Abstract
Background
Brain excitability is changed in migraine but not fully characterized yet. This study explored if somatosensory gating is altered in migraine and linked to migraine chronification.
Methods
Paired electrical stimuli were delivered to the left index fingers of 21 patients with migraine without aura (MO), 22 patients with chronic migraine (CM), and 36 controls. The first and second responses to the paired stimuli were obtained from the contralateral primary (cSI), contralateral secondary (cSII) and ipsilateral secondary (iSII) somatosensory cortices to compute the gating ratios (second vs. first response strengths).
Results
The first and second cSI responses and gating ratios differed in all groups (p < 0.05); the responses were typically smaller in the MO and CM groups. The cSI gating ratio increased as a continuum across controls (0.73 ± 0.04, p < 0.001), MO (0.83 ± 0.04) to CM (0.97 ± 0.06) and was higher in CM vs. controls (p < 0.001). When MO and CM were combined, cSI gating ratio was associated with headache frequency (r = 0.418, p = 0.005). Paired responses and gating ratios of cSII and iSII did not differ among the groups.
Conclusions
Somatosensory gating is altered in migraine and associated with headache chronification. Further studies must clarify if this abnormal sensory modulation is a true gating deficit independent of low preexcitation level.
Keywords
Introduction
Migraine is a brain excitability disorder (1) characterized by habituation deficits to repetitive afferent stimulations, as reported in numerous visual, auditory and somatosensory evoked potential studies (1,2). Specifically, the response amplitude tends to increase across blocks of stimuli lasting minutes during interictal periods of migraine, in contrast to the response attenuation (i.e. habituation) in controls. In migraine pathophysiology, the habituation deficits have been interpreted as a disability to protect the brain from excess stimuli or sensory overload, which elicits metabolic shifts and trigemino-vascular activation, thus leading to migraine attacks (1,2). Intriguingly, the habituation deficits have been reported to normalize during ictal periods of migraine, or when migraine progresses from episodic migraine (<15 headache days/month) to disabling chronic migraine (CM, ≥ 15 headache days/month) (2,3). Furthermore, when CM resolved to episodic migraine, habituation correspondingly switched from a normalized to a reduced pattern (4).
As a feature of brain excitability changes, the habituation phenomenon in migraine has not yet been well characterized. Most habituation studies in migraine have typically used serial blocks of repetitive stimulation lasting minutes. However, the measurement of such “long-term” habituation depends on patients’ attention and technical factors (stimulus number, intensity, frequency etc.) (2,5), which hinder the comparison and interpretation across studies. Moreover, the complex neural mechanism underlying habituation deficits in migraine remains elusive and involves at least low pre-activation excitability (2,6), cortical hyperresponsivity (5), serotonergic dysfunction (7), and abnormal mitochondria energy metabolism (8). Therefore, the clinical relevance of habituation deficits has not been established in migraine or migraine chronification. Some studies have not confirmed habituation deficits in migraine (9). Other central nervous system disorders, such as Parkinson’s disease or tinnitus, may present with this phenomenon (10).
Sensory gating is a habituation-related but more basic protective mechanism against sensory overload of the brain (11). Operationally, sensory gating can be assessed by paired-pulse suppression and is thus considered as “short-term” habituation (12). In response to two identical stimuli in close succession (paired-pulse stimuli), the first stimulus is proposed to activate not only excitatory inputs that cause the first neuronal response but also inhibitory interneuronal pathways that modulate neuronal activity on the following stimulus, hence suppressing the second response (paired-pulse suppression) (11). For measuring brain excitability, sensory gating may be more precise and reproducible than long-term habituation because gating is less dependent on stimulus properties and attentional demands (11,13). In episodic migraine, a few studies have reported gating deficits in visual (14) and auditory (12,15) modalities. The association of migraine with somatosensory gating deficits remains unclear, although a study on somatosensory evoked potential in pediatric migraine reported a shortened recovery cycle, a measure related to somatosensory gating (16). Furthermore, the clinical relevance of gating deficits in migraine and migraine chronification remains undetermined.
The present study used magnetoencephalography (MEG) to explore the somatosensory gating in patients with episodic migraine and CM. MEG is superior to scalp electroencephalography in localization and measurement of cortical activities (17). In our previous studies, MEG has been verified to delineate habituation changes in migraine (3,4) as well as the neural correlates of somatosensory gating in healthy individuals (18). It is hypothesized that somatosensory gating is impaired in migraine, and that abnormal sensory modulation is associated with migraine chronification.
Methods
Subjects
Patients with episodic migraine (migraine without aura; MO) and CM were enrolled from the headache clinic of Taipei Veterans General Hospital. The International Classification of Headache Disorders, third edition, beta version (ICHD-III beta) was used to diagnose MO (code 1.1) and CM (code 1.3) (19). Patients fulfilling the criteria of medication overuse for headache (code 8.2) were excluded. Moreover, patients who used migraine prophylactic drugs, hormones or other medications on a regular daily basis were also excluded. Healthy volunteers who had neither a past or family history of migraine nor any headache attack during the past year were enrolled as controls. All participants were right-handed, had no history of systemic or major neurological diseases, and yielded normal results on physical and neurological examinations. The hospital’s Institutional Review Board approved the study protocol, and each participant provided written informed consent.
Study design
At the first visit, all patients completed a semi-structured questionnaire on demographics and headache profiles. The Migraine Disability Assessment (MIDAS) Questionnaire was used to assess migraine-related disability (20). Anxiety and depression were evaluated with the Hospital Anxiety and Depression Scale (HADS) (21). The patients also maintained a headache diary after recruitment.
Each subject underwent a scheduled MEG recording. For patients with migraine, the recording was conducted during the interictal period, arbitrarily defined as the absence of an acute migraine attack within two days before (days −1 and −2) and after (days +1 and +2) the MEG recordings (day 0). The presence of background or interval headache during this period was allowed for CM patients. The MEG recording was re-scheduled in the case of an acute migraine attack during this period or the use of analgesics, triptans, or ergots for any reason within the 48 hours before the recordings. The temporal relationship between MEG recordings and migraine attacks was determined from the headache diary and by follow-up phone calls.
To elucidate the impact of acute migraine upon somatosensory gating, we additionally enrolled five patients with MO to obtain their MEG recordings both during an acute migraine attack and during an interictal period.
MEG recordings
A whole-scalp 306-channel neuromagnetometer (VectorviewTM, Elekta Neuromag, Helsinki, Finland), composed of 102 identical triple sensor elements, was used to record brain activities in all subjects. Four coils representing the head position were placed on the subject’s scalp, with their positions in the head coordinate frame specified by the nasion, and two pre-auricular points were measured with a 3D digitizer using Cartesian coordinates. These landmarks allowed the further registration of the MEG and magnetic resonance imaging (MRI) coordinate systems. Two electrodes were attached above and below one eye to detect the electrooculography (EOG) activities. During the recordings, the subjects sat comfortably with the head supported against the neuromagnetometer helmet.
For somatosensory evoked fields (SEF) recordings, paired electrical stimuli were delivered to the left index finger by using an electrical stimulator (Konstant-Strom Stimulator). The stimuli comprised two 0.2 ms constant-current square-wave pulses, with an inter-stimulus interval (ISI) of 500 ms and an inter-pair interval of 8 s. The stimulus intensity was two times the subjective sensory threshold without eliciting painful perception or visible twitches of the flexor digitorum superficialis muscle. During SEF recordings, the subjects were asked to watch a silent video without focusing on the electrical stimulation. The signal digitization rate was 600 Hz. The length of each recorded epoch was 500 ms, including a pre-stimulus baseline of 100 ms. Epochs contaminated by prominent electrooculogram signals (>300 µV) or MEG artifacts (>3000 fT/cm) were automatically excluded from further analysis. At least 100 artifact-free SEF responses to the first and second pulses of the paired stimuli (subsequently referred as “first response” and “second response”, respectively) were averaged on-line.
MEG data analysis
The distributed source model of the SEF data was estimated using the weighted Minimum Norm Estimates (MNE) to obtain the current density dynamics of cortical sources. A pertinent methodology was reported previously (18), and comprised three main steps. First, a forward model was constructed from the MRI-derived surface model of each participant's brain to describe the signal pattern generated by a unit dipole at each allowed location on the surface (BrainVISA 4.0.2, http://brainvisa.info). The detailed geometric reconstruction of cortical white matter, gray matter, and brain scalp, as well as the tessellations used to estimate the border between gray and white matter, provided a topographical 3D representation of the surface. The anatomical MRI and reconstructed cortical surface were subsequently co-registered with the corresponding MEG dataset.
Second, the inverse operator of MNE analysis was calculated as follows: (a) the source orientations were constrained to be normal to the cortical surface; (b) a depth weighting algorithm was used to compensate for any bias affecting the superficial sources calculation; and (c) a regularization parameter (λ2 = 0.33) was used to minimize numerical instability, reduce the sensitivity of the MNE to noise, and to effectively obtain a spatially smoothed solution. In addition, a noise-normalization procedure, called dynamic Statistical Parametric Mapping (dSPM), was used to identify the cortical sources that exceeded the noise level. Noise covariance of the SEF data was derived from the pre-stimulation baseline period at −100 to 0 ms.
Finally, for group-level analysis, the cortical activation maps overlaid onto the reconstructed surface of each subject were morphed into the common space, namely the cortex of Colin27, by following BrainStorm Tutorials (http://neuroimage.usc.edu/brainstorm/Tutorials). Then the regions of interest (ROIs) on the cortical surface were manually selected from the grand-averaged dSPM for the first stimulus across all subjects. The selection criteria, as published previously (18), were (a) significant values in the dSPM maps; (b) local maximal source activity in the MNE maps; and (c) somatosensory-related cortical areas.
For both first and second responses, the peak amplitudes of cortical sources were extracted from all ROIs, and the amplitude ratio of second vs. first response (second response amplitude/first response amplitude) was computed as the gating ratio for each ROI.
Statistical analysis
Demographics and clinical profiles were assessed for group differences by chi-squared test or analysis of variance (ANOVA). The activation strengths of first and second responses and the gating ratios for all ROIs were compared among the groups by ANOVA. Pearson’s correlation was used to determine the correlation between MEG measures and headache profiles. This correlation was further verified by a multiple regression analysis to test the effects of age, sex, depression, and anxiety. Finally, the impact of ongoing headache upon paired responses was assessed by comparison of the ictal and interictal data in MO (n = 5, Wilcoxon signed rank test). Paired responses in CM patients with and without background headache were also compared by using a two-sample t-test. Bonferroni correction for multiple comparisons was used throughout if appropriate, and a p value < 0.05 was considered statistically significant.
Results
Demographics and clinical profiles
Demographics and clinical profiles.

MO: migraine without aura; CM: chronic migraine; MIDAS: migraine disability assessment; HADS: hospital anxiety and depression scale; *p < 0.05; **p < 0.01
Cortical responses to paired-pulse stimuli
Dynamic cortical activation derived from the first and second responses were grand-averaged across subjects at −20 to 180 ms and mapped onto the coronal MRIs (Figure 1). The activation in the contralateral primary somatosensory cortex (cSI) peaked at ∼50 ms after the first and second pulse of paired stimuli. Subsequently, clear activation was observed in the contralateral (cSII) and ipsilateral (iSII) secondary somatosensory cortices at 90–130 ms and 110–150 ms, respectively. The source strength is color-coded (Figure 1).
Spatiotemporal activation of the averaged Minimum Norm Estimates (MNE) maps in response to paired-pulse electrical stimulation is shown at −20 ms to 180 ms in controls, MO and CM. These maps are shown on the Colin27 surface. The density of the cortical activation is color-coded, with a larger density being denoted by red. 1st, first; 2nd, second.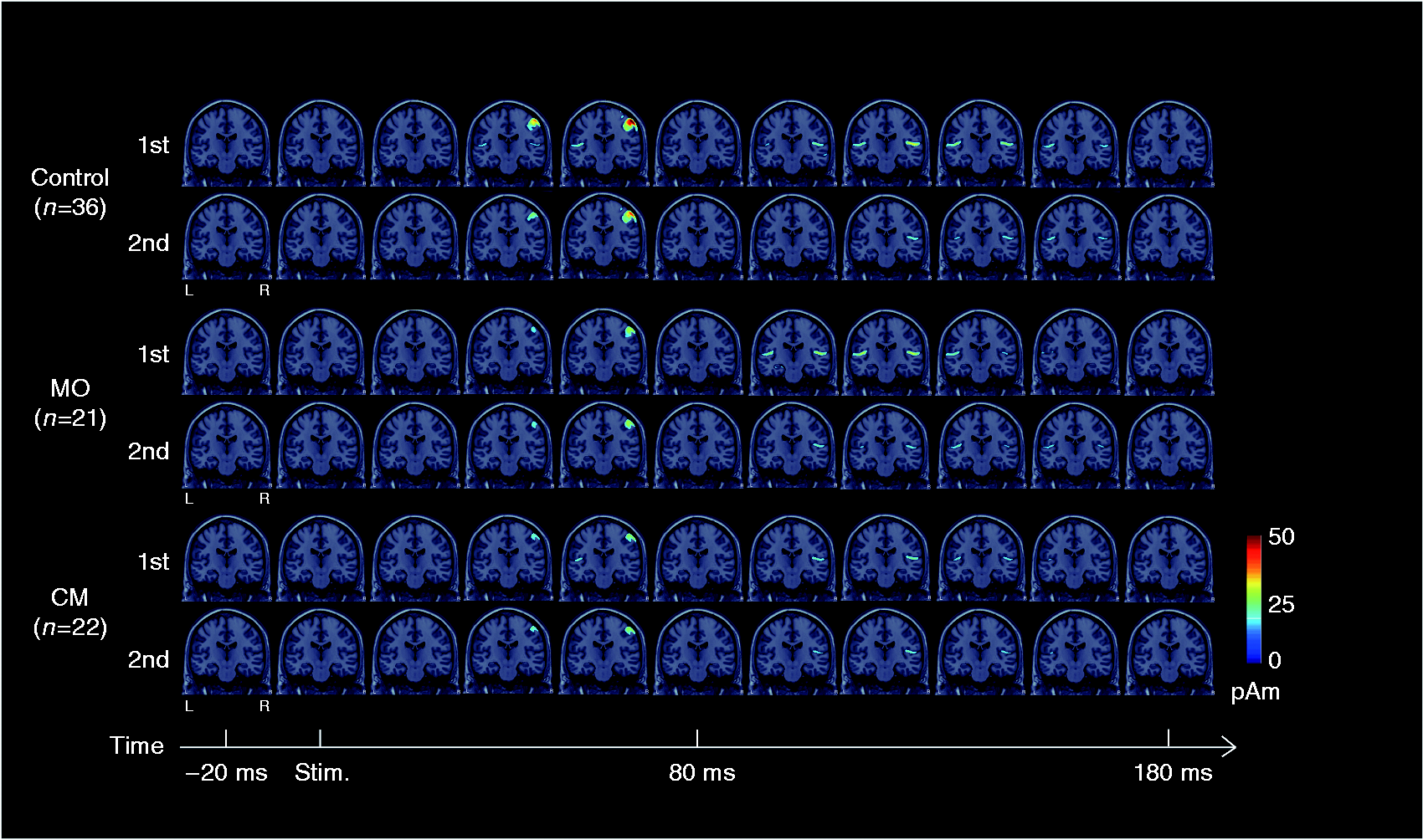
Figure 2(a) shows the grand-averaged dSPMs for the first response. At the time window of −20 to 180 ms, cSI, cSII, and iSII showed significant activations. These regions were manually selected as ROIs for further data analyses (Figure 2(b)).
(a) Averaged dynamic statistical parametric maps (dSPMs) for all study subjects at −20 ms to 180 ms in response to the first stimulus are shown on the flattened cortical surfaces (lateral and top views). The significance level of the cortical maps is color coded. (b) Three regions of interest (ROIs) are selected and mapped onto the MRIs of Colin27, with the center of each region shown in Montreal Neurological Institute (MNI) coordinates. cSI: contralateral primary somatosensory cortex; cSII: contralateral secondary somatosensory cortex; iSII: ipsilateral secondary somatosensory cortex.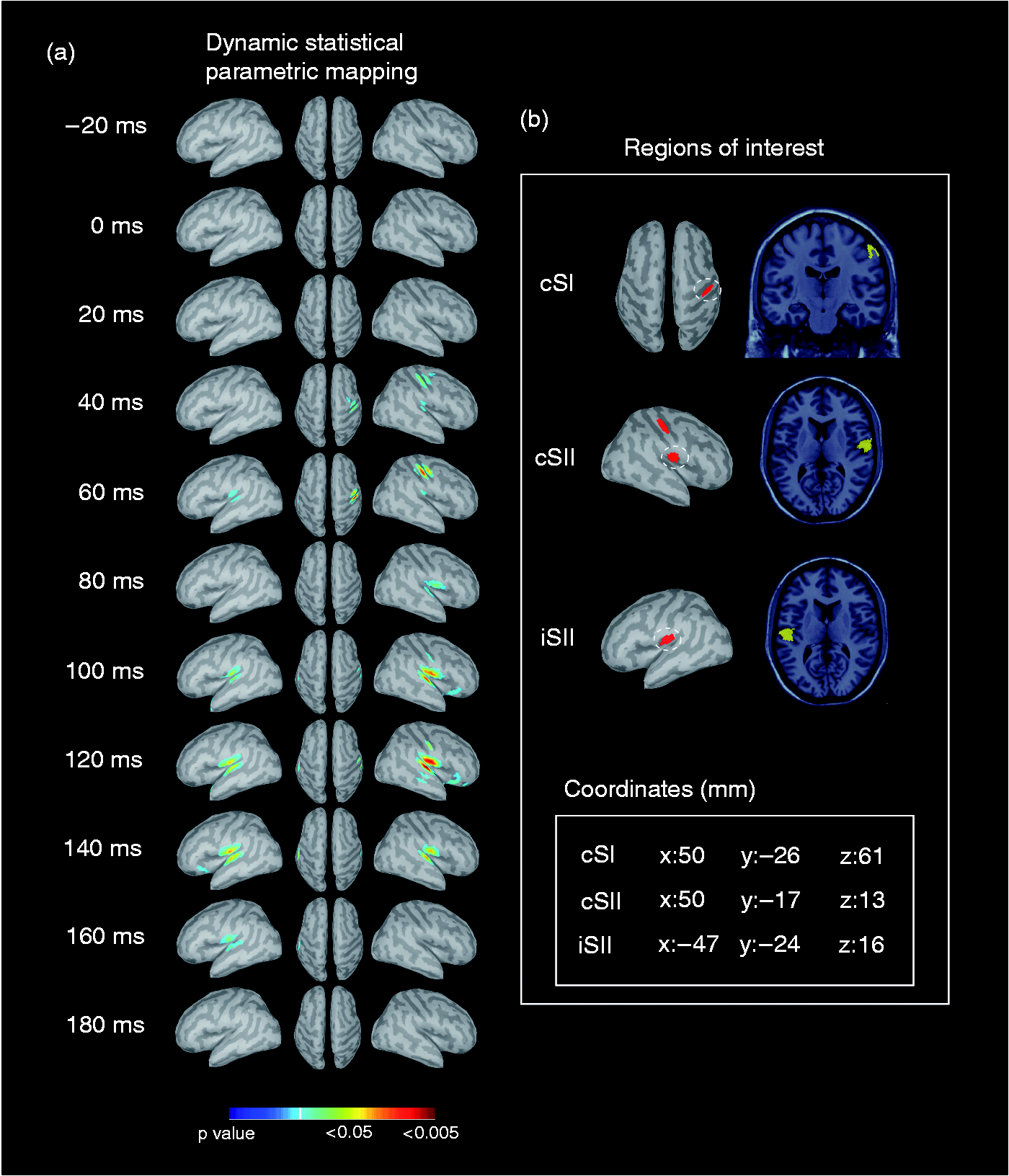
Comparison of activation strength and gating ratio between groups
Based on the dynamics of cortical sources from MNE and dSPM maps, the source strengths in cSI, cSII and iSII were obtained from the peak responses at ∼50 ms, 90–130 ms and 110–150 ms, respectively.
In cSI, the first response strength differed among the groups (F(2,76) = 9.67, p < 0.001] and was smaller in MO (p < 0.001) and CM (p = 0.007) compared to controls. The second response strength also differed among the groups (F(2,76) = 4.93, p = 0.010), and was smaller in MO compared to controls (p = 0.010; Figure 3(a)). The gating ratio increased as a continuum across controls (0.73 ± 0.04, p < 0.001), MO (0.83 ± 0.04) to CM (0.97 ± 0.06). Group differences were observed (F(2,76) = 7.80, p = 0.001), and the cSI gating ratio was higher in CM than in controls (p < 0.001; Figure 3(a)).
The source amplitudes of first and second responses and gating ratios in the regions of interest (ROIs): (a) cSI, (b) cSII and (c) iSII regions. *p < 0.05; **p < 0.01; ***p < 0.001.
In cSII and iSII, the first and second responses did not differ in source strength among the study groups (all p > 0.05; Figure 3(b) and (c)). The gating ratios were all ∼0.8 and comparable across groups.
Clinical correlation
When MO and CM were combined, the cSI gating ratio was positively correlated with headache frequency (r = 0.418, p = 0.005; Figure 4) but not with other clinical parameters (disease duration, headache intensity, MIDAS, HADS-A, HADS-D and total HADS scores; all p > 0.05). The cSII and iSII gating ratios did not correlate with any clinical parameter (all p > 0.05).
The cSI gating ratio is correlated with headache frequency in all patients with migraine.
In multiple regression analysis, the headache frequency in all patients was significantly predicted by cSI gating ratio, age, sex, HADS-A and HADS-D scores (r = 0.518. p = 0.002). Among these factors, cSI gating ratio (β = 0.352, p = 0.015) and depression (β = 0.314, p = 0.028) were independently correlated with headache frequency.
The impact of ongoing headache upon cSI paired responses
In patients MO (n = 5), the cSI first response strength, second response strength, and gating ratio of the ictal recordings tended to be larger in ictal than in interictal recordings, but the differences were not significant (all p > 0.05, Supplementary Figure 1).
On the other hand, CM patients with (n = 10) vs. without (n = 12) background headache also showed tendencies towards a larger first response, a smaller second response, and a smaller gating ratio in cSI without statistical significance (all p > 0.05, Supplementary Figure 1).
Discussion
The present study yielded three main findings. First, patients with MO and CM typically showed reduced cSI responses to paired-pulse stimuli. Second, the somatosensory gating as assessed by paired-pulse suppression presented with a continuum of increased cSI gating ratio across controls, MO and CM, with a significant increase in CM compared to controls. Third, the cSI gating ratio was positively and independently correlated with headache frequency in all patients with migraine. Notably, none of the measurements in cSII and iSII showed group difference or clinical correlation.
cSI gating deficit and clinical relevance
The continuum of increased cSI gating ratio across controls, MO to CM implied a role of gating deficit in migraine chronification. This presumption was further verified by the correlation between cSI gating and headache frequency. In line with this, a previous study using the technique of magnetic suppression of perceptual accuracy (MSPA) also demonstrated a continuum of impaired visual suppression across controls, MO and CM, with CM showing the most deficient suppression (22). According to these findings, MO and CM may be different migraine phenotypes that differ in degree, rather than type.To date, the reason why EM may evolve into disabling CM (23) remains elusive. Studies on the neural substrates of sensory gating have revealed a complex interneuronal inhibition among the primary sensory cortices, thalamus, prefrontal cortex, hippocampus and rhinal cortex (13,18,24). Thus, the gating deficit observed in this study may reflect impaired sensory modulation, or central disinhibition, in a distributed neural network involving intra-cortical, cortico-cortical and subcortico-cortical pathways. Notably, MRI studies in CM echoed this presumption by showing reduced gray matter volume or related structural changes in the above-mentioned gating-related neural structures (25). Clinically, medication overuse is the most important risk factor for migraine chronification (23). Patients with CM and medication overuse also showed sensitization and lack of habituation of somatosensory evoked potentials, highlighting the role of central disinhibition in migraine chronification (2). Depression, another established risk factor for migraine chronification (23), was independently associated with headache frequency in the present study and has also been linked to central disinhibition (26).
Our finding of cSI gating deficits, if interpreted in conjunction with previous data that showed visual (14) and auditory (12,15) gating deficits in MO, suggests gating deficits to be a cross-modal phenomenon in migraine. Notably, an abnormality in cross-modal audio-visual perceptual processing has been recently observed in migraine through sound-induced flash illusions (27). Moreover, visual gating deficits in MO were correlated with migraine frequency (14), in line with the present finding showing correlations between cSI gating and headache frequency. In contrast to the undetermined clinical relevance of habituation deficits in migraine, studies must investigate whether gating deficits, in general or specific modalities, are a potential biomarker for migraine.
Absence of gating deficit in cSII and iSII
In contrast to cSI, we did not observe gating deficits in cSII and iSII in patients with migraine. Notably, it remains controversial whether somatosensory gating in healthy individuals recruits cSII and iSII. Our previous study on paired-pulse suppression (18) and another MEG study using simultaneous stimulations did not reveal the involvement of iSII and cSII in normal somatosensory gating (28). However, two other studies demonstrated an independent engagement of iSII (29) and cSII (30), respectively. Despite the undetermined role of cSII–iSII in sensory gating, the absence of cSII and iSII gating deficits in the present study may imply their minor roles in migraine pathophysiology. Accordingly, few MRI studies in migraine have shown structural or functional changes in the secondary somatosensory cortices. In the information processing of electrical or tactile stimuli, cSII–iSII is functionally ranked higher than cSI because of the serial engagement following cSI and its involvement in tactile learning, object recognition, and sensorimotor integration (29). Therefore, the present finding of normal cSII–iSII gating suggests that somatosensory processing in migraine is impaired at the basic level, in contrast to the higher-order sensory dysfunction in schizophrenia that showed intact cSI gating but impaired cSII–iSII gating (13).
Coexistence of both hyper- and hypo-excitability in migraine
It is somewhat unexpected that the cSI source strength of the first response was lower in both MO and CM vs. controls, given the gating deficits suggesting central disinhibition in these patients. In MO, the first response was reduced after paired-pulse visual stimulation (14). Many evoked potential studies on habituation across modalities also showed a small first-block response in MO (1,2). Taken together, these findings may reflect a reduced cortical preactivation level that has been associated with a functional disconnection of the thalamus from its controlling inputs (e.g. projections from aminergic brainstem nuclei) (2,6). An alternate explanation for this preactivation hypoexcitability is the inhibitory mechanism of the thalamus which compensates for the brain excitability change in migraine (5).
In patients with CM, however, the few available habituation studies have not reported preactivation hypoexcitability. Our earlier MEG studies in visual modality (3) and a recent somatosensory evoked potential study (6) conversely revealed an augmented first-block response that indicated sensitization in CM. This discrepancy may be partly attributed to the methodology of attention control. Despite the preattentive feature of sensory gating, the SEF recordings in this study were conducted during concurrent visual distraction (watching videos), in contrast to the high attentional demands maintained by counting visual cues in our visual evoked field studies (3). It is well known that attention modulates the amplitude of somatosensory activation (31). Moreover, patients with CM may be hypervigilant to sensory stimuli, as suggested by an earlier MEG study showing a rostral shift in the source response to cutaneous laser heat stimulation (32). Therefore, the present methodology may prevent the unwanted attentional modulation inherent to CM, thus yielding preactivation hypoexcitability like MO. The attentional modulation upon evoked brain responses remains uninvestigated in CM across sensory modalities.
The cSI gating deficit observed in the present study was possibly caused by the small first response (preactivation hypoexcitability) rather than the lack of second response suppression (true gating deficit). Technically, it may be impossible to delineate the contribution of both factors to sensory gating. However, our findings are against this assumption, because the first response strength and gating ratio in the cSI tended to be higher in CM than in MO. Moreover, the MO patients with smaller first responses (≤median, averaged 16.7 ± 2.37) and those with stronger first responses (>median, averaged 41.6 ± 2.02) had comparable gating ratios (0.83 vs. 0.82, p = 0.705, Mann-Whitney test). Finally, the paired-pulse responses featured by both hypo- (low preactivation level) and hyper-excitability (increased gating ratio) in this study echoes the previous proposal that migraine is best understood as a disorder of brain “dysexcitability” rather than hyperexcitability (5).
Ictal-interictal fluctuation of brain excitability in migraine
Earlier studies in patients with MO have shown a reduced first-block response and habituation deficit during interictal periods. These changes tended to normalize during ictal periods, suggesting a dynamic change of brain excitability across ictal-interictal periods of migraine (2,3). In the present study, a comparison of the interictal and ictal recordings in MO (n = 5) did show a larger strength of the first cSI response in the ictal recordings (Supplementary Figure 1), in agreement with the trend of ictal normalization. However, the increased gating ratio during ictal periods (vs. interictal) seemed against this trend, and may imply different mechanisms underlying habituation and sensory gatingOf note, when MO evolves into CM, the dynamic fluctuation of brain excitability is lost and the steady-state ictal-like excitability pattern suggests CM as an unresolved or never-ending migraine attack (2,3). In line with this, a recent study applying paired somatosensory stimuli to hands and face in patients with MO yielded an ictal prolongation of somatosensory temporal discrimination that returned to normal interictally (33). In patients with CM, the prolonged temporal discrimination persisted across the interictal-ictal periods and was proposed as a biomarker for CM (34). It may be of concern that the increased gating ratio noted only in patients with CM may result from an unresolved migraine attack rather than a link to headache frequency. However, an unresolved headache and increased headache frequency are difficult to differentiate and both may reflect the phenotype of migraine chronification. Since CM patients with background headache (n = 10) tended to have a lower gating ratio compared to those without background headache (n = 12, Supplementary Figure 1), an unresolved migraine seems less likely to cause increased gating ratio in CM.
Study limitations
Headache lateralization may bias our finding because we only stimulated the non-dominant hand. Although no patients in this study reported a strict lateralization of their headaches, additional studies stimulating both non-dominant and dominant hands in patients with lateralized headaches are mandatory to address the above-mentioned concern.
Although the 500 ms ISI of the paired stimuli has been presumed to be optimal and typically used in most sensory gating studies (12,13,18,24), a systemic exploration is required to determine whether other ISIs yield the same findings.
The present study was unable to investigate the interhemispheric inhibition between cSI and ipsilateral primary somatosensory cortex (iSI) since iSI did not show clear and consistent activation to paired-pulse stimulation (Supplementary Figure 2). Our previous study stimulating the median nerve with the same paired-pulse paradigm did show iSI responses. The present study did not show discernible iSI activation, probably because we stimulated the left index instead of the median nerve to ensure the reproducibility of electrode localization in later follow-up studies.
Limited by the study design, the present study failed to elucidate the causal relationship between sensory gating and migraine chronification. A longitudinal study would be particularly valuable to investigate whether sensory gating switches with the clinical phenotype when CM resolves to MO. Moreover, multi-modal sensory gating studies in the same group of patients may clarify whether the excitability change is truly cross-modal and further differentiate its association with migraine chronification.
Conclusions
Sensory gating of the primary somatosensory cortex is altered in patients with migraine. The excitability changes reflect an altered sensory modulation, and are associated with migraine chronification. The potential of gating deficits as a biomarker for migraine must be investigated in future.
Footnotes
Article highlights
Sensory gating of the primary somatosensory cortex is altered in chronic migraine.
The gating ratio of the primary somatosensory cortex is associated with headache frequency in migraine.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FJ Hsiao, YY Lin, HY Liu, YC Ko, PN Wang and WT Chen report no disclosures. SJ Wang has served on the advisory boards of Allergan and Eli Lilly Taiwan. He has received honoraria as a moderator from local companies (Taiwan branches) of Pfizer, Eli Lilly and Esai. JL Fuh is a member of the scientific advisory board of Novartis, and has received research support from the Taiwan National Science Council and Taipei Veterans General Hospital.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Ministry of Science and Technology (MOST 103-2628-B-075-001-MY3 and 104-2745-B-010-003 to WT Chen; 103-2221-E-010-014 and 105-2221-E-010-005 to FJ Hsiao; and 103-2321-B-010-017, 102-2321-B-010-030, 100-2314-B-010-018-MY3, and 99-2314-B-075-036-MY3 to SJ Wang), Taipei Veterans General Hospital (V104C-115, V105C-092, V105E9-005-MY2-1, and VGHUST105-G7-1-2 to WT Chen, and VGHUST105-G7-1-1, V105C-127, V105E9-001-MY2-1, and VTA105-V1-1-1 to SJ Wang), NSC support for the Center for Dynamical Biomarkers and Translational Medicine, National Central University, Taiwan (NSC 101-2911-I-008-001 and 102-2911-I-008-001 to SJ Wang), Brain Research Center, National Yang-Ming University, a grant from the Ministry of Education, Aim for the Top University Plan, and a grant from the Ministry of Health and Welfare (MOHW104-TDU-B-211-113-003).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
