Abstract
To delineate if the change in cortical excitability persists across migraine attacks, visual evoked magnetic fields (VEF) were measured in patients with migraine without aura during the interictal (n = 26) or peri-ictal (n = 21) periods, and were compared with 30 healthy controls. The visual stimuli were checkerboard reversals with four different check sizes (15',30',60' and 120'). For each check size, five sequential blocks of 50 VEF responses were recorded to calculate the percentage change of the P100m amplitude in the second to the fifth blocks in comparison with the first block. At check size 120', interictal patients showed a larger amplitude increment than controls [28.1 ± 38.3% (S.D.) vs. 8.7 ± 21.3%] in the second block and a larger increment than peri-ictal patients in the second (28.1 ± 38.3% vs. -3.2 ± 19.2%), fourth (22.7 ± 31.2% vs. -5.7 ± 22.3%) and fifth (20.5 ± 30.4% vs. -10.8 ± 30.1%) blocks (P < 0.05). There was no significant difference at other check sizes or between peri-ictal patients and controls. In conclusion, there may be peri-ictal normalization of visual cortical excitability changes in migraine that is dependent on the spatial frequency of the stimuli and reflects a dynamic modulation of cortical activities.
Keywords
Introduction
Migraine is characterized by sensitivity to light, light-triggered attacks and visual illusion to grating patterns (1, 2). Patients with migraine with aura (MA) invariably experience aura of the visual type (3). Cortical spreading depression, a proposed neural event for migraine aura, is strongly suggested to originate from the visual cortex in vivo (4, 5). These observations indicate an alteration of visual cortical excitability in migraine. However, standard visual evoked potential (VEP) studies have failed to corroborate this due to heterogeneous findings in response amplitudes or latencies (6, 7).
In contrast, studies of VEP habituation that calculated amplitude changes of sequential sub-average blocks have yielded more consistent findings (6). In these studies, amplitude attenuation of a sub-averaged block when compared with the first block was defined as habituation, whereas response augmentation was alternatively regarded as potentiation (8, 9). Migraine patients tended to have potentiation of cortical activation in subsequent blocks compared with the initial block (8).
To date, deficient habituation is proposed as an interictal abnormality of migraine (10). This feature of cortical excitability change reflects interictal dysfunction of central neurotransmission, especially the raphe–cortical serotonergic pathway (11, 12). Nevertheless, plasma serotonin levels are increased during headache attacks in migraine patients (13). A recent brain positron emission tomography study visualized in vivo a significant increase of the serotonin synthetic rate during migraine attacks compared with the interictal period (14). An intriguing question thus arises whether the excitability change persists across migraine attacks when serotonergic or other neurotransmissions are dramatically modified.
Judit et al. observed a deficient habituation of VEP in interictal migraine (15). They showed VEP habituation in patients with recordings on or prior to the day of migraine. Another VEP study (16) showed significant VEP habituation when comparing pre-ictal (migraine attacks within 24 h after the recording) with interictal recordings. Unfortunately, the authors failed to confirm the habituation deficit in migraine patients compared with controls. Other studies using contingent negative variation also found no such normalization phenomenon (17, 18).
This study aimed to evaluate the excitability changes of the visual cortex during the interictal and peri-ictal periods of migraine by using magnetoencephalography (MEG). MEG is superior in measuring cortical activations compared with scalp electroencephalography due to the absence of conduction volume effect (19) and ‘paradoxical lateralization’ phenomenon (20, 21). This study also aimed to clarify if the habituation deficit in migraine is dependent on spatial frequencies of stimuli, which may influence the components and habituation behaviour of VEPs (22–24).
Methods
Subjects
Seventy-seven right-handed subjects participated in this study: 47 patients with migraine without aura (MoA) (35F, 12M, mean age 30.1 ± 6.4 years) and 30 age- and sex-matched healthy controls (22F, 8M, mean age 31.1 ± 6.0 years). Recruited from the Headache Clinic of Taipei Veterans General Hospital, the migraine patients were diagnosed according to the International Classification of Headache Disorders, 2nd edn (code 1.1) (25). They had one to eight attacks (1–12 headache days) per month and none received migraine prophylactic therapy. Each completed a semistructured questionnaire on demographics, medical history, medication behaviour, and headache and aura profile during their first visit. They also kept a headache diary after recruitment. The Migraine Disability Assessment Questionnaire assessed any migraine-related disability (26, 27).
The control subjects were medical colleagues, their relatives or friends who did not have past or family histories of migraine and denied any headache attack in the past year. These volunteer subjects were unaware of the study purpose and did not participate in the experimental design, data analysis or manuscript preparation.
All patients denied any history of systemic or other neurological disease, and presented normal physical and neurological examinations. Subjects who took medications or hormone therapy on a daily basis were excluded. The hospital's Institutional Review Board approved the study protocol and each participant provided written informed consent.
The time interval (number of days) between visual evoked magnetic field (VEF) measurement (denoted as day 0) and the most recent migraine attack prior to or after the measurement was determined in all patients based on their headache diaries. Patients who did not have migrainous headache within a period of 2 days before (days −1 and −2) and after (days 1 and 2) the VEF recordings (day 0) were classified as interictal, whereas those with migraine attacks within this period (day −2 to day 2) were classified as peri-ictal. Selection of the time window for the peri-ictal period was based on earlier studies (15, 28).
To minimize any hormonal effect, VEF measurements in women were taken in the luteal phase estimated by their last menstrual cycle and confirmed by the next menstruation according to the headache diaries or telephone interviews.
Visual stimulation
Pattern-reversal white and black checkerboard stimuli were generated using a personal computer. Visual stimuli were delivered via Presentation 0.52 NBS (Neurobehavioral Systems, Inc., Albany, CA, USA) projected onto a screen approximately 110 cm in front of the subject by an LCD projector (PLUS Vision Corporation, Tokyo, Japan). There were four different check sizes of the pattern as stimulation for separate study sessions: 120, 60, 30 and 15 min of arc (′). The checkerboard reversed every second and extended 15° (W) × 22° (H) in the left hemifield of the subject. The mean luminance of the checkerboard pattern was 12 cd/m2, with contrast kept at 0.94. The time lag between trigger onset and stimulus presentation on the screen was 19.0 ± 0.6 ms (mean ±
VEF recording
VEF was recorded in the mornings (08.00–12.00 h). The subject sat comfortably in a magnetically shielded room while the head was supported against the helmet-shaped bottom of a whole-scalp 306-channel neuromagnetometer (Vectorview™; Elekta Neuromag, Helsinki, Finland). Vision was corrected to normal if there was refractive error. During the visual stimulation, the subject gazed at a red fixation point located at the centre of the screen, with the right eye covered by gauze. The subject underwent four separate sessions, with one of the four check sizes randomly displayed within each session. For each session, a total of 250 responses were obtained, with 50 responses sub-averaged in five sequential blocks to evaluate VEF amplitude changes (described below). There was a 3-min interval between sessions to allow the subject to recover from the previous visual stimulation.
The neuromagnetometer comprised 102 identical triple sensor elements, which consisted of two orthogonal planar gradiometers and one magnetometer coupled to three superconducting quantum interference devices. Data analysis of this study was based on signals of the 204 planar gradiometers, which were bandpass filtered (0.1–130 Hz) and digitized at 500 Hz. Epochs coinciding with signals exceeding 300 mV in the simultaneously recorded vertical electro-oculogram were automatically rejected from the analysis.
Dipole source modelling of the 250-trial average response in each check-size session
We analysed the cerebral source of the 250-trial average response in each check-size session by using dipole source modelling. All channels of the measured data were visually searched for signal deflections clearly exceeding the pre-stimulus background level and selected the time windows of cortical responses for further analysis. The single equivalent current dipole (ECD) that best described the measured activities (P100m, the magnetic counterpart of P100 in conventional VEP) peaking at approximately 100 ms after stimulus onset was determined by a least-squares search using subsets of 32–36 channels around the maximal responses. These calculations resulted in a three-dimensional location, orientation and strength (baseline-to-peak amplitude) of the ECD in a spherical conductor. The model's goodness-of-fit was calculated, and only ECDs explaining ≥ 80% of field variance at the selected time periods over a subset of channels were used for further analysis.
The validity of the single-dipole model was then further evaluated by comparing the measured signals with the responses predicted by the model when all channels were taken into account. If the measured signals around the P100m peak were left inadequately explained by the model, the above dipole modelling was re-evaluated for making a more accurate estimation of the source.
The exact location of the head with respect to MEG sensors was determined by measuring magnetic signals produced by current leading to the four head indicator coils at known sites on the scalp. The coil locations with respect to anatomical landmarks on the head were determined using a three-dimensional digitizer to allow further alignment of the MEG and magnetic resonance imaging (MRI) systems.
MRI of the subject's brain was acquired with a 3-T Bruker Medspec300 scanner. Based on the spherical head model, the three-dimensional location and orientations of the ECDs calculated from source analysis were co-registered to the MRI of the subject's brain. The positive x-, y- and z-axes in the head-coordinate system were towards the right pre-auricular point, the nasion and the head vertex, respectively.
Measurement of P100m latencies and amplitudes of subaverage VEF responses
This ECD model for the P100m of the 250-trial average response was then applied to fit the responses in individual subaverage blocks of the same check-size session. Accordingly, a source waveform of P100m was obtained and its strength was allowed to change as a function of time. Here the validity of the single-dipole model for individual blocks was mainly evaluated by comparing the measured signals with the responses predicted by the model. If the measured signals around the peak were adequately explained, the P100m source wave was accepted for further analysis.
Based on the source waveform, the peak latency and baseline-to-peak amplitude of P100m of each subaverage block were obtained. Percentage changes of amplitude in blocks 2–5 compared with block 1 were calculated for each check-size session (referred to subsequently as ‘subaveraged amplitude change’).
Statistical analysis
Group differences in clinical profiles or the 250-trial average responses were analysed by Student's t-test (two groups) or one-way ANOVA (three groups). For subaveraged VEF data, amplitude of block 5 was first compared with block 1 at each check size by paired t-test to identify the trend of VEF change in each subject group, with significant increases regarded as potentiation and decreases as habituation of VEF. Group difference of subaveraged VEF data was also investigated by two-way ANOVA with repeated measures, using the block (n = 5 for subaveraged amplitude or n = 4 for percentage change of subaveraged amplitude) as within-subject factors and the group (n = 3) as between-subject factor. The number of degrees of freedom was adjusted according to Greenhouse and Geisser when necessary (29). Post hoc analyses for all the above comparisons were conducted by Scheffe's test. The Pearson's correlation test was used to search for correlations between subaveraged amplitude change and amplitude of block 1 or clinical profiles. Results were considered significant at P < 0.05.
Results
The 47 migraine patients enrolled in this study were further dichotomized as interictal (n = 26) or peri-ictal (n = 21) patients, both being comparable in demographics and headache profiles, except for a longer time interval to the onset of the most recent migraine attack in the interictal group (P < 0.001) (Table 1).
Demographics and headache profiles (mean ±

P < 0.001 for interictal vs. peri-ictal patients (Student's t-test).
NA, not available; 0–10, numerical scale; MIDAS, the Migraine Disability Assessment Questionnaire.
In all of the subjects, the P100m sources were clearly seen near or around the right striate cortex oriented medially. P100m measurements in relation to check size are shown in Table 2 (grand averaged data). There was no group difference.
Mean (±
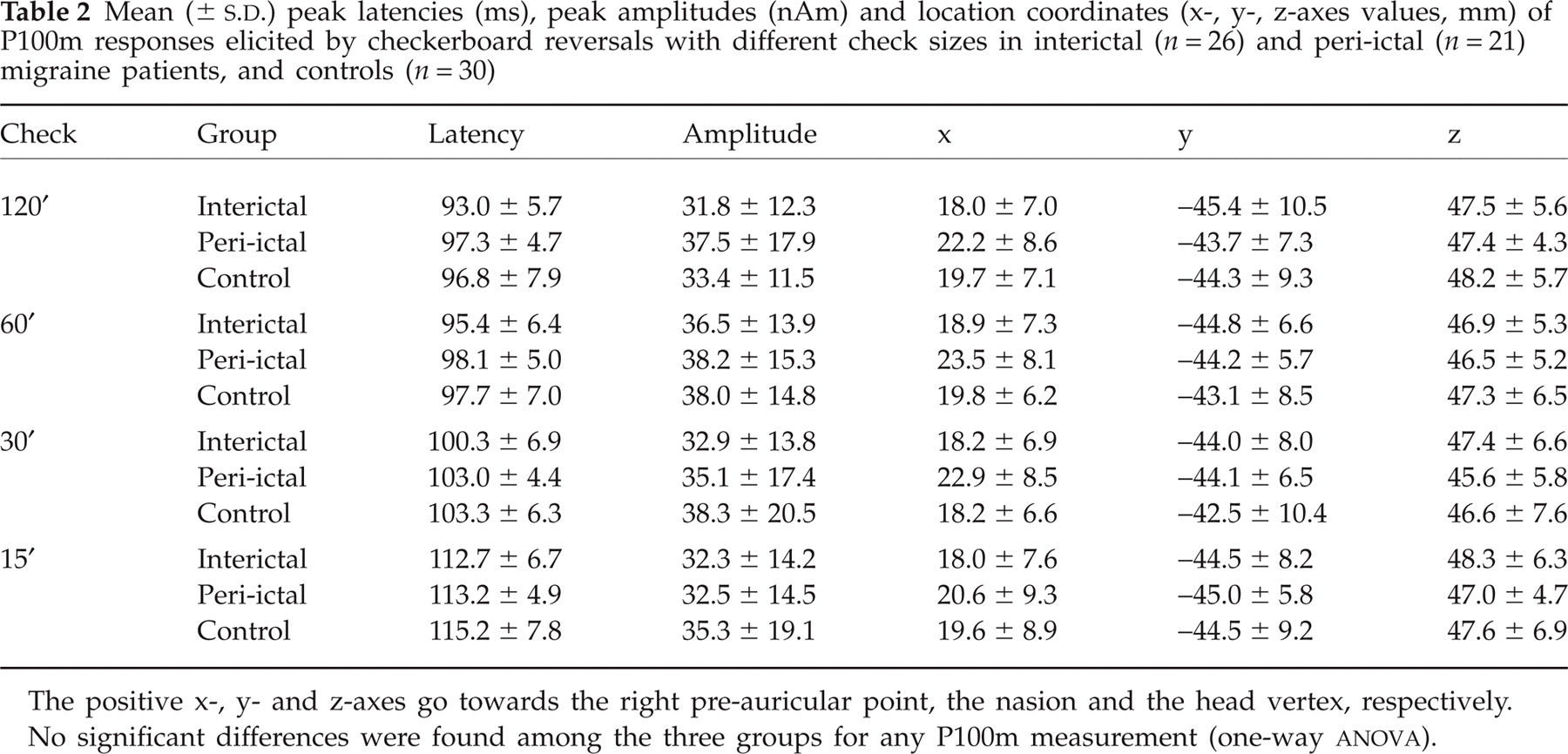
The positive x-, y- and z-axes go towards the right pre-auricular point, the nasion and the head vertex, respectively.
No significant differences were found among the three groups for any P100m measurement (one-way
The source modelling obtained from the total averages of P100m accounted for at least 65% of the field variance of all individual blocks in each subject. The subaveraged P100m amplitudes in sequential blocks are shown in Table 3. There was habituation of VEF at 15′ in controls (P = 0.020) and interictal patients (P = 0.012), although the latter also showed VEF potentiation at 120′ (P = 0.044) and 60′ (P = 0.020). There was no VEF habituation or potentiation in peri-ictal patients.
Mean (±
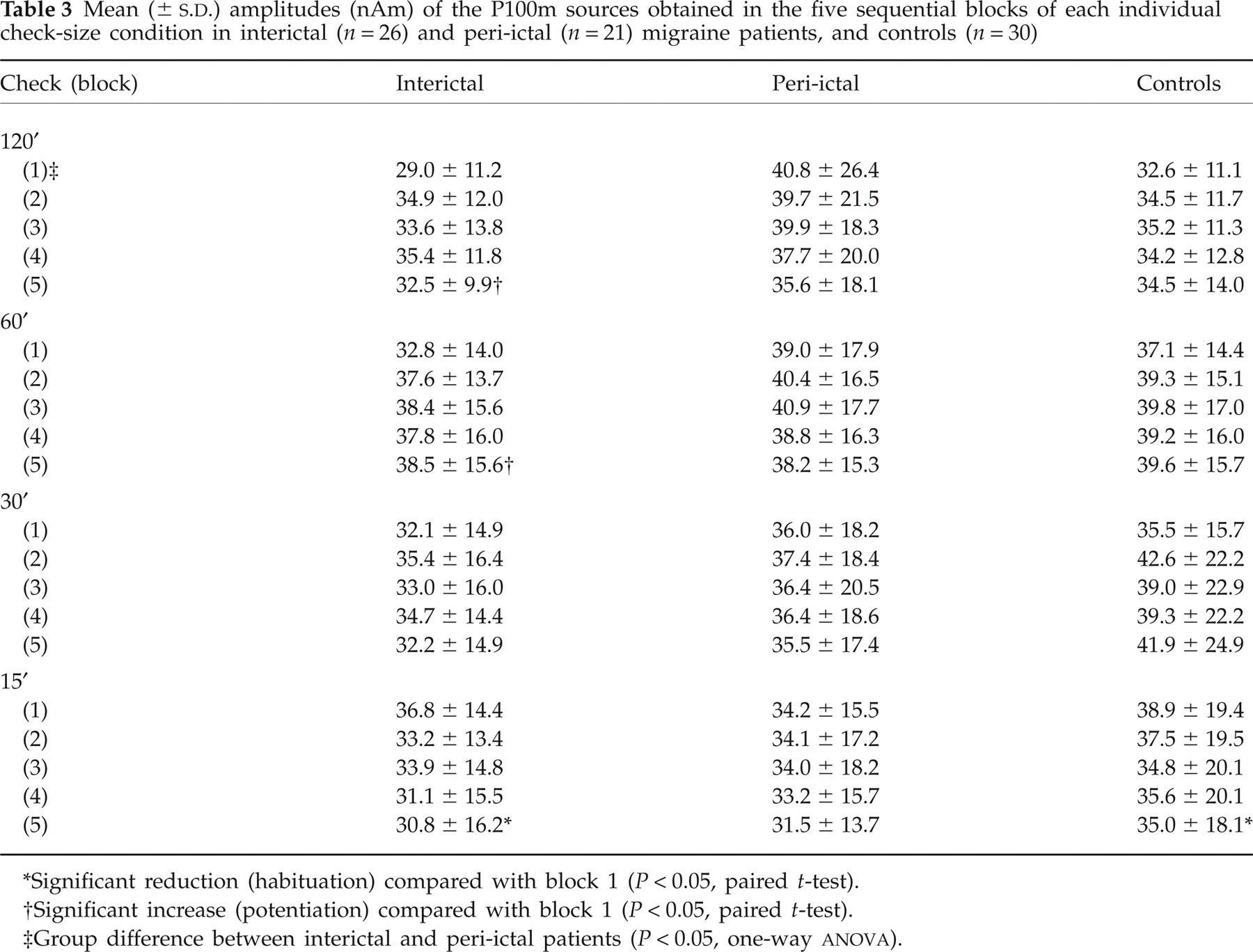
Significant reduction (habituation) compared with block 1 (P < 0.05, paired t-test).
Significant increase (potentiation) compared with block 1 (P < 0.05, paired t-test).
Group difference between interictal and peri-ictal patients (P < 0.05, one-way
In Table 3, the mean amplitudes of P100m ECDs tended to be lower in interictal patients compared with controls and peri-ictal patients in the first block of all check sizes except for 15′. Although there were no group differences in two-way
Percentage changes of individual subaveraged amplitude in blocks 2–5 compared with block 1 are shown in Fig. 1. A group difference in subaveraged amplitude change was found at 120′ (F 2,74 = 8.9, P < 0.001), but not at other check sizes. Group × block interaction was not significant. Post hoc analysis showed that interictal patients had a larger amplitude increment in block 2 (P = 0.040) than controls, and a larger increment than peri-ictal patients in blocks 2 (P = 0.001), 4 (P = 0.002) and 5 (P = 0.001).
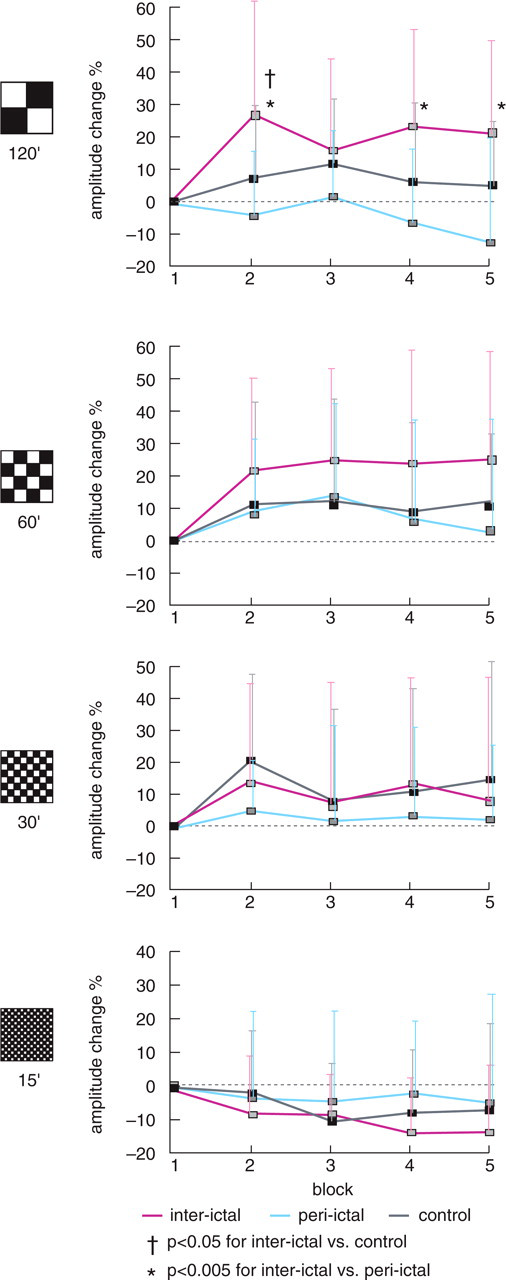
P100m amplitude changes in five sequential blocks expressed as percentages (mean ±
In peri-ictal patients, further categorization by pre-attack (days −1 and −2; n = 8), in-attack (day 0; n = 7) and post-attack (days +1 and +2; n = 6) revealed no group difference in the subaveraged amplitude change of blocks 2–5 at 120′[block 2: −9.0 ± 28.6 (mean ±
In interictal patients, subaveraged amplitude changes in blocks 2 (r = −0.539, P = 0.005) and 5 (r = −0.734, P < 0.001) were inversely correlated with the amplitude of block 1 at 120′ but not with any clinical profile. There was no correlation at other check sizes or subject groups.
Discussion
This study has provided evidence of the excitability change of the visual cortex in migraine and verifies the potential of MEG in identifying this neurobiological change. In line with VEP studies (8, 9, 30), interictal patients at 120′ showed a higher amplitude increment on trial repetition and a tendency for lower amplitude of the first block when compared with controls. In addition, there was an association between the first block amplitude and subaveraged amplitude change in interictal patients but not in the controls. These findings imply initial hypoactivation of the visual cortex in migraine that is followed by a response augmentation on stimulus repetition. In other words, this study does not support the hypothesis of hyperexcitable excitatory neurons as the putative cause of excitability change in migraine, which favours an increased initial activation. Some researchers have proposed ‘hyper-responsiveness’(12) instead of ‘hyperexcitability’ to describe such a complex excitability change dependent on the stimulus number in migraine.
This MEG study has further delineated the excitability change of migraine. First, the excitability change is state-dependent across the ictal–interictal cycle of migraine. Unlike the interictal data, our peri-ictal recordings did not show response augmentation. This suggests peri-ictal normalization of the excitability change, in line with the aforementioned VEP habituation study (15). The exact pathophysiological mechanism remains unclear, but the dynamic variation of cortical excitability between interictal and peri-ictal periods may reflect a disposition change of serotonin transmission (14, 28). Furthermore, migraine patients would resume VEP habituation after treatment with fluoxetine, a selective serotonin re-uptake inhibitor (31), which up-regulates serotonergic disposition.
Second, the excitability change is stimulus-driven and depends on the spatial frequency of the stimulus. The excitability change of migraine and its normalization during the peri-ictal period has been identified exclusively at the check size of 120′. In parallel visual processing, low spatial frequency (or large check size) mainly activates the magnocellular (Y) pathway that processes motion/luminance, whereas high spatial frequency preferentially activates the parvocellular (X) pathway processing contrast/contour (32, 33). Therefore, the excitability change of the visual cortex in migraine preferentially involves the magnocellular pathway. Our findings that the controls and interictal migraine patients both showed P100m habituation at 15′ imply a relatively minor role of the parvocellular dysmodulation.
Early psychophysical studies of migraine support the predominant magnocellular dysfunction (34–36). Magnocellular predominance also echoes the concept of ‘X–Y imbalance’ proposed by a VEP study (24), which attributed increased N2 latency of VEP in migraine patients to attenuated contour-specific N130 with strong predominance of luminance-dependent N180. Notably, the majority of VEP studies have shown deficient habituation in patients using the small check size of 8′, which might imply an excitability change involving the parvocellular pathway. However, this should be interpreted with caution, because these VEP studies adopted a high reversal rate (3.1 Hz) in favour of the magnocellular rather than parvocellular pathway.
On the other hand, several VEP habituation studies in migraine used large check sizes sharing the magnocellular properties with the 120′ checks in this study. Nevertheless, there is habituation difference in two prior studies (15, 30) using 68′ checks and higher reversal rate (3.1 Hz) (‘more magnocellular’), but not in the recent study that used a similar check size (62′) with lower reversal rate (0.95 Hz) (37). The methodology difference, especially the reversal rate, is assumed to be the culprit in the latter (37).
Consequently, many factors should be considered as a whole in interpreting and comparing VEP/VEF studies of the excitability change of migraine (Table 4). This study has highlighted only the effects of the temporal phase of migraine and stimulus spatial frequency.
Factors considered in the interpretation and comparison of visual cortical excitability in migraine patients

Third, the excitability change may fluctuate on stimulus repetition. In this study, the response augmentation was identified at the second blocks (Fig. 1), but not in all of the sequential blocks. This inconsistency has also been noted, although to a lesser degree, in VEP habituation studies conducted at 3.1 Hz reversal rate (8, 9). Since habituation is a complex neurobiological phenomenon involving multiple neurotransmissions (11, 43, 44), the cause for this inconsistency may be the interaction among these transmissions, the rate and number of stimulus repetitions (45), and even lactate accumulation (9, 46). Among these factors, the low stimulus repetition rate of the present study compared with the above VEP studies (3.1 Hz) (8, 9) may especially explain the inconsistent response augmentation across sequential blocks. As stated by the criteria of habituation proposed by Groves and Thompson (47), a high repetition rate speeds up, and a low repetition rate slows down the habituation. Future studies with a higher stimulus rate, longer stimulus time, and concomitant measurements of neurotransmission and cortical lactate levels may help clarify these issues.
Although some features of the excitability change in migraine patients delineated in this MEG-VEF study are consistent with VEP studies, interpreting these data should take different instrumentation and methodology into consideration. The excitability change of the interictal patients at 120′ cannot be interpreted as ‘lack of habituation’ as in earlier VEP habituation studies, because the controls did not show habituation at this check size either. The finding may suggest ‘augmented potentiation’ or ‘relative habituation deficit’. Notably, the small check size of 8′ used by the majority of VEP habituation studies may not elicit a discernible VEF response (48). In contrast, excitability change at 120′ is not reported in VEP studies because this check size is rarely chosen as the spatial frequency of stimulus.
Despite methodology concerns, this study interestingly suggests that the excitability change of migraine may be addressed in quantity (degree of response augmentation) rather than in quality (habituation vs. potentiation). Again, such a finding partly echoes the conception of using ‘hyper-responsiveness’(12) as a better description of the excitability change of migraine.
The current study has some limitations. Interictal and peri-ictal recordings in the same subject were not obtained. Theoretically, a longitudinal study using paired design (interictal vs. peri-ictal) will be more sensitive in determining cortical excitability change, since within-subject comparison reduces intersubject variation. However, a recent longitudinal study (37) has not found any habituation differences in terms of VEP amplitudes. Except for the low reversal rates (0.95 Hz) discussed as the cause of non-significant habituation differences, homogenization of the menstrual phase is also technically challenging for such longitudinal studies.
On the other hand, one may question whether the time interval considered as peri-ictal period (day 0 ± 2) in this study is optimum, because excitability may vary considerably during this period. Our peri-ictal patients did not differ in subaveraged amplitude change when categorized by pre-, in- and post-attack, or by time interval to migraine attack, implying a non-significant excitability fluctuation during this period. Nevertheless, this may be constrained by the small number of subjects and requires validation by consecutive measurements in a larger patient group.
Another limitation is the left hemifield stimulation, which elicited only right visual cortical responses in the patients. Some literature has noted VEP asymmetry in MA patients (49, 50), hence the concern when generalizing the findings to bilateral visual cortices. However, VEP asymmetry in MoA patients remains contradictory (49–51). In fact, we did a pilot study of 13 MoA patients (10F, 3M) also enrolled by this study. In two separate 250-trial interictal recordings (five blocks of 50 trials) stimulated with left- and right-hemifield 60′ checks, respectively, the total-averaged amplitudes of P100m did not differ between the two hemispheres [37.3 ± 20.3 (right) vs. 38.7 ± 14.8 nAm (left), P = 0.676, paired t-test], nor did the subaveraged amplitude changes (unpublished data). We thus followed an earlier VEF study (48) that stimulated only the left hemifield to avoid any adverse effect and attention deterioration after protracted stimulation.
The enrolment of medical colleagues as our controls may also cause weakness in methodology associated with lack of blinding. Nonetheless, they were unaware of the study purpose or design and comprised a small proportion (n = 4) of the controls (n = 30). A 3-year difference in migraine history (Table 1) between our interictal and peri-ictal patients may cause bias because the amplitude of P100 declined with the length of history of migraine in one study (52). However, this change was found in those suffering from MA for > 30 years, much longer than the 3-year difference between patients here. Another study (53) in MoA patients did not show any correlation between length of migraine history and VEP amplitudes, similar to the study here on VEF amplitudes or subaveraged amplitude changes. Lastly, despite cueing by a fixation point and monitoring of eye movement, physiological nystagmus or small deviations of eye fixation of the subjects during visual stimulation cannot be avoided. Nevertheless, results of the source modelling ensured our fixation control, since cortical activation was adequately explained by a single dipole located near or around the right striate cortex.
In conclusion, visual cortical excitability is not a steady biological trait of migraine. There is peri-ictal normalization of the excitability change, and its dynamic fluctuation and stimulus-dependent abnormality may partially explain the discrepancy among prior studies recording cerebral responses of migraine patients.
Acknowledgements
This study was supported by research grants from the National Science Council (Taiwan) (NSC 94-2314-B-038-014 and NSC-95-2314-B-075-121 to W-T.C., NSC-95-2314-B-010-030-MY3 and NSC-96-2628-B-010-030-MY3 to Y-Y.L., NSC-95-2314-B-010-031-MY3 to S-J.W.) and Taipei Veterans General Hospital (V95ER3-006, V96ER3-008, V97ER3-006, VGHUST97-P6-24, V97C1-034 to Y-Y.L.). The authors express their gratitude to Dr Shu-Chiung Chiang for statistical assistance.
