Abstract
Dale showed in 1906 in a seminal work that ergot inhibits the pressor effect of adrenaline. Stoll at Sandoz isolated ergotamine from ergot in 1918. Based on the belief that migraine was due to increased sympathetic activity, ergotamine was first used in the acute treatment of migraine by Maier in Switzerland in 1925. In 1938 Graham and Wolff demonstrated the parallel decrease of temporal pulsations and headache after ergotamine i.v. This inspired the vascular theory of Wolff: an initial cerebral vasoconstriction followed by an extracranial vasodilation. Dihydroergotamine (DHE) was introduced as an adrenolytic agent in 1943. It is still in use parenterally and by the nasal route. Before the triptan era ergotamine and DHE had widespread use as the only specific antimigraine drugs. From 1950 the world literature on ergotamine was dominated by two adverse events: ergotamine overuse headache and the relatively rare overt ergotism. Recently, oral ergotamine, which has an oral bioavailability of < 1%, has been inferior to oral triptans in randomized clinical trials. A European Consensus in 2000 concluded that ergotamine is not a drug of first choice. In an American review of 2003 it was suggested that ergotamine may be considered in the treatment of selected patients with moderate to severe migraine.
Keywords
Introduction
Traditionally both vascular (1–4) and neuronal (5–8) theories of migraine have dominated in different historic periods. In 2002 migraine headache was described as neurovascular (8).
Ergotamine, which for many years was the mainstay of migraine therapy, has contributed to the dominance of the vascular theory, because of its relative specificity for cranial vessels (9, 10). Ergotamine, however, also inhibits neurogenic inflammation in animals (11); and its derivative dihydroergotamine (DHE) has a central effect in the brainstem (12), indicating a possible effect on central trigeminal pain processing.
The use of ergot extract was recommended as a vasoconstrictor drug in migraine in 1868 by Woakes (13, 14). It was used to constrict the dilated vessels. At that time it was also thought that migraine, because of the pale face of the sufferer, was due to hypersympathetic activity (14, 15).
Seminal work by Sir Henry Dale
In 1906 Dale, Nobel Prize winner of 1936, showed that a liquid extract of ergot inhibited the pressor response to adrenaline in anaesthetized cats (16, 17). He further showed that when stimulation of nerves led to contraction in various organs, in situ ergot could inhibit these effects. Conversely, when stimulation or adrenaline led to relaxation, ergot was not effective as an inhibitor. In fact, Dale had then identified the α- and the β-adrenoceptors as proposed 40 years later by Ahlquist (18).
Introduction of ergotamine in migraine treatment
Ergot treatment had been unreliable because of varying alkaloid content, but in 1918 Stoll at Sandoz in Basel isolated ergotamine from ergot (Fig. 1) (19–21). The pharmaceutical product was called Gynergen, since it was envisaged that the drug should be used in gynaecology and obstetrics for its uterotonic effect. Migraine was believed to be due to vasospasm because of the pale face of most migraineurs during attacks. Rothlin at Sandoz thought that the adrenolytic properties of ergotamine (22) would counteract the sympathotonic mechanism of migraine, and his colleague Maier in Zürich did a successful study in 1925 (23), which was confirmed by Trautmann in Germany in 1928, using placebo controls (24). The effect of ergotamine in migraine was also investigated in an open study in 1928 France (25).
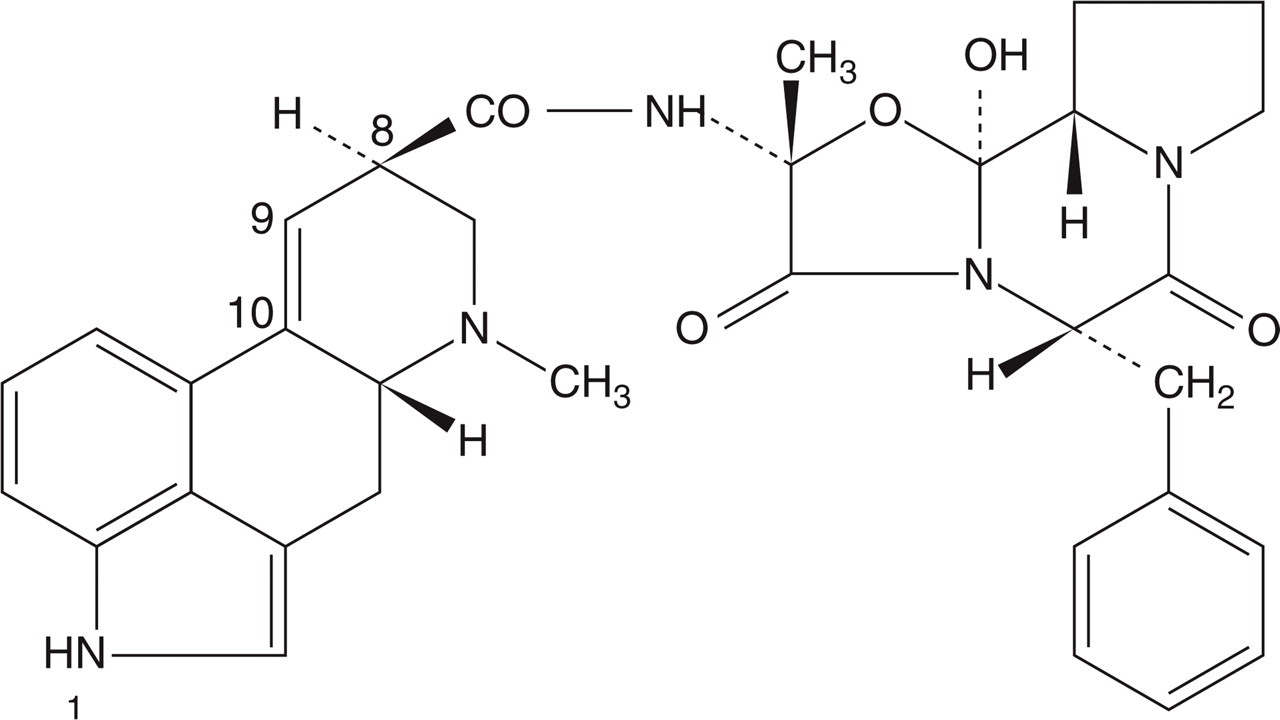
Chemical structure of the alkaloid ergotamine. Dihydroergotamine is hydrogenated at the double-binding 9–10.
Introduction of ergotamine in the USA
Ergotamine was then introduced in the USA, and in 1934 three reports on its use in migraine were published (26–28). Lennox reported relief after injections of ergotamine in 40 of 45 patients (27), and Logan and Allen reported the drug to be effective in 67 of 71 attacks in nine patients (28). Brock et al. reported that ergotamine given subcutaneously was effective in 14 of 18 patients (26). In 1935 Lennox and Von Storch reported the drug to be efficacious in 90% of 109 patients treated with intravenous ergotamine (29). He notes that: ‘As yet unexplained is the fact that after subcutaneous injection, blood pressure changes and uterine contractions begin almost at once but relief of headache does not begin for nearly one hour’. In 1936 O'Sullivan described the effect of ergotamine as outstanding (30), and all but eight of 97 patients experienced benefit from ergotamine. In one of the first efficacy studies on oral ergotamine, the drug, used in a dose of 2 mg/h up to a total of 8 mg, resulted in relief of headache in 41% and improvement in 27% of 56 patients (31). In contrast, 0.5 mg ergotamine i.v. resulted in freedom from headache in 89% and improvement in 5% of 140 patients. This uncontrolled study (31) indicated that parenteral ergotamine was more effective than the oral form. The low oral bioavailability will be discussed below.
Ergotamine and the vascular theory of migraine
Graham and Wolff demonstrated in 1938 that ergotamine decreased migraine headache along with the pulse amplitude measured over the temporal artery (Fig. 2) (32). Migraine was envisaged as a condition with initial cerebral vasoconstriction followed by extracranial reactive vasodilation (3). Later, in 1957 Brazil and Friedman showed, however, that ergotamine and DHE can cause both decrease and increase in temporal artery pulsations during migraine attacks (33). This is most likely due to the fact that pulsations in an artery not only depend on the state of the artery, but also on state of the vascular bed it is feeding (34).
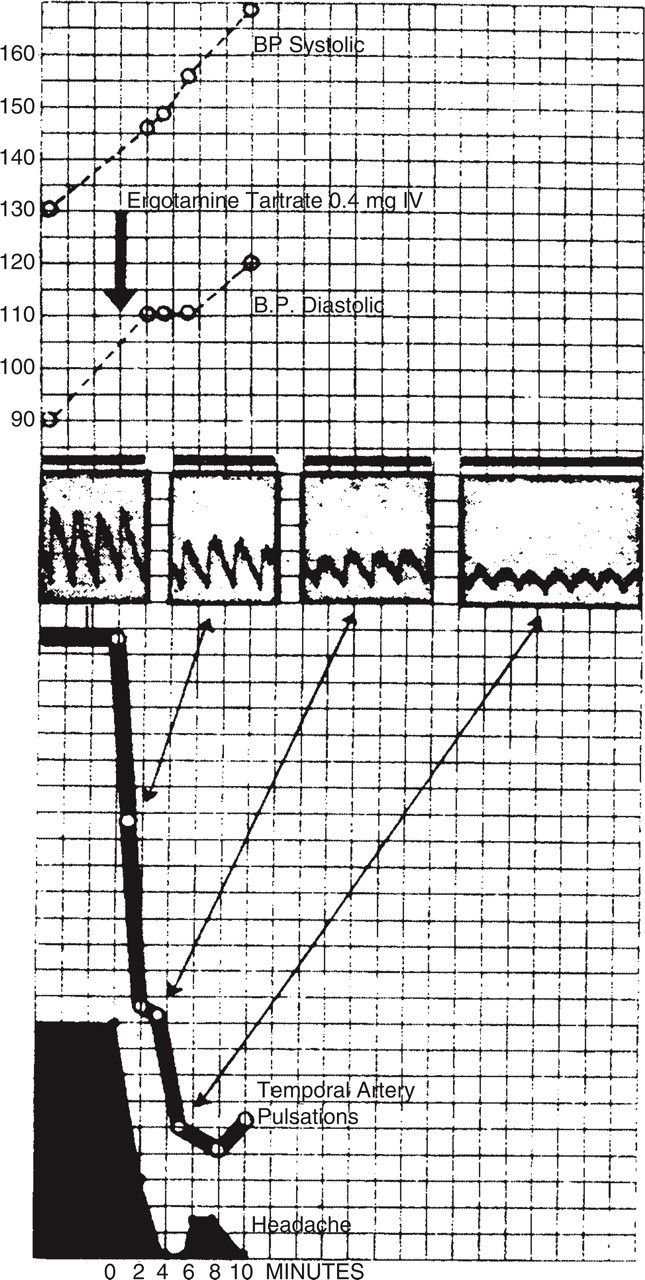
Parallel decrease in pulsation of the temporal artery and headache in a migraine patient treated with 0.4 mg ergotamine tartrate i.v. From Graham and Wolff (32) with permission.
That ergotamine is a relative selective constrictor of cranial vessels in dogs was later shown by Saxena in 1972 and 1974 (9, 10).
Dihydroergotamine
DHE was developed in 1943 (35) and tried in migraine on the basis of the belief that migraine headache resulted from an increase in sympathetic activity (17). Accordingly, DHE was expected to be more efficacious than ergotamine in treating migraine, since it possesses greater α-adrenoceptor activity (17). However, early clinical experience showed that DHE, although effective in the acute treatment of migraine, must be given in higher doses than ergotamine (17, 36–38).
Subsequent studies indicated that the weaker vasoconstrictor effect of the drug could make it suitable for migraine prophylaxis (17, 39–41); and DHE has in some much later randomized clinical trials (RCTs) in the 1980s been shown to be superior to placebo in high (10 mg) doses for migraine prophylaxis (42). It some countries it is still used for this indication.
It was later in 1987 and 2001 shown that intravenous or subcutaneous DHE probably has less effect on peripheral arteries than ergotamine (43, 44). In contrast, DHE has a pronounced effect on veins (45). It has therefore been used, together with heparin, in the prophylaxis of postoperative thrombophlebitis (46), and in orthostatic hypotension (47).
DHE may also inhibit ‘neurogenic inflammation’ in the trigeminovascular system (11) and it has been used parenterally in status migrainosus (48), and by the nasal route in the acute treatment of migraine (49). In 2006 the pharmacology and evidence for efficacy in migraine of DHE was reviewed (50). It was noted that ‘results from published clinical trials and case series have demonstrated that DHE, administered by the intravenous, intramuscular, subcutaneous, or intranasal route, is effective and well tolerated in the treatment of migraine headache’ (50).
In on RCT from 1996, headache relief after subcutaneous DHE 1 mg (86% after 4 h) was quite similar to that after subcutaneous sumatriptan 6 mg (83% after 4 h) with a slower onset of action of DHE (50). DHE resulted in fewer recurrences than sumatriptan (50).
Efficacy of ergotamine in migraine
Being an old drug, ergotamine never underwent a trial programme such as those used nowadays for the triptans. It was for a long period the standard antimigraine drug. From 1970 to 1989, oral ergotamine was studied in several trials and was superior to placebo in five RCTs, whereas it was not better than placebo in two RCTs (51) (see Table 1). Considering the extremely low oral bioavailability of ergotamine (see below), it is amazing that the drug works by the oral route of administration. Active metabolites are probably present. Oral ergotamine was comparable to several other drugs in RCTs (51), but was inferior to oral sumatriptan 100 mg (48% vs. 66% headache relief, P < 0.001), to oral rizatriptan 10 mg (47% vs. 76%, P < 0.001) and to oral eletriptan 40 mg (33% vs. 54%, P < 0.01) in three RCTs (51, 52). In these RCTs, recurrences were less frequent after ergotamine than after the triptan (52).
Efficacy of oral ergotamine vs. placebo in seven randomized, double-blind, clinical trials. For references, see Tfelt-Hansen and Saxena (51)
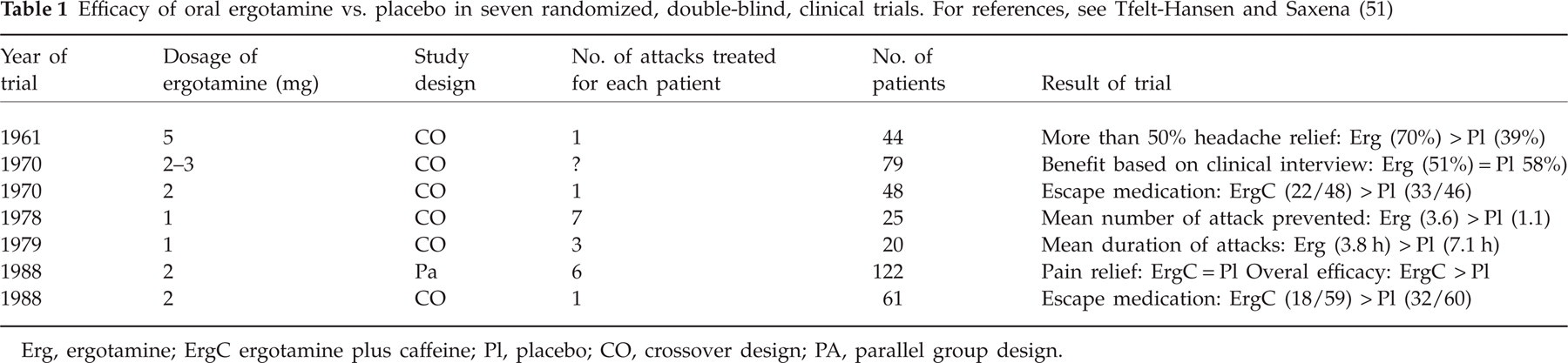
Erg, ergotamine; ErgC ergotamine plus caffeine; Pl, placebo; CO, crossover design; PA, parallel group design.
Based on RCTs, it was questioned in 1993 whether oral ergotamine is at all effective in migraine (53). However, in one RCT rectal ergotamine (73% relief) was superior to rectal sumatriptan (63% relief), the current standard drug for migraine (54, 55) (Fig. 3), demonstrating that ergotamine per se is effective in migraine.

Headache relief (a decrease from moderate or severe headache to none or mild) for rectal ergotamine tartrate 2 mg plus caffeine 100 mg vs. rectal sumatriptan 25 mg in one crossover, randomized, clinical trial (54, 55). Note that sumatriptan was apparently better (NS) in the beginning, whereas ergotamine (73%) was superior to sumatriptan (63%) after 2 h.
A European consensus report in 2000 concluded that ergotamine is not a drug of first choice in the triptan era, but may be useful in long-standing migraine attacks with multiple recurrences (54). If used, ergotamine should be administered by the rectal route (54). In an American review of ergotamine and DHE from 2003, it was stated that ‘ergotamine may be considered in the treatment of selected patients with moderate-to-severe migraine’ (35). ‘Patients with very long lasting attacks or with frequent headache recurrences may be especially suited for ergotamine, as headache recurrence is probably less likely with ergotamine’ (35).
For most patients the more specific drugs, the triptans, are the drugs of first choice, if specific antimigraine drugs are needed. It is our clinical experience, however, that a small minority of migraine patients respond only to ergotamine and not at all to triptans.
From 1950 on, the literature on ergotamine, apart from its use in comparative trials of new antimigraine drugs (51, 52), has been mostly on adverse events (56, 57), including ergotamine-induced headache (58–65) and the relatively rare cases of overt ergotism (56, 66).
Ergotamine overuse headache
In 1949 the vasoconstrictory effect of ergotamine was described as short-lived (67). This was based on the effect of ergotamine on blood flow in an extremity in dogs (67). Ergotamine should therefore be regarded as a safe drug (67).
However, Silverskiöld from Sweden in 1947 (68) and Peters and Horton from the USA in 1951 (69) described ergotamine-induced headache.
Silverskiöld reported on seven patients with possible abuse of ergotamine, but was cautious (68, 70): ‘I do not wish to maintain that in these seven cases Gynergen medication had a specifically deleterious effect, although that is possible’.
Peters and Horton more clearly identified the problem of ergotamine overuse, and in 1951, in their classic paper, reported on 19 patients who had used ergots for prolonged periods (69–71). They wrote: ‘When individuals were taking the drug (ergotamine tartrate) every day, and its effects wore off, headache would recur, necessitating its further administration for relief. The characteristics of the original migraine headache under such conditions became somewhat changed, that is, nausea and vomiting usually were absent, leaving only the periodic recurrence of the hemicranial vasodilating headache’.
They proposed that frequent use of ergotamine induced the ‘ergot cycle’, where vasoconstriction ended up in vasodilation, which required the next dose of ergotamine, an endless cycle causing chronic headache resistant to any treatment except ergotamine withdrawal (69).
Lippman described in 1955 the characteristic headache resulting from prolonged daily use of ergotamine or DHE as a daily dull posterior headache in nine cases (72).
In 1955, Friedman et al. (73) cautioned against frequent ergotamine use: ‘This danger seems to lie in an evocation of a medical paradox. Namely, in some patients the use of ergotamine relieves the headaches for which it is administered but at the same time leads to an increased frequency of these headaches’ (60).
In 1963 Horton and Peters reported on 52 patients with excessive use of ergotamine (58). The maximum used dose of ergotamine was 37 mg in 1 day in a cluster headache patient. He suffered from intermittent claudication in the legs. Toxic symptoms occurred in 37 of 52 patients (58). In hospital, treatment with withdrawal of the drug was suggested (58).
In the next two decades further clinical studies (59–64) of ergotamine overuse were published see (Table 2) (59–62). The duration of ergotamine overuse ranged from 3 weeks to 20 years; the monthly dose of ergotamine ranged from 15 mg to > 300 mg. Most patients (range 50–100%) had daily headaches; constant nausea occurred in 30–50% of patients; and withdrawal headache occurred in 79–91% of patients (59–62). The prognosis of recurrence for pure ergotamine overuse was reasonable good. Thus, only 26% started excessive use of ergotamine again after a median time of 1.5 years (61). In a group of patients with both ergotamine and analgesic overuse, 40% had relapsed within a follow-up time of 5 years (66, 74).
Summary of four reports on patients with ergotamine overuse

Rowsel et al. (59).
Andersson (60).
Tfelt-Hansen and Krabbe (61).
Ala-Hurula et al. (62).
Not relevant since the dose was gradually tapered off.
n.g., not.
If ergotamine is to be used safely in the treatment of migraine, the following points should be observed: (i) ergotamine should be used only for the treatment of migraine attacks; (ii) more important than a single dose or the total dose during a certain period is probably the time interval between doses. Ideally patients should not be allowed more than two doses per week; and (iii) the most important point is to instruct patients properly, and this takes time.
Ergotism (75, 76)
The angiographic findings of ergotism after therapeutic use of ergotamine were first described in 1936 by Yater and Cahill (74). They reported ‘the main arteries of the leg to be smooth in outline and apparently normal down to the lower third of the leg when they faded into a point’ for both their patients (74).
In the Middle Ages two kinds of ergotism caused by ergot occurred: the French form with gangrene of the extremities (St Antony's Fire), due to vasospasms, and the German form with convulsions, due to a central nervous system (CNS) effect (57, 77). What has been reported after clinical use of ergotamine is mainly the French form of ergotism (57, 66), but subclinical functional impairment of long ascending spinal tracts in man has been observed in 1988 in patients with chronic intake of ergotamine (75), and in one abstract on overdosage of ergotamine four cases of convulsions have been reported (57, 76). In addition, central inhibition of pupillary sympathetic activity has been described in ergotamine over-users in 1992 (78).
Taking the widespread use of ergotamine for the treatment of migraine attacks into account, overt ergotism with risk of gangrene is now a relatively rare disease. From 1900 to 1978, 139 cases were reported in the literature (66). Overt ergotism still occurs, but can be treated effectively with nitroglycerin infusion in most cases (79, 80). Based on the relative rarity of overt ergotism, ergotamine has generally been regarded as a safe drug when used properly (81). Methysergide can induce fibrotic disorders with long-term use, including retroperitoneal and heart valves fibrosis (82), and fibrotic heart valves have also been described after chronic ergotamine therapy (83). With the current HIV epidemic there is a risk for ergotism if ergotamine is combined with antiviral antiprotease inhibitors (84–89).
Recent advances in the pharmacology of ergotamine and DHE
It was shown in 1977 that a daily intake of ergotamine suppositories resulted in a small but significant decrease in systolic blood pressure in the legs, as measured with strain gauge plethysmography (90). There was thus a chronic vasoconstriction, a ‘subclinical ergotism’, with daily intake of ergotamine (90). In 1980 it was shown that a single dose of ergotamine caused long-lasting constriction of the arteries of the extremities (34). Following a single therapeutic rectal dose of ergotamine, this vasoconstrictory effect was still present after 24 h, but not after 48 h (34). Former ergotamine over-users reacted in the same way to i.v. ergotamine as did non-overusing migraine patients (91). It was later shown that the vasoconstrictor effect, the increase in systemic blood pressure, mainly due to the effect on kidney and liver blood flow, of i.v. ergotamine in man is short-lived, and gone within 3 h (34, 92). Thus, the effect of a single dose of ergotamine on the resistance vessels, as in dogs (67), is short-lasting, whereas the effect on human arteries is long-lasting (34).
Bioavailability and working mechanism of ergotamine
Using 3H-ergotamine, it was shown in 1977 that its oral absorption amounts to as much as 66% (93). However, using specific high-pressure liquid chromatography (94), it was shown in 1983 that the oral bioavailability of ergotamine is < 1% (95). A study with mass spectrometry indicated an even lower oral bioavailability, notably a relative bioavailability of 5% relative to rectal ergotamine (96). Thus, even if a substantial amount of ergotamine is orally absorbed, the drug undergoes very extensive ‘first-pass’ metabolism during passage through the liver. Even with low bioavailability, oral and rectal ergotamine are pharmacologically active (51). This might possibly be due to the formation of active metabolite(s), similar to DHE: DHE is metabolized to an active metabolite 8'hydroxy-dihydroergotamine, the blood concentration of which is about five to seven times higher than that of the parent drug itself (97). The bioavailability of rectal ergotamine is better than oral ergotamine. It is 1–3% (51), and rectal 2 mg ergotamine plus 100 mg caffeine was superior to rectal 25 mg sumatriptan (Fig. 3) (54).
Extensive studies in animals by Saxena in 1972–1974 showed that the vasoconstrictor effect of ergotamine is particularly marked within the carotid vascular bed (9, 10) and, there too, the selectivity extends to the arteriovenous anastomotic part; tissue blood flow, including that to the brain, is little affected [see, for example (98, 99)]. Similarly, it has been shown that the maximum i.v. dose of 0.5 mg ergotamine was without any effect on acetazolamide-stimulated cerebral blood flow in man (43). The selective effect on the carotid vascular bed of ergotamine was later follow by the demonstration of a similar effect of methysergide by an unknown receptor (100, 101). This result led to the development of sumatriptan in the late 1980s (102, 103).
The mechanism of the effect of the ergot alkaloids in migraine remains to be fully elucidated. Their vasoconstrictor effect is probably involved (51), but other effects may contribute. In rats, i.v. DHE or ergotamine tartrate, in doses similar to those used to treat migraine and cluster headache, prevented the stimulation-induced leakage of plasma proteins within the dura mater (11). Thus, an effect of these ergot alkaloids on neurogenic inflammation was suggested by Markowitz et al. in 1988 (11). In 1996 it was shown that DHE inhibited the trigeminovascular pathway in the CNS (12), indicating a possible effect on central trigeminal pain processing.
The problem of recurrence (after an initial response the migraine pain recurs) after subcutaneous sumatriptan treatment was first observed by Iversen in Denmark in 1989 (104); and in RCTs it has been found that oral and rectal ergotamine (51) and subcutaneous dihydroergotamine (105) caused less, in most cases 50% less, recurrence than the triptans (51, 52). This is most likely due to tight binding of ergot alkaloids to the vascular receptor (106).
Conclusion
Both ergotamine (introduced in 1925) and DHE (introduced in 1943) were for many years the only migraine-specific drugs. The observation by Graham and Wolff in 1938 of constriction of the dilated temporal artery in parallel with a decrease in headache after ergotamine (Fig. 2) resulted in the vascular theory of migraine by Wolff (3), which was the dominating pathophysiological theory for half a century.
Despite extremely low oral bioavailability of ergotamine, there is some evidence for its effect on migraine headache (54). Also for intranasal DHE there is evidence for efficacy (49). Overuse of ergotamine has been a problem (77). Ergotamine was previously considered a safe drug because of a seemingly short-lasting vasoconstrictory effect (67). The duration of the arterial effect in man of a single dose of ergotamine is, however, at least 24 h (34), and daily intake of the drug should be avoided. Overt ergotism is rare (66) and should be treated with i.v. nitroglycerin infusion (79).
It has recently become evident that new antimigraine drugs, the triptans, also can be overused with resulting chronic daily headache (65, 107). Also, overuse of simple analgesics or combination analgesics is well recognized in migraine patients (65). Overall, the current prevalence of drug-overuse headache has been estimated to be 1% of the general population (65). So, despite the large decrease in the use of ergotamine, we still have a big clinical problem.
One of the lessons from this history of ergotamine should be that prevention of medication overuse headache is up to the treating physician: ‘Complications can be avoided if enough time is taken for proper instruction of the patient, so that he can distinguish between vasodilating and nonvasodilating headaches’ (69). This statement by Peters and Horton is as clinically relevant today as in 1951.
