Abstract
The aim of this study was to determine whether subcutaneous octreotide is effective for the treatment of acute migraine. Patients with migraine with and without aura as classified by the International Headache Society were recruited to a double-blind placebo-controlled crossover study. Patients were instructed to treat two attacks of at least moderate pain severity, with at least a 7 day interval, using subcutaneous 100 μg octreotide or matching placebo. The primary endpoint was the headache response defined as: severe or moderate pain becomes mild or nil, at 2 h. The primary endpoint was analysed using a Multilevel Analysis approach. Secondary end-points included associated symptoms and a four-point functional disability score. The study was powered to detect a 30% difference at an α of 0.05 and a β of 0.8. A total of 51 patients were recruited, of whom 42 provided efficacy data on an attack treated with octreotide and 41 with placebo. Modelling the headache response as a binomial determined by treatment, using the patient as the level 2 variable, and considering a possible period effect, and sex and migraine type as other variables of interest, subcutaneous octreotide was not significantly superior to placebo. The two hour headache response rates were 20% for placebo and 14% for octreotide, whilst the two hour pain free rates were 7% and 2%, respectively. Subcutaneous octreotide 100 μg is not effective in the acute treatment of migraine when compared to placebo.
Introduction
The development of new medicines for the acute treatment of migraine is desirable for clinical prac-tice (1). Although a number of triptans, serotonin 5-HT1B/1D receptor agonists, have become available since the discovery of sumatriptan (2, 3) there is still a need for further improvements in abortive agents for migraine. Triptans are contra-indicated in patients with vascular disease, and a group of patients experience universally unacceptable side-effects on these agents (4). Both these issues suggest a need for the development of novel drugs for migraine. Moreover, some patients overuse 5-HT1B/1D-receptor agonists (5) particularly those with long-lasting attacks and headache recurrence, and it is desirable to develop further strategies that may circumvent this problem.
Given the involvement of neuropeptides in migraine (6) there has been interest in the development of new classes of drugs that are specific for certain neuropeptide receptors. Calcitonin gene related peptide (CGRP) receptor antagonists (7, 8) have been developed and early results in the acute treatment for migraine are encouraging (9)
Somatostatin, a 14-amino acid peptide, is known to have an inhibitory effect on a range of neuropeptides, including substance P (15), CGRP (16), and vasoactive intestinal polypeptide (VIP) (17), and has therefore been previously proposed as a potential pharmacological approach to the management of migraine (18). From a therapeutic viewpoint the problem with native somatostatin is that it is an unstable compound, being broken down by endogenous peptidases within a few minutes (19). Octreotide is a somatostatin analogue with longer half of approximately 1.5 h (19). In a study of 29 patients, Kapiciolgu et al. (20) demonstrated that subcutaneous administration of 100 µg octreotide gave headache relief at six hours in 77%, compared to 25% of a parallel placebo-treated group. This observation has not been repeated and six hours is well beyond International Headache Society guidelines (21) and patient expectations (22, 23). Intravenous infusion of somatostatin was shown to be as effective as ergotamine in the acute treatment of cluster headache (24), although this again has not been systematically further studied and there was no placebo control arm.
The analgesic effect of somatostatin analogues, particularly octreotide, in the headache associated with growth hormone secreting pituitary tumours is well recognized (25–27), although the mechanism is unknown. It has been hypothesized that octreotide inhibits the production of a nociceptive peptide by the tumour (25, 28, 29). However, the onset of analgesia in this setting is more rapid than the 30 min required to suppress growth hormone (30) suggesting that octreotide has a direct antinociceptive property (31).
Five somatostatin receptors (sst1−5) have been cloned (32) with octreotide acting predominantly on sst2 and sst5 (33). The distribution of sst2 within the central nervous system strongly suggests that this particular somatostatin receptor has a role in cranial nociception, being highly expressed in the trigeminal nucleus caudalis and periaqueductal grey (34). Octreotide is a nonlipophilic compound and is believed to poorly penetrate the blood–brain barrier (35, 36). This situation is somewhat analogous to the triptans, which also claimed to have the combination of apparent poor central nervous system accessibility (37) and undoubted efficacy in primary headache (3).
We sought to re-examine the finding that octreotide is an effective agent for the acute treatment of migraine (20) by studying a larger group of patients in a double-blind placebo controlled cross-over study using a dose established to be effective in acromegaly associated headache (27), and which we have recently found effective in a parallel study in cluster headache (38). We used the more broadly accepted end point of the 2 h headache response rate, for comparison with previous studies (39), and since patients desire quick relief of symptoms (22, 23).
Subjects and methods
Men or women between 18 and 65 years of age were recruited for the study, the majority of patients responding to trial advertisements published in patient information newsletters (The Migraine Trust and Migraine Action Association). All patients interested in the study underwent an initial telephone interview, followed by a detailed face to face clinical interview, in order to confirm the diagnosis of migraine with or without aura as defined by the International Headache Society (40) and consistent with the revision (41).
Exclusion criteria included pregnancy and lactation; analgesia or triptan overuse; frequent tension-type headaches (> 10 days/month); inability to distinguish between tension-type or migraine headache; diabetes mellitus; or a coexisting condition that might expose the patients to a disproportionately increased risk of a significant adverse event: ischaemic heart disease, peripheral vascular disease, cerebrovascular disease, uncontrolled hypertension (blood pressure > 160/95), epilepsy, use of cimetidine, dopamine agonists, cyclosporin or oral hypoglycaemic agents, renal or hepatic impairment. The study was performed in accordance with the ethical principles of the Declaration of Helsinki, and was given local ethical committee approval (Ref 00/N117), as well as Medicines Control Agency (MCA) approval for the administration of octreotide as part of a clinical trial.
Design and procedures
The design was a randomized, double-blind, placebo-controlled, single-centre two-attack crossover study, involving patients with acute migraine headache of moderate to severe intensity. Octreotide was kindly donated for the purposes of the trial by Novartis who had no input into the design or analysis of the study, and provided no other support for the study of any kind. Octreotide and placebo were packaged for a fee into identical prefilled vials at The Royal Free Hospital pharmacy. All drugs were kept refrigerated in the National Hospital for Neurology and Neurosurgery (NHNN) pharmacy department for the duration of the trial. Normal saline was used as placebo and the dose of octreotide was 100 µg.
At the first visit, patient eligibility for the study was confirmed and a physical examination was performed. Patients who met the inclusion criteria and were willing to participate in the study signed consent agreements authorized by the local ethics committee (NHNN).
Study medication and placebo were provided in two identical prefilled vials, each containing 1 ml of fluid. The vials were labelled Treatment 1 and Treatment 2 for self-administration during a first and second headache attack, respectively, each attack being separated by a minimum of 7 days. The order of active treatment and placebo was randomized by NHNN pharmacy, and each participant was assigned a randomization number. The code was held by the pharmacy until study completion and database locking. All patients were counselled how to draw up the treatment with a 1 ml syringe and taught how to give a self-administered subcutaneous injection. Participants were given the opportunity to practice the injection in supervised conditions during this visit in order to build up confidence. A help line was provided to patients to ring if they experienced trouble during the trial.
Efficacy assessments
Patients were asked to complete forms during both treatments at home and to report to the Unit within 48 h of completing both treatments. Headache severity was measured on a 4-point scale (0 = no pain; 1 = mild pain; 2 = moderate pain; 3 = severe pain) at 0 min, 30 min, 1 h, 2 h, 4 h and 24 h after study intake. The primary end points for clinical efficacy were the relief of headache, from grade 3 or 2 to grade 1 or 0, within 2 h after injection for consistency with recent development programmes in acute migraine (39). Additional efficacy measures included headache free rate (21), functional disability (4-point scale) and the presence or absence of nausea, vomiting, photo- or phonophobia, all of which were monitored for 24 h. Rescue medication was permitted 2 h after the intake of study medication if the headache still required treatment. Ergotamine and triptans were accepted as rescue medication. Adverse effects were documented on the forms provided after both injections.
Statistical analysis
The primary efficacy analysis was based on the number of patients who obtained relief from grade 3 or 2 to grade 1 or 0 within 2 h of treatment. Assuming a treatment difference between placebo and active of 30% based on subcutaneous sumatriptan and placebo response rates (42) it was calculated that 42 patients were needed for the trial to have an 80% power to detect a difference at an α of 0.05 (Sample Power, SPSS Inc.).
The outcome data were to be treated as binary: headache response or none at 2 h. Because the trial was a cross-over study, attacks 1 and 2 were not truly independent because the patients remained the same, a multilevel analysis (43, 44) approach was employed using the software that has been devel-oped by the Multi-level Project, MlwiN (available at http://www.ioe.ac.uk/multilevel). We have used this approach successfully in a previous study (45).
Results
A total of 51 patients were recruited, 2 men and 49 women, with a mean age of 48 ± 12 years (mean ± SD). Patient demographics are shown in Table 1.
Demographic data and migraine characteristics of 43 patients completing study

Disposition of patients
Of 51 patients recruited, 8 withdrew before completion of the study; one was unable to co-ordinate self-injection during a migraine attack, and seven were lost to follow up; whether they treated or did not treat attacks is not clear. We have treated them as if they did not treat any attacks. One patient was excluded from the first arm of the trial, and two from the second arm because of protocol violation, which was to treat mild pain (Fig. 1).

Disposition of the patients in the study.
Clinical features of study cohort
Of 43 patients that successfully completed the study, the frequency of attacks included 4–10 per month (35%), 1–3 per month (60%) and < 1 per month (5%). The mean duration of attack was 53 ± 4.3 h. Of the group, 33 (77%) had migraine without aura and 10 (23%) had migraine with aura. Thirty-eight (88%) had previously used 5-HT1B/1D-agonists for treatment of acute attacks, of which 28(74%) experienced a meaningful response within 2 h.
Overall efficacy
The primary endpoint of the study was the combined, attack 1 and 2, headache response rate to octreotide at 2 h compared to placebo. The Wald test was not significant for the overall regression, suggesting that there was no difference between octreotide and placebo even accounting for treatment order, sex, or migraine type.
Efficacy results
In total, 42 attacks were treated with octreotide and 41 attacks with placebo. In the octreotide-treated attacks, 6 (14%) patients reported headache relief at 2 h, compared to 8 (20%) in the placebo group.
One patient was rendered pain-free at 2 h with octreotide (2%), whilst 3 patients were rendered pain-free with placebo (7%).
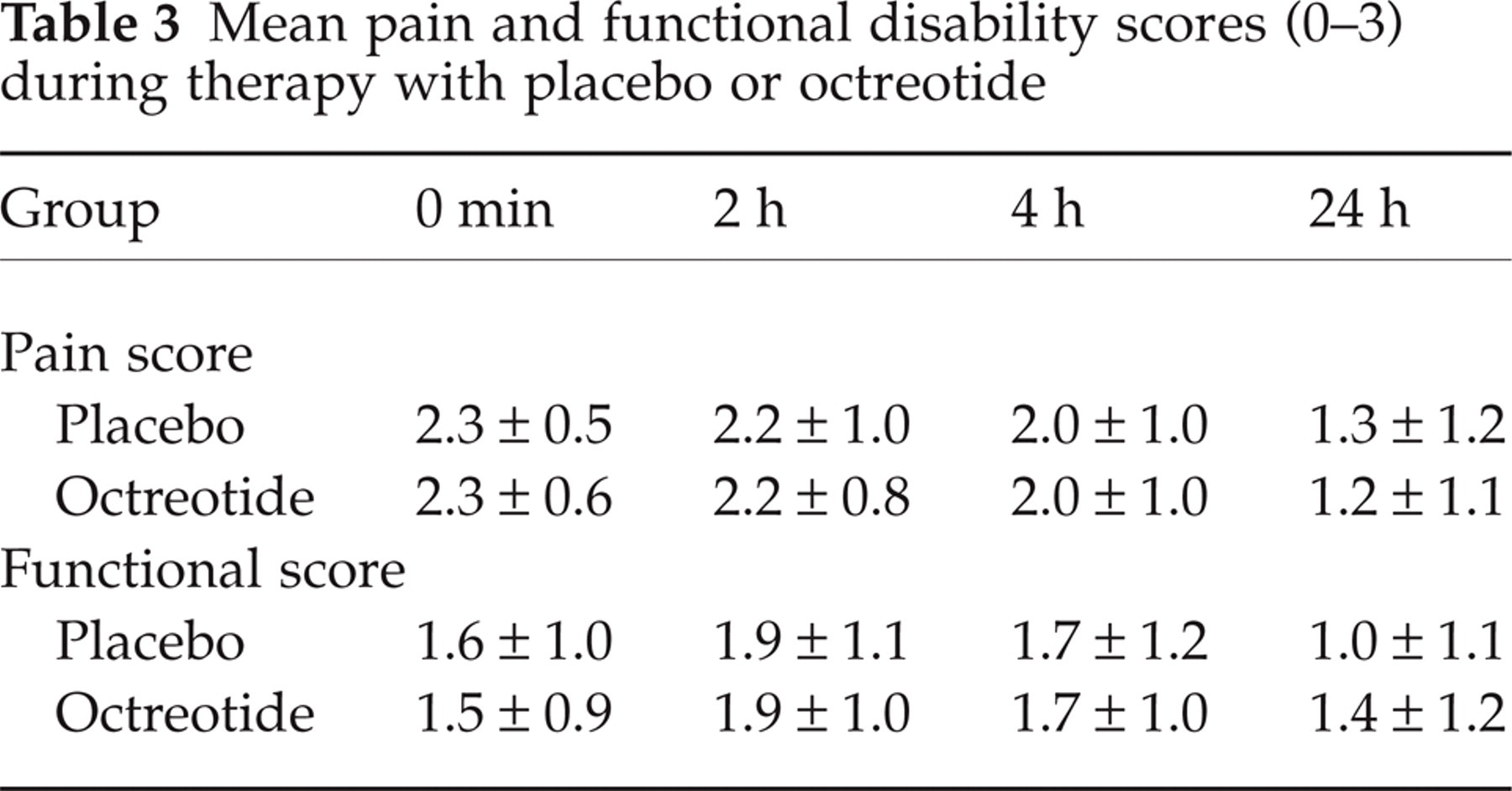
The 2 h- and pain-free response rates are shown in Table 2. Mean pain scores at each time interval are shown in Table 3.
Two hour and pain free response rates of placebo and octreotide for both attacks combined

Headache response, relief of headache from moderate/severe pain to nil or mild pain.
Pain free, relief of headache from moderate/severe pain to no pain.
Mean pain and functional disability scores (0–3) during therapy with placebo or octreotide
Associated symptoms and functional score
To evaluate the associated symptoms, only patients who had the symptoms during an attack were included in the analysis. Octreotide performed less well than placebo for nausea, photophobia, phonophobia and aggravation with movement (Fig. 2). There was no difference in functional disability scores between either group at any of the time points post-treatment (Table 3).

Number of patients reporting persistent associated symptoms at 2 h. ▪ octreotide; □ placebo.
Escape medication
The frequency of the use of escape medication was no different in the octreotide-treated attacks compared to those treated with placebo: 35 (83%) vs. 33 (80%) as shown in Table 4.
Use of escape medication in octreotide- and placebo-treated groups

Tolerability
No serious adverse events were reported in either the octreotide- or placebo-treated attacks. Four patients experienced diarrhoea with octreotide and one patient experienced increased sweating, and another excessive nausea with octreotide, in comparison with their normal attacks. With placebo, one patient reported diarrhoea, one patient experienced light-headedness, and two patients reported excessive tiredness after injection.
Discussion
Contrary to a previous report, the results of this study do not show a difference between 100 µg octreotide and placebo in the acute treatment of migraine. We did not find octreotide to be superior to placebo for relief of headache, associated symptoms or functional disability scores. There was some small excess of adverse events in the octreotide group, suggesting the treatment was biologically active.
It is not clear why our findings are so different to the previous study (20). Our placebo response rate of 20% is not substantially different from the 25% placebo in the previous study (20), suggesting that there was little difference in the patient population group in terms of broad tractability to treatment. However, the relatively low placebo response rates in both studies suggests that there may have been a selection bias towards a group of patients with difficult to treat migraine. This seems likely to us given that patients who volunteered were willing to have an injection for their migraine attacks. The mean attack duration in the study group was 2.2 days, which is longer than that seen in population studies (46, 47). Despite this, 74% of our study group reported a useful response with 5-HT1B/1D-receptor agonists, suggesting that we did not select an entirely refractory group.
In the previous study (20) the primary efficacy end-point was response rate at 6 h (76%), compared to our end-point of response rate at 2 h (14%). Although response rates at 2 h were not provided, a statistically significant difference in pain scores at 2 h was reported, suggesting an analgesic action at this earlier time point. In our study we did not find a difference between octreotide and placebo at the latter time point of 4 h (Table 3), suggesting that the superior response rates observed in the earlier study were probably not related to the difference in time-to-headache relief used in the two studies. It is generally considered that if an acute treatment for migraine is to be useful in clinical practice, it should have an effect within 2 h (22, 48). For an injectable treatment this is considered slow, so our primary end point of 2 h could be considered generous.
The other difference from the previous study (20) was that we employed a cross-over design. It is possible that the excellent response rate found in the treatment group in the previous study (20) was due to a difference in migraine characteristics from the placebo-treated group; this must be an unusual situation if randomization and blinding are preserved. The previous study (20) did not give outcome data for the secondary end-points, such as associated symptoms or functional disability scores; our findings were negative. The gastro-intestinal side-effects observed in five patients in response to 100 µg octreotide in our study is in keeping with the reported experience of side-effects on this dose of the drug (49). In addition we used the same dose of octreotide as in the previous study (20), suggesting that the difference in observed response is not dose-related.
Although our findings were negative, the somatostatin receptor remains a theoretically attractive pharmacological approach to primary headache management. There are in vivo animal data to suggest that the central administration of somatostatin and its analogues results in antinociception, both in peripheral (50) and trigeminal (51) pain models. In conjunction with the localization of somatostatin receptors (particularly sst2) in the trigeminal nucleus caudalis and periaqueductal grey (52), these findings suggest that drugs acting on the somatostatin receptor remain realistic therapeutic options. It is possible that octreotide is simply the wrong compound due to its nonlipophilicity and apparent poor ability to cross the blood–brain barrier (35, 36). The development of a somatostatin analogue with good brain access must be the clearest way to test the somatostatin theory. Novel somatostatin analogues with differing receptor affinities are currently being developed for the treatment of functional pituitary adenomas (53). Some of these compounds are particularly potent at the sst2 receptor, making them potential options as analgesic agents (54). Lastly, our data would not necessarily predict the outcome of a similar study in cluster headache, which behaves in a therapeutically distinct manner in many respects (55), although acute attacks do share triptan sensitivity (56). Our initial analysis of such a study demonstrated a clear effect of octreotide 100 µg in treating acute cluster headache (38).
In conclusion we have seen no effect of the somatostatin receptor agonist octreotide in a placebo-controlled double-blind crossover study in acute migraine. Further tests of the therapeutic efficacy of somatostatin receptor agonists should include compounds with access to the central nervous system.
Footnotes
Acknowledgements
PJ Goadsby is a Wellcome Senior Research Fellow.
