Abstract
Keywords
Introduction
Episodic migraine (EM) may evolve into the more disabling chronic migraine (CM) (1). The exact mechanism of this evolution remains elusive, but EM and CM may exhibit different neurophysiological profiles. A transcranial magnetic stimulation (TMS) study showed a lower phosphene threshold in patients with EM than in controls, with a further reduction in CM (2). In electroencephalographic (EEG) or magnetoencephalographic (MEG) studies, the visual cortex excitability of EM is characterized by pre-excitation hypoexcitability (i.e. lower amplitudes at initial stimulations) and a lack of habituation (i.e. response attenuation) to repetitive visual stimuli during interictal periods (3–5). These response profiles reverted to high initial amplitudes followed by normal habituation during the ictal or peri-ictal period (6,7), reflecting profound changes in serotonergic transmission brought on by migraine attacks (8).
Visual cortex excitability patterns in CM and EM have recently been described in our MEG studies (9,10). An intriguing finding was that the visual neuromagnetic P100m responses in interictal CM patients showed high initial amplitude and normal habituation, which corresponded to the pattern in ictal EM (9,10). The ictal-like excitability in CM that persists throughout the ictal–interictal cycle of migraine may implicate CM as the most severe end of the migraine spectrum underpinned by ‘never-ending migraine attacks’ (11).
Despite the potential to differentiate the disease states of migraine (i.e. EM vs CM), visual cortex excitability still needs longitudinal data to validate its pathophysiological link to CM and its evolution. Visual cortex excitability should ideally be able to reflect the different pathophysiologies underpinning CM and EM and the switch features corresponding with clinical phenotype. Nevertheless, CM is a chronic disorder for many sufferers (1). In population studies, around one-third of CM patients had the same diagnosis at the second year follow-up (12,13); a significant portion may even have disabling symptoms bothering them for a decade (14,15).
Moreover, there has been evidence of structural changes associated with CM, such as iron deposition in the antinociceptive periaqueductal gray matter (16). Therefore, to date, it remains unknown whether the characteristic visual cortex excitability in CM is a modifiable or an inherent physiological trait, given the risk of intractability. Herein, we extended the previous MEG findings in CM to describe the longitudinal change of visual cortex excitability during CM remission. We hypothesized that visual cortex excitability would be a dynamic biological state which varied with clinical migraine status. The specific aims of this study were to:
test if visual cortex excitability was modifiable in remission from CM to EM; investigate if the feature of visual cortex excitability could reflect the disease states (CM vs EM) during CM remission; and explore the potential clinical predictors of the longitudinal change of visual cortex excitability.
Methods
Twenty-five CM patients without medication overuse (20 females and 5 males, 33.7 ± 9.2 years) who enrolled in our earlier MEG study (9) were eligible to participate in this study. To characterize the change in visual cortex excitability associated with CM remission, this study used a longitudinal design to obtain clinical assessment and MEG measurements both before and after remission from CM to EM by means of migraine preventive treatment.
Diagnosis of CM was made according to the revised criteria of the International Classification of Headache Disorders, 2nd edition (ICDH-II) (17) (≥eight migraine days and <15 headache days per month). EM was also defined using the ICDH-II criteria (18) and 15 headache days per month. Subjects who took medications or hormone therapy daily, or received migraine prophylactic therapy before participation were excluded. The hospital’s Institutional Review Board approved the study protocol and each participant provided written informed consent.
The methodology of the clinical assessment and MEG measurement used with these CM patients has been detailed elsewhere (9). Briefly, clinical assessment included a questionnaire survey of headache profiles and the maintenance of a headache diary throughout the study period. Each subject underwent scheduled MEG measurements during the interictal periods (headache-free at least 3 days before and after the recording) to acquire visual neuromagnetic responses elicited by left-hemifield checkerboard reversals (check size: 120 min of arc; six reversals/s). The MEG measurement was rescheduled if there was use of analgesics, triptans or ergots for any reason within the previous 48 h. In each subject, a total of 1500 visual neuromagnetic responses (bandpass filtered 0.1–130 Hz and digitized at 500 Hz) were obtained and sequentially divided into 30 blocks (50 responses each), and were analysed by single dipole modeling to yield the latency, amplitude and location coordinates (x-, y-, z-axis values) of grand- and sub-averaged P100m.
After baseline assessment and MEG recording, all CM patients were treated with topiramate (a migraine preventive agent) at a dose of 50–100 mg per day. Those patients who maintained treatment for at least 3 months with successful remission from CM to EM were asked to repeat the headache assessment and interictal MEG. Topiramate was gradually withdrawn and discontinued at least 3 days before the follow-up recording.
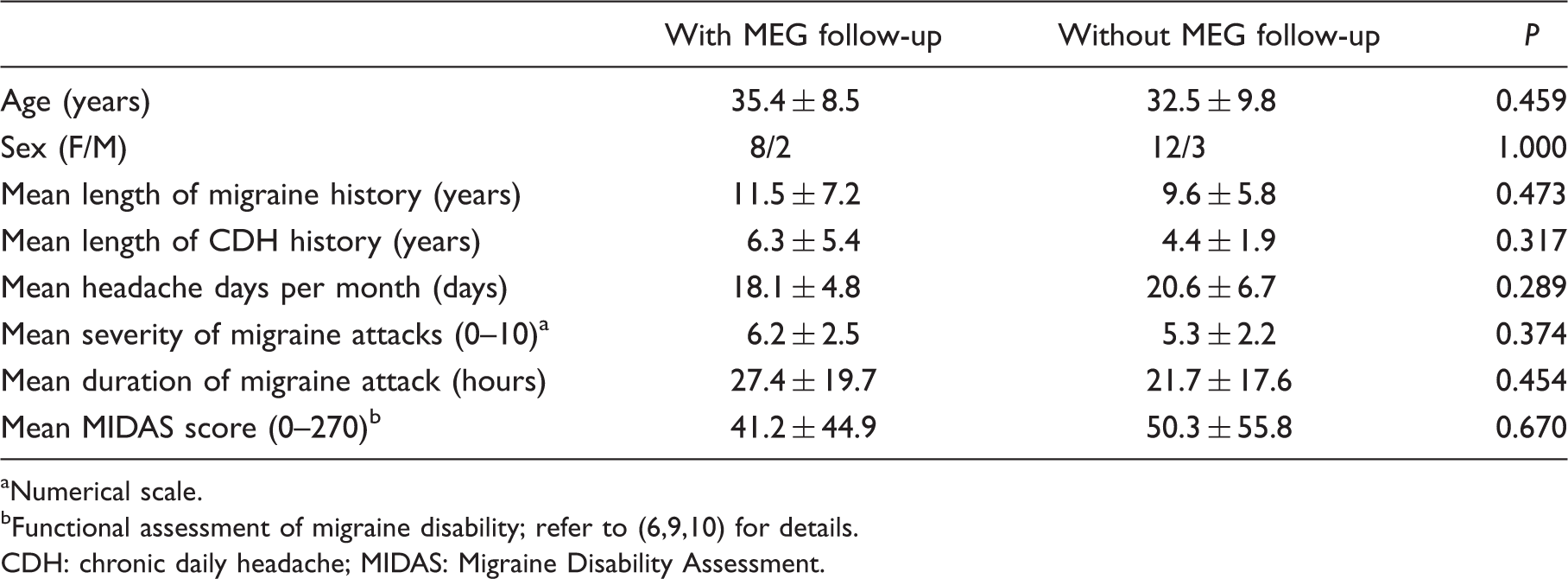
For statistical analyses, group differences of demographics and clinical profiles in CM patients with vs without follow-up were first analysed by means of the Student’s
Results
Comparisons of peak latencies, peak amplitudes and location coordinates (x-, y-, z-axis values) of P100m responses obtained from CM patients before and after remission to EM (

Significant decrease (habituation) or increase (potentiation) compared to block 1 (
The positive x-, y- and z-axes go towards the right preauricular point, the nasion and the head vertex, respectively. CM: chronic migraine; EM: episodic migraine.

Percentage changes of sub-averaged P100m amplitudes in 10 patients (patients A–J) with chronic migraine (CM) before (line) and after (dotted line) remitting to episodic migraine (EM). Sub-averaged P100m amplitudes at blocks 2, 9, 16, 23 and 30 are compared to the initial block (block 1) to yield the relative changes in percentage values. Note that the percentage changes increased in three patients (patients A, E and J) across the whole trial and in all patients at the end of stimulation (block 30) after remission. Pt: patient.

Mean percentage changes of sub-averaged P100m amplitudes at blocks 2, 9, 16, 23 and 30 compared to the initial block (block 1) in 10 patients with chronic migraine (CM) before and after remitting to episodic migraine (EM). Pertinent changes in patients with interictal EM (
P100m and clinical parameters (demographics and headache profiles, including headache intensity, frequency, pattern, duration and associated symptoms) were not correlated either before or after remission. There was also no correlation between the changes of these parameters associated with remission (all
Discussion
Visual cortex excitability varies with clinical migraine status
The main finding of this study is the significant change in MEG measurement of visual cortex excitability when CM remits to EM. The P100m parameters featuring CM, including increased amplitude at the initial block and response habituation to repetitive stimulations, both shifted towards those characterizing EM, i.e. decreased initial amplitude with subsequent potentiation. The significant modification indicates that visual cortex excitability is not a steady physiological trait inherent in CM; rather, it may switch in tandem with the clinical status of migraine (CM vs EM).
The modifiable excitability in CM remission is in line with a TMS study which used magnetic suppression of perceptual accuracy (MSPA) to show altered visual cortex excitability when headache frequency was significantly reduced in CM by topiramate; however, the finding was not independent of a direct pharmacological effect of topiramate due to the lack of a washout period (19). It is also notable that CM, despite potential intractability, still remains a relatively fluid clinical status of migraine in epidemiological studies (12,13). In contrast with the one-third of CM patients maintaining the same diagnosis throughout, one-fourth experienced CM remission at the biannual follow-ups in the American Migraine Prevalence and Prevention study (13).
Changes of visual cortex excitability in relation to CM pathophysiology
The parallel switch of excitability in CM remission further consolidates the pathophysiological link of CM to visual cortex excitability. The reduction of P100m amplitudes after CM remission, especially at the initial block, is compatible with the resolution of visual cortex sensitization that underpins exaggerated P100m amplitudes in CM as shown earlier (9). On the other hand, the finding of a lack of habituation here, when regarded as up-regulated excitability, seems contradictory to the above amplitude decrease which suggests ‘normalized’ hyperexcitability. Nevertheless, this may reflect a resolution of continuous ictal activation in CM due to decreased central sensitization, and a resumed central excitability that features EM, hence an interictal lack of normal habituation. Taken together, the present findings are physiologically in agreement with the shattered steady-state ictal activation and regaining of the dynamic modulation of central excitability after CM remission.
Of note, the reduction of P100m amplitude after CM remission did not approximate the low level as shown in the interictal EM patients in our earlier study (35.6 ± 2.9 nAm in grand average, 33.5 ± 3.0 nAm at block 1) (9). The degree of potentiation (sub-averaged amplitude changes) in remitted CM was also smaller than in the EM patients in our earlier study (Figure 2). The exact reason for such a lesser extent of excitability change in remitted CM than in EM is unknown. Despite the possibility of topiramate effect, as discussed in the next section, a straightforward explanation is that remission was still in its early stage and these ‘remitted’ patients still had high frequency EM (monthly headache days 8.7 ± 2.3 vs 6.5 ± 5.2 in the EM patients in our earlier study) (9).
Studies using positron emission tomography (PET) have revealed ictal-like neural dysfunction in interictal CM because the brainstem activation shown in these studies was also demonstrated during acute migraine (20–23). It was intriguing that brainstem activation persisted when head pain associated with CM was completely suppressed by the short-term use of suboccipital neuromodulation (22). Therefore, ictal-like central activation is independent of headache severity and may linger during transient (or early) remission of CM. The difference in excitability measures between remitted CM (here) and EM (9) might be explained by lingering ictal activity, given the featured excitability change during ictal periods. Nevertheless, the inference needs confirmation by multimodal neuroimaging (or neurophysiological) studies after long-term remission and a significant improvement in headache frequency, and pertinent findings may further explain why the ictal-like MEG activity, as shown in our earlier CM study (9), is independent of the presence or absence or the intensity of headache at the time of recording (11).
The effect of topiramate
Another possibility for the present excitability change is the effect of topiramate. Indeed, central excitability in migraine may be altered by topiramate (24,25). However, the pharmacological effect, or conversely, the withdrawal effect of topiramate as the culprit of the complex excitability changes reported here is not likely. The reasons are threefold. First, the excitability-modulating effect of topiramate seems to be dose-dependent (26,27). A study in patients with chronic neuropathic pain showed that facilitation of muscle-evoked potentials (MEPs) by repetitive TMS can be blocked by topiramate treatment at 75 and 100 mg/day but not at lower doses (25 and 50 mg/day) (27). Of note, the use of topiramate in the present study was tapered then discontinued at least 3 days before MEG follow-up. The majority (nine out of 10) of the CM patients had a washout period at least 5 days before MEG follow-up (except for a 3-day washout in patient G), and the overall washout period averaged 8.9 ± 6.1 days, in contrast with the estimated 19–23 h half-life of topiramate (28). Taken together, the pharmacological effect of topiramate at MEG follow-up, if any, might be negligible in terms of pharmacokinetics, although the threshold level at which topiramate alters visual cortex excitability is undetermined.
Second, the present findings of complex excitability changes seem inexplicable by the inhibitory effect of topiramate. To date, the exact mechanism of topiramate in preventing migraine remains elusive; however, studies on excitability modulation (6) showed that topiramate down-regulates cerebral excitability via reduced central excitation or enhanced central inhibition (19,24–27,29). Although reduced P100m amplitude in CM remission is compatible with down-regulated excitability, the finding of increased sub-averaged amplitude changes seems contradictory. Given the earlier finding that topiramate reversed dishabituation of contingent negative variation in migraine (24), the sub-averaged amplitude changes should be decreased by topiramate. Nevertheless, it is intriguing to note that the inhibitory effect of topiramate, if present after washout, may be an alternative explanation for a lesser extent of excitability change in remitted CM compared to our earlier findings in EM (Figure 2).
Finally, in support of the above assumptions against topiramate effect, our unpublished data did not show any difference in P100m parameters between subjects with 7 (
Lack of a clinical predictor of excitability changes
It is not unexpected that this study failed to find any clinical predictor of the longitudinal excitability change, given the lack of any cross-sectional correlation between MEG results and clinical parameters (9). A TMS study using the MSPA technique also did not yield a clinical/physiological correlation in CM patients treated with topiramate (19). Nevertheless, reduction of headache frequency correlated inversely with an increase of phosphene thresholds in another TMS study treating EM patients with topiramate (29). Besides different methodologies, the complex pathophysiology of CM may thus be the culprit here for the lack of a clinical/physiological correlation.
Limitations of the study
This study was constrained by a small sample size because the MEG follow-up was dependent on topiramate responsiveness. Since this study focused on the changes in visual cortex excitability associated with CM remission, rather than the different excitability patterns between treatment responders and non-responders, we did not follow up MEG in those CM patients who failed to remit to EM. Of note, these ‘intractable’ CM patients might provide further information deciphering potential impacts on MEG measurement resulting from topiramate or migraine pain. Without the comparison with these treatment non-responders, the present finding based on treatment responders might not confirm visual cortex excitability as a faithful biomarker of CM evolution/remission.
However, there were ethical concerns of headache rebound and treatment delay with the discontinuation and washout of topiramate in these patients, for the sake of an interictal MEG follow-up. Moreover, generalization of the present data is justified by comparable demographics, headache profiles and MEG measurements between the CM patients with (
Conclusion
Visual cortex excitability is dynamically modulated in remission from CM to EM. The neuroplasticity reflects the clinical status of migraine and suggests a pathophysiological link between CM and visual cortex excitability. Further studies are needed to investigate if visual cortex excitability can be a faithful biomarker of CM evolution/remission, which would greatly contribute to a timely and precise medical control of this disabling disease.
Footnotes
Funding
This study was supported by the National Science Council of Taiwan [NSC 94-2314-B-038-014 and NSC-96-2314-B-075-073-MY3 to WT, NSC-98-2321-B-010-007, NSC-99-2321-B-010-004 and NSC-100-2321-B-010-004 to YY, NSC-95-2314-B-010-031-MY3NSC and NSC 99-2911-I-008-100 (NSC support for the Center for Dynamical Biomarkers and Translational Medicine, National Central University, Taiwan) to SJ]; Taipei Veterans General Hospital (V98B2-004 to WT, V99C1-063 to SJ, V97ER3-006, VGHUST97-P6-24, V97C1-034, VGH ER3-98-002, VGH-S4-98-018, VGH-C1-99-156 and VGH-ER3-99-006 to YY); Brain Research Center, National Yang-Ming University; and the Ministry of Education, Taipei, Taiwan (Aim for the Top University Plan to SJ).
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.