Abstract
Introduction: Specific problems occur in clinical treatment trials for migraine with aura that differ from those encountered in treatment trials for migraine without aura.
Discussion: Based on our experience with four such trials, we point to a number of possible solutions and outline areas for future inquiry. We make recommendations about subject selection; the choice, definition and assessment of outcome measures; optimal treatments in relation to aura and headache; and we provide samples of study report forms used to record occurrence of aura and headache in this population.
Introduction
The International Headache Society via its committees on the methodology of drug trials has published extensively on the methodology of drug trials in migraine, tension-type headache (TTH) and cluster headache. Guidelines have been provided both on studies of the treatment of acute attacks and on the methodology of preventive drug trials (1–5). While recommendations for migraine have greatly improved the methodological quality of such drug trials, they have failed to recognize a number of issues specific to migraine with aura (MA). The reason for this is primarily that participants with MA often have attacks of migraine without aura (MO) in addition to MA attacks and that aura and headache are two different possible outcome parameters. Furthermore, many participants experience some attacks of aura followed by a migraine headache, some followed by a non-migraine headache and some aura without subsequent headache.
We have previously completed two drug trials (6,7) of the preventive treatment of MA and three drug trials of acute treatment for MA (8–10). In addition, we have gained experience from designing an ongoing study of the acute treatment of MA and from our extensive epidemiological and nosographic studies of MA (11,12). These studies have made it clear to us that a number of methodological problems are specific to MA, and that these have not been addressed in the previous publications of the International Headache Society (IHS) clinical trials subcommittee on drug trial methodology. The aim of the present paper is to direct attention to these specific problems related to studying MA and to present and analyze these problems in the hope that this may improve drug trials in MA in the future and perhaps lead to an official paper from the methodology committee.
Distinction between attacks of migraine with or without aura in clinical trials
While a few studies have tested drugs for MA exclusively (6–10,13–15), the overwhelming majority of studies, for both acute and preventive treatment, have examined the effect of drugs in a mixed population of participants having either migraine with or without aura or both (16). In the mixed MA-MO population, only approximately 25% of the participants on average have MA (17). Furthermore, the participants with aura are usually not selected for their frequency of MA attacks but for their frequency of MO attacks, the MA diagnosis being of minor importance. The effect of a drug on MA attacks may therefore be “drowned” in the effect (or lack of effect) on MO attacks. Whether these two types of attacks occurring in the same patient respond similarly or differently to drugs is impossible to tell unless each attack is separately classified as MA or MO, as discussed later in the present paper. Recent studies with tonabersat have shown the importance of clearly distinguishing between the two, because attacks of aura (with or without a headache) were statistically significantly reduced (7) whereas attacks of migraine without aura were not (18).
Diagnosis, classification and recording of outcome parameters
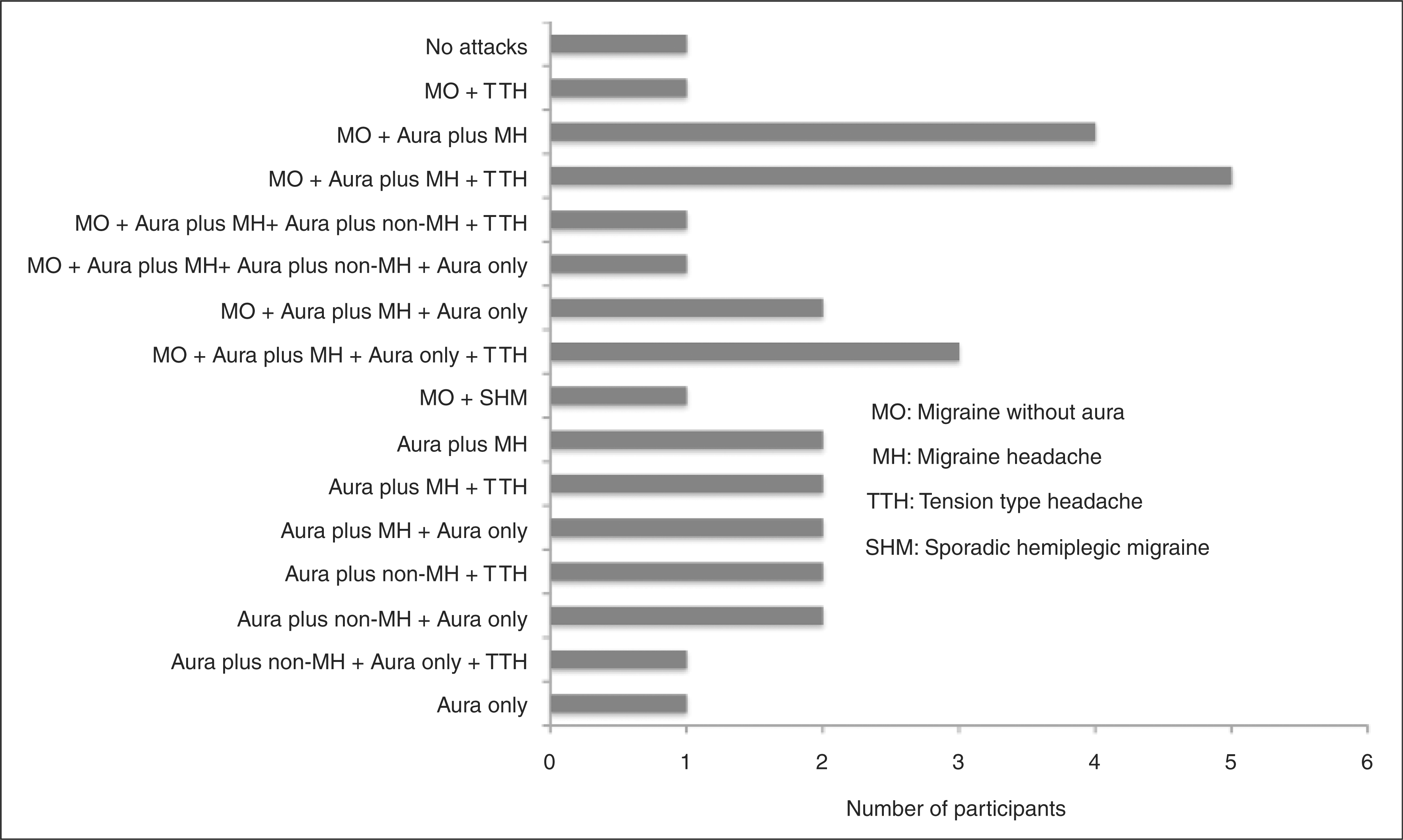
The International Classification of Headache Disorders, second edition (ICHD-II) (19), subdivides group 1.2.1, migraine with a typical aura, into aura followed by migraine headache, aura followed by non-migraine headache and aura not followed by headache. Our epidemiological/nosographic studies have demonstrated that many MA participants do not exclusively have one of these subtypes of attacks (11,12). They have some attacks followed by migraine headache and/or some attacks followed by non-migraine headaches and/or some followed by no headache at all. In drug trials of mixed populations of MA and MO patients, it has not been customary to record each individual attack in a way that makes it possible to distinguish between MA and MO attacks, not to speak of the different subtypes of MA. In our previous study of the preventive effect of tonabersat in MA, number of auras was a main outcome parameter. We therefore designed a headache diary in which the patient recorded all necessary characteristics to classify migraine with or without aura and to further subclassify MA attacks according to the aura symptoms and the subsequent headache (Figure 1). The participants included in this trial were recruited from an outpatient headache clinic and from participants identified for previous genetic research projects. Participants should have one of the six subtypes of migraine with aura. The coexistence of MO and/or TTH was allowed. None of the participants actually included had a previous history of familial hemiplegic migraine (FHM) or basilar-type migraine. The number of different types of attacks recorded is presented in Table 1, and the distribution of participants according to the types of their attacks is presented in Figure 1. It appears from the data shown in Table 2 that the most frequent type of attack in these participants was 1.2.1 (typical aura with migraine headache), whereas 1.2.4 (FHM) and 1.2.6 (basilar-type migraine) were not recorded at all during the study. MA attacks constituted 73% of the attacks recorded and MO attacks constituted 27% of the recorded attacks. Thus, despite selection of participants with a high aura frequency, more than one-quarter of the attacks recorded were attacks of MO. Twenty-nine of 31 participants recorded at least one MA attack, and 12 of these recorded at least two different subtypes of MA. Looking at the classification of participants on the basis of their actually recorded attacks (Figure 2), in combination with previous epidemiological studies (11,12), the wisdom of subdividing participants into 1.2.1, 1.2.2 or 1.2.3 in the ICHD-II is questionable.
Diary for a preventive drug trial in migraine with aura. h = hour; m = minute; 0 = not present; 1 = mild; 2 = moderate; 3 = severe. Combination of different migraine subtypes and tension-type headache recorded in the diaries in the tonabersat study. MO = migraine without aura; MH = migraine headache; TTH = tension-type headache; SHM =sporadic hemiplegic migraine. Number of different types of attacks recorded in patient diaries, tonabersat study Only one patient.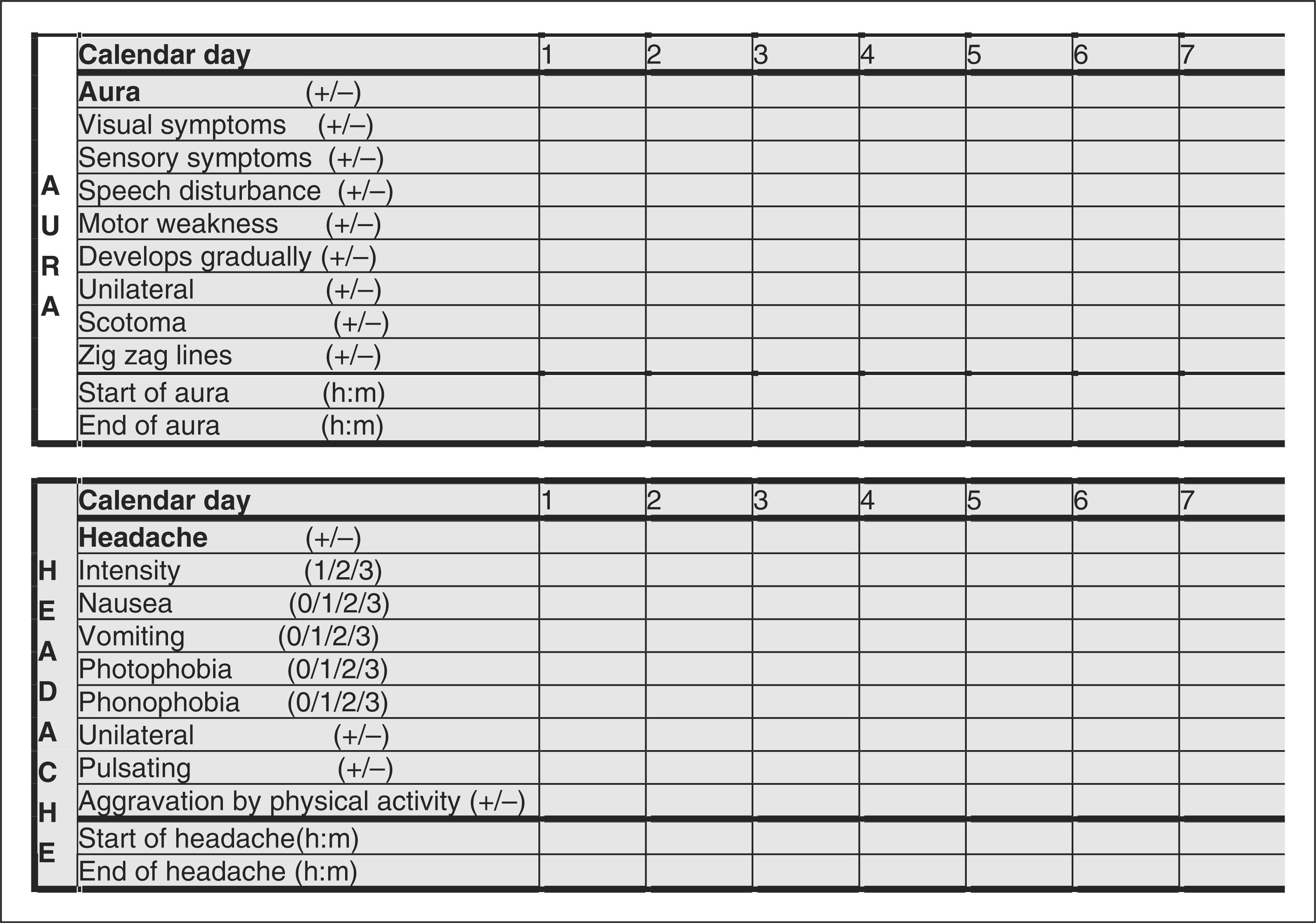

Drug trials dealing with acute treatment
Outcome parameters
In trials evaluating the acute treatment of MA, it should be clearly stated if the aim is to abort or reduce the length of the aura or to reduce or eliminate the headache. In the first case, the drug should be administered just at the onset of aura and by the fastest possible route, as the aura only lasts about 20 minutes in most participants. The main outcome parameter should be duration of the aura. In trials where the effect of treatment on pain is the main question to be answered, the main outcome parameter should be freedom from pain or pain relief at two hours after dosing, or freedom from pain or pain relief 2–24 hours after drug intake. The drug should ideally still be taken at the onset of aura. However, some drugs, like the triptans, are probably not effective taken during the aura (9,10,15), and participants should in that case dose at the onset of pain but the outcome parameter should remain freedom from pain or pain relief at two hours after dosing, or from 2–24 hours after dosing.
Design of the study
There is nothing special regarding design. Both the parallel group and the cross-over designs are applicable. As individuals with many auras are rare, the cross-over design may be preferable because of its superior power (20).
Participants
Participants must have a diagnosis of one of the subtypes of migraine with aura. Given the genetic mutations found in FHM (21), which separate this subtype from the other subtypes of MA, as well as the difference between FHM and migraine with typical aura with regard to sensitivity to glyceryl trinitrate (22,23) and calcitonin gene-related peptide (24), it seems reasonable to consider excluding hemiplegic migraine (both sporadic and familial). Basilar-type migraine is very rare, but our previous studies suggest that it is part of the MA spectrum and thus could be included (25). Participants included can have attacks of MO in addition to their MA attacks, but only attacks of MA should be treated with the test drug.
In trials concerned with the effect on the aura, selection of participants with lengthy auras is necessary. In our opinion the length of the aura should be no less than 20 minutes, even with inhalation or injection, in order to give the drug time to work.
For trials of the effect on headache, it is important to select participants who usually or always have a migraine headache with duration of several hours following the aura. In cross-over trials both participants with migraine headache and participants with TTH following the aura can be included, whereas these groups would require stratification in group comparison trials.
Because recruitment of participants with frequent attacks of MA can be a limiting factor in these trials, we suggest that participants need no more than one attack per eight weeks in order to be included. Limitations on other headaches are not necessary, because only attacks preceded by an aura are to be treated. Thus, chronic and episodic migraine can both be included.
Baseline period
In acute trials no baseline period is necessary, although it can be useful to have participants fill out the attack report form while treating one attack with their own medication in order to familiarize them with the procedure.
Report forms
A detailed recording of each aura symptom and total duration of the aura is mandatory.
Because most participants have some attacks with aura and some attacks without aura, it is absolutely necessary that the attack report form contains enough information to classify each attack treated with an acute treatment in order to sustain the distinction between MA and MO attacks. This will include specific questions regarding duration of the aura, gradual development of aura symptoms, the presence of scotoma and/or zig-zag lines and unilaterality of the symptoms (26). Unilaterality of symptoms greatly improves the accuracy of the diagnosis (19). It is not enough to diagnose participants as MA and then let them treat any migraine attack that occurs. It must also be documented in the attack report form at which time the patient has taken the drug in relation to the onset of aura and in relation to the onset of headache. We therefore recommend using the report form presented in Figure 3, or something similar.
Attack report form for a trial of an acute drug in migraine with aura.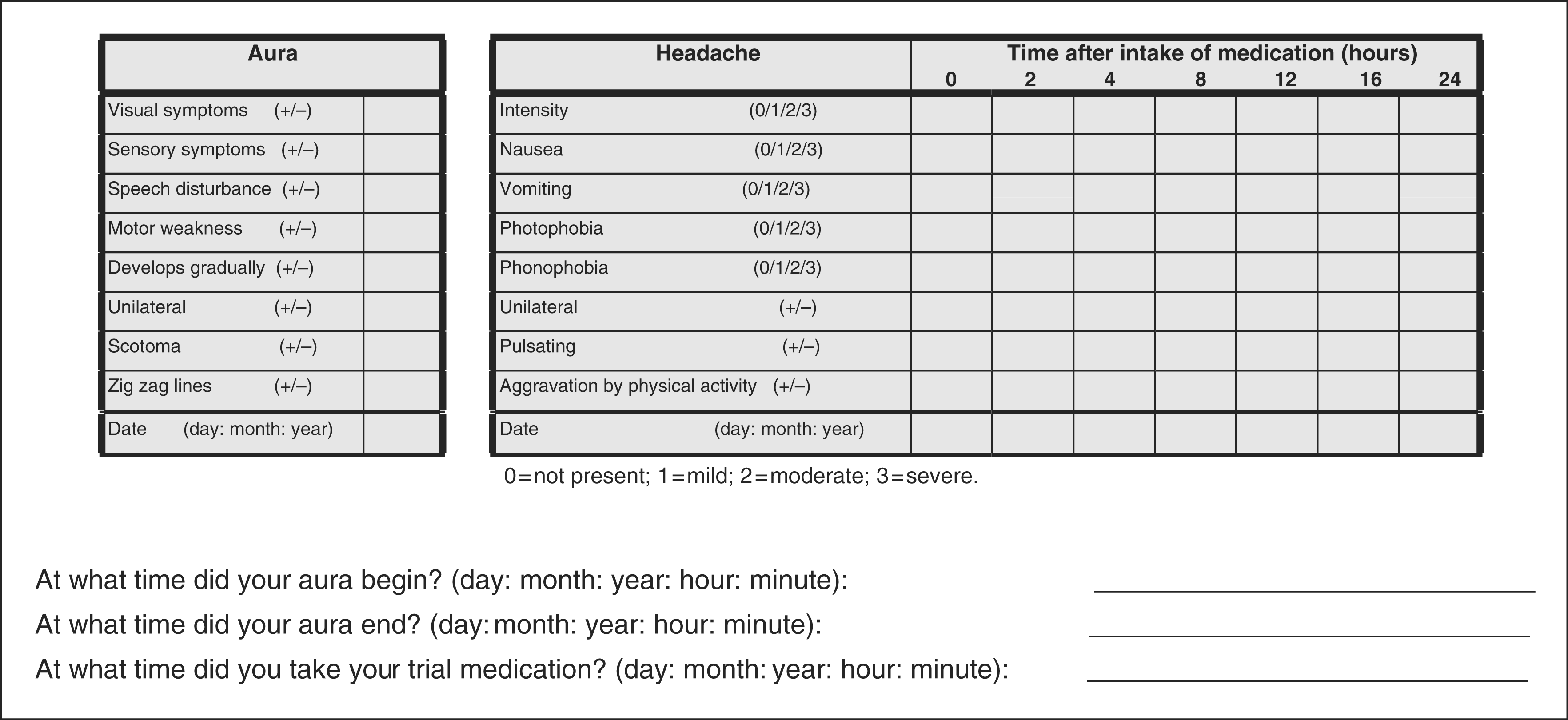
Drug trials dealing with prophylactic treatment
Outcome parameters
In trials evaluating the effect of treatment on MA, the main outcome parameter should be the number of auras. Secondary outcome parameters should be the number of migraine headache days and/or the number of days with any headache. Using an appropriate diary, the number of auras is easy to count. Auras not followed by a headache should be included in the count. In our most recent study, a statistically significant reduction of the number of auras could be shown following tonabersat compared to placebo (7). A migraine headache day is easy to define in MO subjects but more difficult in MA subjects, as discussed later in this paper. We suggest including all headaches following an aura as well as all headaches outside of aura which fulfill criteria for 1.1 migraine without aura (19), including days with headache where the participants take a triptan. Any day with any kind of headache could also be a secondary outcome measure.
Design of the study
In this type of trial, where participants with frequent auras are to be included, we strongly recommend the cross-over design because such participants are rare. The cross-over design achieves the same power as a parallel group design with one-quarter of the participants and hence increases the feasibility of the study (20). Period and carry-over effects, if present, will reduce the power of the study but only exceptionally produce false positive results. In previous trials carry-over and period effects have not been a problem (6,7,27,28).
Participants
With regard to diagnosis, see above under acute trials. According to epidemiological data (11,12), many participants included will have attacks both with and without aura as well as TTH-like attacks. Participants with auras not followed by a headache can be included but will only contribute to the primary outcome parameter (number of auras). Participants must have at least one attack of MA per month.
Baseline period
A one-month prospective baseline recording in preventive trials in migraine is recommended by the IHS Clinical Trials Subcommittee (4). As already mentioned, it is our experience that the attack frequency in MA subjects fluctuates much more than the attack frequency in MO subjects. A one-month baseline period used to exclude participants with too low an attack frequency will result in a number of exclusions due to random fluctuation in attack frequency as well as inclusion of participants who randomly have a high attack frequency during the run-up period. Therefore, a three-month run-up period would be ideal but is rarely feasible. We recommend including participants on the basis of the mean monthly attack frequency during the last three months as recorded by history, even though this makes a comparison between active treatment versus placebo with baseline rather uncertain. On the other hand, such comparison is usually best avoided because comparison between active drug and placebo is always the main outcome parameter.
Duration of the trial
Even though a prolongation of the length of the treatment period could help overcome the difficulties recruiting participants with frequent auras, we find that a three-month treatment period as recommended in MO trials is still preferable because a longer duration will increase the dropout rate.
Report forms (diaries)
Detailed diaries allowing for the classification of each recorded aura and headache according to the ICHD-II are indispensable. We have been satisfied with the diary presented in Figure 1.
Additional suggestions
Based on our experience in the tonabersat trial, we think the following principles are reasonable in conducting treatment trials in migraine with aura.
All episodes of neurological disturbances need to fulfill all the criteria for aura. In the ICHD-II it is recommended that less typical attacks, in participants having a sufficient number of typical attacks to satisfy the diagnosis, are included in attack frequency. In drug trials, where every attack is important for the outcome of the trial, we find that it is reasonable to accept only attacks of aura which fulfill all criteria or all but one criterion (a probable attack).
All aura symptoms must end before a second aura attack can be counted. In order to count as a new attack of aura, all the aura symptoms from the previous attack have to come to an end and this has to be recorded by the patient. An attack of aura lasting for days has to meet the criteria at one time point only.
Any headache following an aura can be considered a migraine headache. An aura can be followed by a headache fulfilling or not fulfilling the criteria of 1.1 migraine without aura. The pathophysiological mechanism triggering such headache is the same whether it fulfils 1.1 or not. All headache recorded in close relation to the onset of aura should therefore be considered as a migraine headache day.
Discussion
Migraine with aura versus migraine without aura
The number of documented differences between MA and MO continues to increase. The concordance between monozygotic twins is higher for MA than for MO (29,30), the increased risk of stroke in migraine individuals is only found in MA (31–35), co-morbidity between migraine and other diseases is in most cases more pronounced in MA than in MO (36,37) and the same is true for MRI abnormalities (38–41). All these findings are relatively new and add to the old observation that these two kinds of attacks are completely different with regard to brain blood flow changes. MA is characterized by pathophysiological changes that very strongly resemble those of cortical spreading depression (42–48). In MO no such changes have been demonstrated (with one possible exception) (49).
Reasons for separating MA and MO individuals in clinical trials
In most previous drug trials, both migraine with and without aura have been included, but since MA represents only a minority of attacks in such trials, it is impossible to conclude that a drug is effective in both types of migraine just because the overall results are statistically significant. Most recently, it has been documented for the first time that a drug (tonabersat) is ineffective in the preventive treatment of MO (18) but nevertheless has statistically significant efficacy against the migraine aura (7). Due to the aforementioned marked differences between MA and MO, we find it advisable in drug trials to study these two types of migraine separately. Safety concerns may also be different in the two types of migraine because of the increased risk of stroke and the marked blood flow changes in MA. So far, all migraine drugs have been tested in mixed populations of MA and MO patients, leading to approval for migraine unspecified. While this is convenient for the pharmacy industry, patient interests as well as scientific interest dictate that the two types of migraine ought to have their own study program. The Food and Drug Administration, or other similar bodies, in the future should not accept a drug for the treatment of MA without specific studies focusing on this disorder.
Issues concerning clinical trials with acute treatment of migraine with aura
In the acute treatment of MA attacks, a number of issues come up. Firstly, is it possible that a drug could be developed that would abort or greatly shorten the aura itself? Such a drug would have to be administered either by injection or by inhalation in order to be immediately available. Furthermore, the drug would have to have a fast onset of action once in the body. No such drug has been formally tested, but in very old case reports, it has been suggested that amylnitrite by inhalation might abort the migraine aura (50). We have tried this in the past, but in our patients it caused such a terrible headache that they were unwilling to inhale the drug again. Besides, our patients reported that it did not abort the aura. Modern technology for inhalation of anti-migraine drugs does, however, make it conceivable that a sufficiently fast action could be obtained if the right drug becomes available. Most likely, a new treatment would, however, be directed against the headache phase of MA. Such a drug would be most useful if it was effective when administrated at the very onset of the aura. In previous studies the triptans were probably not effective dosed in this way (9,10,15). Future acute migraine drugs should obviously be tested given at the onset of aura and would have a great advantage over the triptans if effective taken this early.
Issues concerning clinical trials with preventive treatment of migraine with aura
In preventive trials, several other issues come up. First and foremost, the question is: What should be the main outcome parameter? Should it be the number of auras, or should it be headache related? The auras are easy to count and thus a robust outcome parameter. All patients, whether or not their auras are followed by a headache, can be included. Furthermore, it is difficult to imagine a drug that would reduce the aura without reducing the headache. Finally the pathophysiological mechanism of the aura, cortical spreading depression, is a well-established animal model and an easy target for drug development. If the main outcome parameter is pain related, only patients with an aura followed by a headache can provide an outcome. In real life, it would probably be necessary to include patients of all kinds in a preventive trial similar to what we did in our tonabersat trial (7). This means that some patients would exclusively have aura followed by a migraine headache and others exclusively attacks of aura followed by a non-migraine headache, but many will have a mixture of these attacks as well as attacks of aura without headache (Table 1 and Figure 2). Furthermore, a substantial number of patients will also have attacks without aura and/or headaches not starting with an aura and not fulfilling criteria for migraine but fulfilling criteria for tension-type headache. Thus, the number of auras, whether followed by a headache/migraine headache or not, is the simplest outcome parameter in preventive trials in MA.
Regarding a pain-related outcome: should one count only migraine headaches that follow an aura or should one also count migraine headaches not starting with an aura? Should headache following aura but not fulfilling migraine criteria be counted? Our solution to the problem was to call all headaches a migraine headache if they either fulfilled criteria for 1.1 MO or followed an aura. Furthermore, although our tonabersat study showed a significant response of auras but not of migraine headaches or headaches in general, this was a small trial and future trials should help elucidate whether it is possible for a drug to inhibit the auras but not migraine headaches. In preventive trials of MA, it must be required that participants have at least one aura per month in order to have a reasonable number of outcomes. However, in MO it is considered that at least two attacks per month are necessary to indicate preventive therapy (16). This is different in MA for a number of reasons. These individuals, in addition to their auras, usually have attacks without aura and other headaches, and it is the total burden on the individual that should decide whether the indication is present or not. Secondly, MA has very significant co-morbidity with a number of relatively serious diseases, the most well known probably being stroke (31–34) and depression (36). It is fully possible, but unproven, that effective preventive therapy in MA may benefit the individuals, not only because of a decrease in attack frequency but also because of diminished co-morbidity. We therefore consider an attack frequency of one aura per month as a reasonable indication for the participation in a preventive drug trial.
Conclusions
We have pointed to a number of methodological questions which are important in MA and which differ from MO. The database for answering these questions is still limited, but these questions should be elucidated in future drug trials focusing on MA.
