Abstract
High-frequency repetitive transcranial magnetic stimulation (rTMS) increases and low-frequency rTMS decreases neural excitability. Clinically, rTMS shows beneficial effects in the treatment of neurological and psychiatric disorders. Furthermore, chronic and neuropathic pain has been shown to respond to rTMS treatment. A small pilot study revealed prophylactic effects of high-frequency rTMS in migraine. As there is evidence of neuronal hyperexcitability in migraine, we conducted a placebo-controlled, blinded study to evaluate the therapeutic effects of low-frequency rTMS in migraine. The primary end-point was defined as a reduction of migraine attacks compared with placebo, secondary outcomes were a reduction in the total number of days with headache, hours with headache, pain intensity and a decrease of analgesic intake for migraine. Twenty-seven migraineurs completed the study and were treated with rTMS on five consecutive days. For the verum group, two trains of 500 pulses with a frequency of 1 Hz were applied over vertex with a round coil. For the treatment of the placebo group, a figure-of-eight sham coil was used. A significant decrease of migraine attacks could be observed in the verum group. However, when comparing these effects with placebo, no significance was evident. The same was true concerning secondary outcome measures with regard to days with migraine and total hours with migraine. No effects were evident for pain intensity and use of analgesics. The rTMS treatment was tolerated well. rTMS stimulation over vertex with 1 Hz was not effective in migraine prophylaxis when compared with placebo. The positive effects regarding migraine attacks, days and total hours with migraine in the verum group are encouraging and indicate that further research on this topic is warranted.
Introduction
Repetitive transcranial magnetic stimulation (rTMS) gives the opportunity to modify the activity of cortical neurons locally and non-invasively. In human studies, high-frequency rTMS > 5 Hz leads to an increase and lower rTMS frequency in the range of 1 Hz to a decrease of neural excitability (1,2).
Several studies have underlined the therapeutic benefit of rTMS in psychiatric disorders such as major depression (3). In epilepsy—associated with increased cortical hyperexcitability—Tergau et al. (4,5) and Fregni et al. (6) could demonstrate an anticonvulsive effect of low-frequency rTMS.
Regarding pain diseases, treatment with high-frequency rTMS over motor cortex [e.g. (7); for review see (8)] or low-frequency stimulation with 1 Hz over the right dorsolateral prefrontal cortex (DLPFC) ameliorated experimental (9) or neuropathic pain, respectively [for review, see (10)]. However, the pathophysiological mechanisms underlying these clinical effects are poorly understood. The question whether there are further stimulation sites with antinociceptive properties is still a subject of discussion.
Migraine is a neurovascular headache disorder characterized by severe headache attacks with vegetative symptoms. Clinically, it can be divided into migraine with or without aura. Prophylactic treatment is necessary for patients impaired by frequent or heavy headache attacks. β-Blockers, anticonvulsants or calcium antagonists have been shown to be effective in migraine prophylaxis. However, the use of these medications is accompanied by side-effects and supports the request for non-medicinal alternatives.
There is controversy as to whether cortical hyper- or hypoexcitability is evident in migraine. Electrophysiological studies using TMS indicate contradictory results [for review, see (11–13)]. However, several lines of evidence regarding biochemical data (14–16), familial hemiplegic migraine (FHM) (17) as well as cortical spreading depression (CSD), which is responsible for the aura symptoms in migraine (18,19), support the hypothesis of cortical hyperexcitability in migraine.
Clarke et al. (20) have demonstrated a reduction of pain intensity by TMS administered immediately after the onset of migraine headaches. So far, only one study has examined the effects of rTMS in the prophylactic treatment of migraine (21). Patients with chronic migraine received high-frequency rTMS treatment of the left DLPFC and migraine attacks were reduced significantly in comparison with sham stimulation. However, only 11 patients were included in this pilot study.
Given that migraine is associated with neuronal hyperexcitability as discussed above, low-frequency rTMS could alleviate cortical hyperexcitability as described for epilepsy and thus have therapeutic effects in migraine. Therefore, we conducted this study to evaluate prophylactic effects of rTMS in migraine applying low-frequency rTMS stimulation over vertex with 1 Hz. This approach is different from the design of Brighina et al. (21), who assumed that high-frequency rTMS over left DLPFC might ameliorate chronic migraine by enhancing cortical activity. The setting in our study was chosen according to Tergau et al. (4), where epileptic seizures were reduced successfully. Our primary end-point was defined as a reduction of migraine attacks. Secondary outcome measures included a reduction in the total number of days with headache, hours with headache, pain intensity and a decrease of analgesic intake.
Materials and methods
General procedure
Participants were recruited from the out-patient clinic of the Neurological Department, University of Marburg, as well as by posters and advertisements in local newspapers. For inclusion, subjects had to suffer from migraine according to the guidelines of the International Headache Society (IHS) (22) with at least four migraine attacks per month. Exclusion criteria were: any prophylactic treatment of migraine, cardiac or cerebral pacemaker, metal in the cranium, epilepsy, pregnancy, severe psychiatric or neurological diseases such as Parkinson's disease, depression or schizophrenia as well as complex migraine forms (e.g. FHM). Patients were examined before inclusion by a neurologist specializing in headache.
The experimental protocol was approved by the ethics committee of the medical faculty of the University of Marburg; all study participants provided written informed consent.
Outcome measures
Subjects had to keep a headache diary during an observation period of 8 weeks before stimulation, during the treatment phase of 5 days and during the consecutive follow-up of 8 weeks after study treatment. Participants had to document all possible side-effects as well as headache attacks with the attendant symptoms, onset and ending of pain and intake of analgesics. Furthermore, they had to judge pain intensity on a rating scale from 0 (no pain) to 10 (maximal pain).
Subjects
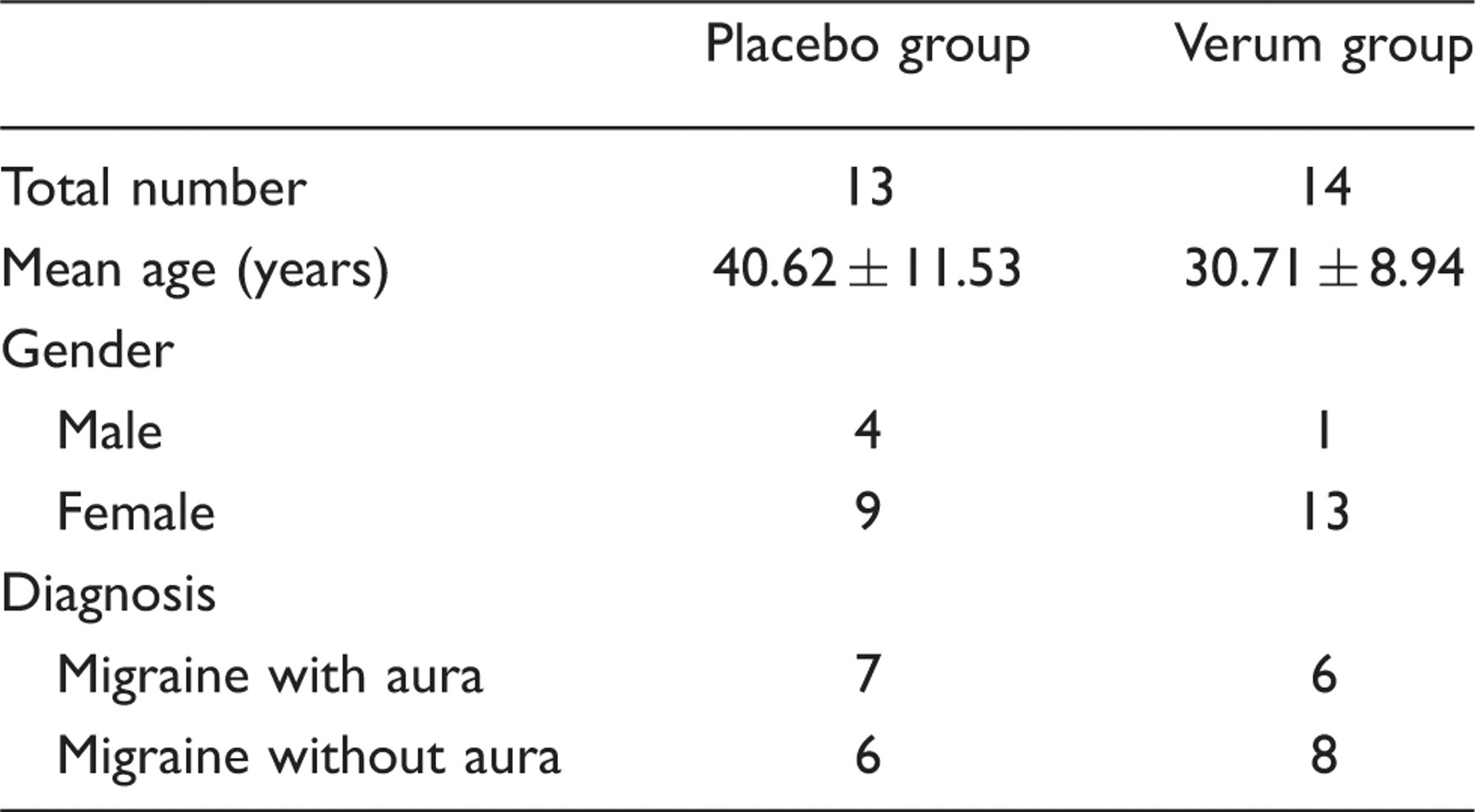
Clinical characteristics of participants
rTMS stimulation
The stimulation took place in a sound- and light-attenuated room of the University of Marburg. The stimulation procedure has been previously described by Tergau et al. (4). Briefly, rTMS treatment was applied on five consecutive days. Subjects were randomized to either the placebo or the verum (rTMS) group.
First, as a gross measure of cortical excitability we assessed the individual motor thresholds of the first dorsal interosseus muscle of the right hand for each subject by visual inspection. We determined the stimulator intensity that produced a visually detectable minimal muscle contraction in the target muscle in at least five out of 10 trials (visMT).
In both treatment groups, a repetitive magnetic stimulator (MagPro compact, Dantec, Denmark) was used to apply two trains of 500 monophasic pulses separated by a 1-min interval between the trains at a frequency of 1 Hz over vertex (Cz, 10-20 EEG System). For verum stimulation, a round coil (diameter 13 cm) was taken in each session. The stimulation intensity was set to ‘visual motor threshold of the dominant hand—2%’, an intensity that corresponds approximately to the resting motor threshold. The treatment procedure of the placebo group was the same as for the verum group, but a sham coil (‘figure-of-eight’, diameter 11 cm; Medtronic, Minneapolis, MN, USA) was used, which produces the same sound and similar sensory feedback on the subject's head without delivering active stimulation.
Statistical analysis
Data are expressed as indicated ± standard deviation (
Sample size calculation was done to detect a reduction in the frequency of migraine attacks by at least 50% in the rTMS group assuming a reduction of 30% in the placebo group and a
For all outcome measures, treatment effects were calculated for each subject by the differences between the baseline values before and after rTMS stimulation. The small number of observations does not allow using t-tests or
Medians for the reductions in placebo and repetitive transcranial magnetic stimulation (rTMS) group
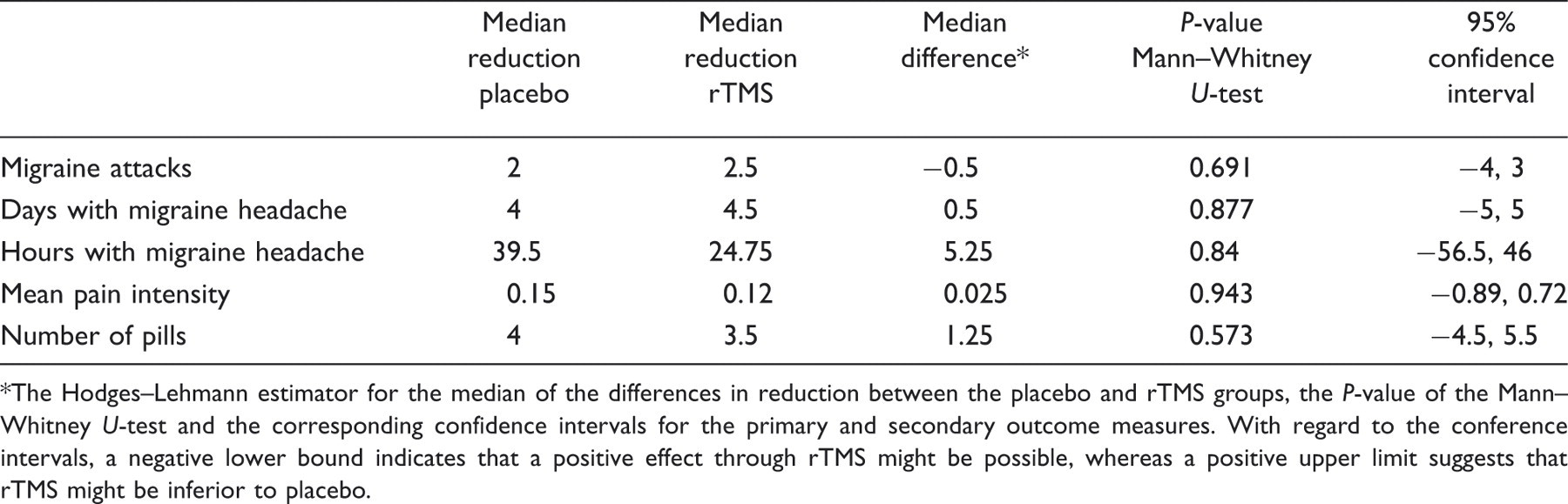
The Hodges–Lehmann estimator for the median of the differences in reduction between the placebo and rTMS groups, the P-value of the Mann–Whitney U-test and the corresponding confidence intervals for the primary and secondary outcome measures. With regard to the conference intervals, a negative lower bound indicates that a positive effect through rTMS might be possible, whereas a positive upper limit suggests that rTMS might be inferior to placebo.
Results
Primary end-points
Migraine attacks (Fig. 1)
In the verum group, the number of migraine attacks during 8 weeks was significantly reduced from 9.36 ± 2.82 days to 6.79 ± 4.28 days (Wilcoxon Signed Rank Test, P = 0.007). In the placebo group, the number of migraine attacks decreased also, but not in a significant manner (Wilcoxon Signed Rank Test, P = 0.216). Comparing the effects between the placebo and verum groups, no significances could be observed (Mann–Whitney U-test, P = 0.698).
Number of migraine attacks during 8 weeks before and 8 weeks after repetitive transcranial magnetic stimulation for placebo and verum group. *Statistical significance (P < 0.05). The effects of the verum group are not significant when compared with placebo.
Secondary end-points
Migraine days (Fig. 2)
There was a significant reduction of migraine days during 8 weeks in both placebo (from 17.69 ± 11.63 days to 13.15 ± 9.27 days; Wilcoxon Signed Rank Test, P = 0.012) and verum groups (from 14.36 ± 5.07 days to 9.50 ± 6.80 days; Wilcoxon Signed Rank Test, P = 0.006). Comparing the treatment effects of the placebo and verum groups, no significant difference was seen (Mann–Whitney U-test, P = 0.884).
Number of days with migraine during 8 weeks before and 8 weeks after repetitive transcranial magnetic stimulation for placebo and verum group. *Statistical significance (P < 0.05). The effects of the verum group are not significant when compared with placebo.
Migraine hours (Fig. 3)
Whereas the verum group showed a significant reduction of migraine hours during 8 weeks by rTMS application from 125.93 ± 80.31 h to 85.36 ± 72.27 h (Wilcoxon Signed Rank Test, P = 0.035), no significance was educible in the placebo group (Wilcoxon Signed Rank Test, P = 0.080) or regarding the effects between the placebo and verum groups (Mann–Whitney U-test, P = 0.846).
Number of hours with migraine during 8 weeks before and 8 weeks after repetitive transcranial magnetic stimulation for placebo and verum group. *Statistical significance (P < 0.05). The effects of the verum group are not significant when compared with placebo.
Migraine—mean pain intensity (Fig. 4)
In the placebo group, a mean pain intensity during 8 weeks of 5.52 ± 1.72 was calculated before and 5.17 ± 2.51 after rTMS stimulation. In the verum group, the values changed minimally from 6.26 ± 1.33 to 6.11 ± 1.26. This difference (placebo: Wilcoxon Signed Rank Test, P = 0.839; verum: Wilcoxon Signed Rank Test, P = 0.455) as well as the effects between the placebo and verum groups were not significant (Mann–Whitney U-test, P = 0.942).
Mean pain intensity judged on a rating scale from 0 (no pain) to 10 (maximal pain) during 8 weeks before and 8 weeks after repetitive transcranial magnetic stimulation (rTMS) for placebo and verum group. There were no significant effects within and between the placebo and verum group, respectively, regarding the rTMS treatment.
Migraine—use of analgesics (Fig. 5)
Following rTMS treatment, the intake of analgesics during 8 weeks changed from 15.15 ± 11.24 pills before rTMS application to 11.81 ± 9.89 pills after rTMS in the placebo group. In the verum group, 12.50 ± 14.65 pills were consumed during 8 weeks after treatment, whereas an average of 14.21 ± 10.13 pills were taken before rTMS stimulation. For all these conditions, no significance was detectable (Wilcoxon Signed Rank Test, placebo: P = 0.094; verum: P = 0.232). Furthermore, there were no significant effects between the placebo and verum groups (Mann–Whitney U-test, P = 0.577).
Number of analgesic pills during 8 weeks before and 8 weeks after repetitive transcranial magnetic stimulation (rTMS) for placebo and verum group. There were no significant effects within and between the placebo and verum group, respectively, regarding the rTMS treatment.
Side-effects
Character and number of side-effects of repetitive transcranial magnetic stimulation (rTMS) treatment

Discussion
There is some evidence that cortical hyperexcitability might be evident in migraine: (i) biochemical data regarding magnesium deficiency (14), disturbed mitochondrial energy metabolism (15) and elevated concentrations of neuroexcitatory amino acids suggest hyperexcitability (16); (ii) in patients suffering from FHM—a special form of migraine with aura—mutations of voltage-gated P/Q type calcium channels (CACNA1A) or natrium channels (SCNA1A) and mutations in ATP1A2, encoding the α2 subunit of the Na+, K+ adenosinetriphosphatase (ATPase), have been described (for review see (17)). FHM is considered as a channelopathy leading to abnormal interictal neuronal excitability (17,23); and (iii) furthermore, the clinical phenomenon of the migraine aura rests upon CSD, a wave of depressed neuronal activity spreading along the cortex at a rate of 2–6 mm/min (18). CSD is related to an elevated release of the excitatory transmitter glutamate in the brain (19).
Assuming that low-frequency rTMS over vertex might be effective in migraine prophylaxis by ameliorating cortical hyperexcitability in migraine, we conducted this placebo-controlled, blinded study. We could observe a moderate and significant reduction by rTMS treatment in the verum group with regard to migraine attacks, days with migraine and total hours with migraine, whereas no effects were evident for pain intensity and the use of analgesics. However, when comparing these results with the effects of the placebo group, no significance was shown. Thus, no superiority of verum rTMS over sham rTMS could be demonstrated regarding any of the primary and secondary end-points (see Fig. 6). In general, rTMS treatment was well tolerated, with mild side-effects in the range of those in the placebo group.
Box plots for the reduction of the number of migraine attacks, migraine days, migraine hours, the mean pain intensity and the number of analgesics between 8 weeks before and 8 weeks after study treatment for the placebo (grey) and verum (white) group, respectively.
Concerning the positive results of the verum group, it is reasonable that changing treatment paradigms would be able to optimise further our effects as previously described for central or neuropathic pain [for review see (8,24)]. For these pain conditions, best results in pain reduction were commonly obtained using a figure-of-eight coil with focal stimulations over motor cortex (7,25–27). Furthermore, high-frequency rTMS was more effective than low-frequency stimulation (7,26). In line with these data, Brighina et al. (21) found significant reduction of headache attacks, the mean number of abortive pills used and the headache index in patients suffering from chronic migraine. They applied high-frequency rTMS with 20 Hz over left DLPFC during 12 sessions. This small-sample study by Brighina et al. (21) is the first to examine rTMS for prophylactic treatment in migraine, therefore no references exist with regard to effective stimulation sites. By applying rTMS with a round coil over vertex one can stimulate widespread, frontal and parietal cortical areas bilaterally. This stimulation paradigm has also been used for the initial rTMS treatment of depression (for review see (2) and for multifocal epilepsy (5,6)). Thus, we might have reached cortical areas known to be pain releasing (e.g. motor cortex or DLPFC as discussed above) in a non-selective manner by this approach.
However, with regard to the overall non-significant results of our approach, one may speculate that low-frequency rTMS over vertex might not be able to ameliorate cortical hyperexcitability in migraine as described for epilepsy (4–6). In line with this, a recent placebo-controlled study investigating rTMS in drug-resistant epilepsy could not demonstrate a statistically significant benefit (28), too, indicating that other stimulation parameters would be required to lessen cortical hyperexcitability. Another reason for our negative findings might be that hypoexcitability—and not hyperexcitability—is the underlying pathophysiological factor in migraine. The literature regarding this topic is controversial as there is evidence for both hypo- and hyperexcitability (12,13). Furthermore, Bohotin et al. (29) investigated the excitability of the visual cortex using 1 Hz or 10 Hz rTMS in healthy volunteers and migraineurs, respectively, and monitored changes by pattern-reversal, visual evoked potentials. In subjects suffering from migraine, 10 Hz stimulation led to an increase in cortical excitability (i.e. an increase of the first block amplitude) and habituation, whereas 1 Hz stimulation had no effect. However, 1 Hz rTMS decreased the first block amplitude and habituation in healthy volunteers. According to the ‘ceiling model’ (30), habituation depends on the pre-activation status of cortical excitability, and in migraine this cortical pre-activation level is low, which might explain why 1 Hz rTMS stimulation did not lead to a decrease in cortical excitability in their study. Therefore, not cortical hyperexcitability, but reduced cortical pre-activation might be present in migraine. If so, high-frequency rTMS (e.g. 10 Hz rTMS) would be necessary for treatment.
Our study might be limited by the following circumstances: (i) subjects included did not take any prophylactic medicinal treatment, although suffering from at least four migraine attacks per month. Most of them were open-minded to alternative or ‘non-chemical’ therapy options, suggesting a positive anticipation for rTMS. Therefore it can not be ruled out that all observed improvements by rTMS treatment were due to a placebo effect; (ii) the rTMS coil used for the verum stimulations (rounded coil) differs from the sham coil (‘figure-of-eight’). Therefore, it was not possible to blind the investigator and conduct a double-blinded study.
Taking all data together, low-frequency rTMS application over vertex was tolerated well but not effective for migraine prophylaxis when compared with sham stimulation in this placebo-controlled and blinded study. There were moderate and significant effects in the verum group concerning the number of migraine attacks, migraine days and total hours with migraine. Therefore, our data are encouraging for further research.
