Abstract
The aim was to apply diagnostic criteria, as published by the International Headache Society (IHS), to the diagnosis of orofacial pain. A total of 328 consecutive patients with orofacial pain were collected over a period of 2 years. The orofacial pain clinic routinely employs criteria published by the IHS, the American Academy of Orofacial Pain (AAOP) and the Research Diagnostic Criteria for Temporomandibular Disorders (RDCTMD). Employing IHS criteria, 184 patients were successfully diagnosed (56%), including 34 with persistent idiopathic facial pain. In the remaining 144 we applied AAOP/RDCTMD criteria and diagnosed 120 as masticatory myofascial pain (MMP) resulting in a diagnostic efficiency of 92.7% (304/328) when applying the three classifications (IHS, AAOP, RDCTMD). Employing further published criteria, 23 patients were diagnosed as neurovascular orofacial pain (NVOP, facial migraine) and one as a neuropathy secondary to connective tissue disease. All the patients were therefore allocated to predefined diagnoses. MMP is clearly defined by AAOP and the RDCTMD. However, NVOP is not defined by any of the above classification systems. The features of MMP and NVOP are presented and analysed with calculations for positive (PPV) and negative predictive values (NPV). In MMP the combination of facial pain aggravated by jaw movement, and the presence of three or more tender muscles resulted in a PPV = 0.82 and a NPV = 0.86. For NVOP the combination of facial pain, throbbing quality, autonomic and/or systemic features and attack duration of > 60 min gave a PPV = 0.71 and a NPV = 0.95. Expansion of the IHS system is needed so as to integrate more orofacial pain syndromes.
Introduction
Orofacial pain is prevalent in the general population, at around 17–26%, of which 7–11% is ‘chronic’ (1–6). However, the term orofacial pain includes a number of individual acute and chronic syndromes, and diagnosis of these requires the ability to identify any one of them from a given set of signs and symptoms. Very often these may present in a partial manner or with atypical features, making diagnosis difficult. Moreover, the diagnosis of orofacial pain is compounded by the close proximity of anatomical structures. These include the eyes, nose, teeth, sinuses, regional muscles, ears and the temporomandibular joints, and each of these structures may be the source of facial pain that may radiate and spread, implicating adjacent but uninvolved areas. This type of cross referral occurs very frequently between headache and orofacial pain entities. Trigeminal autonomic cephalgias commonly radiate to the face and teeth (7–9), and in turn orofacial structures may give rise to ‘headaches’ (10).
Although the head and face are intimately related, diagnostic classifications of headache and orofacial pain are not sufficiently integrated. The International Headache Society (IHS), which publishes the most widely used system for headaches (11), does not adequately cover currently accepted orofacial pain entities such as craniofacial myofascial pain, and various disorders of the temporomandibular joint (12). These are thoroughly described and classified by alternative systems: the American Academy of Orofacial Pain (AAOP) and the Research Diagnostic Criteria for Temporomandibular Disorders (RDCTMD) (13, 14).
Indeed, when employing current IHS criteria 21–29% of patients with facial pain could not be classified (15, 16). However, the IHS criteria were field tested in a neurological tertiary care centre; the patient profile is probably different from that of a dentally based orofacial pain clinic. Thus, the aim of the present study was to apply diagnostic criteria published by the IHS and the AAOP/RDCTMD in the diagnosis of patients observed in an orofacial pain clinic.
Materials and methods
The standard intake form employed in our facial pain clinic includes a pain history that records location, quality, severity, frequency, attack duration, age of onset, associated features, analgesic drug use, and aggravating or alleviating factors. Patients were specifically asked if the pain woke them from sleep. Additionally, demographic data, health status and medication history were recorded. All patients received a 28-day diary to record pain frequency, duration and severity four times daily employing a verbal scale of 0 (no pain) to 10 (maximal pain imaginable). The diary included a section for the notation of prescribed medications and analgesic use (escape drugs). These diaries were used as part of the diagnostic work-up and for follow-up. Clinical examination included a routine physical of the head and neck, the masticatory apparatus and the dental and periodontal tissues, and a basic examination of the cranial nerves. The masticatory apparatus (temporomandibular joint and masticatory/pericranial, sternocleidomastoid and trapezius muscles) was examined for sensitivity to palpation and the results individually recorded. The presence of trigger points with the typical referral pattern was not an essential feature for diagnosis. Muscle tenderness to the application of about 4 kg of digital pressure (examiner calibration was performed) was considered sufficient to classify as a painful muscle. Imaging was requested as needed. The intake was applied for a period of 2 years to all patients attending the Orofacial Pain Clinic at the Faculty of Dentistry, Hadassah-The Hebrew University, Jerusalem. Analysis and use of collected data were approved by the institutional review board.
The orofacial pain clinic routinely employs three diagnostic systems: the HIS and the AAOP, together with the RDCTMD (11, 13, 14). Based on these, one or both of the senior authors (R.B., Y.S.) recorded a clear diagnosis, where possible. Patients undiagnosable by any of these systems were analysed with criteria for neurovascular orofacial pain (NVOP), commonly termed facial migraine, as previously published (17–20).
Statistical methods
Data were tabulated and analysed with SPSS (version 14 for Windows; SPSS Inc., Chicago, IL, USA) with α for significance at 0.05. Interactions between nominal variables were analysed with Pearson's χ2. Differences between means in continuous variables were analysed with analysis of variance (
For the calculation of positive (PPV) and negative predictive values (NPV), disease prevalence was not included as these are unknown for most of the diagnosed entities. Instead, we employed the simplified equations: PPV = number of true positives/number of true positive + number of false positives, and NPV = number of true negatives/number of true negatives + number of false negatives (21, 22).
Results
A total of 328 patients (mean age 43.8 ± 17.7 years) were examined over the 24-month period, consisting of 107 men (32.6%) and 221 women (67.4%).
Diagnoses
The ability of the IHS system in the diagnosis of our cases is shown in Table 1. Employing the IHS classification, it was possible to diagnose definitively 184 (56.1%) of patients. The main groups consisted of anaesthesia dolorosa (n = 46), classical trigeminal neuralgia (n = 31), temporomandibular joint disorders (n = 35) and persistent idiopathic facial pain (PIFP) (n = 34). Less common diagnoses included migraine (n = 6), paroxysmal hemicrania (n = 6) and burning mouth syndrome (n = 6). Rare diagnoses included postherpetic neuralgia, cardiac referred pain, cervicogenic headache, (haemo)dialysis headache, classical glossopharyngeal neuralgia, and pain secondary to salivary gland disorder (all n = 1, total = 7). Other groups consisted of patients with rhinosinusitis (n = 3) and chronic dental pathology (n = 3). Mixed diagnoses included post-traumatic neuropathy with regional myofascial pain (n = 1), or with neurovascular pain (n = 1). In the absence of further classification options, most of the remaining 144 cases would have been undiagnosed, a 44% failure rate. Possibly, some of these cases may have been diagnosable as PIFP; however, it was clear that these 144 cases could be classified more precisely.
Diagnoses assigned to the collected patients

IHS, International Headache Society; AAOP, American Academy of Orofacial Pain; RDCTMD, Research Diagnostic Criteria for Temporomandibular Disorders.
In these 144 (44%) cases we applied AAOP/RDCTMD classifications (13, 14) and were further able to classify 120 (83.3%) as regional (masticatory) myofascial pain. Twenty-four patients remained, 23 of whom were diagnosed as NVOP based on additional criteria (17, 19, 23–25). Trigeminal neuropathy secondary to multiple connective tissue disease was diagnosed in one case (26, 27). This reduced the rate of undiagnosed patients to 0.
Analysis of selected pain entities
Two entities diagnosed in the present series of patients are not included in headache classifications: masticatory myofascial pain (MMP) and NVOP (facial migraine). These are discussed in more detail below.
Masticatory myofascial pain
There were 120 cases diagnosed as MMP with a mean age of onset of 37.6 ± 16.8 years. Of these, 31 were men (mean onset age ±
Reported pain intensity was moderate to severe (7.2 ± 2) and, although higher in women (7.3 ± 1.9) than in men (6.6 ± 2), was not statistically significant (
The ipsilateral masseter and medial pterygoid muscles were involved in all cases with MMP. The ipsilateral temporalis muscle was affected in 60%, and the contralateral masseter or medial pterygoid in 40% and 35%, respectively. In patients with five or more tender muscles the sternocleidomastoid and suboccipital group of muscles were commonly involved bilaterally (30%). Pain was located in the periauricular region or in the periauricular region and mandible in 48 cases (40%). A mandibular location was reported in 34 cases (28.3%) and pain in both jaws with involvement of the frontal region was reported in 11 cases (9.2%). A primarily oral or perioral location was reported in 11 cases (9.2%) and pain in one half of the face in eight cases (6.7%). Unilateral pain was reported in 73 cases (60.8%). Bilateral location was reported in 47 cases (39.2%) and was significantly more common in cases following trauma (50.9%) than in those classified as primary (28.6%, χ2 = 6.3, d.f. = 1, P = 0.012).
Sleep disturbance was found in 23.3% of MMP cases and was more common in post-traumatic (29.1%) than primary (18%) MMP (χ2 = 2, d.f. = 1, P = 0.16). Patients with disturbed sleep reported significantly more severe pain (7.9 ± 1.6) than patients without (6.9 ± 2;
The sensitivity, specificity, PPV and NPV values of various pain characteristics and location are shown in Fig. 1. Analysing the PPV and NPV of facial pain with muscle tenderness, we found that at three or more tender muscles the values were maximized to 0.67 and 0.95, respectively. Pain aggravated by function was associated with a moderate PPV (0.59) but excellent NPV (0.95). The combination of these two factors improved the PPV (0.82) value significantly and slightly reduced the NPV to 0.86.

Positive predictive values (PPV) and negative predictive values (NPV) for selected clinical features in masticatory myofascial pain (MMP). ∗MMP combination included the presence of facial pain with three or more tender muscles and pain aggravated by jaw movement.
Neurovascular orofacial pain (facial migraine)
A total of 23 patients were diagnosed as NVOP with a mean onset age of 39 ± 13.7 years. This group consisted of seven men (mean onset age ±
Pain location was most commonly reported in oral and perioral areas (n = 7, 30.4%), mandibular region (n = 7, 30.4%) or the maxillary region (n = 4, 17.4%). Pain in one half of the face was reported in three cases (13%) and a mandibular/periauricular location in two cases (8.7%). Sixteen patients (69.6%) reported unilateral pain and seven (30.4%) bilateral. Reported pain intensity was not different between unilateral (8.3 ± 1.5) and bilateral (8.3 ± 1.1) cases. Bilateral pain was marginally more frequent (42.9%) in cases that reported pain onset following trauma (n = 7) than in cases classified as primary NVOP (24%, χ2 = 0.7, d.f. = 1, P = 0.4). NVOP patients with disturbed sleep (48%) reported significantly more severe pain (9.1 ± 1.1) than patients without disturbed sleep (7.5 ± 1.1;
Local autonomic signs were observed in eight cases (three ipsilateral tearing, one ipsilateral tearing and conjunctival injection, two ipsilateral eyelid oedema, two rhinorrhoea) and ‘systemic’ signs in seven (four nausea, three photophobia). Patients with local autonomic signs did not report more severe pain (8.4 ± 1.4) than those without (8.4 ± 0.4;
The sensitivity, specificity, PPV and NPV values of various pain characteristics and location in NVOP assessed in this study are shown in Fig. 2. Individually, these parameters are not clinically significant. It was only when facial pain (not head pain), throbbing quality, autonomic and/or systemic features and attack duration of > 60 min were combined that good PPV (0.71) and NPV (0.95) values were attained.

Positive predictive values (PPV) and negative predictive values (NPV) for selected clinical features in neurovascular orofacial pain (NVOP). ∗NVOP combination included the presence of facial pain that was throbbing, accompanied by autonomic signs and/or systemic symptoms and of duration > 60 min.
Discussion
Cursory examination of the results suggests that the ability to diagnose patients when IHS, AAOP and RDCTMD classifications are integrated is 100%. PIFP, previously atypical facial pain, is of unknown origin and is largely employed when chronic facial pain is undiagnosable as one of the other entities. Classifications are useful for interphysician communication, research and in determining therapy. However, PIFP patients do not respond to specific therapy, questioning the clinical value of such a diagnosis. Atypical facial pain, the previous term for PIFP, has been successfully treated with triptans (28), amitriptyline (29, 30), venlafaxine (31) anticonvulsants, non-steroidal anti-inflammatory drugs (NSAIDs), benzodiazepines, psychiatric intervention and physiotherapy (32), none of which has been uniformly successful (33). Thus, when we remove the PIFP cases (n = 34), our ability to reach a clinically significant diagnosis is about 90%. This is marginally better than the best rate of 87% previously reported (15, 16), but less than that reported in a retrospective analysis of an orofacial pain clinic in the USA (34). If we remove the PIFP cases from the series previously reported (15, 16), the rate of ‘accurate’ diagnosis drops to 50%, significantly lower than that in the present study. This is probably due to differences in patient characteristics rather than classification practices as most clinics, whether medically or dentally oriented, apply temporomandibular disorder (TMD) and other classifications (see author reply; (12)). …
Masticatory myofascial pain
Temporomandibular disorder is an umbrella term that includes disorders of the temporomandibular joint and the masticatory muscles. The original Costen syndrome and its historical counterparts are no longer in use, as they all classified muscular and articular entities, together leading to misdiagnosis. The most common TMD is MMP, which is a well-established clinical entity with clear diagnostic criteria (13, 14) but has not been specifically defined in the IHS classification. Essentially, patients with MMP have facial pain, usually at the angle of the mandible and periauricular, aggravated by mandibular function and accompanied by sensitivity to palpation of the regional muscles.
The mean onset age reported in our patients (37.6 ± 17 years) is very similar to the age where symptoms may peak, as reported in the literature (35, 36). The female preponderance in our sample has been previously described in other MMP patient populations (37, 38).
Pain location described by our patients is in line with that described in previous patient series (39, 40). The distribution pattern of pain probably depends on the muscles involved and the intensity of the pain (39–42). Reported pain quality in most of our patients was ‘pressure like’—similar to the descriptions in the literature of pain that is dull, or heavy (43). Dizziness is a common accompanying feature of MMP and was reported by a third of our patients. Reported pain in our series was more severe (7.1 ± 2) than quoted in the literature (43, 44) and may reflect the fact that the clinic, as a secondary/tertiary referral centre, attracts the most difficult and severe cases. This hypothesis is supported by the fact that 23.3% reported disturbed sleep due to pain; this is usually rare in MMP patients (44). Moreover, our patients suffered from long pain attacks with a mean duration of 18 h, although some had daily pain of shorter duration (about 2 h). Mean duration in a group of MMP patients has previously been reported at about 5.5 h (44). Restricted mouth opening was seen in the majority of cases, but in a large proportion (30%) was not; this has been recognized by the RDCTMD, who subclassify MMP as with or without limited mouth opening.
The RDCTMD define the minimum number of muscle sites (three), whereas the AAOP does not include this in its criteria. Indeed, in day-to-day practice the presence of facial pain, aggravated by mandibular function and the presence of two ipsilateral tender muscle sites, would be hard not to diagnose as MMP, particularly if they are in separate masticatory muscles. Notwithstanding, examination of the data reveals that the combination of facial pain with tenderness of three or more muscles and a complaint of pain aggravated by jaw function produced a maximal PPV of 0.82. This reinforces the RDCTMD criteria's philosophy of including a minimum number of muscles or muscle sites. Whether the predictive value of the number of muscles is better than the number of sites needs further research. Interestingly, patients with migraine reported a high number of tender muscles (2.8 ± 2.4), although this was not significantly different from any of the other diagnoses. The presence of tender points or tender regional muscles is well documented in migraine patients (45, 46). In NVOP patients, we found a mean of 1.2 ± 1.5 tender muscles that was not significantly different from any diagnoses other than MMP.
Thus, although the presence of regional muscle tenderness accurately defines MMP patients vs. controls (47), when compared with other orofacial pain patients it is evident from the present study that further signs and symptoms with high sensitivity are needed. The aim has generally been to use signs and symptoms that attain a sensitivity > 0.7 and PPV values > 0.75 (14, 16). Additionally, it has been recommended that for MMP signs with a high specificity are needed (> 0.95); this was due to the invasive therapeutic procedures (surgery, irreversible dental rehabilitation) employed at the time the RDCTMD criteria were established (14). With current conservative therapeutic options for MMP (48), more balanced PPV and NPV values, such as those presented here, are desirable.
Neurovascular orofacial pain (facial migraine)
Patients with migraine may complain of pain referral to the midface, often causing misdiagnosis with sinusitis (23, 49, 50). Referral of pain in migraine to the lower face and oral structures has not been extensively documented, but is a well-known clinical finding (20). Migraine, or a neurovascular-type facial pain, presenting as isolated to maxillary and mandibular regions has been described (17, 19, 23, 25, 49–54) and is currently attracting considerable interest (49).
However, facial pain with ‘migrainous’, or neurovascular, features may also be a trigeminal autonomic cephalalgia (TAC) variant; these may occur exclusively in the facial region (54, 55). Alternatively, neurovascular-type facial pain may be due to referral from atypical primary neurovascular headaches such as migraine (25), cluster headache or other TACs (8, 9, 56, 57). Pain referral implies that typical headache occurs and pain radiates to orofacial structures. Since neurovascular-type facial pain cases complain of isolated pain in the lower two-thirds of the face, with no primary pain in the upper head, referral mechanisms are unlikely. However, it is possible that central mechanisms may be involved that refer pain from the first trigeminal branch to the second and third branches in the absence of pain in the distribution of the first branch (25). Convergence of trigeminal and cervical afferents on nociceptive second-order neurons in the brainstem trigeminocervical complex has been established and explains cervical spread of pain in migraine. Similarly, convergence of dural first branch with somatic second and third branch afferents in trigeminal nucleus caudalis may result in facial pain referral during a typical migraine attack.
It thus remains unclear whether neurovascular-type facial pain is a ‘local’ migraine or TAC variant (58). More data and functional imaging studies are needed, as performed in migraine and TACs (58). For these reasons, we have previously recommended the use of the term neurovascular orofacial pain (NVOP) rather than orofacial migraine, until more concrete pathophysiological data are available (58).
The features of NVOP do suggest that it is a migraine variant, and most authors support this hypothesis (17, 19, 23–25, 58). There are several similarities, but some important differences (see Table 2) between migraine and NVOP. The female : male ratio in NVOP patients reported here is 2.3:1, very similar to that occurring in migraine (2:1) and very different from the male preponderance observed in TACs (11). Migraine onset is usually at an early age, typically < 20 years, but may also occur between 25 and 35 years (59, 60). In contrast, the mean onset age in NVOP, based on patient's reports, is 39.4 ± 13.6 years. Similar onset age has been previously reported in other NVOP patients (17, 19, 25), but the reasons for this are unclear. A history of typical migraine headaches in some NVOP cases suggests that pain may have ‘relocated’ to orofacial structures. Onset of NVOP has also been associated with trauma, such as recent dental extractions or root canal therapy, but the evidence is circumstantial. Penarrocha et al. reported on six of 11 cases (54.5%) with lower half migraine which were historically associated with trauma (19). In the present series, seven cases (30.4%) reported pain onset following trauma. Post-traumatic neurovascular headaches have been extensively reported (11, 61–67), but the underlying mechanisms are unclear.
Differences between neurovascular orofacial pain (NVOP) and migraine

Combining facial pain with throbbing quality, the presence of local autonomic signs and/or systemic signs (nausea and/or photophobia) with attack duration of > 60 min resulted in a clinically relevant PPV of 0.71. The combination of symptoms such as nausea, photophobia, phonophobia and exacerbation by physical activity is routinely used in the diagnosis of migraine (68). We did not assess the effect of physical activity on NVOP cases, but clinical experience suggests that this complaint is rarer in NVOP than in migraine. The ID Migraine questionnaire, a three-item screener consisting of nausea, photophobia, and headache-related disability, has been field tested and has a PPV of 0.72–0.94 (69–71).
Pain intensity in NVOP in this and previous studies (25) is similar to that reported by migraineurs (72), but intensity varies even within the same patient and is an unreliable parameter in diagnosis. Attack duration in migraine is defined as 4–72 h (11); in about half to two-thirds of migraine patients duration is 5–24 h (72, 73). Mean attack duration in NVOP was about 11 h, less than the mean of 29–54 h reported for migraine (72, 74, 75). However, in eight cases the typical attack was shorter, with a mean duration of 1.5 ± 1 h, reminiscent of a cluster headache attack (11). Similarly, migraines can last for < 5 h, but this is admittedly very rare (16%) (73, 76). Migraine is typically located in the upper head, usually in the ocular, temporal and frontal regions (77), whereas all our NVOP cases were located below the orbito meatal line. In most cases migraine is unilateral (76.3%), and we found a slightly lower rate of unilateral NVOP cases (69.6%). Typically, pain in migraine is throbbing or pulsating (47–82%), but may be occasionally pressing (73, 78, 79); similarly, NVOP was throbbing in 65% of cases and 56% reported pressure, mostly as an additional pain feature.
Migrainous features are usually reported less frequently and are milder in NVOP (17, 19, 23–25). In this respect, NVOP may more closely resemble TACs (80). About 50% of migraineurs vomit during an attack and 80% suffer nausea. Photophobia or phonophobia is reported by > 80% of migraineurs (78, 81). However, the presence of accompanying signs is highly irregular; nausea, photo- or phonophobia accompany more than half of headache episodes in only one-third or less of migraineurs (73). Ipsilateral local autonomic signs (usually tearing) are found in up to 50% of migraineurs (82, 83). We found that 35% of NVOP patients had autonomic signs and 30% systemic signs (nausea) during attacks. Although autonomic signs have been linked to headache intensity (82, 83), we found no difference in reported pain intensity between NVOP patients with or without autonomic signs. The majority of migraine patients (71%) report a history of being woken from sleep at least once by a migraine (84), and almost half (47.8%) of NVOP patients reported disturbed sleep that was significantly associated with reported pain intensity.
An interesting finding is the occurrence of cold allodynia in the ipsilateral teeth of NVOP patients with no explanatory pathology. Although we did not quantify this, it is a relatively common finding that causes extensive misdiagnosis with dental pathology (17). In some ways this is a similar phenomenon to the tactile allodynia described in migraine patients (85).
The present study did not examine treatment outcome, and the following is a brief summary of our experience. In chronic cases we have very good results with prophylactic propranolol, divalproex, or, when muscle tenderness is prominent, amitriptyline. Some episodic cases respond quite well to NSAIDs, particularly naproxen sodium. Similarly, we have some cases that respond excellently to triptans, but our impression is that the response rate may be lower than in typical migraine.
The features discussed above have been extrapolated to a tentative set of criteria, as shown in Table 3(20). Whether NVOP is a separate diagnostic entity within the neurovascular headaches remains unclear. Classifications develop as data gather; in the past, cluster was thought a migraine variant, paroxysmal hemicrania a cluster variant and SUNCT an atypical form of trigeminal neuralgia (58).
Criteria for neurovascular orofacial pain
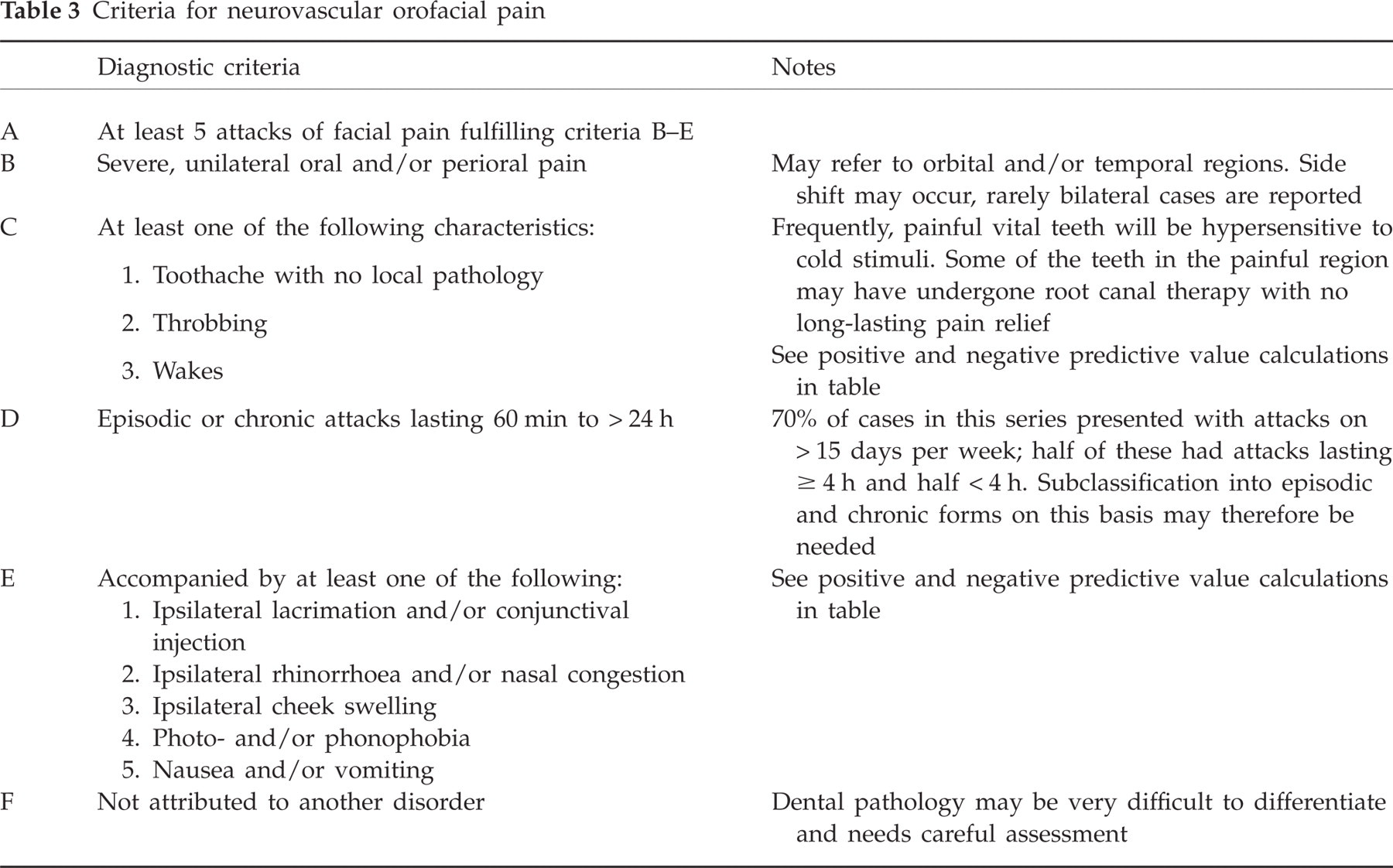
Conclusion
The findings presented call for the expansion of the IHS classification to include more orofacial syndromes. The data presented underscore the need for the integration of orofacial pain and headache classifications.
