shared biological predisposition could also account for the comorbidity and should be explored in future studies (26).
CA refers to the perception of pain after non-painful stimuli. Trigeminal CA is the clinical marker of sensitization at the level of the second order sensory neurons, whose cell bodies are in the trigeminal nucleus caudalis (27,28). Clinic-based studies first suggested that about two-thirds of migraine sufferers develop CA over the course of their attacks (16,29). In the population, it was found that the prevalence of CA was significantly higher in CM (68.3%) than in episodic migraine (63.2%, P < 0.01), and higher in both of these groups compared with other headaches (17). The prevalence of severe CA followed the same pattern. Accordingly, CA has been suggested as a risk factor for CM (30).
The development of CA is influenced by a number of potentially modifiable risk factors and some that are non-modifiable (15). Potentially modifiable risk factors for CA include high attack frequency, high pain intensity and disability, depression and obesity (30). Our data suggest that TMDs may also be modified risk factors for CA. This is of importance because TMDs are highly prevalent in the population, with rates varying from 21.5% to 51.8% (31–33).
It is well established that neurons in the nucleus caudalis integrate nociceptive input from intracranial and extracranial tissues and receive supraspinal facilitatory, as well as inhibitory, inputs. They sum all these inputs and project the net results to the thalamus and onto the cortex (34). Migraine is characterized by activation of the trigeminal nucleus caudalis. Accordingly, several explanations exist to explain the association between migraine, CA and TMD. First, it may be that migraine leads to activation of the trigeminal system, with increased predisposition to allodynia but also to temporomandibular joint pain (because of inflammatory release on the second and third division of the trigeminal nerve). Alternatively, it can be hypothesized that nociceptive inputs from masticatory muscle or temporomandibular joints could lead to activation of the trigeminal nucleous caudalis (35). Further, the presence of proinflammatory factors at the temporomandibular joints could be another form of sensitization. High levels of prostaglandin E2, cytokines such as interleukin 1 and interleukin 6 and tumor necrosis factor have been detected in synovial fluid of inflamed joints and were strongly associated with pain (36). Also, calcitonin gene-related peptide, a major contributor to neurogenic inflammation and nociception, substance P and serotonin have also been detected in the temporomandibular joints (37,38). In addition to these two causal pathways (migraine leading to both TMD and CA vs TMD predisposing to both migraine and CA), it may be that some individuals are predisposed to pain overall (shared biological predisposition). Indeed, several studies reported that TMD patients, like migraine patients, exhibit greater sensitivity to pain in multiple body areas, implicating a generalized dysfunction of the nociceptive systems, and supporting the concept of a generalized upregulation of nociceptive processing (39). Upregulation of central nociceptive pathways is also seen in myofascial pain (40,41) or temporomandibular arthralgia (42). Thus, biological predisposition would lead to both TMD and migraine, and CA would be a marker of the activation imposed by both. In addition, perhaps a confounder such as comorbid depression may contribute to the association of CA with TMD in migraine. In prior studies, CA was more common in migraineurs who also had depression (15). Finally, it has been described that migraine features correlate with the presence and severity of CA, with fairly robust odds (17). If individuals with migraine and TMD have more typical migraine attacks, the presence of CA may be only indirectly related to TMD. These complex relationships should be explored in subsequent studies, because we are underpowered to conduct these analyses in our study. TMD symptoms may cause an excitatory impact on central sensitization, and this would be reflected by an increased prevalence of CA.
Some caution is required when interpreting our results. First, our sample size is modest; though results are consistent and statistically significant we do not have power for multivariate adjustment. Also, we were surprised (because the study was conducted in blinded fashion) by the low number of individuals without TMD. Our sample also presented a low number of individuals with diagnoses of different groups (II or III) of the RDC/TMD. This certainly impacted the power to detect differences in proportions, although we remained powered for detecting differences in normally distributed data. Our study was cross-sectional, weakening causal inferences. Finally, the RDC/TMD also includes a number of non-painful conditions in the TMD diagnosis (e.g. disc displacements and osteoarthritis). We divided the TMD pain into two groups. One was myofascial pain, including myofascial and ‘arthrogenic’ pain, which may attenuate the TMD/CA effect if only myofascial TMD is associated with CA. However, we decided to do this because we found purely arthrogenic TMD to be very rare in our sample (only one case) and the mixed group was composed of 82.3% of the individuals with temporomandibular joint pain and only three individuals (17.6%) without articular pain. Strengths of this study are the blinded measurements, as well as the use of gold standard techniques to assess CA and DTM.
One interesting factor is that the frequency of headache was not different among the different groups. Because CA was reported to be a risk factor for increased migraine frequency (17), and because the TMD groups were more likely to have CA, increased mean monthly headache frequency in these groups should be expected. However, because the study was conducted in a headache centre, all patients were under preventive therapy, and many of them were receiving non-pharmacological interventions, which may have contributed to the lack of difference in headache frequency.
TMDs are associated with increased risk of ictal and interictal, trigeminal and extracephalic CA. Accordingly, TMD and CA are associated in individuals with migraine.
