Abstract
Nitric oxide (NO) donors, which cause delayed headaches in migraineurs, have been shown to activate central trigeminal neurons with meningeal afferent input in animal experiments. Previous reports indicate that this response may be due to up-regulation of NO-producing cells in the trigeminal brainstem. To investigate this phenomenon further, we determined nitric oxide synthase (NOS)-containing neurons in the rat spinal trigeminal nucleus (STN), the projection site of nociceptive trigeminal afferents, following infusion of the NO donor sodium nitroprusside (SNP). Barbiturate anaesthetized rats were infused intravenously with SNP (50 μg/kg) or vehicle for 20 min or 2 h, and after periods of 3–8 h fixed by perfusion. Cryostat sections of the medulla oblongata containing the caudal STN were histochemically processed for detection of nicotineamide adenine dinucleotide phosphate (NADPH)-diaphorase or immunohistochemically stained for NOS isoforms and examined by light and fluorescence microscopy. The number of neurons positive for these markers was determined. Various forms of neurons positive for NADPH-diaphorase or immunoreactive to neuronal NOS (nNOS) were found in superficial and deep laminae of the STN caudalis and around the central canal. Neurons were not immunopositive for endothelial (eNOS) or inducible (iNOS) NOS isoforms. The number of NADPH-diaphorase-positive neurons increased time dependently after SNP infusion by a factor of more than two. Likewise, the number of nNOS-immunopositive neurons was increased after SNP compared with vehicle infusion. Around the central canal the number of NADPH-diaphorase-positive neurons was slightly increased and the number of nNOS+ neurons not changed after SNP treatment. NO donors increase the number of neurons that produce NO in the STN, possibly by induction of nNOS expression. Increased NO production may facilitate neurotransmitter release and promote nociceptive transmission in the STN. This mechanism may explain the delayed increase in neuronal activity and headache after infusion of NO donors.
Introduction
The vasoactive molecule nitric oxide (NO), a short-living free radical that is synthesized under the control of different subtypes of nitric oxide synthases (1, 2), is assumed to be implicated in the pathogenesis of primary headaches (3, 4). NO donors such as glyceryl trinitrate (GTN, nitroglycerin) have long been used to provoke episodes of headaches under experimental conditions (5–7), but the underlying mechanisms are still obscure (8–10). Headaches during or immediately following intravenous (i.v.) application of GTN are normally described as mild to moderate by healthy volunteers, whereas patients suffering from migraine or tension-type headache report more severe immediate pain (9, 11). In addition, patients with a history of migraine or tension-type headache experience a second episode of headache up to several hours later, which fulfil the criteria of migraine or resemble the patients' specific spontaneous headaches (11, 12). These delayed headache episodes cannot be caused by a direct nociceptive effect of NO, because NO is an unstable free radical that is released and rapidly inactivated by reacting with other molecules (2) long before the headache arises. Therefore, it was concluded that patients suffering from primary headaches such as migraine are hypersensitive to NO, which by a yet unknown mechanism can trigger the development of headaches (4). Increased levels of NO metabolites have been found in the venous blood plasma of patients suffering from migraine and other primary headaches both during and, to a lesser extent, between headache episodes (13–16). In conclusion, endogenous NO production may cause hypersensitivity of the trigeminovascular system and trigger headache attacks in a feed-forward manner (9).
In a rat model of meningeal nociception our group has previously shown that i.v. infusion of the NO donor sodium nitroprusside (SNP) increases the activity of neurons with meningeal afferent input in the caudal subnucleus of the spinal trigeminal nucleus (STN), the relay nucleus for nociceptive afferents innervating the head, including intracranial structures (17). At least part of this increase in activity seems to be mediated by a central site, as a recent study has reported facilitating effects on neurotransmission of a NO donor applied iontophoretically to STN neurons with meningeal afferent input (18). The pattern of activation in our previous experiments (17) was reminiscent of the two episodes described for NO-induced headaches in patients (8). A short-lasting increase in activity during the SNP infusion was followed by a second delayed phase of higher and long-lasting activity about 1 h later. This second phase in activity may be explained by delayed endogenous up-regulation in excitability of the central neurons, the time course of which fits that of an altered gene expression. Therefore, we looked for histochemical changes that may reflect the development of delayed hyperexcitability after SNP infusion in the STN region where we had previously found increased neuronal activity.
Based on this concept, we examined if the same dose of SNP that had triggered neuronal activity in the STN (17) may cause changes indicative of an increased level of endogenous NO production. As an indicator of NO production we used an established histochemical method for localization of nicotineamide adenine dinucleotide phosphate (NADPH)-diaphorase in neurons. Neuronal NADPH-diaphorase has been characterized biochemically as a membrane-bound nitric oxide synthase (NOS) of neurons (19, 20). Further studies have shown that NADPH-diaphorase is equivalent to the reductase domain of the NOS, which converts NADPH to NADP+, while at the oxygenase domain NO is synthesized from L-arginine (1, 21, 22). NADPH-diaphorase histochemistry using the conversion of tetrazolium to formazan (19) enables reliable counts of NOS-expressing neurons, but one should be aware of the limits of this method. Although NADPH-diaphorase is regarded as a marker for constitutive NOS of neurons (19), there is uncertainty whether it recognizes all neuronal NOS (nNOS)-producing neurons (23). Conversely, not all neurons positive for NADPH-diaphorase may contain NOS (24). Low levels of the endothelial isoform of NOS (eNOS) immunoreactivity have also been found in medullary neurons (25), and inducible NOS (iNOS) can possibly be expressed in central neurons under pathological conditions such as ischaemia or injury (26–28). Finally, NADPH-diaphorase activity is dependent on the fixation of the tissue (29). During fixation with paraformaldehyde, NADPH-dependent oxidative enzymes are inactivated except for NOS in the cytosol (30), resulting in selective staining of cells that contain NOS. For all these reasons a combination of NADPH-diaphorase histochemistry with immunohistochemistry is of advantage to assess numbers of neurons that express isoforms of NOS.
This study examines the hypothesis that increased neuronal activity in the STN following infusion of a NO donor is paralleled by an increased number in NO-producing neurons, which can be detected by NADPH-diaphorase activity combined with immunohistochemistry to specify NOS isoforms. Increased numbers of NO-producing neurons are likely to increase NO production. NO in the central nervous system is regarded as a retrograde transmitter that facilitates neurotransmitter and neuropeptide release from presynaptic terminals, thereby strengthening synaptic transmission and contributing to central sensitization (18, 22).
Material and methods
General procedures
The study was performed in accordance with the ethical guidelines of the International Association for the Study of Pain and the European Communities Council for the treatment and use of laboratory animals. The experimental procedure was reviewed by an ethics committee and approved by the local district government. Thirty-two adult male Wistar rats (300–460 g) were used. The animals were anaesthetized by intraperitoneal (i.p.) injection of 120–150 mg/kg thiopental (Trapanal® Altana, Konstanz, Germany); the absence of responses to noxious pinching of the earlobes was indicative of deep anaesthesia. The animals' core temperature was maintained constant at 37–37.5°C with a feedback-controlled homoeothermic system (TKM 0902; FMI, Seeheim, Germany). The femoral artery was cannulated for blood pressure monitoring and a catheter in the femoral vein was used for i.v. administration of solutions. The animals were tracheotomized to insert a tracheal tube. For NADPH-diaphorase experiments, spontaneous respiration was supported by a light jet of oxygen blown at the opening of the tube (31). For NOS experiments animals were ventilated with oxygen-enriched room air, and end-expiratory CO2 was held < 5% to suppress spontaneous respiration (17). Blood pressure and body temperature where recorded over the whole experiment, and anaesthesia was supplemented with thiopental (15 mg/kg) if required.
Specific experimental procedures
For NADPH-diaphorase experiments, animals received a 20-min continuous i.v. infusion of either 1-ml isotonic saline (vehicle control group) or the same volume of a SNP solution (50 μg/kg, dissolved in saline). Vehicle animals (n = 5) were sacrificed 6 h after start of the infusion, SNP-treated animals (each group n = 5) after 3, 6 or 8 h. For NOS immunofluorescence experiments, animals were infused intravenously over 4 h with saline (0.5 ml/h, vehicle group, n = 5) or with 1-ml SNP solution (50 μg/kg in saline) over 2 h followed by saline (0.5 ml/h) for 2 h (SNP group, n = 5). Anaesthesia was deepened by injection of 20–50 μg/kg thiopental before the rats were quickly thoracotomized and transcardially perfused with 100–150 ml saline at room temperature followed by 200–300 ml of 4% paraformaldehyde in 0.1-
After opening of the occipital skull and cervical laminectomy, the fixed brainstem with the medulla including the cerebellum and the first two cervical segments was dissected and placed in PBS with 15% sucrose at 4°C overnight. For immunostaining the brainstem was postfixed in paraformaldehyde for several hours before it was incubated in sucrose for cryoprotection. The tissue was then covered by a stabilizing medium (GSV 1—Tissue Embedding Medium; Splee Technik, Mainz, Germany), quickly frozen in methylbutane at −40°C and stored in the freezer at −20°C. The tissue block was cut between the cervical segments and the obex into cross-sections of 30 μm (for NADPH-diaphorase staining) or 20 μm (for immunostaining) using a cryostat (2800 Frigocut N; Leica, Bensheim, Germany). The sections were mounted on poly-L-lysine-coated slides, air dried and stored at 4°C.
NADPH-diaphorase staining
The unstained sections were viewed under a Leica MS5 binocular microscope (Leica) in order to select the sections in the region of interest at the level of 1.2–1.8 mm caudal to the obex. This level was selected because the rostral part of this segment contains few but the caudal part many more neurons with meningeal afferent input according to previous electrophysiological experiments (32). A strict NADPH-diaphorase staining protocol of 50 min was used to achieve homogeneity and to avoid overstaining. The sections on the selected object slides were encircled by drawing a hydrophobic line (Liquid Blocker; SCI, München, Germany) and then the slides were kept dry in PBS buffer. The freshly prepared staining solution consisted of 0.2 mg/ml nitroblue tetrazolium, 1 mg/ml ß-NADPH and 0.3% Triton X-100 in a 0.1-
Microscopy
Digital images were taken from the stained sections at low magnification as templates for the subsequent quantitative analysis using a Leica DFC 320 digital camera connected to the Leica MS5 binocular microscope (Leica). The sections were examined with a Leica Aristoplan bright field microscope (Leica). Neurons were identified using morphological criteria such as size, dendritic extensions and a visible nucleus, which appears light. Neurons showing the diaphorase reaction product (NADPH-diaphorase-positive neurons) within the left and right spinal trigeminal nucleus and around the central canal were counted at high magnification (100–160×), and their position was marked on the low-power digital images. Only neurons with a visible nucleus were included in the counts to avoid double-counting in subsequent sections. During this procedure the examiner was blinded to the experimental group from which the sections derived. Representative micrographs were taken with a charge coupled device (CCD) camera system (Visitron Systems RT color; Diagnostic Instruments, Sterling Heights, MI, USA) connected to the Leica Aristoplan microscope.
NOS immunostaining
For immunohistochemical processing, slides with the mounted sections cut at a distance of 1.8–2.2 mm caudal to the obex were selected. The STN in this medullary segment contains the highest concentration of neurons with meningeal afferent input according to previous electrophysiological experiments (17, 32, 33). The slides were preincubated with 0.1
Following 4 × 10 min rinses in PBS/NaN3, the sections were placed into a solution of specific secondary antibodies (Cy3-conjugated goat antirabbit IgG; Jackson Immunoresearch, Dianova, Hamburg, Germany; code no. 111-165-144) at a dilution of 1:200 at room temperature in the dark for 1 h. Specificity of the NOS immune reactions was controlled by omitting the respective primary antisera in some sections of each series and by blocking peptides containing the respective NOS isoforms as provided by the manufacturer (Calbiochem; cat. no. 482730). For blocking non-specific binding of the second antibody, 5% normal goat serum was used for incubation in some experiments. Alternative fixation methods (acetone/ethanol, freezing without fixation) were applied in additional experiments that did not reveal any non-specific immunoreactions owing to fixation artefacts (data not shown).
After immunostaining, the sections were covered with glycerin gelatine (Merck), coverslipped and sealed with resin to avoid drying. The slides were kept at 4°C in the dark and examined microscopically within 1 week.
Microscopy and image processing
For epifluorescence microscopy a Leica Aristoplan microscope with the filter set N 2.1 (excitation 515–580 nm, emission 580 nm) at magnifications of 65–400× was used. Cells with visible nuclei were counted within the borders of the STN of both sides in one section of each slide stained with the respective antibody. Counting of immunopositive neurons was performed by an examiner who was blinded for the experimental group. Representative micrographs were taken with a CCD camera system (Visitron Systems RT color; Diagnostic Instruments). Low-power images were used as templates in which the positions of detected neurons were drawn to avoid overcounting.
Confocal micrographs were taken with a BioRad MRC1000 confocal laser scanning microscope (BioRad, Hemel Hempstead, UK) equipped with a three-line krypton-argon laser (American Laser Technology, Salt Lake City, UT, USA) and attached to a Nikon Diaphot 300 microscope (Nikon, Düsseldorf, Germany). For visualization of the Cy3 immunostaining an excitation wavelength of 568 nm (filter 605 DF322) was used. To contrast the unstained background, the excitation was changed to 488 nm (filter 522 DF32). Sections were examined using a 20× objective lens and electronic zoom factors of 1.0–2.0. Images of 768 × 512 pixels were taken and with the background merged into 8-bit tiff files using the confocal assistant software 4.02. Further processing of images was done with Corel Photo Paint (Corel, Dublin, Ireland) to adjust for format, contrast and brightness and to add text and scale bars for printing.
Statistical analysis
The Statistica software package (Tulsa, OK, USA) was used for statistics. Differences between groups were analysed by
Results
NADPH-diaphorase staining
In all cross-sections through the medulla oblongata the caudal STN was clearly visible by its darker background staining and an accumulation of NADPH-diaphorase-positive neuronal structures (Fig. 1a). NADPH-diaphorase-positive neurons were manifold in size (maximal diameter 10.9–30.1 μm) and shape, including oval, pyramidal and spindle-like forms that resembled the cell types described after Golgi staining in the STN of the cat (34, 35). The STN was subdivided into three layers (Fig. 1b) based on the histological classification of Gobel (34, 36), which had been derived from the previous classification of feline spinal dorsal horn laminae of Rexed (37). The superficial layer (laminae I and IIa of Gobel) appeared dark and sharply silhouetted against the trigeminal tract owing to many small, mostly diffuse NADPH-diaphorase-positive structures (Fig. 1a,b). Lamina I contained multiple, mostly small (< 15.5 μm) neuronal NADPH-diaphorase-positive cell bodies that rarely revealed longer processes, because these extend preferably in the rostro-caudal direction (Fig. 1c–e). In lamina IIa medium-sized NADPH-diaphorase-positive somata with processes extending both into lamina I and in deeper laminae resembling the stalked cells of Gobel (36) were observed (Fig. 1e). The next layer beneath (lamina IIb of Gobel) appeared light (Fig. 1b) and contained some small to medium-sized spindle-like NADPH-diaphorase-positive neurons, most likely islet cells (36), with fine arborized dendritic trees (Fig. 1d). The deep layer (lamina III/IV of Gobel) passed into the reticular nucleus (lamina V) without a clear boundary (Fig. 1a–c). It characteristically contained large (> 22.3 μm) NADPH-diaphorase-positive somata with long dendritic processes, which were frequently observed to extend radially through the intermediate layer to the superficial layer (Fig. 1c). These cells may be projection neurons (36).
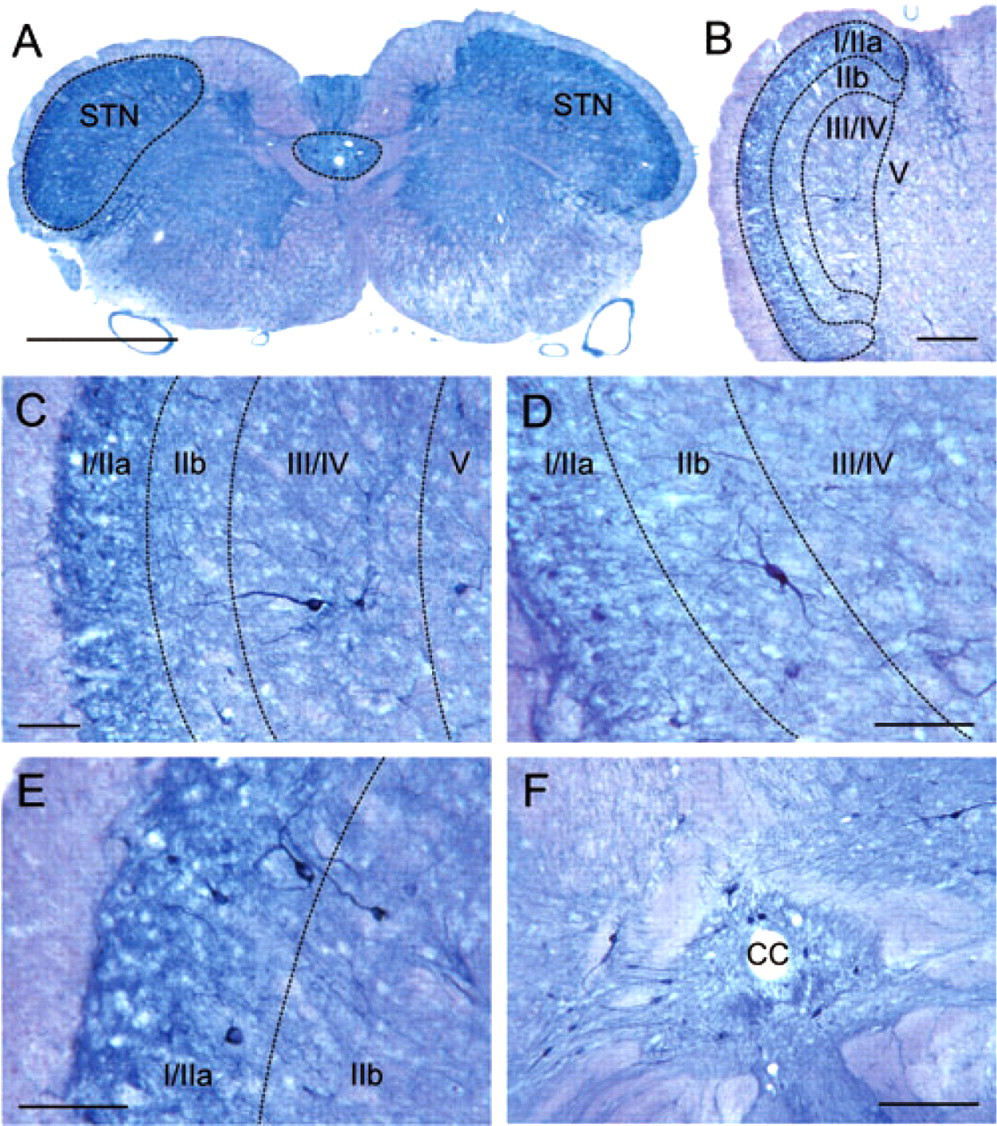
(a) Low-power micrograph of a section through the medulla, 1.65 mm caudal to the obex. The spinal trigeminal nucleus (STN, on left side indicated by dashed line) is clearly visible by the dark blue staining of nicotineamide adenine dinucleotide phosphate (NADPH)-diaphorase-positive neurons and neuronal processes. The stained area around the central canal used for cell counts is also indicated. Size bar: 1 mm. (b) Low-power micrograph of the left STN, 1.5 mm caudal to the obex. Laminae I/IIa, IIb and III/IV as classified by Gobel (36) are indicated by dashed lines. Size bar: 250 μm. (c) Enlarged section of (b). One large NADPH-diaphorase-positive neuron is extending with its dendritic process from lamina III/IV to the superficial laminae. Size bar: 100 μm. (d) Section through laminae I–IV of the STN at a distance of 1.4 mm from the obex. Lamina IIb contains few but a prominent NADPH-diaphorase-positive neuron (resembling an islet cell) with fine dendritic processes. Size bar: 100 μm. (e) Section through the superficial laminae of the STN at a distance of 1.6 mm caudal to the obex. Many small and medium-sized NADPH-diaphorase-positive neurons are visible in lamina I/IIa, some of which (resembling stalked cells) show processes extending in both superficial and deeper laminae. Size bar: 100 μm. (f) Section through the central area (caudal vagal nuclear complex) of the medulla (1.3 mm from the obex) with the central canal (CC) and several NADPH-diaphorase-positive neurons. Size bar: 250 μm.
In 400 serial sections (20 sections per animal) a total of 20.689 NADPH-diaphorase-positive neurons were counted. Most were small to medium in size (max. 15.5 μm in soma diameter) and located in the superficial layer (laminae I/IIa). The largest neurons (max. 30 μm in soma diameter) were located in the deep laminae III/IV. Within the examined brainstem segment of 600 μm (1.2–1.8 mm caudal to the obex) the numbers of NADPH-diaphorase-positive neurons did not vary significantly in rostro-caudal direction either in the control group or in the SNP-treated groups (ANOVA; F
19,60 = 0.10; P = 1.0; see Fig. 2). Therefore all sections within the 600-μm segment were used for comparison of vehicle and SNP-treated groups. The mean numbers of NADPH-diaphorase-positive neurons and their variance (
Number (mean ±

Numbers are specified for the whole spinal trigeminal nucleus (STN), separate laminae of the STN and the caudal vagal nuclear complex (CVNC) around the central canal in a 600-μm segment of the medulla between 1.2 and 1.8 mm caudal to the obex.
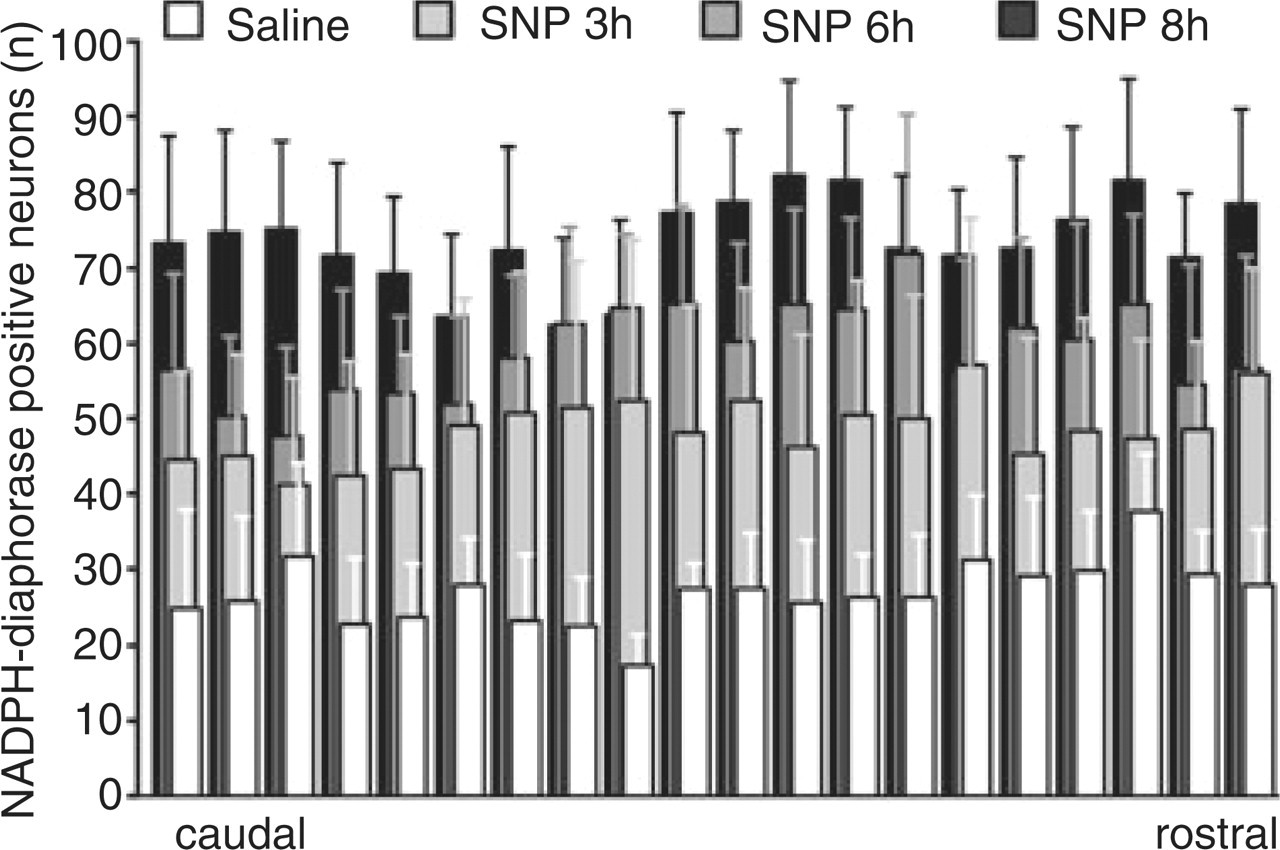
Rostro-caudal distribution of nicotineamide adenine dinucleotide phosphate (NADPH)-diaphorase-positive cells (means ±
Overall, within the STN more NADPH-diaphorase-positive neurons were observed in SNP-treated animals (3, 6 and 8 h after SNP infusion) compared with saline-treated animals (

Micrographs of representative sections through the dorsolateral part of the spinal trigeminal nucleus showing nicotineamide adenine dinucleotide phosphate-diaphorase-positive neurons 6 h after infusion of vehicle (a), 3 h (b), 6 h (c) and 8 h (d) after infusion of sodium nitroprusside. Size bars: 250 μm.

Lamina-specific distribution of nicotineamide adenine dinucleotide phosphate (NADPH)-diaphorase-positive cell counts (means ±
To examine if the effect of SNP infusion on the number of NADPH-diaphorase-positive neurons was specific for trigeminal areas in the medulla, we counted the number of positive cells in an area around the central canal, which comprises caudal portions of the dorsal vagal nuclear complex (38). We observed a time-dependent increase in NADPH-diaphorase-positive neurons after SNP infusion (r = 0.55, P = 0.012, Spearman correlation), which was 141% of the control after 8 h (see Table 1).
NOS immunofluorescence
Immunoreactivity for nNOS was mainly found in cell bodies and dendrites within the STN (Fig. 5a,b,d) and in the area around the central canal (caudal dorsal vagal complex, Fig. 5c). All nNOS+ cellular structures could be identified as neurons with a soma diameter of 10–30 μm. The different shapes of nNOS+ neurons were very similar to the forms found with NADPH-diaphorase staining. The superficial laminae I/II of the STN contained mostly small spindle-like somata, whereas in the deep laminae III/IV large neurons with longer dendritic processes prevailed, some of which extended to the superficial layers. In the area around the central canal mainly large neurons with extended processes were immunostained.

Confocal micrographs of nitric oxide synthase (NOS)-immunoreactive structures (red) and the unspecifically green fluorescent background of the medulla. (a,b) Representative cross-sections through the lateral part of the spinal trigeminal nucleus (STN) showing neuronal (n) NOS-immunoreactive neurons (arrows) and nerve fibres 4 h after infusion of vehicle (a) and sodium nitroprusside (SNP) (b). Small neurons are concentrated in superficial laminae, large neurons are mainly located in deep laminae (see insets at higher magnification). (c) nNOS+ neurons around the central canal (SNP animal). (d) Longitudinal (parasagittal) section through the STN (between lines) showing many nNOS+ neurons and nerve fibres. (e,f) Cross-sections through the dorsolateral segment of the medulla showing inducible (i) NOS (e) and endothelial (e) NOS (f) immunostaining. (e) Glial cells in the spinal trigeminal tract (see inset, arrow) and the outermost laminae show iNOS immunoreactivity, but neurons are not immunostained. (f) Blood vessel endothelium (see inset) and other small structures, particularly in the outer laminae of the STN, show eNOS immunoreactivity, but immunostained neurons are not visible. (g) Staining without primary antibody shows no immunopositive structures. All size bars: 100 μm.
Cellular structures immunoreactive for iNOS or eNOS were not confined to the STN and the area around the central canal, but appeared also in the white matter such as the spinal trigeminal tract (Fig. 5e,f). The iNOS+ structures were all < 10 μm in diameter and were found in superficial layers of the STN and the tractus, where they resembled glial cells (see Fig. 5e, inset). Besides small eNOS-immunoreactive structures of similar appearance and distribution, the endothelium of many small blood vessels throughout the medulla was eNOS+ (see Fig. 5f, inset). In short, none of the iNOS+ or eNOS+ cells could clearly be attributed to a neuronal structure. After infusion with SNP the general appearance of eNOS- and iNOS-immunoreactive structures was similar, although their density seemed to have increased. Therefore, in the present dataset only nNOS-immunoreactive cells were counted to compare experiments of SNP and vehicle infusion. Omitting the nNOS antiserum (Fig. 5g) as well as preadsorption with nNOS protein resulted in no fluorescence but normal background staining, showing that the nNOS immunoreactivity was specific.
In six animals infused with SNP (50 μg/kg) nNOS+ neurons were counted in the STN on both sides and compared with six animals treated with saline as vehicle. After infusion of SNP, the number of immunoreactive neurons was significantly increased, reaching 1.3 times the cell number of vehicle animals (P = 0.008, Mann–Whitney U-test; Figs 5a,b and 6). The number of nNOS+ neurons in the caudal vagal nuclear complex was not significantly different between SNP- and vehicle-treated animals (P = 0.093, Mann–Whitney U-test; Fig. 6).

Numbers of neuronal nitric oxide synthase (nNOS)-immunoreactive neurons in the spinal trigeminal nucleus (STN) and the area around the central canal (CVNC). A significant increase in immunopositive cell numbers was only observed in the STN. SNP, sodium nitroprusside.
Discussion
This study has demonstrated time-dependent histochemical changes that indicate increased neuronal NO activity in the STN caudalis following provocation with a NO donor. We have chosen NADPH-diaphorase histochemistry as a method to detect NO-producing cells because this enzyme is intrinsically tied to NOS, forming a NADPH reductase in NO-producing neurons (1, 21). We have combined this method with immunohistochemistry for NOS isoforms. Although NADPH-diaphorase does not account for a specific subtype of NOS (21), it is very likely that it mostly represents nNOS, because our immunohistochemical stainings indicate that this is the only isoform of NOS present in neurons of the STN. The inducible form of NOS (iNOS) was not found in neurons, even after SNP treatment. We cannot exclude, however, that there is somewhat more NADPH-diaphorase staining than nNOS immunoreactivity, because in other tissues like the adrenal cortex the two stainings have been found not to overlap fully (39). On the other hand, it is clear, even without additional neuronal staining, that all the NADPH-diaphorase-positive cells counted in the present examination represent neurons characterized by the light round nucleus and the typical dendritic extensions. Structures not showing these features were not counted, even if they could have been parts of neurons.
Previous reports are in line with our findings, which show that nNOS immunoreactivity in the rat caudal trigeminal nucleus was increased 4 h after systemic administration of the NO donor GTN (40, 41). In the present study we have in addition looked at the up-regulation of NADPH-diaphorase neurons in the STN. The time-dependent increase in the number of NADPH-diaphorase-positive neurons is not necessarily indicative of de novo expression of NOS. It is possible that some of the NADPH-diaphorase-positive cells are not identified until the enzyme concentration exceeds the level of detection. Second, it is possible that post-transcriptional processes of pre-existing nNOS and not gene expression cause the increase in NADPH-diaphorase activity. Regardless of the underlying process, the number of NADPH-diaphorase-positive cells increased continuously at least for 8 h after NO donor infusion, indicating that active nNOS is produced in a feed-forward and self-maintaining manner for several hours. This is in agreement with the increase in neuronal activity following the same treatment with NO donors. In a recent series of experiments we recorded the activity in STN neurons under slow infusion of 50 μg/kg SNP or 250 μg/kg nitroglycerin for 2 h and compared the effect with previous experiments, in which we infused the same dose of SNP within 20 min (17). In both experiments there was a similar increase in neuronal activity. The slow continuous infusion of SNP or nitroglycerin increased neuronal activity on average within a shorter time (30 min) compared with the fast infusion, in which the effect was significant after 50 min. Therefore, we used the slow SNP infusion over 2 h for the nNOS experiments in the present study. Taken together, both the electrophysiological and the present histochemical experiments indicate that after NO donor treatment a continuing process is induced that increases neuronal activity and the number of presumably NO-producing second-order neurons in the STN.
Regarding the local distribution of NO-producing neurons, the increase in numbers of NADPH-diaphorase-positive nerve cells was most distinct and rapid in the superficial laminae I and IIa. In the deep laminae III and IV, cell counts were lower and a significant increase in NADPH-diaphorase-positive nerve cells was first observed 6 h after SNP infusion. The superficial laminae of the medullary and spinal dorsal horn, in particular lamina IIa (substantia gelatinosa), are known to receive most of the afferent input of thin (Aδ and C) fibres (34, 42–44) and show the most dense immune staining with antibodies against neuropeptides such as substance P or calcitonin gene-related peptide (CGRP) (45, 46), which are regarded as typical for nociceptive afferents. The deep layers are less clearly defined in the STN, and in functional studies are usually not divided into separate laminae (47–49). There is convergent evidence that the deep laminae contain large wide-dynamic range (WDR) neurons that receive both low-threshold and nociceptive information (50–52), and most of our previous electrophysiological recordings are from those neurons (17, 32, 33, 53). It is possible that the large NADPH-diaphorase- and nNOS-positive neurons found in deep laminae belong to the functionally defined WDR neurons. However, several structurally different cell types in all layers of the STN were among the NADPH-diaphorase-expressing neurons, and the increase in NADPH-diaphorase- and nNOS-positive cells was not restricted to a distinct cell type. Ultrastructural studies in superficial laminae of the rat caudal STN have revealed evidence that NADPH-diaphorase-positive cells may be different forms of interneurons and, owing to their vesicle content, may be partly inhibitory in nature (54). In the spinal dorsal horn NO-producing islet cells have been suggested to facilitate transmitter release from primary afferent terminals or interneurons with the released NO operating as a retrograde transmitter (55). We suggest that this physiological function is also attributed to the trigeminal nucleus, where NO release from different types of NO-producing neurons may support synaptic transmission by facilitating the release of neurotransmitters and nociceptive neuromodulators such as CGRP from primary afferent terminals, as depicted in Fig. 7.

Schematic representation of the dorsomedial part of the spinal trigeminal nucleus (STN) showing the localization and typical types of nitric oxide (NO)-producing neurons in superficial (I–II) and deep laminae (III–IV). Laminae I and IIa contain small cells of different shapes with extensions in the rostro-caudal direction (1, 2) and stalked cell-like neurons (3). Lamina IIb contains islet cell-like multipolar neurons of intermediate size (4). Laminae III/IV are characterized by large cells with extensions frequently reaching the outer layers (5) or form multipolar neurons (6). NO may be produced by all these neurons and, as a retrograde transmitter, facilitate neurotransmitter and neuropeptide release from multiple presynaptic terminals of Aδ and C fibres in superficial layers.
Significant up-regulation in nNOS-immunoreactive neurons following subcutaneous (s.c.) administration of the NO donor GTN has previously been reported to occur specifically in the caudal (cervical) part of the STN and not in the thoracic dorsal horn (40). In the present study the number of NADPH-diaphorase-positive neurons after SNP infusion was particularly increased in the STN, whereas only a small increase was seen in the dorsal vagal nuclear complex (38) around the central canal; the latter could not be verified with nNOS immunoreactivity. We conclude therefore that the number of NO-producing neurons increases preferentially in trigeminally innervated areas. The increase in NADPH-diaphorase-positive neurons was observed throughout the examined 0.6-mm segment of the medulla with the STN, which receives nociceptive afferent input. The level of the caudal part of this examined segment (caudal subnucleus caudalis) was the preferred region in our previous electrophysiological recordings (17, 32, 33, 56), because it contains the highest density of neurons with meningeal afferent input, whereas in the rostral part of this segment neurons with meningeal input were rarely found (32). Therefore, we conclude that up-regulation of NOS+ neurons can occur in all areas with trigeminal afferent input. We have immunostained the medullary region containing the caudal subnucleus caudalis to clarify which isoform of NOS is present in the NADPH-diaphorase-positive neurons. The results show clearly that neurons are immunoreactive only to nNOS, and the number of these neurons is up-regulated after SNP infusion.
Concomitant up-regulation of c-fos, a marker of neuronal activation, and NOS-immunoreactive neurons in the rat STN was observed after s.c. injection of low doses (10 mg/kg) of nitroglycerin (40). Tassorelli and her group have examined this issue extensively. They found NADPH-diaphorase activity and Fos expression colocalized in several brain areas including the STN, but s.c. injection of nitroglycerin caused mostly up-regulation of Fos but not NADPH-diaphorase (57, 58). On the other hand, Fos expression in STN neurons after this treatment was significantly lower in animals pretreated with the unspecific NOS inhibitor L-NG-nitroarginine-methylester or the neuronal NOS-specific inhibitor 7-nitroindazol (59), indicating that endogenous neuronal NO synthesis and cyclic GMP, the classical second messenger of intracellular NO signalling, contribute to this response (60). Also, i.p. administration of nitroglycerin (10 mg/kg) but not SNP (4 mg/kg) activated Fos expression in the STN (61). The difference was possibly due to the limited diffusion of this low dose of SNP through the blood–brain barrier, whereas nitroglycerin, which has been reported to accumulate in the brain (62), may well penetrate the blood–brain barrier. The result of Tassorelli et al. (61) seems to contradict our extracellular findings, in which neuronal activity in the STN continuously increased after i.v. infusion of SNP (50 μg/kg) (17). It is yet open whether the higher dose of SNP in our experiments, the slow i.v. administration, instead of i.p. bolus injection or a different detection level of Fos immunohistochemistry compared with electrophysiology might explain the difference.
Taken together, these and our experiments suggest that Fos expression induced by the NO stimulus requires endogenous NO production, but this response is not necessarily followed by up-regulation of the activity of NO-producing enzymes. We suppose that only higher doses of NO donors induce such up-regulation and might cause neuronal activity in adjacent neurons by a spill-over of NO (see Fig. 7). Whether this up-regulation of NO-producing enzymes in STN neurons is functional (involving post-transcriptional processes) or structural (involving expression of nNOS) should be clarified by in situ hybridization and quantitative single-cell polymerase chain reaction in further studies.
According to the present histochemical and functional data, it is now clear that NO donors, including SNP, are able to cause long-term changes of nociceptive processes in the spinal trigeminal system. In dorsal horn slices SNP has been shown to release neuropeptides by NO-dependent and NO-independent mechanisms (63). In a recent study, s.c. injection of nitroglycerin has been found to decrease CGRP immunoreactivity in laminae I–II of the rat STN within 1–4 h (64). Although the underlying mechanism is not clear, it can be speculated that not only NO produced by the injected nitroglycerin but also NO released from second-order neurons is involved in this long-lasting retrograde neuropeptide release. In the same model, nitroglycerin injection has been shown to activate the powerful transcription factor nuclear factor-κB, which is involved in the expression of proinflammatory mediators, in laminae I–II neurons in the rat STN (65).
In conclusion, it is suggested that endogenous production and release of NO in the STN is induced by exogenous administration of NO donors, which may also underlie the induction of headache attacks after infusion of nitroglycerin in patients suffering from primary headaches (4, 6, 11). These changes may occur in the same or in separate types of neurons in the STN, but they probably influence the central release of neurotransmitters and neuropeptides (Fig. 7). Considering a therapeutic application, inhibition of NO production is problematic because of severe side-effects (66, 67). CGRP has been shown to be an important mediator in nociceptive transmission in the STN (68). An increase in synaptic CGRP release might be the result of an increase in NO production. This pathophysiological mechanism can possibly be counteracted by centrally acting triptans (69, 70) and blockade of central CGRP receptors (71, 72).
Footnotes
Acknowledgements
The authors thank Jana Schramm and Birgit Vogler for their competent technical assistance. This work was supported by the Bundesministerium für Bildung und Forschung (German Headache Consortium).
