Abstract
Fibromyalgia syndrome (FMS) is a chronic pain condition of unknown aetiology characterized by diffuse pain and tenderness at tender points. The aim of the study was to assess the prevalence and clinical features of FMS in the different forms of primary headaches, in a tertiary headache centre. Primary headache patients (n = 217) were selected and submitted to the Total Tenderness Score, anxiety and depression scales, Migraine Disability Assessment, allodynia questionnaire, Short Form 36 Health Survey and the Medical Outcomes Study-Sleep Scale. In patients with FMS, the Multidimensional Assessment of Fatigue, the Pain Visual Analog Scale, the Manual Tender Point Survey and the Fibromyalgia Impact Questionnaire were employed. FMS was present in 36.4% of patients and prevailed significantly in tension-type headache and in patients with higher headache frequency. Headache frequency, pericranial muscle tenderness, anxiety and sleep inadequacy were especially associated with FMS comorbidity. In the FMS patients, fatigue and pain at tender points were significantly correlated with headache frequency. FMS seems increasingly prevalent with increased headache frequency, for the facilitation of central sensitization phenomena favoured by anxiety and sleep disturbances.
Introduction
Fibromyalgia (FMS) is a chronic pain syndrome of unknown aetiology characterized by diffuse pain over > 3 months and tenderness in at least 11 tender point sites out of 18. Patients with FMS are more likely to have one or more of the following comorbid conditions: depression, anxiety, headache, irritable bowel syndrome, chronic fatigue syndrome, systemic lupus erythematosus and rheumatoid arthritis (1). Both tension-type headache (TTH) and migraine are diagnosed in the FMS population, although patients with headache do not appear to represent a significantly different subgroup compared with patients without headache (2). Several studies have been performed in primary headache patients regarding FMS comorbidity. In a selected population of migraine patients, FMS was observed in 22.2% of 92 consecutive patients (3). In another recent study on migraine comorbidity factors, FMS was identified as belonging to a cluster of patients with high frequency, depression and anxiety (4). Furthermore, a common pathophysiological basis was also hypothesized for FMS and TTH (5). The prevalence of FMS has been reported to be high in patients with transformed migraine (TM) (6); in the same study, TM patients with FMS comorbidity had more insomnia, were older and had headaches that were more incapacitating than patients without FMS, which was a negative prognostic factor for migraine severity (6). It has been suggested that episodic migraine, chronic daily headache and FMS may actually be a continuum of the same disorder (7, 8). The rationale for this hypothesis is that a dysfunction of the mechanisms of pain modulation, which enhances the expression of central sensitization phenomena in both migraine (9) and TTH (10), leads to a topographical diffusion of pain. It is known that symptoms attributable to central sensitization are diffusely pronounced in FMS patients, who show increased sensitivity to mechanical, thermal and electrical stimuli, probably subtended by abnormal central pain mechanisms with augmented pain experience (11–13). The diffusion of central sensitization phenomena to extracephalic sites (3) may be the cause of FMS comorbidity in both migraine and TTH. No study has yet compared FMS comorbidity across different primary headaches, and its association with symptoms of central sensitization.
The aims of the study were: (i) to assess FMS prevalence in migraine, TTH and other forms of primary headache in a cohort of consecutive patients referred to our tertiary headache centre, in the light of headache frequency, disability and presence of allodynia; (ii) to evaluate psychological features, the number of allodynia symptoms, muscular tenderness, quality of life and sleep in headache patients plus FMS compared with headache alone sufferers; and (iii) to evaluate how the headache variables (frequency, disability, allodynia severity) correlate with the severity of FMS.
Methods
Subjects
Two hundred and ninety consecutive outpatients who had been examined for the first time at the Neurophysiopathology of Pain Unit (Neurological and Psychiatric Sciences Department, Bari University) from 1 January 2007 to 30 June 2007 were screened for admission into the study. The Neurophysiopathology of Pain Unit is a tertiary referral centre where patients are referred by primary physicians as well as by neurological and other specialty clinics. All participants gave written informed consent after receiving a detailed explanation of the purpose and design of the study. The study was approved by the local Ethics Committee of the Policlinico General Hospital.
During the first visit, all subjects had a standardized interview and underwent clinical neurological and psychiatric examination. The diagnosis of primary headache was made by three neurologists with special experience in headache, according to the International Classification of Headache Disorders, 2nd edn (ICHD-II) criteria (14) and was supported by a 3-month observation time with the headache diary and allodynia questionnaire.
At the first visit, patients with general medical, neurological or psychiatric diseases, according to the Diagnostic and Statistical Manual of Mental Disorders, 4th edn (DSM-IV) (15), were excluded from the study, as well as patients on central nervous system-active drug therapy to rule out any drug effect on diffuse pain. The exclusion criteria were finalized specifically to screen out patients suffering from various conditions with diffuse pain, such as arthritis, diabetes or other metabolic causes of neuropathic pain. Patients on preventive treatment for primary headache which proved effective against headache recurrence were invited to continue the treatment but were not enrolled. Patients who reported that their preventive therapy was ineffective were instructed to gradually interrupt it over the following 15–30 days, after which the 3-month observational period started.
All patients were asked to take symptomatic medication whenever they had a headache, i.e. triptans (almotriptan or sumatriptan) for migraine and non-steroidal anti-inflammatory drugs (acetylsalicylic acid or ibuprofen) for TTH. The same recommendation was made to patients (off preventive treatment) reporting at the first visit a headache frequency of > 15 days/month, who were advised of the opportunity to start prophylactic drugs after the observational period. Patients reporting medication overuse were encouraged to change their usual symptomatic treatments over the following 3 months and/or submitted to in-hospital detoxicant treatment. All patients were requested to keep a headache diary and to note whether they had any allodynic symptoms per single episode, using an Italian version of a previously validated questionnaire (16). A new visit was then scheduled after the 3-month observation period.
Clinical evaluation
Headache diagnosis
All the patients were diagnosed according to the ICHD-II criteria (14). Patients reporting different types of headache at the first visit were diagnosed on the basis of the symptoms occurring during the observation period. During the follow-up visit, all patients underwent the following clinical assessment.
Fibromyalgia diagnosis tender point count
For classification purposes, patients were said to have FMS if both criteria, presence of diffuse pain and tender point count, were satisfied (17). Pain was considered widespread when all of the following were present: pain in the left side of the body, pain in the right side of the body, pain above the waist and pain below the waist. In addition, axial skeletal pain (cervical spine or anterior chest or thoracic spine or low back) had to be present to confirm the diagnosis of FMS. In this definition, shoulder and buttock pain was considered as pain for each involved side. ‘Low back’ pain was considered lower segment pain.
The tender point count was performed by the clinician at the follow-up visit. This assessment checks for the presence of pain at a minimum of 11 of 18 specific locations (tender points). Tender points were evaluated by palpation with the pulp of the thumb or the first two or three fingers, at a pressure of approximately 4 kg/cm. The 18 sites (nine pairs) examined were identical to those defined in the American College of Rheumatology (ACR) criteria (17), which require a history of widespread pain present for ≥ 3 months with pain in 11 of 18 tender point sites on digital palpitation. For a tender point to be considered ‘positive’ the patient had to state that the palpation was painful. ‘Tender’ was not considered painful. Pain on digital palpation had to be present in at least 11 of the following 18 tender point sites:
Occipit (bilateral, at the suboccipital muscle insertions);
Low cervical (bilateral, at the anterior aspects of the intertransverse spaces at C5–C7);
Trapezius (bilateral, at the midpoint of the upper border);
Supraspinatus (bilateral, at origins, above the scapula spine near the medial border);
Second rib (bilateral, at the second costochondral junction, just lateral to the junctions on upper surfaces);
Lateral epicondyle (bilateral, 2 cm distal to the epicondyles);
Gluteal (bilateral, in upper outer quadrants of buttocks in anterior fold of muscle);
Greater trochanter (bilateral, posterior to the trochanteric prominence); and/or
Knee (bilateral, at the medial fat pad proximal to the joint line).
Frequency of headache
The frequency of headache was reported as the average number of days with headache/month, computed in the previous 3 months. We also stratified patients into three frequency classes (class 1 from 0 to 10 days/month, class 2 from 10 to 14 days/month, class 3 > 15 days/month).
Total tenderness score
The total tenderness score (TTS) was also assessed in all patients to evaluate pericranial tenderness, following the procedure described by Langermark and Olesen (18). Assessment was performed with manual palpation by a neurologist with experience in headache, who was experimentally blinded to the patient's diagnosis, after at least 30 min from the tender point count and the Manual Tender Point Survey (see below). The right frontal muscle, masseter muscle, temporal muscle, pterigoid muscle, sternocleidomastoid muscle, sternocleidomastoid muscle insertion, neck muscle insertion and trapezius muscle were examined using the TTS system. This method used a combination of behavioural and verbal items, each of which is scored on a four-point Likert scale, defined as: 0, denial of tenderness, no visible reaction; 1, verbal report of discomfort or mild pain, no visible reaction; 2, verbal report of moderate pain, with or without visible reaction; 3, verbal report of marked pain and visible expression of discomfort. We obtained a total score in all headache patients.
Allodynia questionnaire
The presence of allodynia symptoms during the headache episodes was inferred from the notes the patients made on their diaries while answering the questionnaire we had given them to complete after each headache episode. An Italian version of the allodynia questionnaire was employed, recently reported by Ashkenazi et al. (19). Patients were classified as allodynic if they confirmed the presence of at least one symptom reported in the questionnaire, over ≥ 50% of the headache episodes (19). Allodynia symptoms were also considered in the cases with a diagnosis other than migraine to distinguish patients with allodynia from those without. In addition, we computed the mean of the total number of symptoms reported by patients across headache episodes.
Short-Form 36 Health Survey
All patients were asked to complete the Short-Form 36 Health Survey (SF-36), which is a self-administered questionnaire that measures each of the following eight health concepts: physical functioning, role limitations due to physical problems, social functioning, bodily pain, mental health, role limitations due to emotional problems, vitality and general health perception (20). Higher scores reflect better patient status. We evaluated the Physical Component Summary (PCS) and Mental Component Summary (MCS) scores (20).
Depression and anxiety scales
All patients were assessed with the Zung Self-Rating Depression (SDS) and Anxiety (SAS) Scales, as they are considered reliable tools to detect symptoms of anxiety and depression in a general non-psychiatric patient population (21, 22).
Medical Outcomes Study—Sleep Scale
The Medical Outcomes Study—Sleep Scale (MOS) was relied on to assess the quality of sleep. The MOS is a self-administered questionnaire consisting of 12 items that assess key constructs of sleep (23). Instrument scoring yields seven subscales (sleep disturbance, snoring, awaken short of breath or with a headache, quantity of sleep, optimal sleep, sleep adequacy and somnolence) as well as a nine-item overall sleep problems index. With the exception of sleep adequacy, optimal sleep and quantity, higher scores reflect greater impairment in the MOS subscales. In this study, we evaluated the sleep disturbances (SLD) and sleep adequacy (SLA) subitems.
Migraine Disability Assessment scale
The Migraine Disability Assessment (MIDAS) scale (24), in the Italian version (25), was used to quantify headache-related disability in patients with a diagnosis of migraine. Based on the score, one of four disability grades was assigned: grade I, minimal or infrequent disability, corresponded to a MIDAS score of 0–5; grade II, mild or infrequent disability, corresponded to a MIDAS score of 6–10; grade III, moderate disability, corresponded to a MIDAS score of 11–20; and grade IV, severe disability, corresponded to a MIDAS score of ≥ 21 (24). We used the MIDAS in patients with migraine without aura (MoA) (ICHD II code 1.1), migraine with aura (code 1.2) and chronic migraine (code 1.5.1).
Patients presenting FMS comorbidity, according to the ACR criteria (17), were submitted to the following examinations.
Manual Tender Point Survey
The Manual Tender Point Survey (MTPS) provided a rate of the severity of pain patients felt upon palpation of the specific 18 tender points defined by the ACR and of the three control sites (26, 27). The three control points were palpated and recorded to provide baseline documentation of the patient's pain perception, but pain at control sites did not excluded FMS diagnosis (28). The purpose of the MTPS was to describe the global severity of pain at tender and control points in FMS patients. It was based on an 11-point numeric scale, where 0 is no pain and 10 is the worst pain the patient ever had. Separate scores (FMS intensity score) were calculated for each of the nine bilateral tender points and the three control sites (Mid-forehead, dorsum right forearm, junction of proximal two-thirds and distal third and left thumbnail). A total score was then computed.
Multidimensional Assessment of Fatigue
The Multidimensional Assessment of Fatigue (MAF) scale is a self-administered survey that yields a Global Fatigue Index (29).
Fibromyalgia Impact Questionnaire
The Fibromyalgia Impact Questionnaire (FIQ) is a fibromyalgia-specific patient-reported outcome instrument designed to assess health status, progress and outcomes in patients with fibromyalgia. It contains 10 subscales that are combined to yield a total score (30).
Pain visual analogue scale
The visual analogue scale (Pain VAS) is a 100 mm long horizontal line, where patients rate their global impression of muscular-skeletal pain, with 0 being ‘no pain’ and 100 ‘worst possible pain’ (31). The Pain VAS did not include the self-rating of headache intensity.
Statistical analysis
Pearson's χ2 was used to assess the prevalence of FMS in primary headache patients stratified by the following features: type of headache (migraine, tension type, mixed form, other) frequency classes, disability (MIDAS score), presence of allodynia symptoms and gender. The patient's age, headache duration, the TTS, SAS, SDS, SF-36 and MOS results were evaluated by a one-way
In FMS patients, the MAF, FIQ, Pain VAS and MTPS were evaluated, by means of
Results
Two hundred and seventeen patients were entered into the study because of a diagnosis of primary headache (Table 1). The presence of allodynia was similar in all headache diagnosis categories (Pearson's χ2 test: 6.63 d.f. 6: NS) (Table 1). Of 12 patients that had been classified at the first visit as ‘Probable medication overuse headache’ (code 8.2.7), five changed their symptomatic treatment (all of them from analgesics to triptans) and three were finally classified as chronic migraine patients (1.5.1) and two as MoA (1.1), whereas two were lost to follow-up. Five patients were submitted to detoxicant treatment, but were not admitted to the study because they started preventive treatments with anti-epileptic drugs. Of the remaining patients that were not included in the study, 15 did not fulfil the inclusion criteria at the first visit, 20 were lost to follow-up, 15 did not keep a reliable headache diary according to our recommendations, and the remainder were not able to exhibit any written documentation of their headache occurrence.
Clinical features of primary headache patients
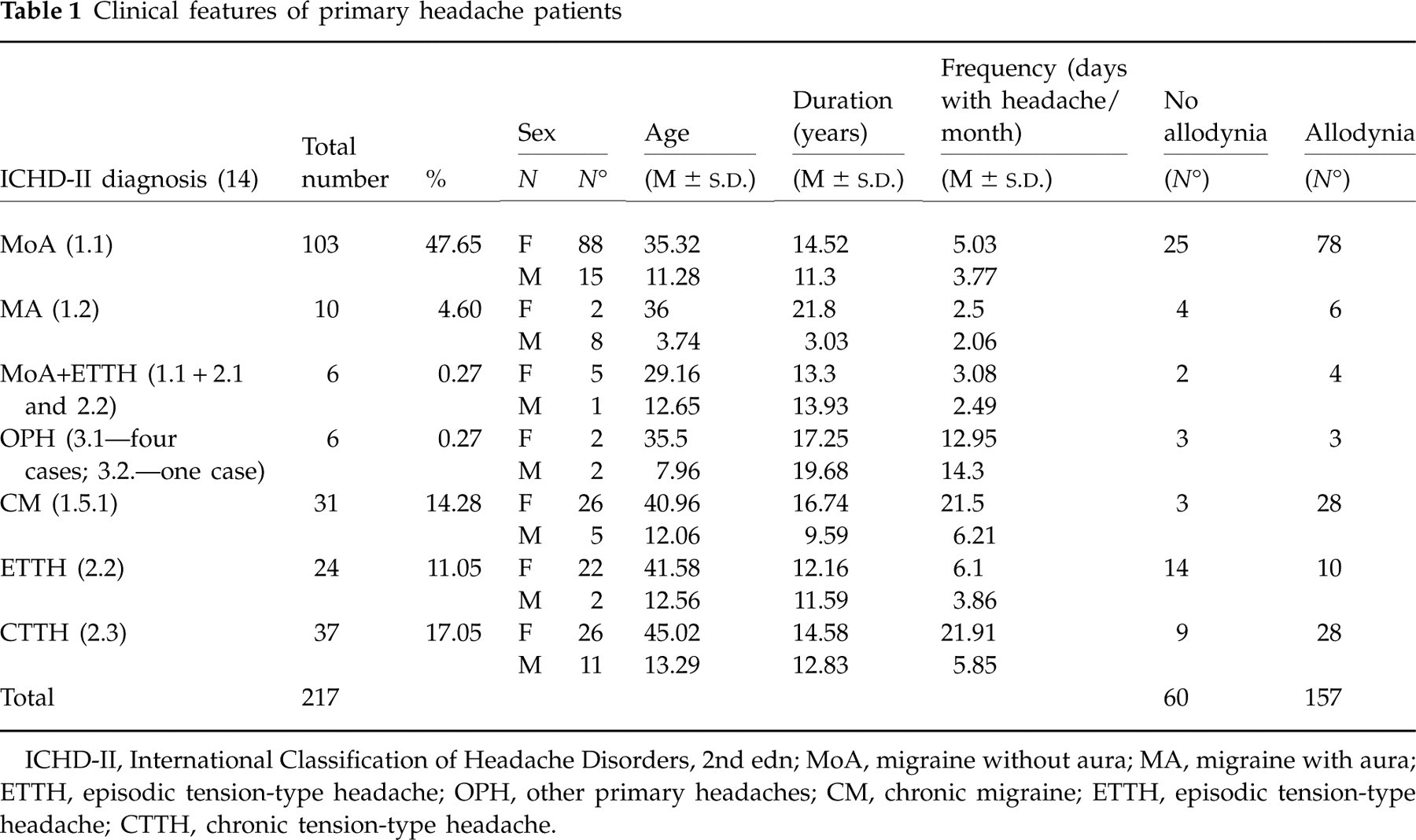
ICHD-II, International Classification of Headache Disorders, 2nd edn; MoA, migraine without aura; MA, migraine with aura; ETTH, episodic tension-type headache; OPH, other primary headaches; CM, chronic migraine; ETTH, episodic tension-type headache; CTTH, chronic tension-type headache.
Most of admitted patients were female (178; 82%).
FMS comorbidity
FMS comorbidity was present in 79 of 217 patients (36.4%). It prevailed in patients with TTH, compared with the other types of headache (Table 2), in those with higher headache frequency and in migraine patients with the higher MIDAS scores (Table 2). FMS comorbidity was equally distributed across patients with and without allodynia and across the two sexes (Table 2).
Frequency of fibromyalgia across different headache subtypes

Groups where there was a statistically relevant prevalence of FMS comorbidity:
∗ P < 0.05;
∗∗∗∗ P < 0.0001, Pearson's χ2.
ICHD-II diagnosis (14): MoA, migraine without aura; MA, migraine with aura; ETTH, episodic tension-type headache; OPH, other primary headaches; CM, chronic migraine; ETTH, episodic tension-type headache; CTTH, chronic tension-type headache.
Age and headache duration
Patients with FMS comorbidity were significantly older than those without diffuse pain (Fig. 1A). TTH patients were also older than the others, but the interaction FMS × headache diagnosis was not statistically significant (Fig. 1A). No difference was found in headache duration between FMS and non-FMS patients (Fig. 1A).

Mean values and standard deviations of (A) age and illness duration in primary headache patients; (B) number of allodynia symptoms and total tenderness score. The
Number of allodynia symptoms
The number of allodynia symptoms was significantly greater in patients with FMS comorbidity, irrespective of their headache diagnosis (Fig. 1B).
Total Tenderness Score
The TTS score was higher in patients with FMS symptoms than in those without FMS comorbidity, independent of headache type (Fig. 1B).
Depression and anxiety scales
Anxiety levels were significantly higher in patients with FMS comorbidity, especially in migraine and TTH sufferers (Fig. 2A), as well as depression symptoms (Fig. 2A).
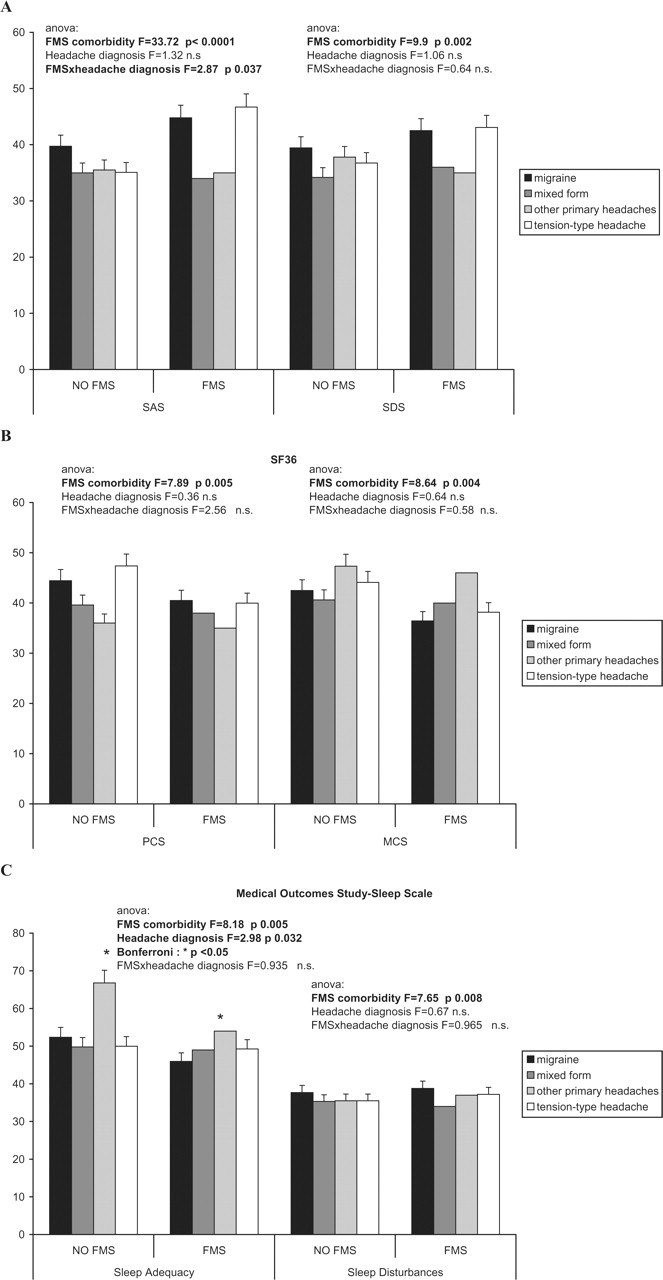
Mean values and standard deviations of (A) Zung Self-Rating Anxiety (SAS) and Depression (SDS) scales; (B) SF-36 Physical Component Summary (PCS) and Mental Component Summary (MCS); (C) Medical Outcomes Study-Sleep Scale subitems. The
Short-Form 36 Health Survey
The PCS was significantly lower in patients with symptoms of diffuse pain, irrespective of their headache diagnosis (Fig. 2B). The Mental Component Summary was also significantly decreased in patients with diffuse muscular-skeletal pain (Fig. 2B).
Medical Outcomes Study—Sleep Scale
SLA levels were significantly lower in headache patients with FMS and they were consistently so across the headache subtypes, although patients with other primary headaches exhibited better sleep adequacy (Fig. 2B). The SLD rates were higher in FMS patients, irrespective of their headache type (Fig. 2C).
Step-wise discriminant analysis was run by introducing all the variables that were present to a different extent in patients with FMS compared with those without. The frequency of headache, the SAS, the TTS and the SLA MOS scores were selected in this order as the best discriminating variables, since they provided correct classification in almost 80% of cases (Table 3).
Step-wise discriminant analysis of headache patients for FMS (fibromyalgia) comorbidity
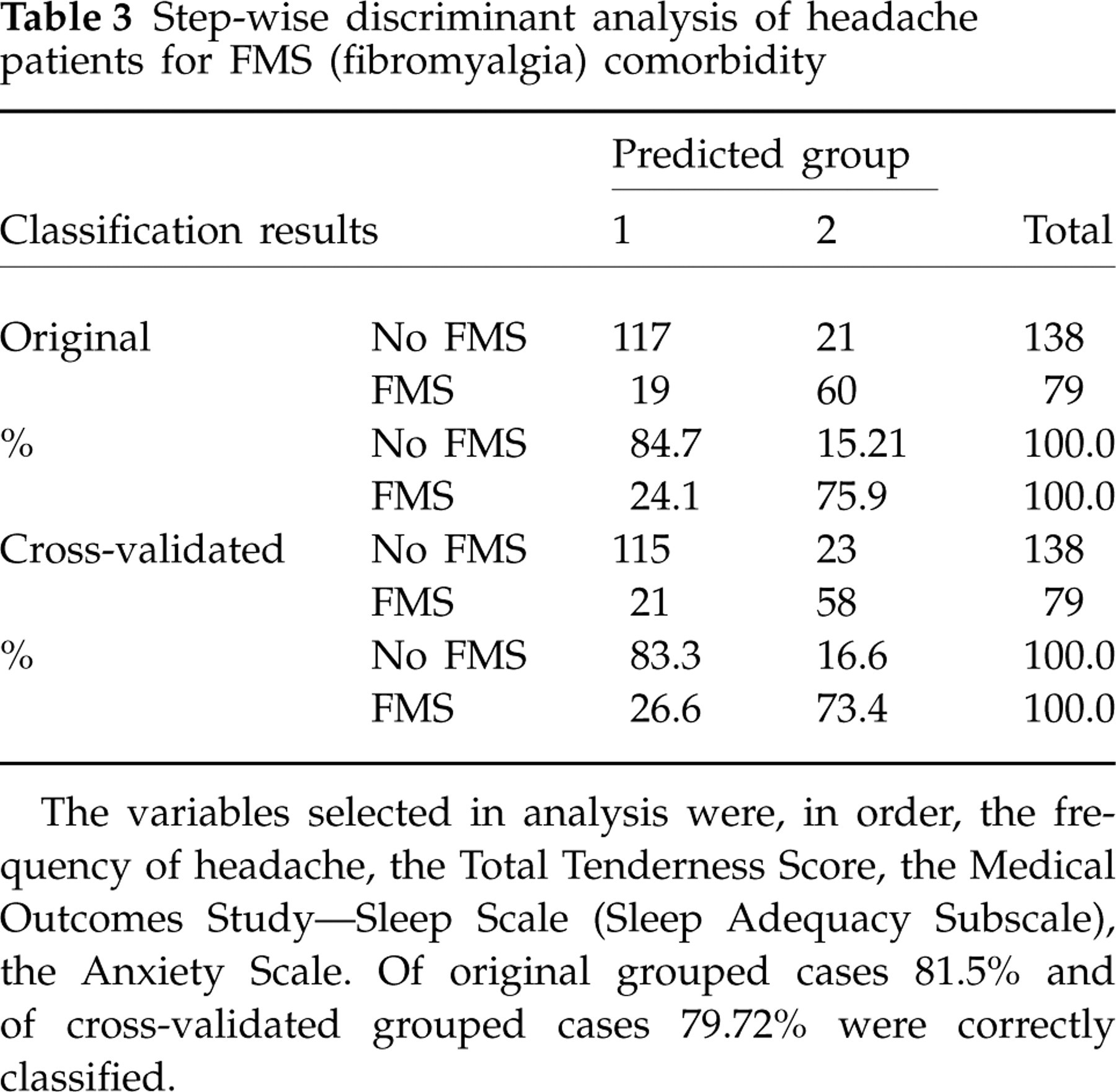
The variables selected in analysis were, in order, the frequency of headache, the Total Tenderness Score, the Medical Outcomes Study—Sleep Scale (Sleep Adequacy Subscale), the Anxiety Scale. Of original grouped cases 81.5% and of cross-validated grouped cases 79.72% were correctly classified.
In patients with FMS comorbidity, considering groups with more than one patient, the MAF was not significantly different across the headache diagnoses (
Discussion
Prevalence of fibromyalgia comorbidity in headache patients
In this study we have compared the frequency of FMS across different primary headache diagnoses given that most previous studies regarded the analysis of FMS prevalence in specific primary headache groups (3, 6). The prevalence of FMS in our headache patients (36.4%) was high compared with that in the general population, which is estimated to be 4–13% (32–34), although our population had been selected in a tertiary headache referral centre and may not share the features of a general headache population. Although patients screened as fibromyalgic came to our centre for their headache, the symptoms of FMS were relevant, as shown by high levels of pain at tender points and severe fatigue, especially in chronic headache sufferers. There was no difference in gender distribution across the headache with and without FMS groups, where there was a similar female prevalence. FMS seemed to prevail in TTH patients, according to a possible common pathophysiological origin, as previously suggested (5). In studies describing headache features in patients with FMS, both migraine and TTH have been observed (2, 35). The prevalence of FMS in migraine patients was 16.8%, similar to the rate reported by Ifergane et al. (3). FMS was observed in one patient affected by chronic cluster headache, but no conclusions can be drawn regarding the possible prevalence of FMS in this primary headache given the small number of cases in our series.
Clinical features of headache patients with FMS comorbidity
A clear predominance of FMS comorbidity in chronic headache patients emerged from our data, as also observed in studies regarding headache features in fibromyalgic patients (2). The results for the MIDAS score confirmed that patients with FMS had the highest level of migraine severity. Although TTH patients seemed prone to diffuse muscular-skeletal pain, more than the other headache patients, in migraine patients the headache frequency conditioned the association with FMS symptoms, which seemed a feature of chronic headache in general.
The common factor for FMS comorbidity and chronic headache may be the expression of central sensitization phenomena. There is a growing body of evidence that central sensitization phenomena have a role in FMS pathogenesis (13, 36). It is also known that headache frequency is related to sensitization (37). Allodynia at the trigeminal and cervical levels was present in both migraine and TTH patients, and this finding is in agreement with the most recent theories about the pathogenesis of these headaches (9, 10) and with similar findings that have recently been reported for different types of headache (38). Allodynia was found in 74.33% of our migraine patients, similar to other recent reports on migraine (19). The variability registered in the occurrence of allodynia across headache subtypes may be partly caused by differences in the ways data were collected from patients. Lovati et al. (38) retrospectively questioned patients about allodynia during their first visit, and this may have led to underestimation of skin discomfort symptoms during and between headaches, but our findings are in agreement with the results of that study regarding a more frequent occurrence of allodynia in the forms of chronic headache. Lipton et al. (39) have recently validated a new version of the allodynia questionnaire to improve further the reliability of the clinical examination, but unfortunately our study started before the new tool was available. Although the presence of allodynia was not different in patients with and without FMS comorbidity, the severity of allodynia symptoms was increased in headache patients with diffuse pain, thus suggesting that predisposition to pain spreading in the body is related to the severity of central sensitization expression. Myofascial tenderness upon palpation (18) was also greater in patients with FMS comorbidity. Muscle tenderness has also been recognized to be a symptom of central sensitization at the trigeminal and cervical levels (10) that is very frequent in patients with chronic headache, as is hyperalgesia, shown by the reduced pain-pressure threshold (37). Pericranial muscular tenderness, as well as allodynia, may also predispose to the expression of diffuse muscular-skeletal pain in primary headache patients.
Psychopathological features seemed to be linked with FMS comorbidity in headache patients. Our patients were not affected by psychiatric comorbidity, as coded by the DSM-IV (15). However, the SAS and SDS tools that are used to test psychological features in the general population clearly indicated increased levels of anxiety and depression in headache patients with diffuse pain. Psychiatric comorbidity has been observed in both TTH and migraine (40), with maximal expression in chronic headache populations (41–43), as a reflection of the burden of the disease rather than a hallmark of a specific headache category. In our study, anxiety prevailed in patients with FMS comorbidity in all the headache groups. The reason underlying the marked expression of chronic pain in headache patients with symptoms of anxiety and depression may be a disturbed pain modulation with facilitation of central sensitization (44).
On the other hand, it is also likely that chronic pain may enhance anxiety and depression symptoms (45), thus resulting in a self-perpetuating circuit that causes pain severity to increase.
Another factor associated with FMS comorbidity in our headache patients was the poor quality of sleep. Sleep disorders are a frequent symptom in headache; they prevail in chronic forms and in patients with psychiatric comorbidity (46). Although chronic pain per se may be a cause of poor sleep quality, which is also mediated by the same psychiatric comorbidities, there is an attractive hypothesis suggesting that sleep deprivation is a factor producing hyperalgesic changes (47).
The quality of life indices were also lower in our headache patients with FMS comorbidity, both for physical and mental health. Reduced quality of life has also been found in chronic headache patients (48), but the presence of FMS may cause further life worsening, as already reported in previous studies of migraine patients (3).
Fibromyalgia symptoms in relation to headache features
In fibromyalgic patients, headache frequency was associated with a more severe clinical outcome, expressed by increased fatigue and pain severity at the tender points. The degree of headache severity heightened the intensity of diffuse pain and fatigue. Although the findings reported by Marcus et al. (2) did not provide evidence that the presence of headache was an aggravating factor for FMS, our data suggest that when headache is present its severity is linked to an increase in expression of FMS symptoms.
General remarks and conclusion
The results of our study have also shown that the best discriminating factors for FMS comorbidity were headache frequency, followed by anxiety, pericranial muscular tenderness and reduced sleep adequacy. Although the prognostic implications of the present results are hypothetical, precise investigation of these features and an adequate therapeutic approach in patients with headache may improve the expression of diffuse and chronic pain and quality of life.
