Abstract
Using diffusion tensor (DT) tractography, we quantified optic radiation (OR) structural changes in seven migraine patients with (MA) and eight without visual aura (MoA) and their relation to clinical manifestations and T2-visible burden. The corticospinal tract and the corpus callosum were studied as ‘control’ white matter (WM). No difference was found for any of the WM fibre bundles metrics between controls and MoA patients. MA patients had reduced average fractional anisotropy (FA) of both OR compared with controls and reduced average FA of the right OR compared with MoA patients. They also showed higher right OR mean diffusivity than controls. OR metrics were not correlated with clinical and magnetic resonance imaging (MRI) metrics. DT tractography reveals OR changes in MA patients that might represent a phenotypic biomarker of the disease given the lack of correlation with clinical and structural MRI metrics.
Introduction
The advent of modern magnetic resonance-based techniques characterized by a high sensitivity towards central nervous system (CNS) structural changes is providing important insights into the pathophysiology of several neurological conditions. Among these techniques, diffusion tensor (DT) magnetic resonance imaging (MRI) provides quantitative data on water molecular motion, a marker of tissue structure (1). Water diffusion in the brain is affected by the presence of barriers to translational motion such as cell membranes and myelin. Pathophysiological processes, including ischaemia, can modify the integrity of the tissue microstructure, resulting in significant alterations of the diffusivity characteristics. This notion has been supported by the results of several studies performed on patients with chronic vascular diseases, including stroke (2), leukoaraiosis (3) and cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) (4). More recently, DT MRI has also been applied to the assessment of ‘occult’ brain damage in patients with migraine. These studies have shown that brain damage in migraineurs may extend beyond T2-visible lesions and involve diffusely the normal-appearing brain tissues (NABT) (5, 6). Involvement of visual-processing areas (7) and of pain-related pathways (8, 9) has also been demonstrated.
Visual disturbances, mainly in the form of aura, are commonly described in migraineurs and are thought to reflect brain hyperexcitability. Recently, a study in healthy individuals has shown a correlation between functional MRI measures of neuronal activity within the visual cortex and DT MRI markers of neuronal structure within the subserving optic radiation (OR) (10), suggesting that there might be a strict relationship between function and structure.
In this study, we hypothesized that selective diffusion changes of posterior visual pathways might be detected in patients with migraine and that the quantification of such changes might be different in patients with and without visual aura. To test our hypothesis, we used MR tractography to explore the diffusivity and atrophy characteristics of the OR in these patients. To provide clues as to the nature of the detected changes, if present, we also investigated their correlation with patients’ demographic characteristics, clinical manifestations of the disease, and extent of MRI-visible lesions. In addition, to evaluate whether or not CNS diffusion changes in these patients were selective, we also quantified diffusivity and atrophy metrics of the corticospinal tracts (CST) and the corpus callosum (CC).
Patients and methods
We studied 15 migraine patients [seven with visual aura (MA), eight without aura (MoA); 14 women, one man, mean age 43.3 years, range 30–58 years; mean disease duration 25.6 years, range 2–48 years; mean number of attacks per year 21.0, range 12–244, mean time elapsed from the last attack 30 days, range 15–45 days]. All patients met the criteria of the International Classification of Headache Disorders for the diagnosis of migraine (11). In the MA group, aura was bilateral in four patients, right-sided in two patients and left-sided in the remaining patient. Attack duration could not be estimated precisely because the majority of patients used medications for pain control. In order to have definite evidence of structural subcortical pathology in these patients, the presence of at least four discrete brain MRI abnormalities was an additional inclusion criterion. The cut-off of four lesions was chosen to minimize the risk of including subjects with incidental MRI signal abnormalities (12). The patients were recruited consecutively from the migraine population attending the Outpatient Clinics, Department of Neurology, Scientific Institute and University Ospedale San Raffaele, after having screened 57 patients. Patients with hypertension, hypercholesterolaemia, diabetes mellitus, vascular/heart diseases or other major systemic and neurological conditions were excluded. At the time MRI was performed, four patients were taking prophylactic treatment for migraine. Eleven healthy volunteers, with no family history of migraine, no previous history of neurological dysfunction (including migraine), normal neurological examination, and a normal brain MRI (to exclude possible confounding factors such as hypertension, hypercholesterolaemia, diabetes mellitus, etc.) served as controls (10 women and one man, mean age 41.6 years, range 29–55 years). All subjects were assessed clinically by a single neurologist, who was unaware of the MRI results. Local Ethics Committee approval and written informed consent from all subjects were obtained prior to study initiation.
Using a 3-T Intera scanner (Philips Medical Systems, Best, the Netherlands), the following scans of the brain were obtained: (i) T2-weighted turbo-spin echo images [repetition time/echo time (TR/TE) = 3000/120 ms, flip angle 90°, matrix size = 512 × 512, field of view (FOV) = 230 mm, 28, 4 mm thick, contiguous, axial slices]; (ii) fluid attenuated inversion recovery (FLAIR) sequence (TR/TE = 11000/120 ms, TI = 2800 ms, flip angle 90°, matrix size = 256 × 256, FOV = 230 mm, 28, 4 mm thick, contiguous, axial slices); (iii) high-resolution 3D T1-weighted fast field echo (FFE) sequence (TR/TE = 25/4.6 ms, flip angle 30°, matrix size = 256 × 256, FOV = 230 mm, voxel size = 1 × 1 × 1 mm3); and (iv) pulsed-gradient spin-echo planar pulse sequence with sensitivity encoding (SENSE) (acceleration factor = 2.5; TR/TE = 8300/80 ms; acquisition matrix size = 96 × 96; FOV = 240 × 240 mm2; 55, 2.5 mm thick axial slices; after SENSE reconstruction, the matrix dimension of each slice was 256 × 256, with in-plane pixel size of 0.94 × 0.94 mm) and with diffusion gradients applied in 32 non-collinear directions, using a gradient scheme which is standard on this system (gradient overplus). Two optimized b factors were used for acquiring diffusion-weighted images (b1 = 0, b2 = 1000 s/mm2) (13). Fat saturation was performed to avoid chemical shift artefacts.
Lesion volumes (LV) were measured on T2-weighted images using a local thresholding segmentation technique (14). FLAIR scans were always used to increase confidence in lesion identification. T1-weighted images were used to measure the normalized brain volumes (NBV), using the cross-sectional version of the Structural Imaging Evaluation of Normalized Atrophy (SIENAx) software (15).
Diffusion gradient directions were corrected for scanner settings (i.e. slice angulation, etc.) before DT estimation (16). From DT images, the diffusion tensor was estimated by non-linear regression (Marquardt–Levenberg method), assuming a mono-exponential relationship between signal intensity and the b-matrix components (17). After diagonalization of the estimated tensor matrix, the two scalar invariants of the tensor, mean diffusivity (MD) and fractional anisotropy (FA), were derived for every pixel. Then, using the VTK Computational Imaging Science Group Registration Toolkit (18), the rigid transformation needed to correct for position between the b = 0 images (T2-weighted, but not diffusion-weighted) and T2-weighted images was calculated. Normalized mutual Information (19) was the similarity measure used for image matching. The same transformation parameters were then used to coregister the MD and FA images to the T2-weighted images. The final step consisted of automatic transfer of lesion outlines onto the MD and FA maps and calculation of average lesion MD and FA.
A streamline tractography algorithm (20) was used to reconstruct the anatomy of brain white matter (WM) fibre bundles. Since the direct application of tractography in patients with brain pathology is hampered by the presence of regions with decreased FA, tractography was used to construct the probability maps for each WM fibre bundle analysed, using data from 40 healthy subjects (31 women and nine men, mean age 42.3 years, range 34–62) (reference group), including those of the present study. These probability maps were then applied to subjects’ data to derive diffusivity measures inside the WM fibre bundle of interest, as previously described (21).
Tract trajectories were obtained by placing regions-of-interest believed to contain a section of the desired WM fibre bundles on both FA and colour-coded maps for the principal diffusion directions (22), by cross-referencing neuroanatomical (23, 24) and previous tractography studies (21, 25–28). In addition to the OR, the CST and the CC were also studied as ‘control’ WM fibre bundles. Trajectories were checked by qualitative visual assessment after reconstructing them in three dimensions.
Finally, the non-linear transformation between the FA map of each subject and the FA atlas obtained from healthy volunteers (reference group) was computed: in the reference control group, it was applied to tracts obtained from tractography, before their average to produce tract probability maps; in patients and controls of the study, it was used to transform MD and FA maps and to calculate the determinant of the jacobian (J) of the transformation (29), which summarizes the point-wise volume changes produced by the deformation. Values less than unity reflect atrophy, whereas values greater than unity reflect hypertrophy of the WM fibre bundles of interest (29, 30). Figure 1 shows an example of the WM fibre bundle reconstructions in an individual subject prior to constructing the probability map. Figure 2 shows the probability map in axial, coronal and sagittal orientations, superimposed on the FA atlas. The probability maps, thresholded at 0.4 (31), were then applied as a binary mask to the transformed MD, FA and to J maps and average values obtained.
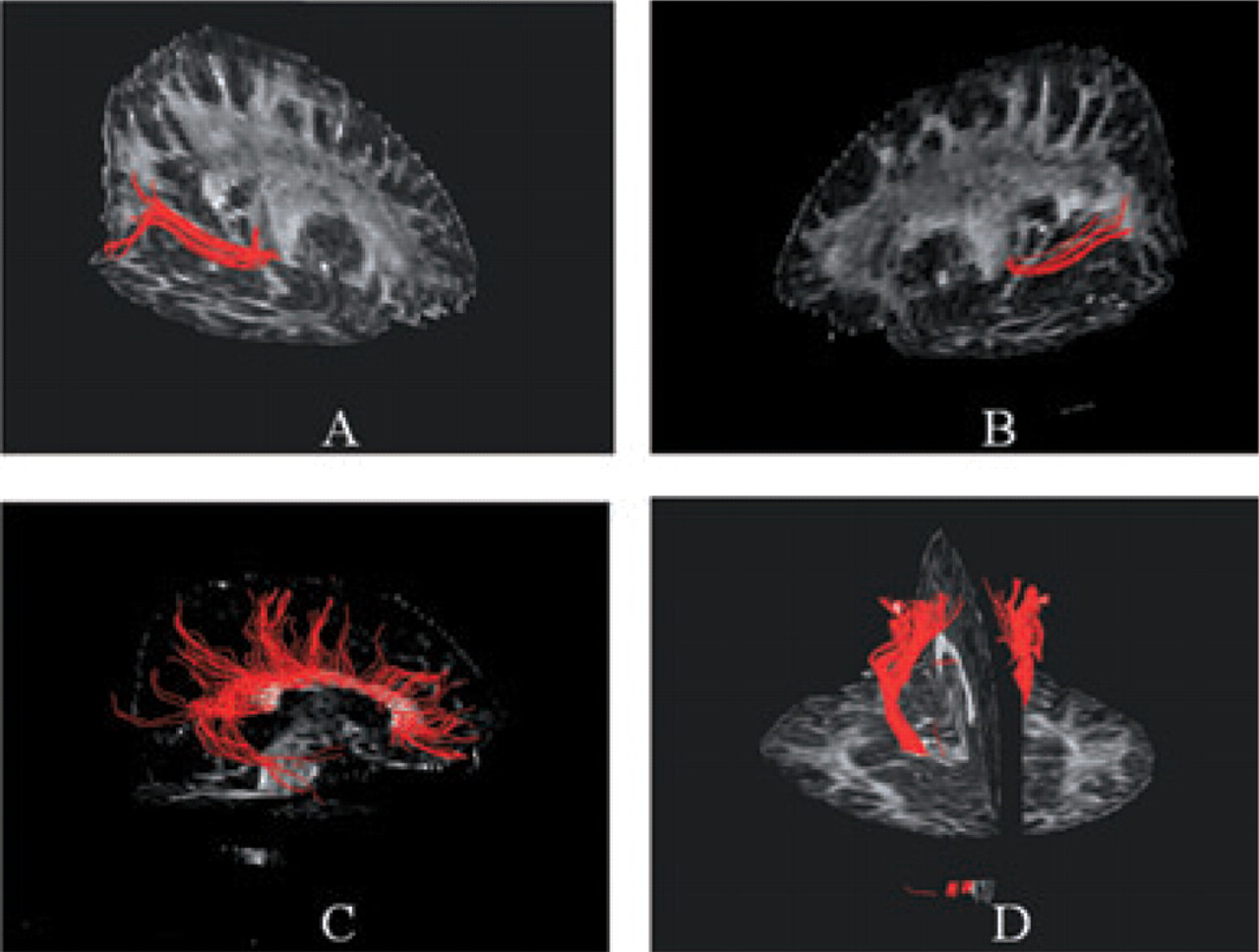
Illustrative behaviour of the main white matter fibre bundles studied in a single healthy control, prior to spatial normalization: (A) right optic radiation (OR), (B) left OR, (C) corpus callosum (CC), (D) corticospinal tracts (CST).
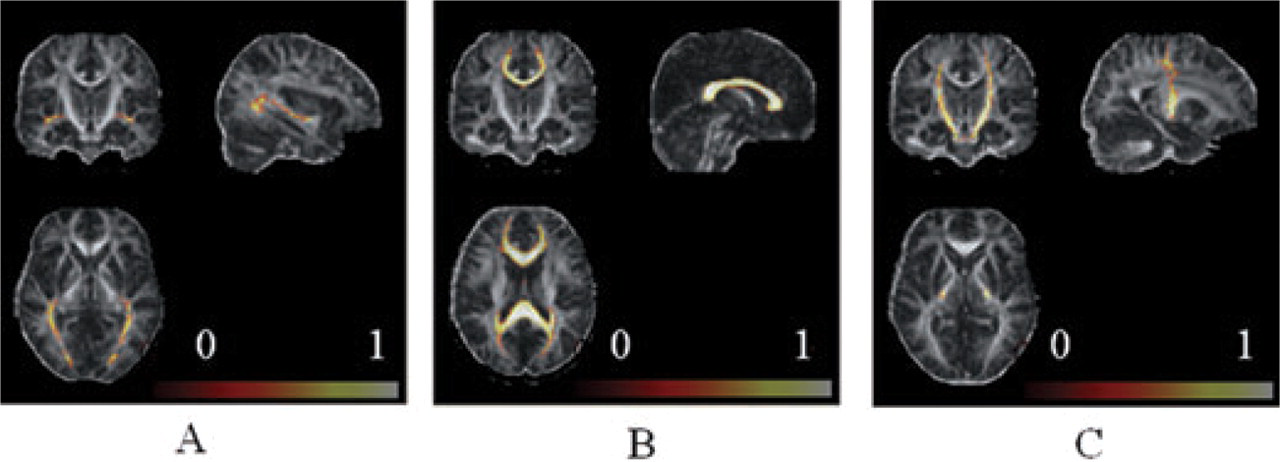
White matter fibre bundle probability maps of the optic radiations (A), corpus callosum (B) and corticospinal tracts (C) superimposed as a colour overlay to the fractional anisotropy (FA) atlas. The overlay indicates the probability (frequency of occurrence normalized for the number of subjects) of a pixel for individual subjects being part of the investigated tract.
T2-visible LV in the different WM fibre bundles were derived by applying fibre bundles probability maps obtained using DT tractography and calculating the volume of lesions inside the WM fibre bundles. The method is described in detail elsewhere (21).
All statistical analysis was performed using SPSS for Windows (version 13.0; SPSS Inc., Chicago, IL, USA). One-way analysis of variance was used to compare WM fibre bundles derived measures among the three groups. Post-hoc analyses were performed using an unpaired t-test. Univariate correlations were explored using Spearman's rank correlation coefficient.
Results
In migraine patients, the mean T2-weighted LV was 2.1 ml (range 0.02–11.2 ml), average lesion MD was 0.92 × 10−3mm2 s−1 (range 0.85–1.10 × 10−3mm2 s−1) and average lesion FA was 0.35 (range 0.24–0.42). Mean LV was 0.07 ml (
In Table 1, mean MD, FA and J determinants of the WM fibre bundles analysed from migraine patients and controls are reported. The three groups had different mean FA of the OR (P = 0.009). On post-hoc analysis, no difference was found for any of the WM fibre bundles metrics analysed between healthy controls and patients with MoA. Conversely, patients with MA had reduced average FA of the OR when compared with the other two groups (P = 0.009 vs. healthy controls and P = 0.05 vs. MoA patients). The analysis of right and left OR, separately, showed between-group differences for the FA values of both OR (P = 0.003 for the right OR and P = 0.05 for the left OR), and disclosed between-group differences for the MD values of right OR (P = 0.003) (Table 2). On post-hoc analysis, no difference was found in any of the WM fibre bundles metrics analysed between healthy controls and patients with MoA. Conversely, MA patients had reduced average FA of both OR when compared with healthy controls (P = 0.003 for the right OR and P = 0.05 for the left OR) and reduced average FA of the right OR when compared with MoA patients (P = 0.02). They also showed increased MD of the right OR in comparison with healthy controls (P = 0.02).
DT MRI derived metrics and Jacobian determinants of the different white matter fibre bundles studied from healthy volunteers and patients with migraine

One-way
Average MD of the diffusivity histogram is expressed in units of mm2 s−1 × 10−3, FA and J determinants are dimensionless indices.
DT, diffusion tensor; MRI, magnetic resonance imaging;
DT MRI-derived metrics and Jacobian determinants from the right and left optic radiations from healthy volunteers and patients with migraine

One-way
Average MD of the diffusivity histogram is expressed in units of mm2 s−1 × 10−3, FA and J determinants are dimensionless indices.
DT, diffusion tensor; MRI, magnetic resonance imaging;
No atrophy was detected in the OR of migraine patients (Tables 1 and 2).
In migraine patients, no significant correlation was found between OR-derived metrics and age, disease duration, frequency of attacks and T2 LV of the entire brain and of the selected WM fibre bundles (data not shown).
Discussion
In this study, we used DT MR tractography and a 3-T scanner to quantify the integrity of tissue microstructure of the OR in patients with migraine and to investigate whether the visual disturbances often experienced by these patients, mainly under the form of aura, might be associated with underlying structural changes. Given the preliminary nature of this study, we selected patients with evidence, albeit modest, of brain damage—in the case of patients with completely normal brain MRI scans and negative results, it would have been indeed impossible to know whether additional subtle changes were not present or the technology used not sensitive enough to detect them. The main advantage of using a 3.0-T system is the gain in signal-to-noise ratio (SNR) over lower field MR scanners and, as a consequence, the possibility of achieving a higher scan resolution that permits better visualization of the different anatomical structures. The improvement in SNR is particularly important for DT MRI, because the application of diffusion gradients causes attenuation of the signal and intrinsically limits the SNR available. In addition, since the direct application of streamline tractography algorithms in patients with brain lesions might be hampered by the presence of regions with decreased FA, resulting in uncertainty as to the principal diffusion direction, probability maps of the WM fibre bundles of interest were constructed from data of healthy controls and then applied to those of patients. This method has already been validated in patients with clinically isolated syndromes suggestive of multiple sclerosis and has been shown to be able to ameliorate the relationship between MR-derived metrics and clinical findings (21).
Using histogram-based analysis, previous DT MRI studies have shown the presence of subtle changes in the NABT of patients with migraine (5, 6). More recently, three studies have investigated changes of WM structures which were a priori considered to be a possible target of the disease (i.e. some visual processing areas and some pain processing related regions) and found these structures to be abnormal in patients both with and without aura (7–9). OR involvement was not selectively analysed in these studies, which recruited patients without macroscopic brain lesions (7–9).
In this study, we hypothesized that diffusion changes to the OR could be detected in patients with migraine and that the quantification of these changes might be different in patients with and without visual aura. To this end, we applied an objective and reproducible DT MRI-based method. In addition, in order to have a complete picture of the structural changes of the OR, we also used a recently developed approach, based on intersubject non-linear registration of FA images, which allows atrophy of the major WM fibre bundles in the brain to be spatially analysed (29). By using such an approach, we have demonstrated that MD and FA changes can be detected in the OR of patients with MA, and that these changes are more pronounced on the right side. These results confirm the exquisite sensitivity of DT MRI in revealing microscopic disease-related changes in migraineurs and prompt the application of this technique to the assessment of larger and more heterogeneous groups of patients in order to gain additional information for the understanding of migraine pathophysiology.
Although this study can give only a partial answer to the pathological substrates of OR diffusion changes in patients with MA (but definitive histopathological correlations in these patients are unlikely ever to be obtained), it is worth noting that we found significantly decreased OR FA, slightly increased OR MD (albeit significant only for the right side), but no change of OR volumetry. Since the major determinants of anisotropy in neuronal structures are thought to be axonal membranes, whereas the contribution of myelin is likely to be minor (32), axonal pathology should be considered as a possible explanation of our findings. However, since axonal loss alone would result in abnormal MD and FA values and in a reduction of OR volumetry, the mismatch between OR intrinsic changes and volumetry suggests that axonal damage might be followed by proliferation and hypertrophy of astrocytes. Alternatively, the decreased OR FA, with preservation of volumetry, might be secondary to an increased axonal diameter (33). Although this latter explanation fits with the theory of a sort of experience-dependent structural plasticity (34, 35), the lack of a correlation between OR diffusion changes and disease duration argues against this hypothesis.
Although the partial lateralization of OR diffusion changes we found in patients with MA should be considered with caution, given the relatively small sample of patients studied and the heterogeneity of lateralization of visual aura in these patients, our findings support the ‘right hemi-ageing model’, which proposes that the right hemisphere is more vulnerable to age-related changes than the left hemisphere (36), as well as other MRI studies showing that an asymmetric distribution of tissue changes is not uncommon in several neurological disorders (37, 38).
Having established that OR diffusion changes exist in patients with MA, the second aspect deserving consideration is whether these changes are congenital or secondary to the repetition of migraine attacks over time. Although the results of the correlation analysis have to be considered with caution, given the relatively small number of subjects studied, the absence of correlation between OR diffusion changes and number of attacks, disease duration and T2-visible lesion load suggests that two of the mechanisms that have been considered by several authors to explain MRI findings in migraine, i.e. the repetition of episodes of oligaemia (39) and retrograde degeneration of axons passing through macroscopic WM lesions (5, 6), are unlikely to explain our findings. Conversely, the lack of these correlations suggests that the observed DT MRI changes might represent a phenotypic biomarker of the disease, reflecting a congenital condition rather than a process related to disease progression over time. This hypothesis is supported by the recent study of DaSilva et al. (9), who found no correlation between changes of cortical thickness in the somatosensory cortex and clinical data (disease duration, age at onset and attack frequency) in patients with migraine, and is in line with previous theories of intrinsic increased susceptibility to spontaneous depolarization in migraine, which might reflect abnormal presynaptic P/Q-type calcium channels (40) and low brain magnesium levels (41), as well as a disorder of mitochondrial energy metabolism (42, 43). It is also supported by twin and family studies showing that a genetic component is at work in this disease, which is more evident in patients with MA (44–46).
