Abstract
Neck muscle nociception probably plays a major role in the pathophysiology of tension-type headache. Recent studies have demonstrated sustained facilitation of brainstem nociception due to noxious neck muscle input evoked by nerve growth factor (NGF) or α,β-methylene ATP (ATP) in mice. Hypothesized different afferent pathways in NGF and ATP models were addressed by local application of tetrodotoxin (TTX) in neck muscles. Brainstem nociception was monitored in 55 anaesthetized mice by the jaw-opening reflex elicited by electrical tongue stimulation. Sole administration of 100nmol/l ATP or 0.8 μmol/l NGF evoked sustained reflex facilitation for at least 95 min. Preceding TTX administration prevented ATP-induced facilitation, but was without effect on NGF. Subsequent administration of 100 nmol/l TTX reversed ATP-evoked facilitation, but was ineffective on NGF. Divergent effects of TTX suggest preferential excitation of group III muscle afferents by ATP and group IV by NGF. Thus, both models address different pathways in pericranial pain.
Introduction
Tension-type headache (TTH) is the most frequent primary headache with an important socioeconomic impact, but its pathophysiology is still unclear (1–5). Pericranial tenderness of neck muscles is the most significant abnormal finding in patients suffering from TTH (1, 6–11). Therefore, recent studies have addressed the development of an animal model for the investigation of the physiology and pharmacology of myofascial nociceptive processing in the neck and may open up new vistas into the pathophysiology and treatment of TTH (4, 12–14). Single injections of human β-nerve growth factor (NGF) or α,β-methylene ATP (ATP) into mice semispinal neck muscles induce strong and sustained facilitation of brainstem somatosensory processing as recorded by the jaw-opening reflex (JOR) under general anaesthesia (12, 14). Both algogenic substances are common in myofascial pain studies (15–20).
Whereas facilitatory effects of intramuscularly administered NGF and ATP on brainstem nociception are very similar, molecular mechanisms differ. NGF may act via tyrosine kinase A (trkA) and p75 receptors in muscles (21), ATP probably interacts with P2X3 receptors on muscle nociceptors (12, 22). Nociceptive primary afferents are classified into peptidergic and non-peptidergic fibres (23, 24). Peptidergic neurons contain peptides (e.g. substance P, calcitonin gene-related peptide, somatostatin) and express trkA. Non-peptidergic neurons contain fluoride-resistant acid phosphatase, typically bind plant lectin IB4 and express P2X3 receptors. Thus, NGF and ATP effects are probably mediated by distinct afferents.
The excitatory effect of NGF on nociceptors involves sodium currents which are resistant to tetrodotoxin (TTX). There is evidence that NGF enhances nociceptive afferent excitability by exerting both an acute influence on the gating of TTX-resistant sodium current and a prolonged influence by increased expression of TTX-resistant sodium channel Nav1.8 (25, 26). Local administration of TTX to peripheral nerves is used to differentiate between TTX-sensitive and TTX-resistant mechanisms in sensory physiology (27–30). Local TTX administration to the saphenous nerve in rat differentially affects action potential propagation in A and C fibres with IC50 of 57 nmol/l and 372 nmol/l, respectively (27). Experimental studies in rabbits and cats have confirmed selective blockade of neural conduction in A fibres by low concentrations of TTX (28–30).
The present study addressed the hypothesis that brainstem facilitation evoked by local administration of NGF and ATP in semispinal neck muscles involves different classes of nociceptive afferents with different configuration of TTX-resistant and -sensitive sodium channels. Therefore, possible inhibition of NGF and ATP effects by intramuscular low-dose TTX application was tested.
Materials and methods
Experiments were conducted in 55 adult male C57BL/6 mice (about 12 weeks old; 21–27 g; Charles River Laboratories, Sulzfeld, Germany). All procedures received institutional approval from the local ethics committees. The principles of laboratory animal care and use of laboratory animals [European Communities Council Directive of November 24, 1986 (86/609/EEC)] were followed. All efforts were made to minimize animal suffering and to use only the number of animals necessary to produce reliable scientific data.
A detailed description of anaesthesia, surgery and electrophysiological recording in mice has recently been published (12, 31). Mice were deeply anaesthetized by an initial intraperitoneal injection of a 0.5% pentobarbital sodium salt solution (Sigma-Aldrich, Munich, Germany) with a dose of 70 mg/kg. Depth of anaesthesia was checked by ensuring that noxious pinch stimulation (blunt forceps) of the hindpaw, the forepaw and the ear did not evoke any sensorimotor reflexes. When the mouse was sufficiently deeply anaesthetized the skin of the throat was carefully shaved and Lidocaine gel (Astra Zeneca, Wedel, Germany) was applied to induce local anaesthesia. Dexpanthenol eye ointment (Roche, Grenzach-Wyhlen, Germany) was applied to the cornea of both eyes to protect it from drying. The right external jugular vein was catheterized for continuous administration of a 2% methohexital sodium salt solution (Lilly, Bad Homburg, Germany) with a dose of 60 mg/kg per hour corresponding to a flow rate of about 0.07 ml/h for a 23-g mouse. A pair of Teflon-coated stainless steel wires (140 µm diameter) was inserted into the right anterior digastric muscle (Dig) to record electromyographic (EMG) activity via a differential amplifier. After tracheotomy, animals were placed in a stereotaxic frame and were artificially respired with a stroke volume of about 150 µl and about 200 strokes per minute for the duration of the experiment (MiniVent Model 845; Harvard Apparatus, Holliston, MA, USA). The percentage of expiratory carbon dioxide was continuously monitored (Capstar-100; CWE Inc., Ardmore, PA, USA). Body core temperature was maintained at 37.5 °C with a heating blanket and a fine rectal thermal probe (FMI, Seeheim-Ober Beerbach, Germany). One platinum needle electrode each (300 µm diameter) was subcutaneously inserted into the left forepaw and right hindpaw to record the electrocardiogram (ECG) via a differential amplifier. Two stainless steel needle electrodes (150 µm diameter) were longitudinally inserted into the tongue musculature (parallel, 2 mm distance) in order to evoke the JOR of digastric muscles by electrical stimulation of the tongue. The oral cavity was filled up with white Vaseline (Riemser, Greifswald-Insel Riems, Germany) to protect the oral mucous membrane from drying. Semispinal neck muscles on both sides were carefully exposed. A pair of Teflon-coated stainless steel wires (140 µm diameter) was inserted into the right semispinal neck muscle to record EMG activity during intramuscular injections. One injection canula each (0.4 mm diameter) was inserted into the muscle belly of both semispinal neck muscles. Each canula was connected via thin tubing to a glass microsyringe (1 ml) that was fixed into a CMA/102 microdialysis pump (CMA Microdialysis, Solna, Sweden). This procedure allowed bilateral induction of noxious input from neck muscles in order to mimic bilateral neck muscle pain in TTH patients. Via these canulas intramuscular injections of ATP (100 nmol/l; Sigma-Aldrich), tetrodotoxin citrate (TTX; 30, 50 or 100 nmol/l; Tocris.com) and recombinant human NGF (0.8 µmol/l; Calbiochem.com) in both semispinal neck muscles were performed with a flow rate of 20 µl/min. Considering a possible postsurgical hormonal stress response, the anaesthetized animal had a rest for at least 1 h after completion of surgery and placement of all electrodes. In this time period, levels of anaesthesia (reflexes) and heart rate were routinely checked and documented, and depth of anaesthesia was maintained. After this rest period anaesthesia was adjusted to a level that allowed stable JOR recording. Three toes each of hindpaw and forepaw were successively pinched by blunt forceps with an interstimulus interval of 1 s. Anaesthesia was adjusted to a level that provided for a weak motor response to pinch stimulation of third toe without any increase of heart rate. This reflex level was maintained through the entire experiment monitored by periodical pinch stimulations. Electrophysiological experiments started about 1 h after completion of surgery and electrode placement at the earliest.
All electric signals (EMG, ECG) were recorded by bioamplifiers and led into a data collection system (CED micro 1401) and a personal computer using the Signal and Spike2 software programs (CED, Cambridge, UK).
Somatosensory processing and neuronal excitability in the brainstem and the upper cervical spinal cord was monitored by the JOR evoked by electrical stimulation of tongue musculature (31–35). The JOR was elicited by rectangular electric pulses of 500 µs duration with a stimulation frequency of 0.1 Hz. The electric threshold of the JOR was determined by applying increasing and decreasing stimulus intensities from 0 to 2 mA in steps of 100 µA. The lowest stimulus intensity that just evoked a reflex response was defined as the JOR threshold (IJOR). Test stimulus intensity was adjusted to about 125% of the IJOR. The JOR was evoked in series of eight stimuli each. Reflex series were repeated every 5 min. After three stable baseline JOR series, TTX (30–100 nmol/l) was applied into semispinal neck muscles. After another three reflex series, ATP (100 nmol/l), NGF (0.8 µmol/l) or isotonic saline (saline) were intramuscularly administered and JOR series were continued every 5 min for 1 h (Fig. 1a). In order to characterize the role of TTX-sensitive and TTX-resistant sodium channels in the maintenance of ATP and NGF effects, in a second experiment TTX (100 nmol/l) was subsequently applied after ATP or NGF injection. JOR series were repeated for 1 h after intramuscular TTX administration (Fig. 1b).
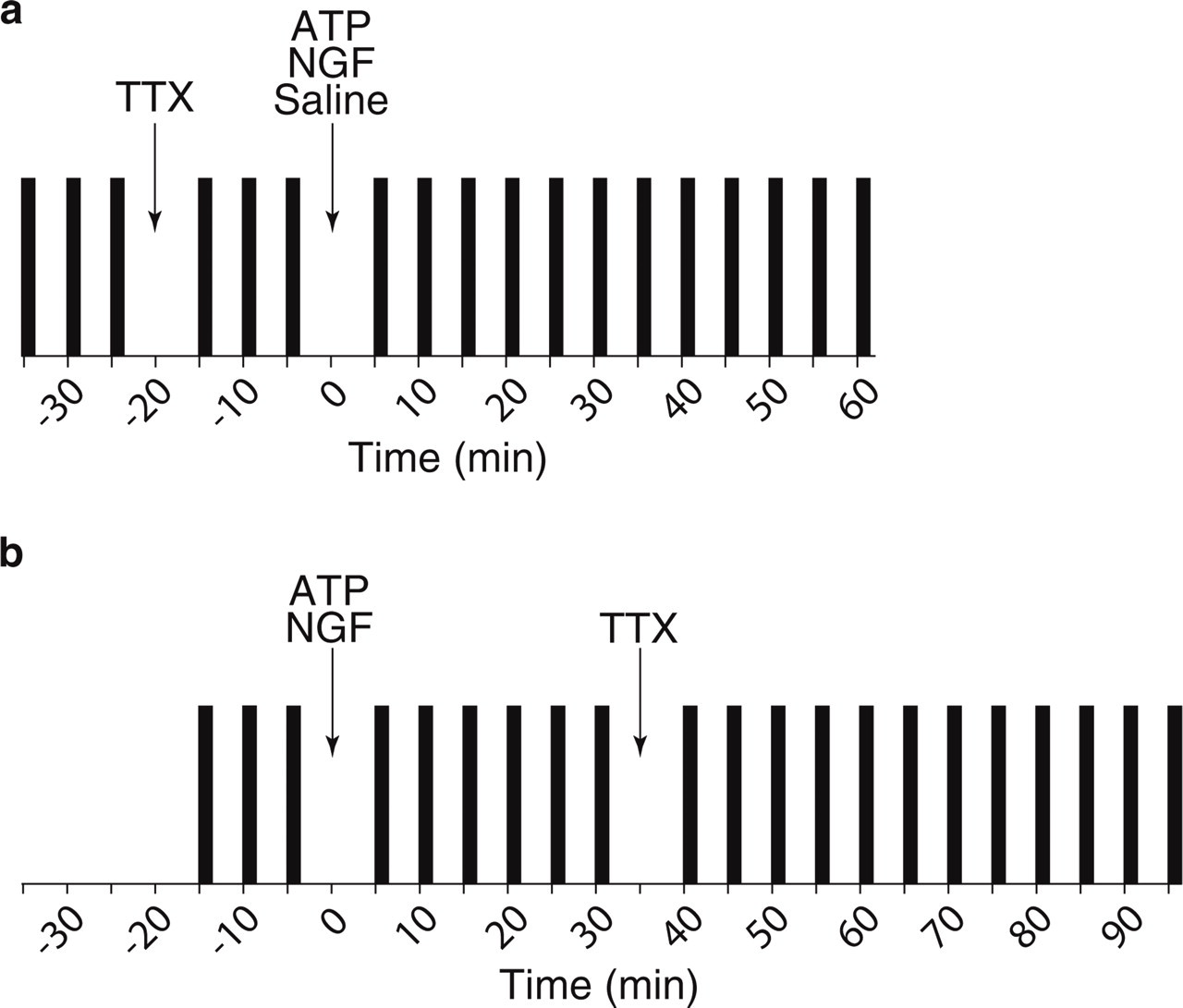
Stimulus protocol. The jaw-opening reflex was evoked in series (vertical black bars) of eight electrical stimuli each. Reflex series were repeated every 5 min. (a) Preceding administration of tetrodotoxin (TTX) was performed after three baseline reflex series and 20 min before subsequent injection of α,β-methylene ATP (ATP), nerve growth factor (NGF) or isotonic saline (saline). Reflex series were continued for 1 h after ATP, NGF or saline injection. (b) ATP or NGF were administered after three baseline reflex series and 35 min before additional local injection of TTX. Thereafter, TTX reflex series were continued for 1 h.
Onset latency, duration and integral of JOR were analysed in each single sweep. Arithmetic mean and standard error were calculated (mean ± SEM). One-way repeated measures analysis of variance (
Results
The experiments were performed on 50 mice under general anaesthesia. The JOR was elicited by electrical tongue stimulation with an intensity of IS = 878 ± 30 µA (mean ± SEM) corresponding to 129 ± 1% of IJOR (680 ± 22 µA).
In order to investigate the role of TTX-sensitive fibres in ATP- and NGF-induced JOR facilitation, different concentrations of TTX (30, 50 and 100 nmol/l) were intramuscularly applied 20 min before injection of ATP (n = 15), NGF (n = 5) or saline (n = 5). In the time window after TTX application and before administration of ATP, NGF or saline, statistical analysis (one-way repeated measures

No effect of tetrodotoxin (TTX) on baseline reflexes. Local injections of different concentrations of TTX solution (30–100 nmol/l) into semispinal neck muscles did not induce any statistical changes of reflex integral (mean ± SEM). ○, 30 nmol/l TTX (n = 5), NS; □, 50 nmol/l TTX (n = 5), NS; Δ, 100 nmol/l TTX (n = 15), NS.
After sole administration of 100 nmol/l ATP into semispinal neck muscles (n = 5), JOR integral (F = 19.48, P < 0.001) and duration (F = 2.63, P < 0.01) significantly increased by 65.5 ± 7.7% and 7.7 ± 2.6%, respectively, and JOR latency significantly decreased by −8.5 ± 1.8% (F = 14.19, P < 0.001) within 1 h (Fig. 3a). JOR thresholds under baseline conditions and 1 h after ATP administration were 740 ± 51 µA and 480 ± 49 µA, respectively (P > 0.05). Preceding intramuscular TTX administration modulated the facilitatory ATP effect on the reflex in a dose-dependent manner (Fig. 3). Under 30 nmol/l TTX and subsequent ATP injection, reflex integral (χ2 = 32.38, P < 0.01) and duration (F = 1.97, P < 0.05) increased by 68.2 ± 8.2% and 20.6 ± 9.1%, respectively, and JOR latency decreased by −6.7 ± 2.4% (F = 4.37, P < 0.001). After preceding administration of TTX with concentrations 50 nmol/l (n = 5) and 100 nmol/l (n = 5) and additional ATP or saline injections, reflex parameters including thresholds remained unchanged. Statistical comparison of ATP effects supported significant differences between groups (F = 29, P < 0.001) with significantly lower integral changes after combinations of 100 nmol/l TTX and ATP or 100 nmol/l TTX and saline compared with all other groups (t > 4.5, P < 0.001) (Fig. 3b). Furthermore, the effect after combined administration of 50 nmol/l TTX and ATP significantly differed from ATP application alone (t = 2.4, P < 0.05) and combined 30 nmol/l TTX and ATP injections (t = 2.7, P < 0.05).

Dose-dependent prevention of α,β-methylene ATP (ATP) effect by preceding tetrodotoxin (TTX). (a) Percentage changes of reflex integral with sole intramuscular administration of 100 nmol/l ATP (○), with combined TTX (30–100 nmol/l) and subsequent 100 nmol/l ATP administration, or with combined 100 nmol/l TTX and subsequent isotonic saline (saline) injections (mean ± SEM). ATP injection alone (○) and combined 30 nmol/l TTX and 100 nmol/l ATP (□) induced a significant increase in reflex integral as analysed by one-way repeated measures analysis of variance. ○, ATP alone (n = 5), P < 0.001; □, 30 nmol/l TTX, ATP (n = 5), P < 0.01; Δ, 50 nmol/l TTX, ATP (n = 5), NS; •, 100 nmol/l TTX, ATP (n = 5), NS; ▪, 100 nmol/l TTX, saline (n = 5), NS. (b) Percentage changes of reflex integrals after administration of ATP alone (□), combined TTX (30–100 nmol) and ATP injections, or combined 100 nmnol/l TTX and saline injections (mean ± SEM). Group differences are significant according to one-way analysis of variance (F = 29, P < 0.001). ∗∗∗Significant (P < 0.001) differences compared with combination of 100 nmol/l TTX and ATP. †††Significant (P < 0.001) differences compared with combination of 100 nmol/l TTX and saline. Furthermore, effects under 50 nmol/l TTX and ATP differed from sole ATP (P < 0.05) and combination of 30 nmol/l TTX and ATP (P < 0.05). Post hoc comparisons were performed by Holm–Sidak test.
In order to address the role of TTX-sensitive sodium channels in maintenance of reflex facilitation, ATP alone or combined ATP and TTX (100 nmol/l) were intramuscularly administered in 10 experiments each, and reflex recording was continued for 95 min after ATP injection (Fig. 4). After ATP alone, reflex integral and duration increased by 105.1 ± 17.6% (χ2 = 155.85, P < 0.001) and 14.8 ± 4.6% (χ2 = 50.24, P < 0.001), respectively, and latency decreased by −10.4 ± 1.6% (χ2 = 160.60, P < 0.001). Subsequent administration of TTX 35 min after preceding ATP injection (compare Figures 1b and 4a) prevented further reflex facilitation and reversed augmented reflex down to baseline values within 1 h. Statistical comparison of 30 min averages after ATP (Fig. 4), documented an increase of reflex integral with ATP alone (F = 32.77, P < 0.001) and a decrease of reflex integral with combined ATP and TTX injections (F = 4.31, P < 0.05). Reflex integral changes with ATP alone and combined ATP and TTX significantly differed in time windows between 35 and 65 min (t = 3.25, P < 0.01) and between 65 and 95 min (t = 61.0, P < 0.01). Whereas IJOR significantly decreased from 760 ± 43 µA to 540 ± 50 µA (t = 4.49, P < 0.01) with application of ATP alone, IJOR remained unchanged with combined ATP and TTX injection.

Reversal of α,β-methylene ATP (ATP) effect by subsequent tetrodotoxin (TTX). (a) Percentage changes of reflex integral (mean ± SEM) by administration of 100 nmol/l ATP alone (○) or by combined 100 nmol/l ATP and subsequent 100 nmol/l TTX injections (Δ). ○, ATP alone (n = 10); Δ, ATP, 100 nmol/l TTX (n = 10). (b) Percentage changes (mean ± sem) within 30-min intervals after administration of 100 nmol/l of ATP alone (□) or combined 100 nmol/l ATP and 100 nmol/l TTX injections (▪). ATP alone induced significant increase of reflex and combined ATP and TTX induced significant decrease of reflex according to one-way repeated measures analysis of variance. Asterisks mark significant differences between groups (∗∗P < 0.01). □, ATP alone (n = 10), P < 0.001; ▪, ATP, 100 nmol/l TTX (n = 10), P < 0.05.
After administration of 0.8 µmol/l NGF into neck muscles (n = 5), reflex integral and duration increased by 170.1 ± 31.0% (F = 16.82, P < 0.001) and 27.8 ± 6.0% (F = 9.30, P < 0.001), respectively, and latency decreased by −12.4 ± 2.0% (F = 33.63, P < 0.001) within 95 min (Fig. 5). Integral increase of 138.8 ± 27.4% (χ2 = 61.62, P < 0.001) within 60 min after NGF injection was also significant. After NGF, IJOR decreased from 880 ± 97 µA to 540 ± 60 µA (t = 3.9, P < 0.05). Neither preceding administration of 100 nmol/l TTX 20 min before NGF (n = 5, Fig. 5a) nor subsequent intramuscular TTX injection 35 min after NGF caused any modulation of NGF-evoked reflex facilitation. After combined administration of TTX and subsequent NGF reflex integral increased by 88.5 ± 14.3% (F = 12.77, P < 0.001) within 1 h after NGF. Preceding NGF and TTX induced an integral increase of 127.1 ± 24.2% (F = 7.03, P < 0.001) within 95 min after first injection. Statistical comparison of reflex integral changes after application of NGF alone, of combined 100 nmol/l TTX and NGF, or combined 100 nmol/l TTX and saline resulted in a clearly significant difference (F = 15.2, P < 0.001). Whereas combined TTX and saline administration clearly differed from NGF alone (t = 5.5, P < 0.001) and combined TTX and NGF injection (t = 3.5, P < 0.01), the two latter conditions showed no statistical differences.

No effect of tetrodotoxin (TTX) on nerve growth factor (NGF)-evoked reflex facilitation. (a) Percentage changes of reflex integral (mean ± SEM) due to administration of 0.8 µmol/l NGF alone (Δ), combined injections of 100 nmol/l TTX and subsequent 0.8 µmol/l NGF (•), or combined injections of 100 nmol/l TTX and subsequent isotonic saline (saline; □). Δ, NGF alone (n = 5), P < 0.001; •, 100 nmol/l TTX, NGF (n = 5), P < 0.001; □, 100 nmol/l TTX, saline (n = 5), NS. (b) Percentage changes of reflex integral (mean ± SEM) due to administration of 0.8 µmol/l NGF alone (Δ) or combined injections of 0.8 µmol/l NGF and 100 nmol/l TTX (•). All combinations induced significant increase of reflex integral as calculated by one-way repeated measures analysis of variance. Δ, NGF alone (n = 5), P < 0.001; •, NGF, 100 nmol/l TTX (n = 5), P < 0.001.
Discussion
The present data demonstrate prevention and reversal of ATP-induced facilitation of brainstem nociception by topical administration of TTX into semispinal neck muscles in anaesthetized mice. In contrast, TTX was not able to affect NGF-evoked facilitation. This divergent effect of TTX suggests different mechanisms of facilitation after intramuscular injection of ATP and NGF.
Nociceptive processing in the brainstem can be assessed by the JOR. This brainstem reflex can be elicited by electrical, thermal and mechanical stimulation of the craniofacial region (32–37). Primary trigeminal afferents synapse on excitatory sensory neurons of the spinal trigeminal complex. These neurons project ipsilaterally and contralaterally to excite digastric motoneurons (38, 39). The reflex is a commonly accepted model to investigate alterations of excitability in sensory brainstem neurons with convergent afferent input from different craniofacial tissues such as neck muscles. Sustained reflex potentiation by noxious input from neck muscles, evoked by ATP or NGF, indicates heterosynaptic facilitation due to access of nociceptive muscle afferents to the reflex neural network in the brainstem (12–14).
TTX is known to block action potential generation and propagation in sensory neurons (25, 40, 41). Voltage-gated sodium channels (Nav) transmit electrical signals through action potentials from peripheral endings of primary sensory neurons to the central nervous system. Ten isoforms of Nav channels have been cloned from various tissues, and at least seven are known to be expressed in the mammalian nervous system. Whereas channels Nav1.1, 1.2, 1.3 and 1.6 are preferentially located in the central nervous system, channels Nav1.7, 1.8, and 1.9 are more frequently found in peripheral neurons (40, 42). Nav1.7 is located on many different nociceptive and non-nociceptive primary sensory neurons (25, 42, 43), Nav1.8 shows higher expression in small-diameter neurons (25, 42, 44) and Nav1.9 is predominantly present in small-diameter nociceptive neurons that give rise to unmyelinated axons (25, 42, 45). From these Nav channels located on sensory neurons, TTX is able to bind to α-subunits of Nav1.1, 1.2, 1.3, 1.6 and 1.7 and to block sodium currents highly effectively. In contrast, Nav1.8 and 1.9 are resistant to TTX blockade (25, 40, 42).
In rat dorsal horn ganglia, neurons have been classified according to conduction velocity and action potential duration using intracellular microelectrode recording (41). Electrically evoked action potentials in large Aα/Aβ neurons or medium-sized Aδ neurons were always or partially blocked by TTX, whereas action potentials in all C cells were TTX resistant. Thus, in vitro electrophysiological data and sodium channel pharmacology argue for a close link between TTX-resistant NaV channels and nociceptive C neurons and suggest TTX as an appropriate drug in order to differentiate between A and C fibre-mediated actions (40, 41).
In rat saphenous nerve, local administration of TTX differentially affects action potential propagation in A and C fibres with IC50 of 57 nmol/l and 372 nmol/l, respectively (27). With concentrations of TTX solution up to 100 nmol/l, compound action potentials of C fibres are unaffected, whereas potentials in A fibres drastically decrease by about 70%. In decerebrated rabbits, application of 30 nmol/l TTX to the desheathed sural nerve completely blocks A fibre potentials and reduces C fibre potentials by about 40% (28). Experimental studies in cats seem to confirm selective blockade of neural conduction in A fibres by TTX (29, 30, 46, 47). Corresponding to published experiments in rat saphenous nerve, the present study in mice applied TTX in concentrations ranging from 30 to 100 nmol/l.
Divergent effects of TTX on NGF- and ATP-induced myofascial nociception may be due to differential excitation of group IV (corresponding to C) and group III (corresponding to Aδ) fibre afferents in neck muscles by these substances. On the one hand, NGF primarily interacts with petidergic neurons bearing trkA receptor and on the other hand ATP interacts with P2X receptors probably located on IB4-positive non-peptidergic neurons containing fluoride-resistant acid phosphatase (23, 24). The excitatory effect of NGF on nociceptors involves sodium currents which are resistant to TTX. NGF enhances nociceptive afferent excitability by exerting a heavy influence on the gating of TTX-resistant sodium current (25, 26). Recent experiments applying ATP in neck muscles have demonstrated mediation of sustained brainstem effects by P2X receptors (12). Immunohistochemical studies in primary sensory neurons supplying pericranial muscles in rodents have indicated a larger impact of heteromeric P2X2/3 receptors than homomeric P2X3 receptors (48). P2X2/3 receptors are known to be primarily localized on medium-sized sensory neurons (22, 49, 50). Thus, mediation of ATP-effect by P2X2/3 receptors may correspond to the blockade of this effect by TTX that predominantly affects A fibres.
In summary, modulation of neck muscle nociceptive processing by local TTX administration suggests different mechanisms of intramuscular NGF and ATP. Both models of neck muscle pain may be useful for future studies addressing pathophysiological mechanisms of pericranial pain and tension-type headache.
Acknowledgements
The authors appreciate the technical assistance of Claudia Panfil and Andreas Isaak. This research project was supported by grants of the German Headache Consortium (Federal Ministry of Education and Research, 01EM0516, project A3) and the Interdisciplinary Centre for Clinical Research BIOMAT of the Medical Faculty of RWTH Aachen University.
