Abstract
Stress is a provoking factor for both tension-type headache and migraine attacks. In the present single-blind study, we investigated if stress induced by norepinephrine (NE) could elicit delayed headache in 10 healthy subjects and recorded the cranial arterial responses. NE at a dose of 0.025 μg kg−1 min−1 or placebo was infused for 90 min and the headache was followed for 14 h. Blood flow velocit in the middle cerebral artery (measured with transcranial Doppler) and diameters of the temporal artery and the radial artery (measured with ultrasound) were followed for 2 h. There were no changes in these arterial parameters after NE. In both treatment groups three subjects developed delayed headaches. Thus, stress by NE infusion did not result in delayed headache.
Introduction
When migraine patients are questioned what might trigger their attacks, the most frequently mentioned precipitating factor is ‘stress’ (1). In the most comprehensive study published, Sorbi et al. (2) collected data on headache symptoms and stressfulness of daily problems and sleep quality. In comparison with headache-free days, the incidence of daily problems was greater during the afternoon preceding an evening or night-time attack of migraine. The timing of evening and night-time attacks is consistent with the notion of migraine developing during a period of relaxation after stress.
Stress can be experimentally induced by physical exercise, mental stress or by pharmacological intervention such as norepinephrine (NE) infusion. In the present single-blind study, we investigated if stress induced by NE could elicit headache in healthy subjects in an established headache model (3–8).
We found that NE did not induce headache and cranial hemodynamics were unchanged.
Methods
Ten healthy volunteers were recruited (seven female, three male), age 20–29 years (mean 24 years), body weight 53–90 kg (mean 69 kg) with no history of migraine and who never suffered or suffered fewer than one/week any other non-migraine headache such as tension-type headache. Before inclusion the participants underwent physical and neurological examination. Written informed consent was obtained before randomization. The study was performed in accordance with the Helsinki II Declaration and was accepted by the local ethics committee. No subject was replaced.
Subjects were studied in the supine position and blood pressure (BP) and heart rate were measured automatically every 5 min using an inflatable arm cuff (Tonoprint; Speidel und Keller, Jungingen, Germany). No medication, coffee, tea, alcohol, chocolate or tobacco was allowed for the last 8 h prior to the examination, which began at 08.30 h. A brief medical examination was performed and a cannula was inserted into the left cubital vein for infusion of NE or saline.
Following a single-blind, placebo-controlled crossover design, NE or placebo (isotonic saline, 0.9% NaCl) was randomly administered continuously intravenously during 90 min on two separate days, at least 1 week apart. The starting dose of NE, 0.025 μg kg−1 min−1, was increased to 0.010 μg kg−1 min−1 until mean arterial pressure (MAP) was increased > 15% within 30 min. If MAP later decreased < 15% or exceeded 25%, infusion was adjusted with 0.005 μg kg−1 min−1. Different doses were given by altering the speed of the infusion, as a volume directed pump (Syringe Pump P2000) was used.
The frontal branch of temporal and the radial artery diameters were measured by a high-resolution ultrasound unit (Dermascan C; Cortex Technology, Hadsund, Denmark: 20 MHz, bandwidth 15 MHz) (9, 10). Blood flow velocity in the middle cerebral artery (MCA) was recorded bilaterally by transcranial Doppler (TCD) (2 MHz, Multidop X Doppler: DWL, Sipplingen, Germany) using handheld probes as previously described (11). A fixed point for measurements of mean maximal blood velocity (V mean) was chosen along the middle cerebral artery as the point that was as close as possible to the bifurcation between the middle cerebral and the anterior cerebral arteries. This fixed point was then used throughout the study in each individual (Table 1).
Time schedule for procedures on the day of experiment

Artery diameters refer to radial end temporal measured by high frequency ultrasound. Mean arterial pressure (MAP) is mean arterial blood pressure. The order of procedures each 10 min was first scanning of radial artery, then recordings of mean blood velocity (V mean) with simultaneous measurement of pCO2 and finally scanning of temporal artery.
End-expiratory pCO2 (12) was measured simultaneously with the transcranial Doppler measurements (POET; Crisicare Systems Inc., Waukesha, WI, USA) (11). V mean was corrected for changes in pCO2 according to Markwalder et al. (13):

Headache was scored every 10 min in the laboratory on a 0–10 point scale (0 = no headache, 5 = moderate headache, and 1 = worst possible headache). After leaving the laboratory the subjects scored headache and possible associated symptoms hourly for the next 12 h in a headache diary.
Statistics
For each subject, data were reduced to mean values at baseline (t =−10), two values during continuous infusion (t = 40, t = 80) and after termination of infusion (t = 120). Vascular results are presented as absolute mean ±
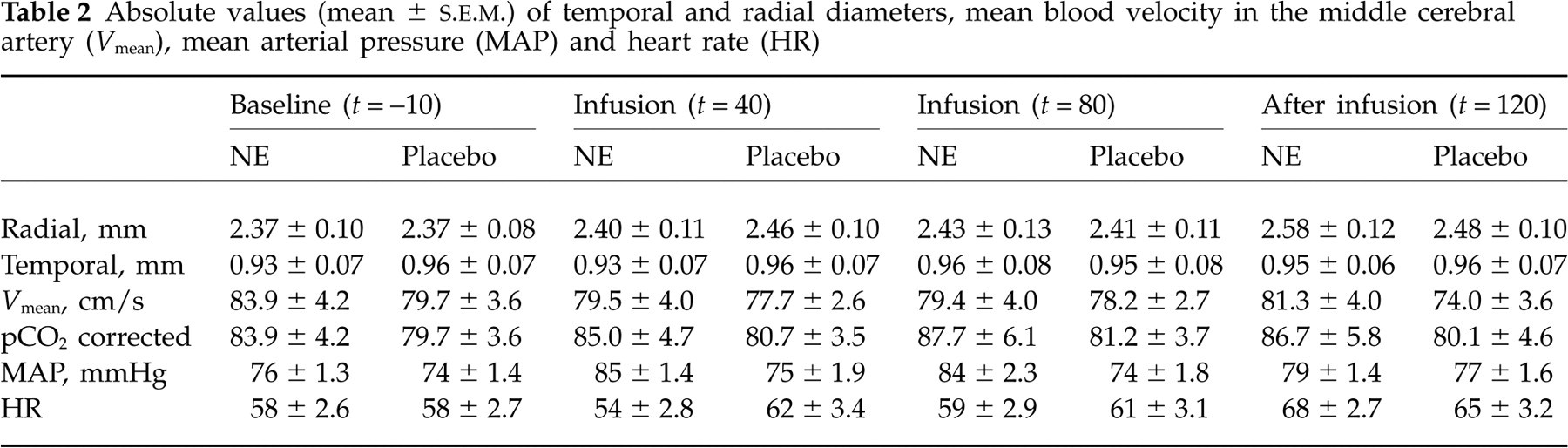
Absolute values (mean ±
Results
Three subjects experienced delayed headache after placebo infusion and three after NE infusion. One person had mild headache during placebo infusion and no person had headache during NE infusion.
The mean final dose of NE used was 0.056 μg kg−1 min−1 (range 0.03–0.085), the mean final increase in BP was 9 mmHg and the mean heart rate was unchanged (Table 2). Arterial diameters were unchanged during infusion of NE.
The diameter of the radial artery increased after termination of the NE infusion compared with both baseline (P < 0.05) and placebo (P < 0.05) (Table 2). No significant difference in mean temporal artery diameter was detected between the 2 days at baseline, during or after infusion.
Blood flow velocity V
mean in MCA on each side was analysed as the mean of left and right values. Mean pCO2 corrected baseline value of V
mean was 83 cm/s on the day of NE infusion and 80 cm/s on the day of placebo (P = 0.48). Velocity during NE infusion V
mean did not change significantly over time (

Relative mean (±
Discussion
The present experimental headache model has been used to show the headache-inducing effect (or lack thereof) of vasodilators such as glyceryl trinitrate (6, 14, 15), histamine (16), calcitonin gene-related peptide (5), adenosine (8), sildenafil (7) and vasoactive intestinal peptide (17). Both an immediate headache (14) and a delayed migraine-like headache, occurring hours after infusion of vasodilators, have been observed (6, 18). In the present study the general vasoconstrictor norepineprine was used. It was hypothesized that ‘vascular stress’ could initiate a slow counteractive process at the cellular level resulting in a delayed headache. Possible vascular effects of NE, blood flow velocity in MCA, diameters of temporal and radial arteries, and mean BP were monitored during NE infusion and headache was monitored during infusion and 12 h after. The general result was that NE, at the dose used (mean 0.056 μg kg−1 min−1), caused no arterial changes, an increase in mean BP and no more delayed headache after infusion than after placebo (three subjects in each arm experienced delayed headache). There was thus no indication of delayed headache caused by NE in these healthy subjects.
We aimed at an increase of 15% in mean BP and in fact achieved an 11% increase after 40 min of NE infusion (Table 2). There was thus a clear pharmacological effect of the dose chosen.
NE causes constriction in vitro of human temporal arteries (19). In the present study we found no effect on the diameter of the temporal artery (Table 2). This could theoretically be due to too low a dose of NE or could reflect that the artery in vivo reacts differently from in vitro.
A study was recently published in which the blood flow velocity in MCA was measured after a similar final dose of NE 0.06 μg kg−1 min−1 with a resulting increase of 25% in mean BP (20). The dose was thus similar to that used in the present study. There was no effect on blood flow velocities (53 cm/s before and 54 cm/s after NE) in MCA (20).
Thus, with the highest doses of NE that are possible in healthy volunteers, with close monitoring of BP, there is no vasoconstrictor effect on cranial arteries. The chances of subsequent delayed headache induced by dilatory counterregulation are therefore small. Only in the radial artery did we observe a possible rebound phenomenon (see above).
In conclusion, we observed no changes in cranial arterial parameters and no headache induction after NE.
Footnotes
Acknowledgements
We thank laboratory technician Lene Elkjær for skilful assistance. Supported by the Lundbeck Foundation Centre for Neurovascular Signalling (LUCENS).
