Abstract
Tension-type headache is associated with noxious input from neck muscles. Due to the importance of purinergic mechanisms in muscle nociception, experimental studies typically inject α,β-methyleneadenosine 5′-triphosphate (α,β-meATP). In contrast to native adenosine 5′-triphosphate (ATP), α,β-meATP has a narrow receptor profile and remains stable in tissue. The present study administered α,β-meATP or ATP in semi-spinal neck muscles in anaesthetized mice (n = 65) in order to address different effects in neck muscle nociception. The jaw-opening reflex monitored the impact of neck muscle noxious input on brainstem processing. Injection of α,β-meATP induced reflex facilitation in a dose-dependent manner. In contrast, only the lowest ATP dosage evoked facilitation. Preceding P2Y1 receptor blockade revealed facilitation even under high-dosage ATP. Ongoing facilitation after α,β-meATP injection neutralized under subsequent activation of P2Y1 receptors. Results demonstrate opposing excitatory P2X and inhibitory P2Y effects of ATP in neck muscle nociception. These mechanisms may be involved in the pathophysiology of neck muscle pain in man.
Introduction
Neck muscle nociception and pain probably play a decisive role in the pathophysiology of tension-type headache (TTH) (1–5). Consequently, a recently developed experimental animal model addressed the impact of nociceptive afferent input from neck muscles on central nervous system pain processing (6–11). Intramuscular (i.m.) injection of an adenosine 5′-triphosphate (ATP) analogue induces a sustained increase of neuronal excitability in the brainstem. This long-term excitatory effect on neck muscle nociception is mediated by P2X receptors (9).
Involvement of ATP in nociception and pain was recognized first in 1966 (12). Meanwhile, ATP is known to be an important neurotransmitter that interacts with ionotropic P2X receptors and metabotropic P2Y receptors (13). Of all P2X receptors especially homomeric P2X3 and heteromeric P2X2/3 subtypes mediate nociception, as shown in genetically manipulated mice and by pharmacological studies (14–17). Whereas ionotropic P2X3 and P2X2/3 receptors transmit excitatory actions, metabotropic P2Y1 receptor mediates inhibitory effects (18–20). Under in vitro conditions P2Y1 receptors inhibit P2X3 receptor channels in dorsal root ganglion neurons, indicating an antagonist interaction at cellular level (19).
ATP seems to be particularly suitable for experimental induction of noxious input from muscles, for various reasons. Interstitial ATP concentration in muscles increases with muscle contraction (21–24). ATP excites nociceptive group III and IV afferents in skeletal muscles (25, 26). Ionotropic P2X3 and P2X2/3 receptors are localized in pericranial muscles (27). Finally, ATP injection into human trapezius muscle induces local tenderness and strong pain (28).
Due to several decisive pharmacological differences, experimental studies usually favour administration of the ATP analogue α,β-methyleneadenosine 5′-triphosphate (α,β-meATP) over native ATP. ATP and α,β-meATP differ mainly in enzymatic degradation and receptor interaction. Degradation studies have demonstrated dephosphorylation of ATP, adenosine diphosphate (ADP) and adenosine monophosphate (AMP) with half-lives of about 15–20 min (29, 30). These studies have also shown that α,β-meATP is very slowly degraded to α,β-meADP, with approximately 80% remaining in tissue after 1 h. Whereas native ATP interacts with most P2X and P2Y receptors, α,β-meATP exclusively addresses P2X receptors with high sensitivity to P2X1, P2X3 and P2X2/3 (13, 31, 32). Biochemical stability and narrow receptor profile are the main reasons for selecting the P2X receptor agonist α,β-meATP in most experimental studies.
A single local injection of α,β-meATP into murine neck muscles increases neuronal excitability in the brainstem for many hours. This sustained effect is obviously mediated by ongoing excitation of P2X receptors on peripheral nociceptors in neck muscles (9). Due to the stability of α,β-meATP in tissue and its consequential long residence time in muscle, the experimental model with a single injection mimics a more prolonged application. The short half-life of native ATP and a broad receptor profile suggest different effects of ATP in the mouse model compared with α,β-meATP. In vitro studies, in particular, predict inhibitory actions of ATP. Thus, the present study addressed different effects of α,β-meATP and ATP in neck muscle nociception.
Methods
Electrophysiological experiments were performed in 65 adult male C57BL/6 mice (approximately 12 weeks old; 20–28 g; Charles River Laboratories, http://www.criver.com). All procedures received institutional approval from the local ethics committee (ref. no. 50.203.2-AC 15, 16/03). The principles of laboratory animal care and use of laboratory animals (European Council Directive of November 24, 1986(86/609/EEC)) were followed. All efforts were made to minimize animal suffering and to use only the number of animals necessary to produce reliable scientific data.
A detailed description of anaesthesia, surgery and electrophysiological recording has been published (33). Mice were anaesthetized by an initial intraperitoneal (i.p.) injection of a 0.5% pentobarbital sodium salt solution (Sigma-Aldrich, http://www.sigmaaldrich.com) with a dose of 70 mg/kg. The depth of anaesthesia was checked by ensuring that noxious pinch stimulation (blunt forceps) of hindpaw, forepaw and ear did not evoke any sensorimotor reflexes. When the animal was sufficiently deeply anaesthetized, the skin of the throat was carefully shaved and lidocaine hydrochloride gel (Xylocaine® 2%; AstraZeneca, http://www.astrazeneca.com) was applied to induce local anaesthesia. Dexpanthenol eye ointment (Bepanthen®; Roche, http://www.roche.com) was applied to cornea and conjunctiva of both eyes to protect them from drying. The right external jugular vein was catheterized for continuous administration of a 2% methohexital sodium salt solution (Brevimytal®; Hikma, http://www.hikma.de) with a dose of 60 mg kg−1 h−1 corresponding to a flow rate of about 0.07 ml/h for a 23-g mouse. A pair of Teflon-coated stainless steel wires (140 µm diameter) was inserted into the right anterior digastric muscle (Dig) to record electromyographic activity (EMG) and the jaw-opening reflex (JOR) via a differential amplifier. After tracheotomy, animals were placed in a stereotaxic frame and were artificially respired with a stroke volume of about 150 µl and about 200 strokes per minute during the complete experiment (MiniVent Model 845; Harvard Apparatus, http://www.harvardapparatus.com). Body core temperature was maintained at 37.5°C with a heating blanket and a fine rectal thermal probe (FMI, http://www.fmigmbh.de). One platinum needle electrode each (300 µm diameter) was subcutaneously inserted into the left forepaw and right hindpaw to record the electrocardiogram (ECG) via a differential amplifier. Two stainless steel needle electrodes (150 µm diameter) were longitudinally inserted into the tongue musculature (parallel, 2 mm distance) in order to apply electrical stimuli and to evoke JOR. The oral cavity was filled up with white vaseline (Riemser, http://www.riemser.de) to protect oral mucous membrane from drying. Semispinal neck muscles on both sides were carefully exposed. One injection canula each (0.4 mm diameter) was inserted into muscle belly of both semispinal neck muscles. Each canula was connected via thin and short tubing to a liquid switch (CMA/110, http://www.microdialysis.se). Glass microsyringes (1 ml) were connected to the liquid switch by thin tubing and were fixed in a microdialysis pump (CMA 102, http://www.microdialysis.se). Use of the liquid switch enabled repeated injections at the same i.m. site without changing canula and without interruption of flow during the in vivo experiment. This procedure allowed bilateral induction of noxious input from neck muscles in order to mimic bilateral neck muscle pain in TTH patients. The following substances were intramuscularly administered with a volume of 20 µl per muscle during a time period of 1 min (corresponding to flow rate of 20 µl/min): α,β-meATP (100 nmol/l, 1 µmol/l; Sigma-Aldrich, http://www.sigmaaldrich.com), ATP (100 nmol/l, 1 µmol/l, 7.6 mmol/l; Sigma-Aldrich, http://www.sigmaaldrich.com), the competitive P2Y1 receptor antagonist 2′-deoxy-N6-methyladenosine 3′,5′-bisphosphate tetrasodium salt (MRS2179, 1 µmol/l; Tocris, http://www.tocris.com), the P2Y1 receptor agonist 2-(methylthio)adenosine 5′-diphosphate trisodium salt hydrate (2-MeSADP, 1 µmol/l; Sigma-Aldrich, http://www.sigmaaldrich.com) and isotonic saline (0.9%, Delta-Select GmbH, http://www.deltaselect.de). A maximum of two injections were performed in one muscle during one experiment.
After surgery and placement of all electrodes, the anaesthetized animal was rested for at least 1 h. During this time period the level of anaesthesia and heart rate were routinely checked and documented, and the depth of anaesthesia maintained. All electrical signals (EMG, ECG) were recorded by bioamplifiers and led into a data collection system (CED Micro1401, http://www.ced.co.uk) and a personal computer using Signal® and Spike2® software programs (CED, http://www.ced.co.uk).
The JOR was elicited by rectangular electrical pulses of 500 µs duration with a stimulation frequency of 0.1 Hz. The electrical threshold of the JOR was determined by applying increasing and decreasing stimulus intensities from 0 to 2 mA in steps of 100 µA. The lowest stimulus intensity that just evoked a reflex response was defined as the JOR threshold (I JOR). Test stimulus intensity was adjusted to about 140% of the I JOR. The JOR was evoked in series of eight stimuli each. These series were repeated every 5 min. After three stable baseline JOR series, substances were administered intramuscularly and the effect was monitored in three different time schemes (Fig. 1).
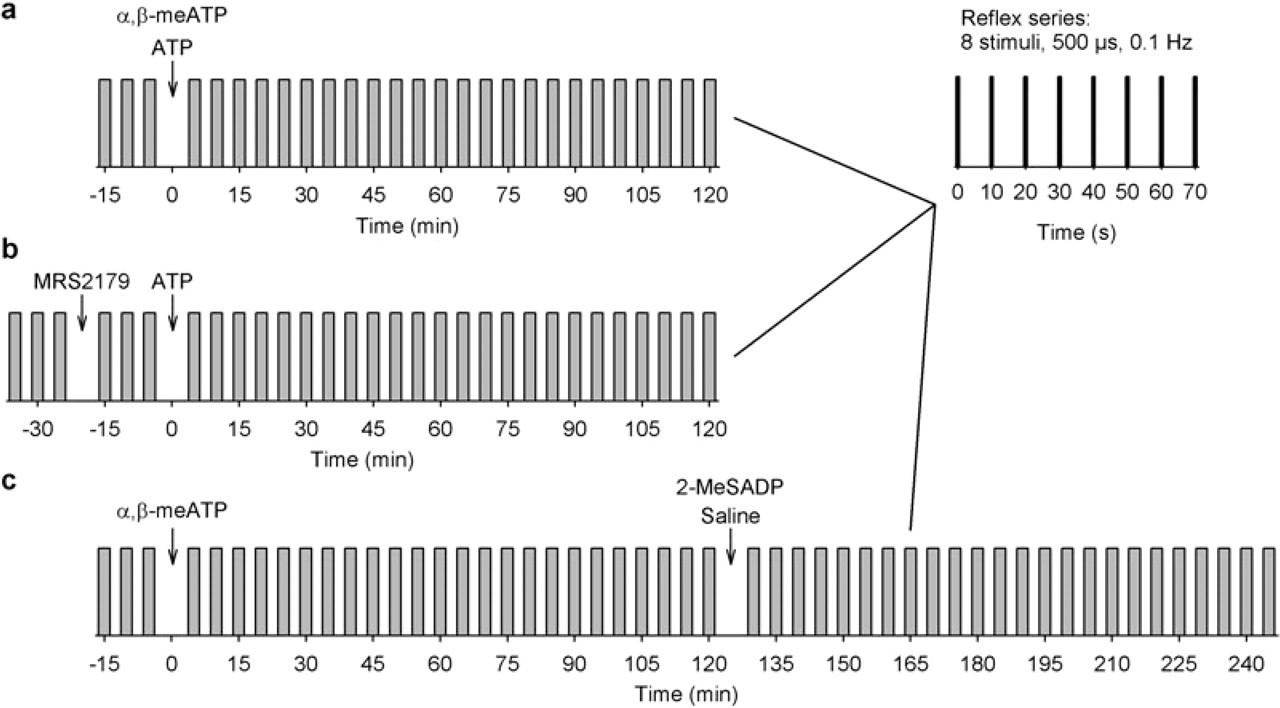
Stimulation protocol. The jaw-opening reflex (JOR) was evoked in series (vertical bars) of eight stimuli each (top right-hand corner). The reflex series were repeated every 5 min (grey bars). In three different experimental groups, substances were injected into both semspinal neck muscles (as marked by arrows). (a) After three baseline series either α,β-meATP (100 nmol/l, n = 10; 1 µmol/l, n = 10) or native ATP (100 nmol/l, n = 10; 1 µmol/l, n = 10; 7.6 mmol/l, n = 10) was intramuscularly (i.m.) administered and the effect on JOR was monitored for at least 2 h. (b) After three baseline series MRS2179 (1 µmol/l, n = 10) was injected intramuscularly and the reflex was recorded for three series. Subsequently, native ATP (1 µmol/l) was administered intramuscularly and effects on JOR were monitored for 2 h. (c) After three baseline series α,β-meATP (1 µmol/l, n = 10) was injected intramuscularly and the effect was monitored for 2 h. 2-MeSADP (1 µmol/l, n = 5) or saline (n = 5) was then administered intramuscularly and reflex effects were monitored for another 2 h.
In the first scheme (Fig. 1a), after three baseline series either α,β-meATP (100 nmol/l, n = 10; 1 µmol/l, n = 10) or native ATP (100 nmol/l, n = 10; 1 µmol/l, n = 10; 7.6 mmol/l, n = 10) was administered intramuscularly and the effect on JOR was monitored for at least 2 h. In the second scheme (Fig. 1b), after three baseline series MRS2179 (1 µmol/l, n = 10) was injected intramuscularly and the reflex was recorded for three series. Subsequently, native ATP (1 µmol/l) was administered intramuscularly and effects on JOR were monitored for 2 h. In the third scheme (Fig. 1c), after three baseline series α,β-meATP (1 µmol/l, n = 10) was injected intramuscularly and the effect was monitored for 2 h. 2-MeSADP (1 µmol/l, n = 5) or saline (n = 5) was then administered intramuscularly and reflex effects were monitored for another 2 h.
Onset latency, duration, and integral of JOR were analysed in each single sweep. In recent studies reflex integral turned out to be the key parameter of reflex alteration in the present animal model (6, 7, 9, 10). Reflex integral, indeed, partly depends on duration of reflex. For the sake of clarity and readability, the present study focuses on statistical analysis of reflex integral. Arithmetic mean and standard error were calculated (mean ±
Results
In 65 mice electrical tongue stimulation elicited the JOR with a threshold intensity of I
JOR = 694 ± 23 µA (mean ±
Sole administration of α,β-meATP
In 20 mice α,β-meATP solution (100 nmol/l, 1 µmol/l, n = 10 each) was injected into semispinal neck muscles (Figs 1a and 2). Reflex integrals significantly increased within 2 h after injections (Fig. 2). A higher concentration of α,β-meATP caused stronger facilitation of the reflex (100 nmol/l: median +97.5%; 1 µmol/l: median +171.2%; Mann–Whitney rank sum test: T = 77, P < 0.05).

Reflex facilitation after sole intramuscular (i.m.) injection of α,β-meATP. (a) Integral of jaw-opening reflex (JOR) increased significantly within 2 h after i.m. administration of 100 nmol/l α,β-meATP (black triangle; Friedman
Sole administration of ATP
In 30 mice native ATP was injected in semispinal neck muscles (Figs 1a and 3). Within 2 h after administration of 100 nmol/l ATP, reflex integral significantly increased (Fig. 3). After 1 µmol/l, ATP reflex remained unchanged. With 7.6 mmol/l ATP reflex integral decreased in the first hour without any effects in the second hour.

Reflex facilitation after sole intramuscular (i.m.) injection of native ATP. (a) Integral of jaw-opening reflex (JOR) significantly increased within 2 h after i.m. administration of 100 nmol/l ATP (white circle; Friedman
Combined administration of MRS and subsequent ATP
I.m. injection of native ATP in semispinal neck muscles induced reflex facilitation only with a low concentration of 100 nmol/l but not with 1 µmol/l and 7.6 mmol/l (Fig. 3). This might be due to inhibitory effects of native ATP and its metabolites on nociceptors via metabotropic P2Y1 receptors. In order to test masking of a suggested excitatory effect of ATP by a competitive inhibitory effect of ATP and its metabolites (e.g. ADP), the potent P2Y1 receptor antagonist MRS2179 (MRS) was applied intramuscularly 20 min before homotopic injection of native ATP (1 µmol/l) in 10 mice (Figs 1b and 3). MRS itself did not induce any changes of baseline reflex. Within 2 h after ATP administration, the reflex integral increased significantly (Fig. 3). Within the first and second hours after ATP injection, mean reflex integrals significantly differed between various ATP concentrations and combined MRS and ATP injection (Fig. 3b). Facilitatory effects on JOR within the first (SNK: q = 5.5, P < 0.05) and second hour (SNK: q = 6, P < 0.05) were significantly greater after combined administration of MRS and ATP than after sole injection of 1 µmol/l ATP (Fig. 3b).
Combined administration of α,β-meATP and 2-MeSADP
Preceding blockade of P2Y1 receptors revealed a facilitatory effect of i.m. ATP injection (see above, Fig. 3). The result seemed to support competitive antagonism between purinergic inhibitory and excitatory effects on myofascial nociception mediated by P2Y1 and P2X3 receptors, respectively. This concept predicted inhibition of α,β-meATP-evoked facilitation by subsequent activation of P2Y1 receptors. In order to test the concept, the P2Y1 receptor agonist 2-MeSADP (1 µmol/l) was intramuscularly administered during established reflex facilitation 2 h after injection of 1 µmol/l α,β-meATP in semispinal neck muscles (n = 5) (Fig. 1c). The results were compared with experiments where a first injection of 1 µmol/l α,β-meATP was followed by a saline injection in order to exclude any influence of dilution by second injection (n = 5) (Fig. 1c). Within 2 h after i.m. administration of 1 µmol/l α,β-meATP, the reflex integral similarly increased in both groups (saline group:

Subsequent administration of 2-MeSADP reverses reflex facilitation. (a) After three baseline jaw-opening reflex (JOR) series, 1 µmol/l α,β-meATP was intramuscularly (i.m.) injected in both semispinal neck muscles (n = 10). In five experiments each, either saline (black triangles) or the P2Y1 receptor agonist 2-MeSADP (white triangles) were administered intramuscularly 2 h after injection of α,β-meATP. Reflex integrals are given as mean ±
Discussion
Local administration of α,β-meATP in murine semispinal neck muscles induces sustained facilitation of brainstem nociception in a dose-dependent manner. In contrast, native ATP injection evokes facilitation only with low dosage. Preceding i.m. blockade of P2Y1 receptors reveals reflex facilitation even under a high dosage of native ATP. Ongoing facilitation after injection of α,β-meATP is abolished by subsequent activation of P2Y1 receptors. Results indicate significant interactions between excitatory P2X and inhibitory P2Y1 receptors in myofascial nociceptive processing from neck muscles in mice.
Experimental studies on myofascial nociception usually apply the ATP analogue α,β-meATP due to its prolonged chemical stability, with 80% remaining active in the tissue after 1 h. One major reason for prolonged stability in tissue is the ability of α,β-meATP to inhibit endogenous ecto-ATPases located on the cell surface and thus limiting emergence of metabolization products (34). α,β-meATP has a narrow receptor profile with high affinity to homomeric P2X1, P2X3, and heteromeric P2X2/3 receptors (13, 31, 32). Several animal studies have documented the algogenic character of α,β-meATP (15, 35–38). In a previous study applying the same mouse model of neck muscle nociception, direct evidence for involvement of P2X3 receptor as well as indirect evidence for an involvement of P2X2/3 receptor in reflex facilitation after α,β-meATP injection were demonstrated (9, 39). Due to lack of homomeric P2X1 receptors in skeletal muscles and rapid desensitization of P2X3 receptors after ligand interaction, α,β-meATP effects seemed to be mainly mediated by heteromeric P2X2/3 receptors (13, 16, 27, 40, 41).
On the other hand, native ATP is under in vivo conditions immediately degraded into its main metabolites (ADP, AMP, adenosine) within 15–20 min (29, 30, 42) and couples to a broad range of various ionotropic and metabotropic purinergic receptors (32, 43, 44).
In general, purinergic receptors are subdivided into ATP-gated ion channels (P2X) and G-protein-coupled P2Y receptors. Today, seven P2X and eight P2Y receptors have been identified (45, 46). Purinergic P2X receptors in particular play an important role in nociceptive excitatory processing (16, 40, 47, 48), with ATP being the most potent native agonist (40). Homomeric P2X3 and heteromeric P2X2/3 receptors have previously been identified to mediate the primary sensory effects of ATP (40, 49). P2X receptors are localized in a wide variety of tissues, including skeletal muscle as well as the central and peripheral neuronal system (32, 44). In muscle, the nociceptive properties of ATP were shown to be conducted via P2X receptors, located on group III and IV muscle afferents. Single nerve fibre recordings in decerebrate cats have demonstrated excitation of group III and IV muscle afferents from the triceps surae muscle under application of α,β-meATP into the poplietal artery (25). Studies in P2X2 and P2X3 double-knockout animals have shown a loss of α,β-meATP-induced currents in dissociated dorsal root ganglion neurons (17). In rat, P2X receptors are located in the spinal dorsal horn and especially in the substantia gelatinosa (50). With focus on trigeminal sensory neurons, P2X2 and P2X3 receptors were identified to be predominantly located on small to medium size nociceptive neurons and attributed a critical role in the modulation of craniofacial pain (51). Afferents from neck muscles in mice synapse in superficial layers of the upper cervical cord (8, 52). Immuno-histochemical techniques indicate that 22% of masseter muscle afferent neurons were positive for P2X3 receptor, and 77% of these neurons were classified as heteromeric P2X2/3 neurons (27).
Regarding the source of ATP involved in purinergic receptor activation, it is accepted that ATP is not only released during cell damage, but is actively released from skeletal muscle and neuronal cells (53–55). Recent studies have shown that in skeletal muscle the interstitial ATP concentration increases with muscle contraction (21–24) and compression (56). In various types of resting cells, extracellular ATP metabolism is balanced by constitutive release (57).
Metabotropic P2Y receptors were previously identified to interact with ionotropic P2X receptors. In nociceptive processing, the P2Y1 receptor, in particular, seems to play an important role. Several in vitro studies have proposed an inhibitory effect of the metabotropic P2Y1 receptor onto the excitatory effects being mediated through P2X3 receptor (18–20).
In the present study, the blockade of the P2Y1 receptor revealed the excitatory effects of higher concentrations of native ATP. Confirming the inhibitory role of the P2Y1 receptor, injection of an agonist (2-MeSADP) led to a complete reversal of P2X-mediated facilitation.
Various studies using immunostaining techniques have shown that P2Y1 receptor antibodies stained > 80% of dorsal root and trigeminal ganglia cells (58, 59) and that P2Y1 and P2X3 receptors are frequently colocalized on the same neuron (58, 60). P2Y1 and P2Y2 purinergic receptors were shown to be typically expressed in small, putatively nociceptive cells corresponding to known distribution of P2X3 receptors (58, 61). In rat, distribution in skeletal muscle and neuronal cells has been identified (58, 62).
The physiologically most potent agonist at the P2Y1 receptor is ADP as well as ATP at high concentrations, whereas MRS2179, which was also used in this study, is a highly selective antagonist (43, 63).
Due to rapid degradation of ATP, effects of application in tissue are probably induced by ATP and its metabolites. In order to minimize possible side-effects of ATP metabolites, an inhibitor of the endogenous ecto-ATPases (ARL67156) was applied in the present study (data not presented). Because of endogenous agonistic properties of ARL67516 (dirty drug) at both the excitatory P2X3 and the inhibitory P2Y1 receptor, no coherent results could be obtained (64). Since no other substances with similar effects on enzymes are available, experiments were stopped.
Different molecular mechanisms are involved in interaction of P2X3 and P2Y1 receptors (18–20). P2X3 receptors are ion channels and are selectively permeable to cations (32, 65–68). Metabotropic P2Y1 receptors are known to exert their effects through a G-protein-coupled cascade (69, 70). P2Y receptors modulate a number of voltage- and ligand-gated membrane channels such as M-type K+ channels, voltage-activated Ca2+ channels, N-methyl-D-aspartate receptors, as well as vanilloid-, pH- and heat-sensitive TRPV1 receptors (18). Recent studies have demonstrated that activation of metabotropic P2Y1 receptors inhibits the conductance of P2X3 receptor channels through facilitation of their desensitization and suppression of their recovery from the desensitized state (18–20).
ATP binds with high affinity to the P2X receptors (in higher concentrations also to P2Y receptors), whereas its main metabolite ADP shows greater affinity to the P2Y1 receptor (43, 71). ADP has been shown to exhibit an EC50 value at purified P2Y1 receptors of approximately 1 µmol/l (72). This matches with our results, where lower concentrations of ADP (merging from the breakdown of ATP, 100 nmol/l) did not show significant effects. At that concentration, excitatory effects of native ATP at P2X receptors appear to predominate. For higher concentrations of native ATP and resulting higher concentrations of the metabolite ADP, the amount of ADP binding to the inhibitory P2Y1 receptor is probably sufficient to counterbalance the ATP effect at the excitatory P2X receptor.
The present study has applied a recently developed model of neck muscle nociception in mice (6–9, 11). The suggested link between TTH on one hand and the animal model on the other may be the importance of neck muscle pain processing in patients. TTH patients typically suffer from pericranial tenderness and hardness of head and neck muscles (1, 73–75). Shoulder and neck pain is induced by static exercise in TTH patients more frequently than in healthy controls (76). Tonic muscle activation with subsequently induced ATP release may therefore be connected to the pathophysiology of TTH. The present study has demonstrated that even low intramuscular levels of ATP may facilitate neck muscle nociception.
By using native ATP as a pharmacological tool in the present study, interactions between P2X and P2Y1 purinergic receptors in muscle nociception could for the first time be shown under in vivo conditions in mice. Divergent interactions of purinergic P2X and P2Y1 receptors might be useful for a future pharmacological strategy in the treatment of TTH.
Acknowledgements
This research project was supported by grants of the German Headache Consortium (Federal Ministry of Education and Research, 01EM0516, project A3). This paper is part of the doctoral thesis at the Medical Faculty of RWTH Aachen University by cand. med. M.R.
