Abstract
In a large retail business group the ID Migraine Screening Test was sent to employees with three or more absences from work in the past year (n = 2893). Employees with positive results were invited for a neurological consultation and migraine patients were randomly assigned to: first attack ‘treated as usual’ and the second attack treated with 40 mg eletriptan, or reversed order. Of the 2893 employees, 799 responded (28%), 260 were positively screened for migraine (33%), 84 patients were diagnosed by a neurologist and 41 of the 75 included patients completed the protocol. Eletriptan induced pain-free response in 33.3% of the patients at 4 h compared with 0% after ‘non-specific’ treatment (P = 0.03). Eletriptan also significantly improved quality of life, but differences in absence from work and productivity loss could not be detected. In conclusion, in-company screening can be beneficial for undertreated employees, but implementation obstacles can reduce the effectiveness of screening.
Introduction
There is considerable evidence that migraine is associated with reduction in quality of life and productivity loss in employees (1–8). The availability of triptans since the early 1990s has been a significant advance for patients (9), and their efficacy in aborting a migraine attack has been proven (10, 11). Although migraine is burdensome for work performance, many patients do not seek adequate medical treatment (12). The relevance of a two-step approach, in which a first step of screening migraine patients is followed by a second step of prescribing of migraine-specific medication, has also been demonstrated (13, 14). A number of screenings instruments have been constructed in order to select patients who might benefit most from migraine-specific treatment, such as the Migraine Disability Assessment (15), the Migraine-Act (16) and the ID-Migraine Screening Test (17). The last is a three-item screener that is easy to use for migraine recognition in primary care (see Table 1). These findings suggest that both employees and their companies might benefit from interventions aimed at improving migraine recognition and thereby reducing the burden of migraine in the workplace. However, this assumption is not supported by a firm body of empirical evidence obtained in daily practice. Burton et al. reported in a review nine pre-post test trials, but patients were not randomized, nor were results compared with a control group (18). The median days of productivity loss per patient per month was 0.67 days for triptan users and 1.69 days for non-triptan users. Kwong et al. reported lower migraine-associated productivity loss due to sumatriptan, but the results were obtained in a clinical setting and not through an intervention organized at the work floor (19). Vicente-Herrero et al. organized a trial on the work floor, but the pre-intervention comparison data were collected retrospectively and therefore liable to recall bias (20). They found that migraine-specific treatment led to a mean reduction of €20 (£15.90) in productivity costs per attack. The aim of our study was therefore to test the feasibility and effectiveness of screening for migraine on the work floor, and subsequently providing appropriate pharmaceutical treatment. Two issues were particularly relevant: (i) how many and which type of patients would participate in this real-life setting on the work floor; and (ii) which participants may benefit and which not? The primary end-point of this study was headache response as defined by the percentage of patients pain free 4 h after treatment. Secondary aims were to reduce productivity loss and the migraine-specific burden in quality of life. It was hypothesized that employees who normally treated migraine with over-the-counter medication only or used no medication at all (the ‘non-specific’ group) would benefit most from the intervention (12).
The ID migraine screening test

Methods
Patient inclusion
A crossover multicentre effect study was implemented in Maxeda, a large retail business group in the Netherlands with 30 978 employees. The screening focused on those employees with three or more periods of work absence (≥ 1 day) in the last year. Von Korff et al. found in their population-based diary study that 20% of migraine patients accounted for 77% of the days lost at work as due to migraine (8). This group also explained 50% of the days with reduced work productivity. Therefore, it was expected that the group with high absenteeism would contain those migraine patients, who made the largest contribution to productivity loss. Another important reason to restrict the screening procedure was the high prevalence of migraine. It was anticipated that company-wide screening would result in an unmanageably high response. For example, if the prevalence of migraine is 10% and half of the patients would respond to the screening (n = 2000), and the ID-Migraine Screening Test would detect 80% of the patients (sensitivity is 0.81) (17), then > 1500 medication subscriptions would be needed. This amount and sample size was not needed for answering the questions of the study.
After the Board of Directors from Maxeda agreed to the study, the research group first wrote an article in the company's magazine to announce and explain the study. Next, company doctors sent the ID-Migraine Screening Test (13) to employees who were recorded in their system as having had three or more periods of work absence in the last year. When it was known from the records that a life-threatening disease or a stressful life event caused the absenteeism, the employee was excluded. Ethical approval was obtained from the Medical Ethical Committee of the Erasmus University Medical Centre and the works council of Maxeda. Data collection took place from September 2005 until May 2007.
Responders completed the three items of the ID-Migraine and answered the question whether a neurologist was currently treating them. A positive result on the ID-Migraine Screening Test was determined if two of the three questions about the last two headache episodes during the last 3 months were answered positively (17). Responders with positive test results were invited for clinical consultation with a local neurologist specialized in headache (number of participating neurologists, n = 18), provided that a headache specialist was not treating them at the time. Exclusion criteria for participation in the study were: (i) no migraine according to the criteria of the International Headache Society (IHS) (21); (ii) no distinction possible between migraine and other headaches; and (iii) contraindication for medication. If the neurologist considered a new migraine-specific treatment advisable, patients were randomly assigned to a condition in which the first attack was ‘treated as usual’ (see Table 2) and the second attack treated with eletriptan (condition A), or to a condition with the reversed order (condition B). Patients were treated for a single migraine attack with eletriptan 40 mg and were allowed to take a second tablet if the first dose was ineffective. The randomization was determined by a random sequence table and the neurologist handed the concealed envelop with condition A or B, study medication and questionnaires to the patients. Eletriptan is a potent and selective 5HT1B/1D agonist, whose efficacy, onset and patient acceptability has been proven very effective (22, 23).
The treatment as usual of the selected patients

The feasibility of the study approach was evaluated at the end of the study by a semistructured telephone interview conducted by the second author. The participating neurologists were interviewed on the following topics: logistics, study procedure, workload, patient satisfaction and clinical relevance.
Outcome measures
Pain was measured on a four-point Likert scale as defined by the guidelines for controlled trials of drugs in migraine (24). The primary end-point of the study was pain-free response, operationally defined as the number of patients with a change in pain intensity 4 h after the time of intervention (either eletriptan or ‘treatment as usual’) from moderate or severe to none. A 4-h unit was chosen instead of the usual 2 h, since this reflects a common unit in the workplace (half a working day).
Secondary end-points were designed to assess the effectiveness on work productivity, other headache outcomes and quality of life:
Productivity loss: assessed by PRODISQ version 2.1, Productivity and Disease Questionnaire (25, 26). Absence was measured by a question that asked about absence from work in the first 2 days after a treated migraine attack. Productivity was measured by the quality and quantity of work in the first 2 days after a migraine attack. Employees assessed their work performance on a 10-point scale compared with a regular workday.
Pain relief: the percentage of patients with a change in pain intensity from moderate or severe to mild or none.
Pain reduction: the raw difference score on the pain scales before and after treatment.
Quality of life: migraine-specific quality of life was measured by the 24-h Migraine Quality of Life questionnaire (27, 28) which consists of 15 items measuring five domains that are affected during a migraine attack, i.e. work functioning, social functioning, energy and vitality, migraine symptoms and feelings and concerns (27, 28). Patients answered the questions 24 h after the onset of a treated migraine attack. Each question was answered on a seven-point scale, therefore the maximum score per domain (three items) was 21, indicating best health-related quality of life.
Data analysis
Effectiveness was determined by testing the difference in outcome between the ‘treatment as usual’ and treatment with eletriptan. According to our hypothesis the experimental group was the group of patients that did not use medication or only analgesics to treat migraine. This group was labelled as the ‘non-specific’ treatment. Patients who were already using a triptan as usual treatment were analysed separately, as this group was not the primary interest of the study. For the primary outcome ‘pain free’ the number of patients per treatment group was calculated and differences between the ‘non-specific’ treatment and eletriptan were tested using Wilcoxon's signed rank test. The secondary headache outcome pain relief was analysed with Wilcoxon's signed rank test, difference in pain reduction was analysed by T-tests and T-tests adjusted for order effect were applied for quality of life (29). Wilcoxon's signed rank test was also applied to compare absence from work in the first 2 days after treatment (‘non-specific’ or eletriptan) and, as such, absence was tested within subjects. Productivity was analysed by multiplying the scores of quality of work by quantity of work, as described in the manual of the PRODISQ questionnaire (26). For example, a quality of 60% compared with a regular workday and a quantity of 60% compared with a regular workday constitute a productivity of 36%. Again, Wilcoxon's signed rank test was used to test the differences. The norm for a relevant effect size was determined at 0.30 on the basis of two effect (cost-) effectiveness studies, and 276 patients were necessary to provide sufficient power (α, two-sided = 0.025, β= 0.10) (30, 31). Given an expected 10% of drop-outs a total of 310 patients were needed. The results are presented of patients who completed both questionnaires and thus adhered to the study protocol. In all cases, P-value of 0.05 was chosen (two-sided). The interviews were analysed by grouping the positive and negative aspects as reported by the neurologists.
Results
Patients
Company doctors sent out screening tests to 2862 employees and 799 responded (28%). Of the 260 responding employees screened positive (33%), 128 (49%) agreed to consult a neurologist and 84 (32%) were actually diagnosed. Of the latter group, 75 patients could be included in the trial and 41 completed two questionnaires according to the protocol (see Fig. 1, flow chart). The drop-out analysis showed that there were no significant differences between completers and non-completers on demographic, socioeconomic or headache outcomes. The characteristics of the patients that entered the trial are described in Table 3. In the ‘non-specific’ group 16/17 were female, mean age was 41 years, mean net income was €1204 (£957) per month and patients were on average absent for 3 days in the past 3 months. Furthermore, nine patients did not use any medication and eight used only analgesics to treat an attack.
The characteristics of patients who entered the trial and completed the protocol

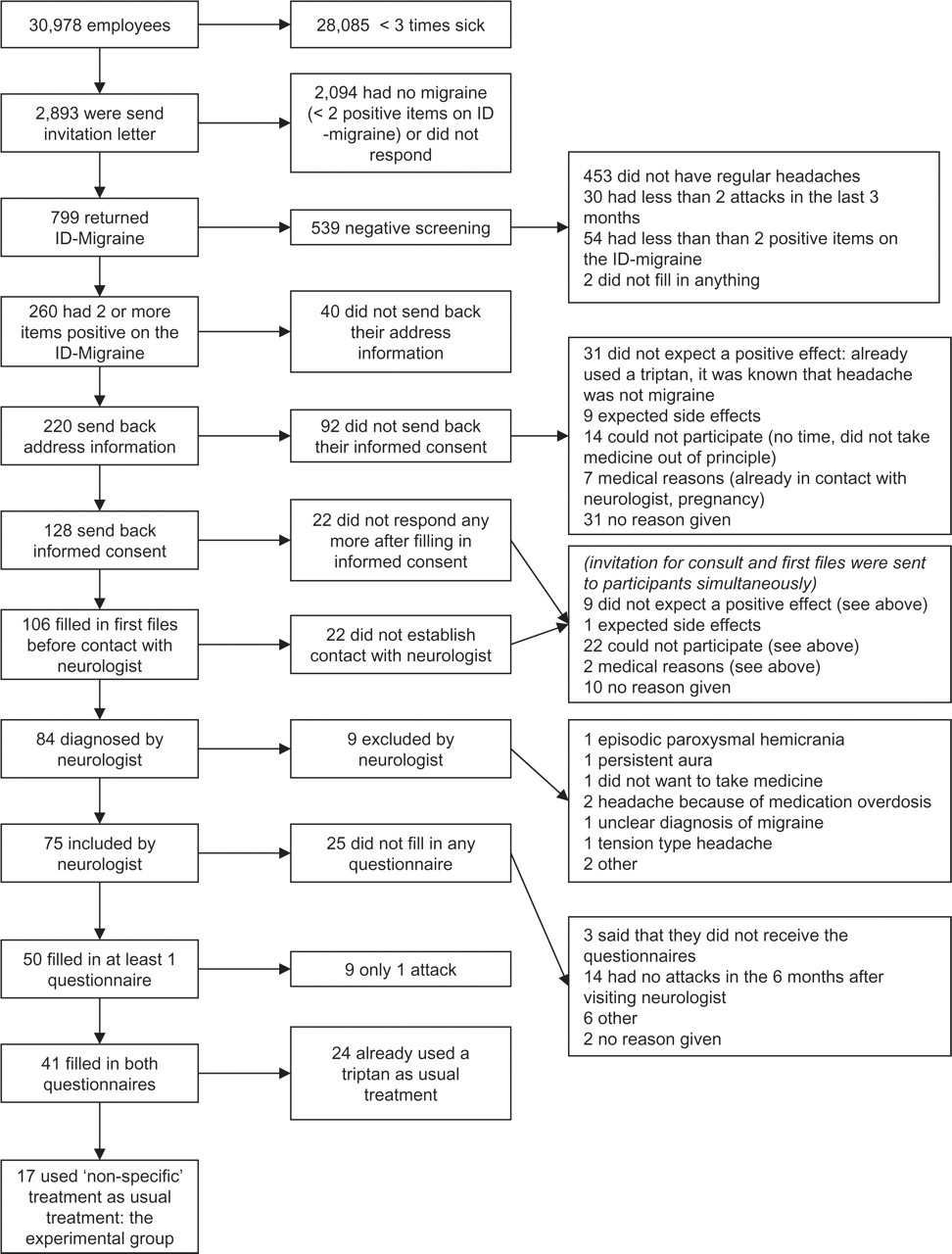
Flow chart.
Implementation obstacles
During the screening, a number of unexpected problems occurred in different divisions of Maxeda when employees with three or more periods of work absence in the last year were screened. First, a minority of the human resources departments included from their files only those with ‘absenteeism probably caused by migraine’ instead of excluding only those with general absenteeism caused by stressful life events or life-threatening diseases. As a result, these departments hardly send out any invitation letters. A second problem was that in some of the larger divisions absenteeism was registered inadequately: if an employee got sick during the working day and returned home earlier, possible subsequent absenteeism the next day was registered as a second period of absenteeism. The personnel departments of the two largest divisions tried to compensate for this flaw by tightening their inclusion criteria: they autonomously decided to change the selection criterion to ‘more than four absences’ instead of ‘three or more’. Obviously, this was a deviation from the research protocol. This affected 71% of the employees of the company. For this reason, the ‘correct’ query was run again in the two largest divisions, which represented 68% of the employees. All the included employees were sent the invitation letter again, which means that many of these employees received the letter twice.
Effects
Table 4 presents the effects on the patients (n = 17) that did not use migraine-specific treatment. As can been seen, the number of pain-free responders after treatment with eletriptan was significantly higher than after their ‘non-specific’ treatment (33% vs. 0%, P = 0.03). Eletriptan also improved quality of life significantly for the subscales ‘feelings and concerns’, ‘energy and vitality’ and ‘social functioning’ compared with ‘non-specific’ treatment. Table 5 shows the results on productivity; in both groups four patients were sick on the first day after a migraine attack, while on the second day one patient was sick after use of eletriptan vs. two patients after ‘non-specific’ treatment. Because one-third of patients were free from work, the sample size was too small to detect any possible differences. Productivity was on the first day in favour of the ‘non-specific’ treatment and on the second day in favour of eletriptan. In patients who were already using a different triptan, there were no statistically significant differences when using eletriptan (see Table 4).
Headache response and quality of life after the ‘non-specific’ treatment vs. eletriptan and the ‘other triptan’ vs. eletriptan

∗ P < 0.05.
†Two missing values.
‡One missing value.
Absence and productivity on the first 2 days after a migraine attack with the ‘non-specific’ treatment vs. eletriptan

Feasibility
Fifteen of the 18 neurologists participated in the evaluation, and their comments were mainly focused on the logistics and length of the study. They had expected to include more patients. There was a great delay between the response of employees to the invitation for clinical consultation and the actual visit to their clinic. Neurologists would also have liked more frequent and rapid feedback by the researchers. Positive factors that were reported included that all patients were satisfied that their headache and work-related problems received serious attention. Employees were often underdiagnosed and some thought that they suffered from tension-type headache. The workload for neurologists was minimal and they all wanted to participate in future projects. The clinical relevance was confirmed, as employers did not have sufficient knowledge and understanding of headache disorders.
Discussion
Summary
This study was an open randomized trial comparing eletriptan with ‘usual treatment’ on the work floor. The study showed that an in-company screening programme reached fewer migraine patients than was expected on the basis of prevalence figures. However, the small group who consulted a neurologist and did not use migraine-specific medication benefited from the intervention. Their headache response was significantly improved as well as the quality of life in the 24 h after a migraine attack. This study could not show that absenteeism and productivity loss were reduced. Furthermore, implementation obstacles in the company reduced the potential sample size and consequently the impact of large-scale screening.
Strengths and limitations
A strong point of this study was the effort to test the effects of a screening programme in a real-life situation on the work floor. The screening was indeed beneficial for the group of undertreated employees. In addition, the number needed to be screened to treat one patient adequately was 1 to 579 (n = 2893 were screened with an absolute difference in pain-free response of 5). Although this number seems high, it is in line with the results of other screening programmes (32, 33). Both neurologists and patients were positive about the genuine attention to migraine in the workplace, as they often experienced a lack of understanding of headache disorders. Despite these positive outcomes, the study design did not provide the desired response rate and protocol adherence. In this section we will discuss several factors contributing to the low response rate and protocol deviations. Regarding the low response rate, the following comments can be made. The primary response rate, defined as the reaction to the invitational letter, was only 28%, and this was certainly a limitation of the study. In the present design, the research group did not select and approach the patients themselves, but relied on the human resources departments of the (sub-) companies. This design was chosen because the Medical Ethical Committee required that the initiative for participation belonged to the patients, not to the researchers. This is understandable from the perspective of having an autonomous choice for study subjects, but inevitably leads to losing potential participants who feel any threshold to take action. In addition, patients had to take the initiative on several other occasions. For example, employees had to read, fill in and send in the screening questionnaire on their own. Without assistance, employees then had to understand from the accompanying letters that the screening was not meant as a check or control of the company physician, but as an effort to help the employee in an academic trial setting. This complex and indirect way of approaching patients using the infrastructure of the company might have contributed to the lower than expected participation. Also, this study may have been seen as a drug trial, instead of a trial promoting screening and state-of-the-art treatment of migraine. As drug trials may suffer from negative connotations, this might have lowered the motivation to participate.
Another limiting factor was that the screening procedure was restricted to employees with known high absenteeism. The motivation for this restriction was the expected high prevalence of migraine and the wish to study the influence of the screening programme on productivity loss. However, the numbers turned out much lower than expected: only 3% (84/2893) of the workforce with high absenteeism could be diagnosed as having migraine. This finding questions the claim that migraine is a major cause of frequent absenteeism, the same assumption that made us choose to select only employees with three or more periods of work absence in the last year. Many migraine sufferers might have had fewer than three periods of work absence in the last year, which would imply that many did not receive an invitational letter in the first place. We think that our investigation contributes to the cumulative evidence that migraine indeed interferes with work, but it is probably not a major cause of absenteeism (34, 35). However, in the past and during the design of this study, the link between migraine and absenteeism was seen as much more substantial. This line of reasoning was based on the often reported 10–12% 1-year prevalence of migraine in the general population (36). Moreover, it is estimated that the direct costs of migraine are considerable (37) and the prevalence in the working force population is high (18% of the Dutch population has migraine in the age category 25–44 years and 14% in the age category 45–64 years; Statistics Netherlands). This would mean that in a large company such as Maxeda with > 35 000 employees a response rate of 50% could lead to an overwhelming response of 2800 (50% response of 16% prevalence of 35 000). Also for this reason, we selected migraine sufferers with high absenteeism. Finally, the many part-time workers within the retail business make it possible to change days with colleagues, and employees might also compensate for migraine episodes in their leisure time.
Regarding the protocol deviations, the most cumbersome difficulty was the deviation from the selection protocol by some of the human resources departments. As a result of this, it became impossible to compute exactly the percentage of responders on the screening test. It was not exactly clear how many employees received the invitation letter, as the employees might have joined and left the division between the two mailings. Furthermore, we did not know from which division the participants came, as the works council asked us not to collect that information because of privacy reasons. The interpretation of the amount of response on the invitation letters with the screening instrument (the second box in the flowchart) must therefore be seen as an estimate. Another protocol deviation was the drop-outs during the trial phase. Of the 128 positively screened patients who gave informed consent, only 84 (66%) were actually diagnosed by a neurologist. This lower consultation rate might have been caused by the fact that the neurologist was unfamiliar with the employee. Obviously, this is often the case when visiting a specialist, but normally, especially in the Netherlands, this reference is given face-to-face by a known professional, such as the general practitioner. Also, of the 75 included patients, 41 completed the study protocol (55%). This drop-out rate was caused mainly by migraine itself, which was of a too low frequency to appear twice during the study time (23/75 patients).
Despite the lower than expected group of participants in the clinical trial, positive effects of eletriptan were found for pain and several quality of life effects in undertreated patients (‘non-specific’ users). We could not detect an effect on productivity as others have found (18). Although our design was different in the sense that we tested the effect of screening on the work floor instead of in medical wards, we think that this difference does not explain why we were not able to reproduce these results. We do think that we are dealing here with a lack of power for this measure. First, the final response was much lower than expected. Second, we measured only one attack per patient per treatment, whereas other designs such as that of Kwong et al. measured several attacks before switching to the alternative treatment (19). Of note, effects on pain-free response and quality of life were found, suggesting that sufficient power for productivity losses relates to higher inclusion rates then the usual outcomes in migraine research (38).
Implications of the findings
Our study design was based on the assumption that many undertreated migraine patients would be reached through screening on the work floor and that the patient could be easily motivated to consult headache specialists, who would provide the best quality for their migraine diagnosis and treatment. However, the complexity of the logistics and indirect ways to approach the patients caused a high threshold to participate and hampered compliance. The following remedies might be considered to prevent the difficulties encountered in this type of study. One could remove the hurdles for participation by implementing a broad screening procedure, in which all employees who suffer from migraine are invited to participate (38). This invitation might also be more appealing when placed in a company periodical or through information on their website. Further, in accordance with the scientific guidelines of the IHS, migraine should be of sufficient frequency, i.e. at least once per month.
Another solution might be to approach employees more directly. First, screening could be conducted on site, including the presence of a researcher at the company to assist and to give a face to the unfamiliar and abstract trial setting. Second, the company doctor could personally invite the positively screened patients. In this way the stepped care approach will be extended to three levels. After the first step of screening, the company doctor could diagnose the patients and prescribe the migraine-specific medication. The third step would be referral of the complex patients to headache specialists. Another remedy to increase the cooperation of employees may be to educate human resource managers and executives on headache disorders and effective treatments. Some employees were not able to visit the neurologist during working-time, but had to take a day off. If the managerial board were better informed, employees would feel more supported, and this approach might also lower the threshold to ask for assistance. Last, recruiting the Trade Union may be a fruitful strategy to enhance the credibility of the call.
Recommendations
Given that in this trial setting, screening and state-of-the-art treatment did have an effect on quality of life, we would suggest that further investigation focuses on ways to narrow the social distance and shorten the procedure. One way to achieve this is to avoid the trial setting and implement the ID-Migraine Screening tool applied by a general practitioner or the company doctor at an early stage. The response rate may also be increased when the screening procedure is standardized, as in our investigation of children with chronic pain, where the screening is part of the standard screening procedure of the municipal Youth Health Service in Rotterdam. Incentives such as a free half-day to participate might facilitate the cooperation of employees also. If one is to provide more evidence that improving treatment of migraine reduces productivity loss, a new randomized trial will need a large number of patients. In any case, focus on productivity loss on the work floor might be more rewarding, as absenteeism might not be the most common reason during and directly after a migraine attack.
Conclusions
In-company screening may reach fewer migraine patients than expected, but can improve treatment and quality of life for those employees who are not using migraine-specific medication. Adequate screening and implementation might be improved by implementing a broad screening procedure and using a more direct and simple approach for migraine-specific treatment. It is also recommended that general practitioners and company doctors screen employees with headache complaints for migraine at an early stage and refer complex patients to a neurologist.
Footnotes
Acknowledgements
Pfizer supported the research with an unrestricted grant. We thank T. J. Steiner for his constructive comments on the manuscript. The opinions expressed and the remaining flaws remain the responsibility of the authors. The authors thank the Dutch Society of Headache Centres for their valuable cooperation, in particular the neurologists J. W. M. ter Berg, J. S. P. van den Berg, M. A. M. Bomhof, P. A. D. Bouma, C. A. J. Broere, J. P. ter Bruggen, P. A. T. Carbaat, A. J. H. van Diepen, M. F. Driessen-Kletter, P. Eekers, J. D. M. van der Meulen, W. M. Mulleners, M. Padberg, P. H. M. Pop and J. L. van der Zwan. Maxeda is thanked for their willingness and cooperation to implement the screening procedure.
