Abstract
Introduction
The efficacy of several antiepileptics in the preventive treatment of episodic migraine in adults has been systematically reviewed. Because many trial reports have been published since then, an updated systematic review was warranted.
Methods
We searched the Cochrane Central Register of Controlled Trials, PubMed/MEDLINE (1966 to January 15, 2013), MEDLINE In-Process (current week, January 15, 2013), and EMBASE (1974 to January 15, 2013) and hand-searched
Results
Mean headache frequency on topiramate and sodium valproate is significantly lower than placebo. Likewise, topiramate and divalproex demonstrated favorable results for the proportion of subjects with ≥50% reduction of migraine attacks. For topiramate, 100 mg and 200 mg outperformed 50 mg, but this was paralleled by a higher adverse event rate. For valproate/divalproex, a dose-effect correlation could not be established. There was no unequivocal evidence of efficacy for any of the other antiepileptics.
Conclusion
Topiramate, sodium valproate and divalproex are effective prophylactic treatments for episodic migraine in adults. In contrast to previous reports, there is insufficient evidence to further support the use of gabapentin.
Introduction
The prevention of disease-related disability is the ultimate goal of successful migraine treatment. Currently available prophylactic remedies for migraine may at best lead to only partial reduction of frequency of headache attacks, often at the expense of significant adverse experiences, and improving patient functioning and quality of life to a limited extent only. Moreover, they are used by only an estimated 3%–12% (1–3) of patients, hence failing to meet the needs of migraine sufferers worldwide (4).
Evidence-based guidelines on the drug treatment of migraine have been developed and published by the European Federation of Neurological Societies (EFNS) (5). These guidelines suggest that prophylactic therapy should be considered for patients with migraine when quality of life, business duties, or school attendance are restricted; when attack frequency exceeds one per month; in case of a lack of response to acute drug treatment; and when frequent, long, or uncomfortable auras occur. Moreover, patient preferences and treatment costs should be considered when making treatment decisions.
Based on a shared primary indication of use or rationale for development, drug treatments for epilepsy are collectively known as “antiepileptics.” The use of antiepileptics for the prophylactic treatment of migraine is theoretically warranted by several known modes of action, which relate either to the general modulation of pain systems (6) or more specifically to systems involved in the pathophysiology of migraine (7). However, it is not currently possible to state with any certainty which particular mode or modes of action of antiepileptic drugs are relevant to the prophylaxis of migraine. Thus, with respect to a systematic review of migraine-preventive treatments, the argument for considering them as a class fails. It was therefore decided that the update of an earlier Cochrane review (8) should be split into four.
This review considers the evidence for the efficacy and tolerability of antiepileptics for preventing migraine in adults and is a companion paper to four recently published systematic reviews in the
Methods (additional details are in the companion papers in the Cochrane Database of Systematic Reviews (9–12))
Characteristics of eligible studies
Studies were required to be prospective, (pseudo-)randomized controlled trials of self-administered preventive drugs in adults 16 years or older. Trials were not required to be blinded but results needed to be published in full; short conference and journal abstracts were not included. Diagnosis needed to be based on at least some of the distinctive features of migraine, preferably a specific set of diagnostic criteria (e.g. Ad Hoc Committee (15), International Classification of Headache Disorders, first edition (ICHD-I) (16) or second edition (ICHD-II) (17)).
Studies evaluating treatments for chronic migraine, medication-overuse headache or any other chronic daily headache were unsuitable for this review. Studies were required to have at least one arm with an antiepileptic drug. Acceptable comparator groups included placebo, no intervention, active drug or non-pharmacological treatments (with proven efficacy), and same drug treatments with a clinically relevant different dose. Only drugs used for the treatment of epilepsy or status epilepticus, commercially available and suitable for outpatient use in either Europe or the United States (US) were considered.
Types of outcome measures
Data on headache frequency (both in continuous and dichotomous format), quality of life and adverse events (AEs), obtained directly from patients, were collected.
For outcomes measured on a continuous scale (headache frequency), we preferred number of migraine attacks to number of days with migraine. We anticipated reporting in a variety of ways, e.g. as mean pre-treatment, post-treatment, and/or change scores. When variance associated with these means was not reported or provided by the authors upon request, we attempted to estimate variances based on primary data, test statistics, and/or error bars in graphs.
When efficacy outcomes were reported in dichotomous form (success/failure), we chose a ≥50% reduction in headache frequency as the clinically relevant outcome. Reviewers recorded, for each treatment arm, the number of patients included in the analysis and the number with each outcome.
As we anticipated that frequency outcomes would be measured over various units of time (e.g. number of attacks per two weeks versus per four weeks), and for numerous different time points (e.g. four-week headache frequency at two months versus at four months), we attempted to standardize the unit of time over which headache frequency was measured at 28 days (four weeks) wherever possible.
For quality-of-life data, we envisaged that the Migraine-Specific Quality of Life Questionnaire (MSQ) and the Medical Outcomes Study 36-item Short-Form Health Survey (SF-36) would most likely be the preferred outcome scales. However, other types of outcomes related to quality of life (e.g. work absenteeism) were not excluded a priori.
In addition, we tabulated AEs for each included study. Where data on AEs were missing or inadequate, we intended to obtain these data by correspondence with authors, but in several cases this was not possible because of the age of the studies.
Identification of studies
Databases searched were (a) The Cochrane Pain, Palliative & Supportive Care Trials Register; (b) The Cochrane Central Register of Controlled Trials (CENTRAL) (Issue 12, 2012); (c) PubMed 1966–December 2005; (d) MEDLINE (via OVID) 2005–January 15, 2013; (e) MEDLINE In-Process (via OVID) January 15, 2013; (f) EMBASE 1974–December 2005; and (g) EMBASE 2005–January 15, 2013. In addition, the reference lists of review articles and included studies, books related to headache, and two journals (
Titles and abstracts of studies identified by the literature search were screened for eligibility by two independent reviewers (WM, ML). Papers passing this initial screening process were retrieved, and the full text was reviewed independently by the reviewers. Disagreements at the full-text stage were resolved through discussion.
Data extraction and analysis
Information on patients, methods, interventions, outcomes and AEs were abstracted by two independent reviewers.
For outcomes reported on a continuous scale (change in migraine frequency), pre- and post-treatment means and variances were analyzed and reported as mean difference (MD) with 95% confidence interval (CI).
Dichotomous data meeting our definition of a clinically significant threshold were used to calculate odds ratios (ORs) with CIs. AEs and withdrawal rates due AE were assessed, and data on the proportion of patients reporting specific AEs were used to calculate numbers-needed-to-harm (NNH).
Estimates of efficacy (both MDs and ORs) were tested for homogeneity. If study estimates were homogeneous, they were combined using a fixed-effect model. When significant heterogeneity was present, an attempt was made to examine the differences based on the clinical characteristics of the included studies. Only when studies with statistically heterogeneous results appeared to be clinically similar were the study estimates combined using a random-effects model.
Results
The PubMed search strategy for the original review (8) yielded 1089 potentially eligible citations, while the EMBASE and CENTRAL searches yielded 290 and 6952 citations, respectively. No additional citations were retrieved from the Cochrane Pain, Palliative & Supportive Care Trials Register or from other sources. After title-and-abstract screening, we obtained 58 published papers on antiepileptics for full-text scrutiny (Figure 1).
PRISMA flow diagram: paper selection, stages and numbers.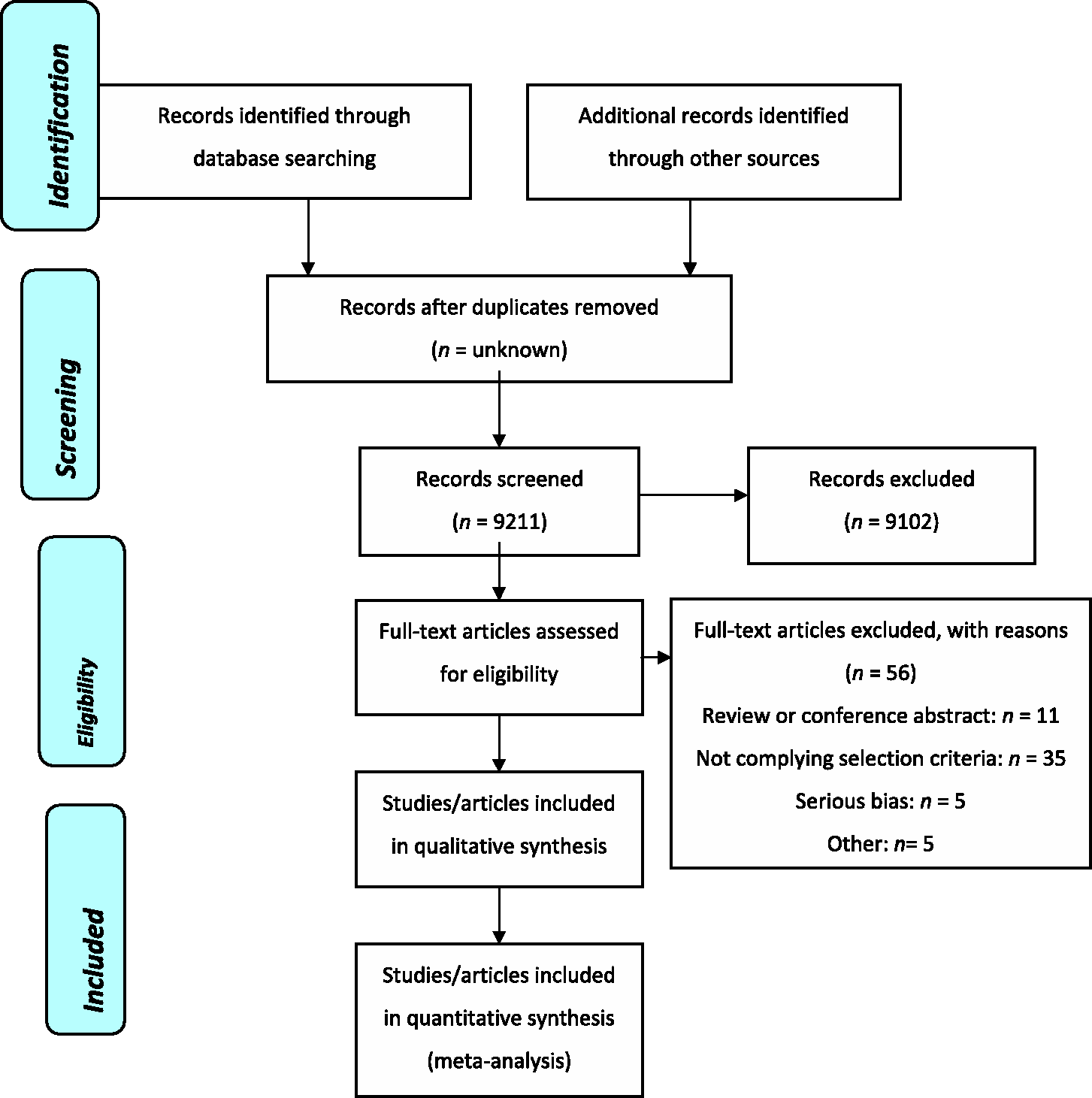
The MEDLINE search strategy for the present update (from 2005 on) yielded 188 citations as possible candidates for the current series of reviews on antiepileptic drugs for migraine prophylaxis; the search of MEDLINE In-Process identified an additional 20 citations. The EMBASE and CENTRAL updates identified 484 and 85 citations, respectively. Three additional study reports (all unpublished and all pertaining to gabapentin) were identified from other sources. After title-and-abstract screening, we obtained 37 published and three unpublished papers on antiepileptics for full-text scrutiny.
Gabapentin and pregabalin (Table 1)
Gabapentin main outcomes.
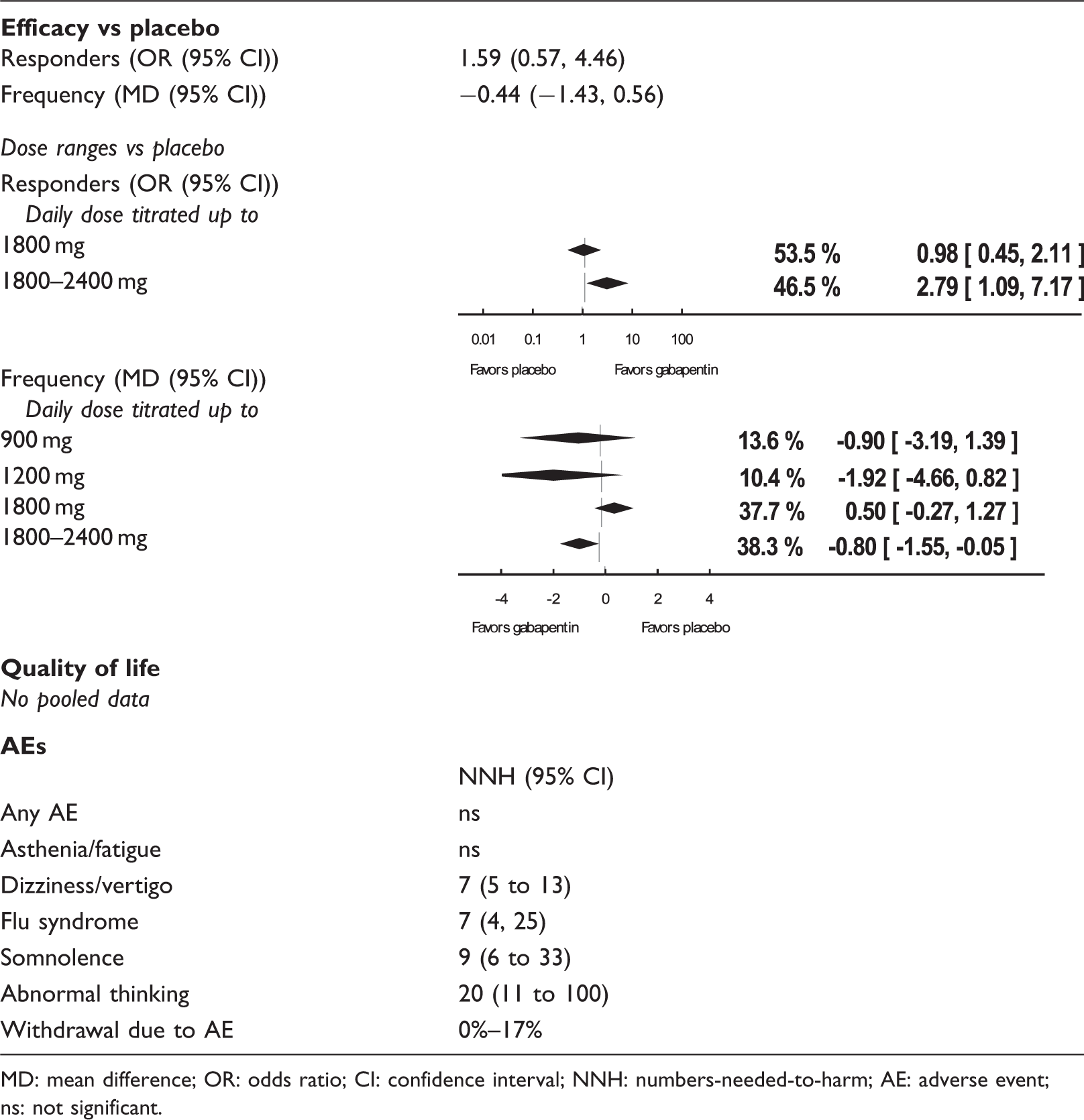
MD: mean difference; OR: odds ratio; CI: confidence interval; NNH: numbers-needed-to-harm; AE: adverse event; ns: not significant.
The risk of bias in the studies was generally judged as low or unclear. However, three studies were at high risk of selection bias (20), performance bias (20,22), attrition bias (22) or reporting bias (20,21).
The pooled results did not demonstrate a significant difference between gabapentin and either placebo or different gabapentin doses on the outcomes “headache frequency” or “responder rate” (Table 1). The sole trial of gabapentin enacarbil (21) failed to demonstrate a significant difference in responder rate across all dose ranges.
Because the only placebo-controlled paper that reported data on quality of life reported no significant difference with regard to reduction of headache frequency, no further analyses were undertaken of these data (23).
Overall, 68% of subjects on active treatment reported one or more AEs, as opposed to 57% on placebo. Withdrawal due to AEs ranged between 0% and 17%. NNHs for any as well as the five most prevalent AEs for the pooled analyses of gabapentin 900 to 2400 mg versus placebo are presented in Table 1.
Topiramate (Table 2)
Topiramate main outcomes.

MSQ: Migraine-Specific Quality of Life Questionnaire; MD: mean difference; OR: odds ratio; CI: confidence interval; SF-36: Medical Outcomes Study 36-item Short-Form Health Survey; NNH: numbers-needed-to-harm; AE: adverse event; ns: not significant.
We judged nine studies to be at high risk of bias for at least one of six attributes (randomization, allocation, performance, detection, attrition and reporting) (29–32,35,38–41).
The combined analysis of nine trials (25–31,33,34) showed a significant reduction in headache frequency in the active group compared to placebo of about one attack per 28 days. Likewise, patients were twice as likely to experience a ≥50% reduction in frequency with topiramate as with placebo. The 100 and 200 mg target doses were significantly superior to 50 mg in both outcomes, but did not differ from each other (25,33).
The combined analyses of two studies found favorable results of topiramate across dose ranges for quality of life on various domains of the disease-specific MSQ, but the generic SF-36 was more equivocal (only two of 24 analyses pointed in this direction) (25,33). A correlation between dose and increasing response was not apparent.
Based on the risk differences for the various doses of topiramate versus placebo, we calculated NNHs where appropriate, and these are reported in Table 2. AE rates increased with the topiramate dose, the mean percentage of participants withdrawing because of AEs at 100 mg was 20%.
Seven trials examined several doses of topiramate against active comparators (amitriptyline 50–100 mg (37), flunarizine 5 mg (38), propranolol 80 mg (36) and 160 mg (27), sodium valproate 400 mg (35,39) and relaxation therapy (40)). Mean headache frequencies demonstrated a slight but significant advantage for topiramate over valproate, and relaxation was superior in change from baseline in MSQ. It should be noted that most of these studies were probably not powered to detect superiority of any compound, and that the doses in some studies were lower than those used in routine clinical management.
Valproate (Table 3)
Valproate main outcomes.

MD: mean difference; OR: odds ratio; CI: confidence interval; NNH: numbers-needed-to-harm; AE: adverse event; ns: not significant.
We judged seven studies as having a “high” risk of bias for at least one item in the domains allocation (48,49), randomization (48,49), performance (35,43,48,49), detection (43,48), attrition (43,47–49) and reporting (43,48,49).
Two cross-over trials of sodium valproate (46,47) showed a significant reduction in headache frequency in the active compared to the placebo group of approximately four headaches per 28 days. Four placebo-controlled divalproex sodium trials (42–45) showed that with active treatment patients are twice as likely to experience a ≥50% reduction in headache frequency. One trial (47) found that sodium valproate was significantly superior to placebo for this outcome. We have not identified any placebo-controlled studies reporting quality-of-life outcome measures. One small trial compared different doses of sodium valproate by measuring serum valproate concentrations (48), showing that lower serum levels gave rise to slightly but significantly lower headache frequencies. Comparisons with flunarizine (49) and propranolol (43) were not significantly different between treatments.
AE rates for sodium valproate and divalproex sodium were higher than for placebo and resulted in withdrawal rates between 8% and 19%. NNHs for any AE, and for the five specific AEs we judged to be of greatest clinical importance, are summarized in Table 3.
Other antiepileptics (Table 4)
Other antiepileptics main outcomes.
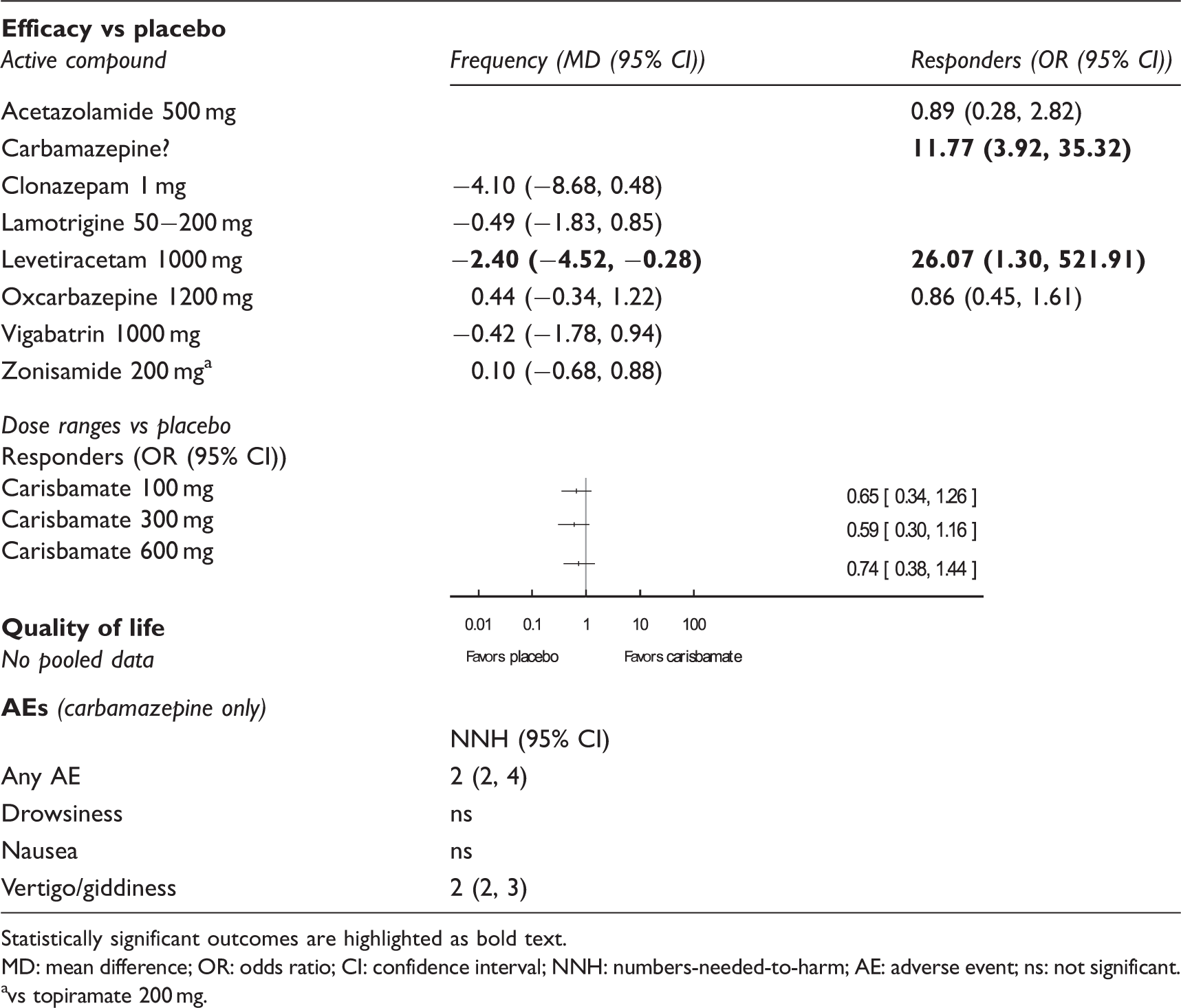
Statistically significant outcomes are highlighted as bold text. MD: mean difference; OR: odds ratio; CI: confidence interval; NNH: numbers-needed-to-harm; AE: adverse event; ns: not significant. avs topiramate 200 mg.
The duration of the treatment phase of the included trials varied from four to 15 weeks, with a mean of 10 weeks. We judged eight studies as having a “high” risk of bias for at least one of the items performance bias (30), detection bias (54), attrition bias (52,56,57), and reporting bias (51,53,55,57).
Of all investigated compounds, two drugs were better than placebo, while zonisamide was not different from topiramate 100–200 mg. Carbamazepine (dose unknown) had a higher responder rate than placebo (52), levetiracetam 1000 mg outperformed placebo with regard to post-treatment mean headache frequency and responder rate, while headache frequency was slightly but significantly higher with levetiracetam compared to topiramate 100 mg (26).
One trial provided data on quality of life but was not further analyzed as it failed to reach its primary endpoint (55). Analyses of AEs were undertaken for carbamazepine only, as that was the sole drug with a significant effect that provided safety data that enabled a comparison to placebo. For this compound, NNHs are reported in Table 4.
Discussion
In this review, the results of the meta-analyses of 43 unique studies (57 reports) of 15 antiepileptic compounds are summarized. The results may be boiled down to the following highlights:
Both sodium valproate and topiramate significantly reduced mean monthly headache frequency compared to placebo by approximately four and one days, respectively. Furthermore, and perhaps of greater clinical relevance (though less informative scientifically), patients were more than twice as likely to have a ≥50% reduction in headache frequency with divalproex sodium or topiramate than with placebo. All three target doses of topiramate significantly improved three or more domains of quality of life compared to placebo; for valproate, no placebo-controlled quality-of-life data were available. For topiramate and valproate, there is insufficient evidence to support a relation between dose and effect size. There may be a slight but significant advantage of topiramate 50 mg daily over valproate 400 mg (both doses lower than routinely used). Neither topiramate nor valproate appear to give rise to an unexpectedly high rate of AEs when used for migraine prophylaxis. For the other currently marketed antiepileptic drugs, there is no robust evidence to support the use of any of them in the preventive management of episodic migraine. Although single studies of other antiepileptics may attest to the efficacy of carbamazepine and levetiracetam, this evidence is considered insufficient because of methodological issues (small sample size (26,52), dose used not reported (52)), and efficacy offset by AEs (52). Despite a small but significant effect in one trial of gabapentin 1800–2400 mg (26), this evidence is considered insufficient because of the results from the pooled analyses, findings from other trials, and lacking evidence for a clear dose-response trend.
There are, however, several findings that require special emphasis. For topiramate, some of the included trials were almost certainly underpowered (26,29,34). The studies including more than one dose of topiramate were generally not designed to enable direct dose comparisons, and the results of the dose comparisons reported here should therefore be viewed with some caution. For valproate, the largest trial of divalproex sodium reported a statistically significant difference in favor of active treatment (42). The clinical relevance of this effect was, however, less compelling, as both the investigators’ analysis and our own found no significant difference between divalproex sodium and placebo in the proportion of responders. Furthermore, the finding that lower serum valproate levels produced lower headache frequency than higher serum levels (48) is somewhat counterintuitive, has not been replicated, and should be regarded as preliminary.
Considering gabapentin, although the pooled results of all gabapentin trials argue against a significant effect, the highest gabapentin dose studied (1800 to 2400 mg/day) demonstrated a small but significant effect in one trial both with regard to reduction of mean headache frequency and proportion of responders (24). However, an evident dose-response relation was lacking. It is remarkable that our analyses would have been biased in favor of gabapentin had it not been for the inclusion of previously confidential research reports (22–24) that after several years became available in the public domain by virtue of being entered into evidence in a legal proceeding.
For the trials with acetazolamide, clonazepam, and vigabatrin, these were certainly too small to allow any conclusion. The finding that low-dose lamotrigine (50 mg) was effective in lowering headache frequency (30) while the high dose (200 mg) was not (50) should also be interpreted with caution. The age of the single carbamazepine trial is reason for concern and may partly explain the outlying OR (52). The placebo-controlled levetiracetam study was not primarily designed to obtain clinical outcome data of the interventions (26). The equivalence in effect of zonisamide and active comparator should be further investigated in placebo-controlled trials (57).
Two trials using active comparators found a slight but significant advantage of topiramate over valproate. It should be noted that these trials have various and variable sorts of bias, notably insufficient statistical power, performance, attrition reporting, and reporting bias. Further well-designed trials, both of antiepileptics against other drug categories, and comparing different antiepileptics, are desirable.
Antiepileptic drugs do not appear to give rise to an unexpectedly high rate of AEs when used for migraine prophylaxis, although clearly (a) nausea is a problem when trials of sodium valproate and divalproex sodium are considered together, (b) a large percentage of patients taking topiramate reported paresthesia, and (c) a large percentage of patients taking carbamazepine reported vertigo/giddiness. Given the currently available evidence, in our view the latter finding outweighs the very weak evidence in favor of carbamazepine. It should also be noted that dropouts because of AEs are somewhat higher in trials of topiramate than would generally be expected on the basis of trials of other anticonvulsant drugs, particularly sodium valproate or divalproex sodium.
It can be concluded from this review that sodium valproate, divalproex sodium, and topiramate are of proven efficacy in migraine prevention and are suitable for routine clinical use. Convincing evidence for efficacy differences with amitriptyline, flunarizine, propranolol or relaxation is lacking, although topiramate may be marginally better than valproate. One important caveat should be noted: These drugs are known to or may be teratogenic, and appropriate caution must be used when prescribing to women of child-bearing age. Although AEs are reported by a large proportion of migraine patients treated with sodium valproate/divalproex sodium or topiramate, these are usually mild and of a non-serious nature. On a case-to-case basis, rational prescriber preferences may be appreciated because of differences in side effect profiles.
The evidence derived from trials of gabapentin provides insufficient grounds for advocating their use in clinical practice, even at higher dosing schemes. Similarly, for all other antiepileptic drugs there is, at present, either insufficient or no evidence concerning either efficacy or tolerability to warrant their use in clinical practice. Although a large number of antiepileptics, including the newer ones, appear to be amply used in the management of headache, as judged by the vast number of publications of small retrospective series and studies in chronic daily headache or transformed migraine, these results cannot be translated as compelling evidence, and thus we would like to argue against the liberal use of antiepileptics other than topiramate and valproate.
Clinical implications
Both sodium valproate and topiramate significantly reduce mean monthly headache frequency approximately four and one days, respectively. Patients are more than twice as likely to have a ≥50% reduction in headache frequency with divalproex sodium or topiramate than with placebo. Topiramate significantly improves quality of life compared to placebo. There is insufficient evidence to further support the use of any dose of gabapentin. For the other currently marketed antiepileptic drugs, there is no robust evidence to support the use of any of them.
Notes
This paper has been adapted from four systematic Cochrane reviews published in the
Linde M, Mulleners WM, Chronicle EP, et al. Topiramate for the prophylaxis of episodic migraine in adults.
Linde M, Mulleners WM, Chronicle EP, et al. Valproate (valproic acid or sodium valproate or a combination of the two) for the prophylaxis of episodic migraine in adults.
Linde M, Mulleners WM, Chronicle EP, et al. Gabapentin or pregabalin for the prophylaxis of episodic migraine in adults.
Linde M, Mulleners WM, Chronicle EP, et al. Antiepileptics other than gabapentin, pregabalin, topiramate, and valproate for the prophylaxis of episodic migraine in adults.
Footnotes
Acknowledgments
Professor EP Chronicle, PhD, sadly passed away February 9, 2007. We wish to acknowledge Professor Chronicle’s major contribution and tremendous effort in compiling all statistical analyses and much of the text of the original review on antiepileptics.
We thank the following members of the Cochrane Editorial Unit (CEU) and the Cochrane Pain, Palliative and Supportive Care Review Group (PaPaS) for their assistance: Ruth Foxlee (CEU), Jane Hayes (PaPaS), and Joanne Abbott (PaPaS) for assistance in designing search strategies and running searches, Prof Timothy Steiner (PaPaS) for editorial guidance, and Dr Rebecca Gray (PaPaS) for editorial assistance and technical support.
Funding
Lifting The Burden: The Global Campaign against Headache and the International Headache Society provided financial support for the editorial process for the original Cochrane reviews.
Conflicts of interest
Dr Mulleners has nothing to disclose. Dr Linde was involved as an investigator in a clinical trial sponsored by AstraZeneca and comparing candesartan, propranolol, and placebo in the prophylaxis of migraine. Dr Linde also received a travel grant from Allergan. Dr Linde was senior investigator in one of the included studies (Varkey 2011). Dr McCrory received personal fees from the Plaintiff's Sttering Committee in the Neurontin Marketing and Sales Practices. Moreover, he received financial reimbursements for committee and consultant activities from Cleveland Clinic, MEDACorp and The College of American Pathologists.
