Abstract
Wind-up is a progressive, frequency-dependent increase in the excitability of trigeminal and spinal dorsal horn wide dynamic range (WDR) nociceptive neurons evoked by repetitive stimulation of primary afferent nociceptive C-fibres. The correlate of wind-up in humans is temporal summation, which is an increase in pain perception to repetitive constant nociceptive stimulation. Although wind-up is widely used as a tool for studying the processing of nociceptive information, including central sensitization, its actual role is still unknown. Here, we recorded from trigeminal WDR neurons using in vivo electrophysiological techniques in rats and assessed the wind-up phenomenon in response to stimuli of different intensities and frequencies. First, we found that the amplitude of C-evoked responses of WDR neurons to repetitive stimulation increased progressively to reach a peak, then consistently showed a stable or slightly decreasing plateau phase. Only the first phase of this time course fitted in with the wind-up description. Therefore, to assess wind-up, we measured a limited number of initial responses. Second, we showed that wind-up, i.e. the slope of the frequency-dependent increase in the response to C-fibre stimulation, was linearly correlated to the stimulus intensity. Intensities of brief C-fibre inputs were thus coded into frequencies of action potentials by second-order neurons through frequency-dependent potentiation of the evoked responses. Third, wind-up also occurred at stimulation intensities below the threshold for C-evoked responses in WDR neurons, suggesting that wind-up can amplify subthreshold C-fibre inputs to WDR neurons. This might account for the observation that sparse, subliminal, neuronal activity in nociceptors can become painful via central integration of neural responses. Altogether, the present results show that wind-up can provide trigeminal WDR neurons with the capability to encode the intensity of short-duration orofacial nociceptive stimuli and to detect subthreshold nociceptive input. Thus, not only may wind-up play a physiological role in trigeminal sensory processing, but its enhancement may also underlie the pathophysiology of chronic orofacial pain conditions.
Introduction
Although pain is clearly a subjective experience that is generated within the brain and integrally associated with emotional, cognitive and learned behaviour, there is one site apart from the brain that is actively involved in the early stages of producing trigeminal pain: the central neurons in the trigeminal brainstem sensory system. These neurons analyse and decipher the significance of peripheral input before transmitting the flow of nociceptive information to higher centres of the brain that, in return, regulate this flow by descending pathways through the brainstem (1). Among these neurons, the population of wide dynamic range (WDR) nociceptive neurons plays a critical role in the sensory discriminative analysis of trigeminal nociceptive stimuli. Indeed, they respond to both innocuous and nociceptive stimuli in a graded manner, and pathways arising from these neurons provide direct input to brain structures involved in perception (2, 3). In the rat, trigeminal WDR neurons are preferentially located within the deep laminae of the medullary dorsal horn (3) and within the subnucleus oralis of the spinal trigeminal nucleus (Sp5O) (4, 5).
Trigeminal as well as spinal WDR neurons display a striking form of activity-dependent plasticity that is called wind-up (5, 6). Wind-up manifests as a progressive increase in the response of WDR neurons to repetitive stimulation of primary afferent nociceptive C-fibres leading to neuronal hyperexcitability (7, for review see [8]). The correlate of wind-up in humans is thought to be perceptual wind-up, which is also commonly referred to as temporal summation (9). Perceptual wind-up is described as an increase in pain perception to a train of stimuli of the same intensity when delivered at ≥ 0.3 Hz. It develops in parallel with the temporal summation of nociceptive reflexes such as the RIII nociceptive flexion reflex (10) or the nociceptive blink reflex response (11). Temporal summation of electrical (12, 13), mechanical (14) and thermal (15) painful stimuli has been demonstrated. It has been used as a proxy for central sensitization to explore pathophysiological mechanisms in chronic pain conditions such as fibromyalgia syndrome (16), temporomandibular joint disorders (17) and headaches (18). The enhanced temporal summation demonstrated by these patients has led authors to conclude that central sensitization contributes to pain hypersensitivity. Thus, wind-up has been suggested to be responsible for the induction and maintenance of central sensitization (8, 9, 19).
However, it has become clear that wind-up is not equivalent to central sensitization, nor is it sufficient for the induction of all of the characteristics of central sensitization (8, 9, 19). Indeed, central sensitization may be evoked in the absence of wind-up, e.g. in visceral pathways. On one hand, spinal neurons with visceral input show the characteristic signs of central sensitization after noxious visceral stimuli, i.e. increased receptive field size, decreased thresholds and increased background activity (8), but, on the other, they do not show wind-up to electrical stimulation of visceral afferent C-fibres (20, 21). Similarly, Bolton et al. (6) have recently shown that wind-up does not occur following stimulation of dural afferents compared with facial skin stimulation. Furthermore, whilst wind-up-evoking stimuli increase the excitability of spinal neurons, not all the characteristics of secondary hyperalgesia are evoked by wind-up. Indeed, wind-up increases responses to C-fibre but not to A-fibre stimulation (22). Another major difference is that wind-up does not persist for more than a few minutes after the end of the conditioning stimulation, whereas central sensitization is very long lasting (8). Finally, numerous clinical studies have shown that temporal summation of pain is unchanged after induction of central sensitization (23, 24). Thus, although the two phenomena share similarities, they are different (8, 19), and the physiological role of wind-up is still unknown. Here, we investigated the role of the frequency and intensity of stimulation on wind-up of trigeminal WDR neurons. We infer from our results a possible role of wind-up in trigeminal sensory processing.
Methods
Adult male Sprague Dawley rats (280–300 g) were obtained from Charles River (L'Arbresle, France) and maintained in a controlled environment (lights on 07.00–21.00 h, 22°C) with food and water ad libitum. All efforts were made to minimize the number of animals used. The experiments followed the ethical guidelines of the International Association for the Study of Pain and the European Community Council directive of 24 November 1986 (86/609/EEC).
As previously described (5), animals were anaesthetized with 2% halothane in a nitrous oxide/oxygen mixture (2:1). After intraperitoneal injection of 100 μg atropine sulphate, a tracheal cannula was inserted and the carotid artery and the jugular vein were cannulated. Animals were then paralysed by an intravenous perfusion of pancuronium bromide (0.5 mg/h) and artificially ventilated with a volume-controlled pump (54–55 strokes/min). The levels of halothane, O2, N2O and the end-tidal CO2 (3.5–4.5%) were monitored by an anaesthetic gas monitor (Artema MM 200, Sundbyberg, Sweden) during the entire experimental period. The arterial catheter was attached to a calibrated pressure transducer (UFI, Morro Bay, CA, USA) connected to an amplifier (Stoelting, Wood Dale, IL, USA) for continuous monitoring of blood pressure. The analogue output from the blood pressure amplifier was connected to a computer data sampling system (Cambridge Electronics Design 1401 computer interface; Cambridge, UK). Heart rate was monitored continuously and cutaneous vascularization was periodically checked by observing the colour of the paw extremities and the rapidity by which they regained normal colour after pressure application. Core temperature was maintained at 37 ± 0.5°C with a homeothermic blanket system. Variations in heart rate and blood pressure were used as indices of nociception.
The rats were placed in a stereotaxic frame and a craniotomy performed to give access to the Sp5O. After surgery, the levels of halothane (0.7–0.8%), O2, N2O and end-tidal CO2 (3.5–4.5%) were monitored together with heart rate, blood pressure and core temperature, under the control of alarms.
Unitary extracellular recordings were made with glass micropipettes (8–10 MΩ) filled with a mixture of 5% NaCl and pontamine sky blue. Single unit activities were amplified and displayed on an oscilloscope and also led into a window discriminator connected to a CED 1401plus interface and a computer (CED and Spike 2.5 software; Cambridge Electronics Design) to allow sampling and analysis of neuronal activity. Neurons were classified as WDR on the basis of their responses to both mechanical and percutaneous electrical stimulation applied to their receptive field. Innocuous mechanical stimuli to the skin, mucosa and teeth included air puff, brushing with a soft brush, gentle stroking and light pressure with a blunt probe. None of these stimuli induced changes in heart rate or blood pressure. Nociceptive mechanical stimuli consisted of heavy pressure, pinprick and pinching with fine forceps (tip area 1 mm2), which evoked a painful sensation when applied to the experimenters' skin. They all evoked transient and small increases in heart rate and blood pressure. Once a neuron had been identified, the extent of its receptive field was determined and mapped, and its location was designated in terms of its involvement in intraoral, perioral or more peripheral regions of the face.
Electrical square-wave stimuli (0.66 Hz, 2 ms duration) were applied through a pair of stainless steel stimulating electrodes inserted subcutaneously into the centre of the previously delineated receptive field. Thresholds for obtaining A- and C-fibre responses were determined; increasing the current to a suprathreshold value induced reproducible responses. Poststimulus histograms were analysed to distinguish responses caused by A- and C-fibre inputs, according to their latencies. The latency of the responses was used to compute the conduction velocity of afferent inputs after making allowance for the conduction distance (∼50 mm) and 1 ms for the central synaptic delay, the delay in activation of the peripheral axons, and the narrowing of afferents in the trigeminal spinal tract. Neurons responding with a discharge burst at a latency > 30 ms were considered to be excited by C-fibres (4–6). The C-fibre-evoked responses were taken as all spikes recorded between 30 and 300 ms after the stimulus.
The experimental procedure for wind-up consisted of sequences of 15 or 30 electrical shocks applied repeatedly at 0.1 or 0.66 Hz to the excitatory receptive field of the recorded neuron at 0.5, 1, 1.5 or 3 times C-fibre activation threshold. Sequences were delivered every 5 min. One single cell was tested in each animal. At the end of the experiment, injection of pontamine sky blue allowed further histological delineation of the recording and injection sites. The animal was then killed by injection of a lethal dose of pentobarbital, its brainstem removed and fixed by immersion in a 10% formaldehyde solution before further histological processing.
Results are expressed as mean ±
Results
General properties of Sp5O WDR neurons
A total of 40 WDR neurons were recorded within the Sp5O. They were located throughout the dorsoventral extent of the nucleus, between frontal planes P 1.1 and P 2.4 (25). Figure 1b illustrates a histologically confirmed recording locus in ventrolateral Sp5O. Most neurons were not spontaneously active. All had an ipsilateral receptive field within the intraoral or perioral region (Fig. 1a). They were sensitive to both innocuous and noxious mechanical stimuli and responded by increasing their firing rate as stimulus intensity increased into the noxious range (Fig. 1c).
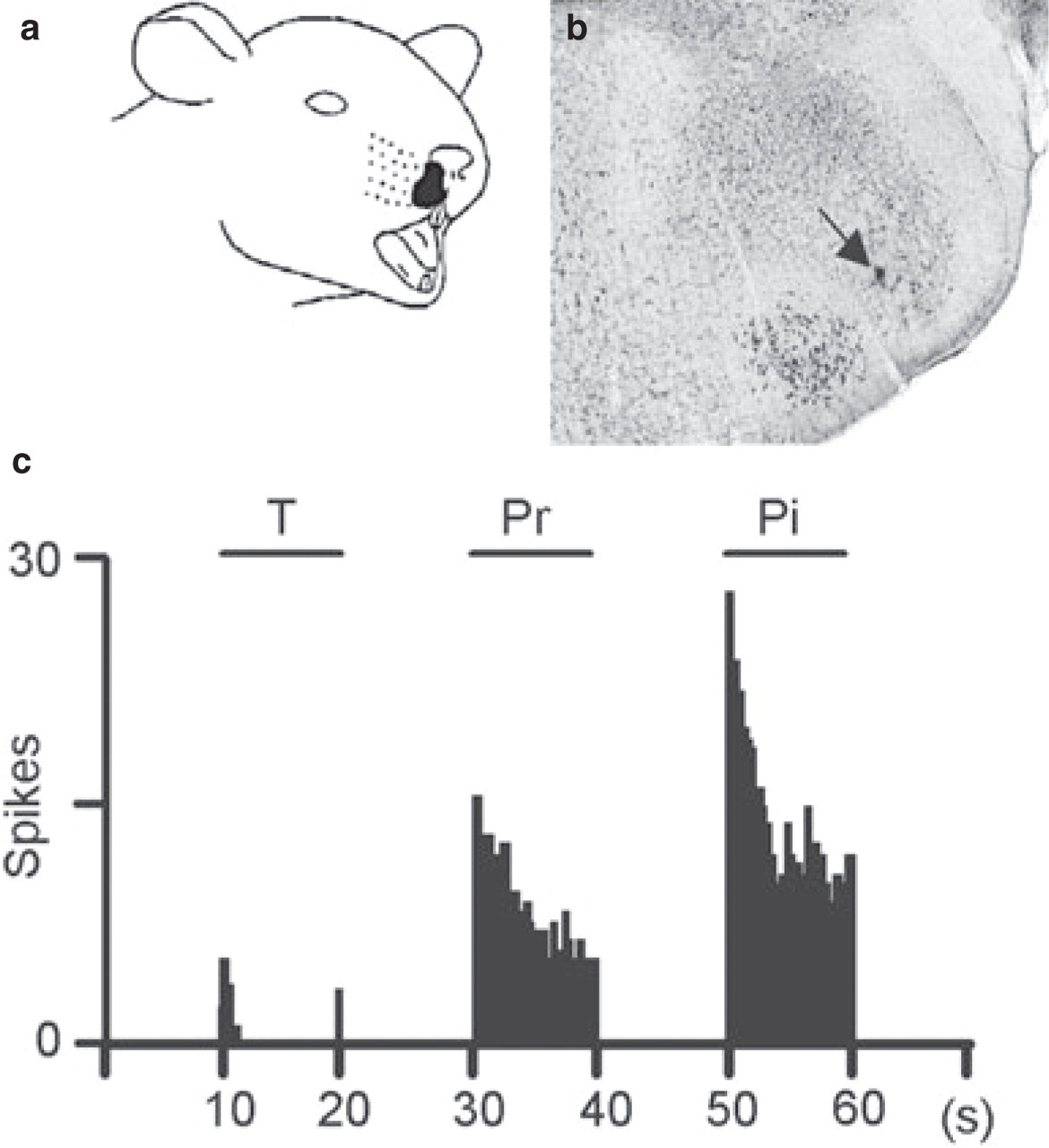
Responses properties of a spinal trigeminal nucleus (Sp5O) wide dynamic range (WDR) neuron to mechanical stimuli applied to its perinasal receptive field (a). (b) Microphotograph showing the histologically confirmed (arrow) recording locus of a Sp5O neuron. (c) Responses of a neuron to mechanical stimuli (T, touch; Pr, pressure; Pi, pinch).
When we applied 2-ms percutaneous electrical stimuli to the centre of the excitatory field of the neurons, responses attributable to peripheral activation of A- and C-fibres could be recorded (Fig. 2). The responses attributable to C-fibres were obtained at a mean threshold of 8.18 ± 0.65 mA and with a mean latency of 84.32 ± 2.34 ms, which corresponds to peripheral fibres with a conduction velocity of about 0.6 m/s.

Wind-up phenomenon. Consecutive single sweep recordings showing the evolution (from bottom to top) of the responses of a single spinal trigeminal nucleus (Sp5O) wide dynamic range (WDR) neuron following repetitive supramaximal percutaneous electrical stimulation (3 × threshold, 0.66 Hz) applied to its receptive field.
Wind-up of C-fibre-evoked responses
All recorded Sp5O WDR neurons exhibited wind-up following repetitive constant percutaneous electrical stimulation applied to their receptive field at C-fibre threshold and suprathreshold intensities (Fig. 2). During repetitive stimulation, C-fibre-evoked responses of trigeminal WDR neurons progressively increased, thereafter reached a maximum or peak and then slightly decreased (Fig. 3a). Such a secondary decline of C-fibre-evoked responses was consistently observed. It could be best fitted using a simple linear regression (Fig. 3a). It is important to note that, nevertheless, final responses (from peak to the 30th one) remained significantly higher than initial ones. As previously reported (for review see [8]), there was no wind-up of A-fibre-evoked responses following repetitive stimulation of primary afferent nociceptive A-fibres (see Fig. 2). Finally, in contrast to our previous findings (4, 5), we did not find here any WDR Sp5O neurons not displaying wind-up.

Wind-up is linearly correlated with stimulus intensity. (a) Cumulative results illustrating the dynamic properties of the successive responses to the 30 first stimulations at C-fibre threshold (T) and suprathreshold (3T) intensities at 0.66 Hz. Superimposed on the second half of the plots are the linear fits of the data (3T, n = 10, y =−0.131x + 16.483; 1T, n = 10, y =−0.260x + 22.624). (b) Cumulative results illustrating the dynamic properties of the successive responses to the 15 first stimulations at different intensities (T, C-fibre threshold intensity) at 0.66 Hz. Superimposed on the plots are the polynomial fits of the data (3T, n = 10, y =−0.183x2 + 4.113x + 1.18; 1.5T, n = 10, y =−0.110x2 + 3.088x − 1.159; 1T, n = 10, y =−0.023x2 + 1.573x − 1.725; 0.5T, n = 10, y = 0.001x2 + 0.419x + 0.142). (c) Enlargement of (B) illustrating the dynamic properties of the successive responses to the 15 first stimulations at 0.5T and 0.66 Hz. (d) Values of the initial slope required for fitting the values of spikes plotted in (A) as a function of stimulation intensity. (e) Cumulative results illustrating the dynamic properties of the successive responses to the 15 first stimulations at different intensities at 0.10 Hz. Superimposed on the plots are the polynomial fits of the data (3T, n = 10, y =−0.017x2 + 0.571x + 5.591; 1.5T, n = 10, y =−0.040x2 + 0.883x + 2.791; 1T, n = 10, y =−0.010x2 + 0.283x + 0.993). (f) Enlargement of (e) illustrating the dynamic properties of the successive responses to the 15 first stimulations at 3T and 0.1 Hz.
Wind-up can be induced by stimulus intensities below the threshold for evoking C-fibre responses in WDR neurons
When the stimulation intensity was applied at half the intensity required to evoke C-fibre responses, WDR neurons initially did not produce C-fibre-dependent responses (Fig. 3b,c). However, following repetitive stimulation, spikes at C-fibre latencies were triggered, the number of which progressively increased. The number of spikes increased significantly (P < 0.003) from 0.3 ± 0.2 (first stimulation) to 6.3 ± 1.3 (15th stimulation). This suggests that wind-up could amplify subthreshold input to WDR neurons.
Wind-up increases with stimulus intensity
To determine the role of wind-up in encoding the sensory attributes of orofacial nociceptive information, we examined whether wind-up depends on the intensity of peripheral stimulation. At constant frequency (0.66 Hz), the peak of C-fibre-evoked response strongly increased with stimulus intensity. At the same time, its delay—the number of stimuli required to reach this maximum—decreased. Peaks were observed at around the 15th, 14th, 12th and 10th stimulation for intensities of 0.5, 1, 1.5 or 3 times the C-fibre activation threshold, respectively.
The dynamic properties of the successive responses were best fitted using a second-order polynomial regression (R 2 ranking from 0.95 to 0.99, P = 0.01–0.0001), indicating that in all cases there was a strong correlation between the amplitude of the response and the number of repetitive stimuli. When we used first-order linear curve-fits from the first to the peak response, such significant correlation coefficients were not obtained (R 2 ranking from 0.70 to 0.98, P = 0.05–0.001). Since the initial slope of the polynomial fit curves corresponded to the ability of the neuron to amplify its response, it was used to quantify wind-up. As shown in Fig. 3d, the initial slope of the response curves, and therefore wind-up, was linearly correlated with the intensity of the stimulation (R 2 = 0.97, P = 0.001). Interestingly, this allowed the neurons to increase the difference between responses to inputs of distinct intensities through repetition. Indeed, although the first responses to stimulation of distinct intensities were close to each other, the difference between them increased with stimulation number, even more with higher intensities of stimulation (Fig. 3b).
Such amplification was clearly related to the phenomenon of wind-up. As shown in Fig. 3e, at very low frequency (0.1 Hz), repetitive stimulation did not produce wind-up and did not significantly amplify the WDR neuron responses. For example, during 0.10 Hz stimulation at three times the intensity required to evoke C-fibre responses, the number of spikes increased—although not significantly (P < 0.06)—from 5.3 ± 1.2 (first stimulation) to 11.1 ± 3.4 (15th stimulation) (Fig. 3e,f). In such cases, the slopes of the fit curves no longer varied as a function of stimulus intensity (R 2 = 0.045, P > 0.05).
Discussion
Wind-up is a progressive, frequency-dependent increase in C-fibre evoked responses of trigeminal and spinal dorsal WDR neurons to repetitive nociceptive stimuli of constant intensity. We show here that the time course of wind-up is actually a multiphasic phenomenon with an initial increase leading to a peak and followed by a slightly decaying plateau phase. We also report that wind-up increases with stimulus intensity and that it can be induced at stimulation intensities below the threshold of C-evoked responses of WDR neurons.
Time courses of wind-up similar to that reported here have been observed while recording WDR neurons in the medullary (6) and the spinal dorsal horn (22, 26, 27) as well as in psychophysical studies (15). Although a gradual decline in the number of spikes was observed following the peak response, neurons still remained hyperexcitable. A similar decrease following long-lasting facilitation of spinal nociceptive reflexes produced by temporal summation of nociceptive inputs has been described, which results from supraspinally mediated inhibitions (28). In the case of WDR neuron wind-up, intrinsic cellular mechanisms may also be at work, as suggested by the negative value of the second-order parameter of the polynomial fit of in vitro wind-up curves provided by Morisset and Nagy (29). These results suggest that several mechanisms are likely to be overlapping during wind-up. Therefore, the frequency-dependent increase in C-fibre-evoked responses should be assessed by measuring only a limited number of initial responses, the exact number depending on the stimulus frequency and intensity.
Previous studies have clearly established that wind-up is a frequency-dependent phenomenon obtained between 0.3 and 3 Hz (for review see [8]). Here, we provide the first report that wind-up is also intensity-dependent. Indeed, the value of the slope of wind-up is linearly related to stimulus intensity. Thus, the stronger the stimulus, the steeper is the increase in firing to the next stimulus. As such, the slope code of wind-up can contribute to the perception of stimulus strength. As a consequence, wind-up can provide help in distinguishing between isolated intense brief events that are unlikely to threaten the integrity of tissue and repeated events that have a potential noxious meaning (30). Consistently, we have shown that the relationship between the intensity of stimulation and the slope of temporal summation disappears when the stimulation frequency is lowered and wind-up is not evoked. As previously reported for neurons recorded from the medullary (31) and spinal (27, 32) dorsal horn, Sp5O neuron responses to initial stimulation of different intensities are different, but close. Similarly, psychophysical studies have shown that following a single, brief noxious stimulus, normal healthy subjects cannot perform a reliable pain estimate (12, 15, 33). In our study, after repetition of stimulations, differences between responses evoked by different intensities increased. Clearly, in psychophysical experiments, ratings of pain intensity increase with repetition of brief electrical, mechanical or heat stimulation, and then subjects become able to discriminate between different intensities of stimulation (12, 15, 33, 34). Interestingly, the relationship between the encoding properties and wind-up phenomenon is also suggested by recent data that found that these characteristics disappear simultaneously after deletion of the dorsal horn NK1 receptors (35) and also in mice lacking the NK1 receptor (36, 37). Thus, an essential function of wind-up is to enhance the encoding properties of WDR neurons. Other nociceptive neurons rarely display wind-up (26, 38, for review see [3]). However, nociceptive cells located primarily in the superficial dorsal horn (lamina I) and that receive input only from nociceptors could also contribute the qualitative estimation of pain intensity (39). For instance, polymodal nociceptive lamina I neurons respond to repeated heat stimuli with an augmenting discharge that could account for the increasing second pain report in human psychophysical studies (27).
In our experiments, wind-up could be obtained by stimulation intensities below the threshold for C-fibre-evoked responses of WDR neurons. Such a finding suggests that wind-up may also amplify subthreshold excitatory synaptic input to WDR neurons. At 0.5 times the threshold, only a few C-fibres are recruited, and this is not sufficient to produce action potentials in WDR neurons. Indeed, intracellular recordings have revealed that a population of spinal neurons displays a ‘subthreshold wind-up’, which takes the form of progressive cumulative depolarization with no significant increase in spike count (see References in [8]). Interestingly, clinical studies have shown that stimuli that are normally felt only as warmth can result in very high pain when delivered repeatedly, hence producing temporal summation (15, 40). Similarly, repetitive low-intensity, non-painful electrical stimuli can result in severe pain (13, 41). These results are consistent with the idea that wind-up can amplify subthreshold inputs to WDR neurons. Even sparse, subliminal, neuronal activity in nociceptors can become painful via central integration of neural responses (13). This fits with our interpretation that wind-up helps in distinguishing between non-significant isolated brief events and repeated events that acquire a potential noxious meaning.
If wind-up helps to discriminate between short-duration nociceptive stimuli of different intensities, then any changes in this phenomenon should be associated with variations in sensory discrimination capabilities. Accordingly, clinical studies have shown that temporal summation of heat pain is greater (42, 43) and sensory discrimination higher in women compared with men (44), and greater temporal summation of pain is correlated with reports of more intense clinical pain within a group of patients with fibromyalgia (45). Moreover, if normally non-significant stimuli can evoke aches and pain because of temporal summation, then a more pronounced summation of pain should contribute to pain pathophysiology. Accordingly, patients with either temporomandibular disorders (17, 46) or chronic headache (47, 48) show enhanced temporal summation of pain to repetitive noxious (thermal, mechanical or electrical) stimuli compared with control subjects. This is also the case in migraine patients, suggesting that, although wind-up is not observed in response to stimulation of the dura mater, central hyperexcitability caused by noxious dural stimulation can lead to an increased wind-up of responses evoked by cutaneous facial stimulation (18).
In summary, the present study has shown that wind-up enhances the encoding of short-duration orofacial nociceptive stimuli intensity and allows the detection of subthreshold nociceptive input. As a consequence, wind-up endows trigeminal WDR neurons with a greater ability to assess the intensity of nociceptive information. Wind-up thus may play a physiological role in trigeminal sensory processing, and enhanced wind-up may underlie the pathophysiology of several common chronic headache and facial pain conditions.
Footnotes
Acknowledgements
We thank Pr. A. Artola for helpful comments on this manuscript and A. M. Gaydier for secretarial assistance. This work was supported by funding from Inserm, MENESR, Direction Générale des Armées (01.34.012.00470.75.01), Fondation Benoit and Université Clermont1 (France).
