Abstract
Background
In about half of migraine patients, anti-calcitonin gene-related peptide monoclonal antibodies reduce monthly migraine days by >50%. In these patients, this class of drugs may change cortical functions by decreasing nociceptive afferent barrage. This prospective study investigated functional connectivity changes in treatment responders after three-month treatment with galcanezumab.
Methods
Resting-state functional magnetic resonance imaging data were acquired for patients with high-frequency episodic or chronic migraine (N = 36) before and after treatment. Of these, 19 patients were classified as treatment responders (≥50% reduction in monthly migraine days) and 17 were considered non-responders (<50% reduction). Functional connectivity across cortical regions was assessed using a region-of-interest (ROI)-to-ROI analysis approach.
Results
At baseline, there were no significant differences between treatment responders and treatment non-responders. In the treatment responder group, reduced functional connectivity was observed after treatment between regions of the primary somatosensory and motor cortices, insula, and several occipital and temporo-occipital areas (within the visual network). In contrast, no such changes were seen in the non-responder group.
Conclusion
These findings suggest that even a relatively short period of reduced nociceptive signals may be sufficient to initiate a cortical recovery process in which its resting hyperexcitable mode shifts to a less excitable state.
This is a visual representation of the abstract.
Introduction
Migraine is a complex neurological disorder characterized by recurrent disabling attacks of headache, frequently accompanied by abnormal sensitivities to light, sound, smell, and touch (1). Mounting evidence suggests that these abnormal sensitivities are caused by amplification of sensory signals along the different routes they use to reach cortical areas involved in generating perception (2,–6).
Cortical hyperexcitability is likely to play a role in the initiation and chronification of certain migraine attacks (6,7). Neuroimaging studies have reported enhanced responsivity of the somatosensory and visual cortices in migraine patients, and a possible correlation between the degree of excitability and the frequency of attacks (8,–15). Enhanced or sensitized interactions between these brain regions could be interpreted as either increased cortical responsiveness or compensatory mechanisms aimed at stabilizing the cortical excitability level (16). These changes are constant in high-frequency and chronic migraine, potentially driving maladaptive plasticity processes in the brain (3,4,17).
It is widely accepted that recurring migraine attacks can alter brain functions (5,18,20–). Peripherally acting drugs (e.g., galcanezumab) used for migraine treatment may restore/normalize altered brain functions in patients by decreasing nociceptive afferent barrage (21). In about half the patients who suffer from migraine, galcanezumab and other anti-calcitonin gene-related peptide monoclonal antibodies (anti-CGRP mAbs) are capable of reducing monthly migraine days by >50% (22,23). As such, these patients are considered treatment responders (24,25). In line with this, we recently reported that in responders, a reduction of 50% in monthly migraine days over a three-month treatment with galcanezumab reduced cortical thickness in regions associated with migraine attacks, including the somatosensory cortex (26). Based on these latter findings, we proposed that secondary to the reduction in exposure to pain, cortical neurons became less excitable and consequently their soma volume, degree of myelination and axonal sprouting – all known contributors to alterations in cortical thickness – decreased (27,28). Previous functional MRI studies have also demonstrated that a short period of treatment with erenumab (a mAb that targets the CGRP receptor instead of the peptide) or galcanezumab is sufficient to drive brain changes in response to pain, and in functional connectivity in episodic and chronic migraine patients (29,–32).
In the present longitudinal study, functional connectivity analysis was performed in individuals with either high-frequency episodic or chronic migraine who were treated for three months with galcanezumab. Our primary aim was to identify functional organization changes in patients whose monthly migraine days decreased by 50–90%. We hypothesized that after three months of treatment with galcanezumab, changes in functional connectivity, mainly between the primary somatosensory and visual cortices, would be more prominent among treatment responders than treatment non-responders.
Methods
The present study was part of a prospective observational open-label study investigating the effects of galcanezumab on the incidence of headache following the occurrence of premonitory symptoms and triggers, and its impact on brain structure and function (23,26). The study was approved by the local institutional review board of the Beth Israel Deaconess Medical Center (BIDMC), Boston, MA (Committee on Clinical Investigations; CCI Protocol # 2019P001081) and was conducted according to Good Clinical Practice and the Declaration of Helsinki. Patients provided written informed consent before inclusion in the study.
Participants
Sixty-four participants, who were initially contacted and screened, met inclusion criteria, and agreed to participate in the MRI study. They were enrolled at the BIDMC Comprehensive Headache Center in Boston, MA, between September 2020 and March 2022, and all fulfilled the criteria for (high-frequency) episodic or chronic migraine according to the third edition of the International Classification of Headache Disorders (ICHD-3) (1). High-frequency episodic migraine was characterized by 8–14 migraine days per month, whereas chronic migraine was defined by at least 15 migraine days per month (1). Confirmation of migraine diagnosis was conducted by a neurologist and a headache specialist through a clinical interview and completion of a four-week electronic diary (e-diary). Thirty-six of the 64 participants (aged between 21 and 63 years) completed all aspects of the study and were thus included in the data analysis.
The inclusion and exclusion criteria were similar to those outlined in our prior studies (23,26). Inclusion criteria comprised individuals aged 18 to 65 years, with previous diagnosis of migraine (with or without aura) based on the ICHD-3 criteria, and onset of migraine at age 50 years or younger. Exclusion criteria included current use of one or more migraine preventive treatments, positive pregnancy test, breastfeeding, significant cognitive impairment, psychiatric disorders and/or behavioral problems that could interfere with the study, other significant pain problems (e.g., cancer pain, fibromyalgia, other head or facial pain disorder), severe cardiac disease (e.g., symptomatic coronary artery disease, prior myocardial infarction, congestive heart failure), cerebrovascular disease (e.g., prior stroke or transient ischemic attack, symptomatic carotid artery disease, prior carotid endarterectomy or other vascular neck surgery), report of an abnormal electrocardiogram within the last year (e.g., second or third-degree heart block, prolonged QT interval, atrial fibrillation, atrial flutter, history of ventricular tachycardia or ventricular fibrillation, clinically significant premature ventricular contraction), uncontrolled high blood pressure (systolic >160 mm Hg, diastolic >100 mm Hg), a known history or suspicion of secondary headache, a known history or suspicion of substance abuse or addiction (within the last five years), current use of marijuana or has used marijuana (including medical marijuana) or cannabidiol oil within the last year, claustrophobia, or MRI incompatible implants. Additional exclusion criteria included current use of opioids for headaches or other body pain, current use of simple analgesics or non-steroidal anti-inflammatory drugs (NSAIDs) for 15 days or more per month, or use of triptans, ergots, or combined analgesics for 10 days or more per month, receiving nerve blocks (occipital or other) in the head or neck within the last three months, and receiving onabotulinumtoxinA or anti-CGRP or anti-CGRP mAb treatment within the last six months. During the study, participants were asked not to start or modify any medication, including changing type, dosage, or frequency of any medication (e.g., antidepressants, anticonvulsants, beta-adrenergic blockers).
Study design and data collection
Study participants were scheduled for three visits:
Visit 1: During the initial visit, participants received detailed information about the study, provided consent, gave full medical and headache history, and received training on utilizing a daily electronic headache diary. The headache history was documented through a self-completed questionnaire, covering demographics, body mass index (BMI), disease duration, family history, headache location, attack frequency, associated symptoms (those appearing during but not before headache onset), premonitory symptoms (those appearing before headache onset, including aura), triggers, a list of headache medications, and any comorbid conditions. The e-diary was conducted through a Research Electronic Data Capture (REDCap) survey, accessed by participants through an email link on their personal computer or electronic device. It comprised a questionnaire designed to assess the daily/monthly occurrence of headache and migraine, laterality (unilateral or bilateral), pain intensity (rated on a 0–10 visual analogue scale), and the daily occurrence of symptoms required to establish migraine criteria (such as nausea, vomiting, throbbing, photophobia, phonophobia, and osmophobia). The impact of migraine was evaluated with the Migraine Disability Assessment Scale (MIDAS) (33). MIDAS score was determined by adding up the total days of missed work/school and days of reduced productivity due to headache from work/school, household tasks, and nonwork activities (including family, social, leisure) over a period of three months.
Visit 2: During the second visit, which took place after 30 days of completing the e-diary without initiating any prophylactic treatment, participants underwent an MRI scan of brain at the hospital's Imaging Research Center. After the MRI scanning, participants returned to the Headache Center, where they received the first galcanezumab treatment, an initial loading dose of 240 mg, administered by the study physician (S.A.) who also provided training on how to self-administer galcanezumab at home.
Visit 3: During the third visit, scheduled three months after the second visit, participants who self-administered the injections at home (or came back to the headache clinic to receive the 2nd and 3rd injections, 120 mg each) at 30 and 60 days after the first injection, and completed the e-diary for the entire 120 days (one month before and three months after treatment initiation), returned to the hospital for their post-treatment MRI scanning. All post-treatment MRI scans were conducted within three days from day 90 of the treatment period (specifically between days 87 and 90 of the treatment period or between days 117 and 120 of the entire study period). Considering the occurrence of migraine among many of the participants, we allowed them to undergo MRI scanning (during visits 2 and 3) either when they were free of headaches or experiencing mild background headache that did not meet any of the migraine criteria, and the pain was so mild that no pain or headache medication had been taken in the 12 h leading up to the scanning.
Classification of responders and non-responders
Based on their response to treatment at the end of the study, participants were classified as treatment responders or treatment non-responders. Treatment responders were those whose monthly migraine days decreased by 50% or more over the three-month treatment period (calculated based on the information recorded in their daily e-diary during the 30 days before and 90 days after treatment initiation). Non-responders were those whose monthly migraine days decreased by less than 50% during the three-month treatment period. These definitions are in accordance with current guidelines and recommendations for clinical trials (24,25,34).
MRI data acquisition
MR images were obtained utilizing a 3T MR750 (software version DV26.0) and Signa Premier (DV29.1) scanners (GE Healthcare, Chicago, IL) equipped with a 32-channel NOVA proton head coil. T1-weighted structural and resting-state functional images were acquired at baseline, prior to the start of treatment, and then again, three months after treatment initiation for each participant. For each functional imaging run, a pair of spin-echo echo-planar images (SE-EPI) with opposing phase encoding polarity was acquired in order to compute B0 field maps for correcting susceptibility distortions in the fMRI images using the FSL topup tool. The T1-weighted structural imaging used an accelerated sagittal Inversion Recovery – Fast Spoiled Gradient (IR-SPGR) sequence with echo time (TE) = 3.06 ms, inversion time (TI) = 400 ms, repetition time (TR) = 7.39 ms, flip angle (FA) = 11°, in-plane resolution = 1 × 1 mm, number of sagittal slices = 196, slice thickness = 1 mm, no gap, matrix = 256 × 256, field of view (FOV) = 256 × 256 mm, voxel size = 1 × 1 × 1 mm3, pixel bandwidth = 244.14 Hz, acceleration factor = 2. Resting-state functional images were acquired using a multiband accelerated 2D Gradient Echo EPI sequence with the following parameters: TE = 30 ms, TR = 0.8 s, FA = 60°, slice thickness = 2.5 mm, no gap, matrix = 88 × 88, FOV = 220 × 220 mm, voxel size = 2.5 × 2.5 × 2.5 mm3, volumes = 750. The parameters for SE EPI images used for computing field maps were: TE = 60 ms, TR = 8 ms, flip angle = 90°, FOV = 220 × 220 mm, matrix = 128 × 128, slice thickness = 2.5 mm, 64 slices, voxel size = 1.7 × 1.7 × 2.5 mm3. Participants were instructed to rest without moving, keep their eyes open, and focus on a fixation cross visible via a mirror attached to the head coil.
MRI data processing
Resting-state fMRI image processing and data analysis were performed using the FMRI Expert Analysis Tool (FEAT), part of the Functional Magnetic Resonance Imaging of the Brain (FMRIB) Software Library (FSL, version 6.0.6.5) (http://fsl.fmrib.ox.ac.uk/fsl/fslwiki/, London, UK) and the Functional Connectivity (CONN) toolbox (version 21.a) (35), implemented in Statistical Parametric Mapping (SPM12) (http://www.fil.ion.ucl.ac.uk/spm/, Wellcome Department of Cognitive Neurology, University College London, London, UK). The first 10 volumes of each fMRI dataset were excluded from the analysis to allow the fMRI signal intensity to reach a steady state. Preprocessing steps included visual inspection; removal of non-brain structures (i.e., skull and surrounding tissues) using Brain Extraction Tool (BET); distortion-correction using fieldmaps (B0 unwarping); head motion correction using Motion Correction FMRIB's Linear Image Registration Tool (MCFLIRT); interleaved slice timing correction; spatial smoothing using a Gaussian kernel with a 5 mm full-width at half-maximum (FWHM), and grand-mean intensity normalization of the entire 4D dataset by a single multiplicative factor (36,–38). No participant's fMRI dataset was removed from the analysis due to head motion (exclusion criteria: motion parameters exceeding 3 mm of translation or 3° of rotation).
After preprocessing, the functional MRI images were first co-registered to structural images using boundary-based registration (BBR) in the FSL linear registration tool (FLIRT) (36,39). The structural images were then normalized to the Montreal Neurological Institute 152 (MNI-152) 2-mm standard template using 12-parameter affine transformation (36,39), followed by a nonlinear registration (FNIRT) (40), with a warp resolution of 10 mm.
Denoising of resting-state fMRI data
Given the sensitivity of resting-stat fMRI data to motion (41), further measurements and corrections were applied. Independent component analysis with automatic removal of motion artifacts (ICA-AROMA) (42) was used to identify and remove motion-related artifacts from the resting-state data. Subsequent signal denoising was performed using anatomical component-based noise correction method (aCompCor; implemented in CONN software) (43) to estimate physiological noise from white matter (WM) and cerebrospinal fluid (CSF) for each participant. Functional data were denoised in CONN to remove motion parameters (based on realignment of functional data during FSL preprocessing; 12 parameters) and physiological components (2 × 5 parameters). Linear detrending was performed and temporal high-pass filtering was applied to remove low-frequency drifts (<0.01 Hz). Denoised images were registered to 4 mm isotropic voxel size.
Statistical analysis
Demographic and clinical data analysis
Prior to the study, no statistical power calculation was carried out. The sample size was determined based on available data, and all analyses were performed in accordance with the a priori analysis plan. The effects of galcanezumab treatment on brain morphometry in cortical regions in both responders and non-responders were previously assessed in this patient group (26). The present study is the primary analysis of the resting-state functional connectivity MRI study data.
Statistical analyses of demographic and clinical data were performed with the Statistical Package for Social Sciences version 27.0 software (SPSS, Inc., Chicago, IL, USA). Summary statistics (including means, standard deviations [SDs], medians, and interquartile ranges [IQR] for continuous variables; frequencies and percentages for categorical variables) were computed to describe the sample characteristics. Normality assumptions were evaluated using histograms, Q-Q plots, and Shapiro-Wilk tests, while the homogeneity of variances was assessed using Levene's test. Mann-Whitney U tests were performed to examine the differences between the responder and non-responder groups in age, BMI, and other clinical characteristics. Fisher Exact Test was used to compare the categorical variables (sex, aura, handedness, and migraine classification) between groups. Differences were considered significant if p < 0.05.
Functional connectivity analysis
An atlas based region-of-interest (ROI)-to-ROI approach was applied to evaluate the level of cortical functional connectivity among 91 (cortical) ROIs from the default Harvard-Oxford Atlas implemented in CONN software. Cortical regions were selected for the connectivity analyses based on cortical hyperexcitability observed in migraine (6,–9) and our previous study demonstrating cortical thickness changes after the three-month treatment with galcanezumab (26). In order to perform analyses that encompass and correct for all connections between the cortical ROIs (4095 connections), the ‘Group analysis results: all ROIs’ option was selected. Second-level analyses were conducted to assess baseline differences (cross-sectional evaluation) and changes between condition contrasts (post- vs. pretreatment) (longitudinal evaluation) in the responder and non-responder groups. The functional network connectivity (FNC) method (44,45) implemented in CONN was used to identify clusters of significantly different connections. More specifically, ROIs were sorted automatically by applying a data-driven hierarchical clustering procedure (complete-linkage clustering), based on ROI-to-ROI anatomical proximity and functional similarity metrics (45,46). Once the sets/clusters of connections were defined, the entire set of connections between all pairs of ROIs was analyzed using a multivariate parametric general linear model analysis. ROI-to-ROI analyses were corrected for multiple comparisons using the default cluster threshold of Benjamini and Hochberg false discovery rate (FDR)-corrected p < 0.05 (47). Notably, all default criteria in CONN are defined to ensure proper analysis-wise error control, and the applied correction adjusts the p-value for multiple comparisons arising from using multiple ROIs (all ROIs were tested in the same group-analysis). All brain maps and graphs were visualized using the CONN graphics interface. Mean connectivity values (Fisher's Z-transformed) were computed from the obtained significant results.
Results
Participants’ characteristics
This study included 36 participants (29 females, age range: 21–63, mean [M] age = 34.7, standard deviation [SD] ± 11.3), from whom 19 participants (15 females, M age = 33.1, SD ± 9.7) were classified as responders and 17 (14 females, M age = 36.7, SD ± 12.9) as non-responders. There were no statistically significant differences between the two groups for age, sex, BMI, headache days per month, migraine days per month, years with migraine, headache intensity, impact of headaches (MIDAS score), and aura symptoms at baseline (Table 1). There was no missing data in the reported dataset. Treatment impacts on the clinical characteristics (including migraine days per month, associated symptoms, prodromes and triggers and their ability to predict/initiate a headache) in responders and non-responders were previously reported (23).
Baseline demographics, migraine classifications, and disease characteristics of all patients (non-responders and responders).

Note. Data are expressed as mean (standard deviation [SD]), median (interquartile range [IQR]) and number of participants (N; proportion [%]).
a Average headache intensity reported by patients.
BMI, body mass index; CM, chronic migraine; EM, (high-frequency) episodic migraine, MMD, monthly migraine days; MIDAS, Migraine Disability Assessment Scale.
Global functional connectivity changes after three months of treatment in all participants
Within the whole group, after three months of treatment with galcanezumab, decreased functional connectivity was found between the right lateral occipital cortex (inferior division) and the left precentral gyrus and bilateral postcentral gyrus. Significant increase in functional connectivity was not observed after treatment. Results are summarized in Table 2, Figure 1.
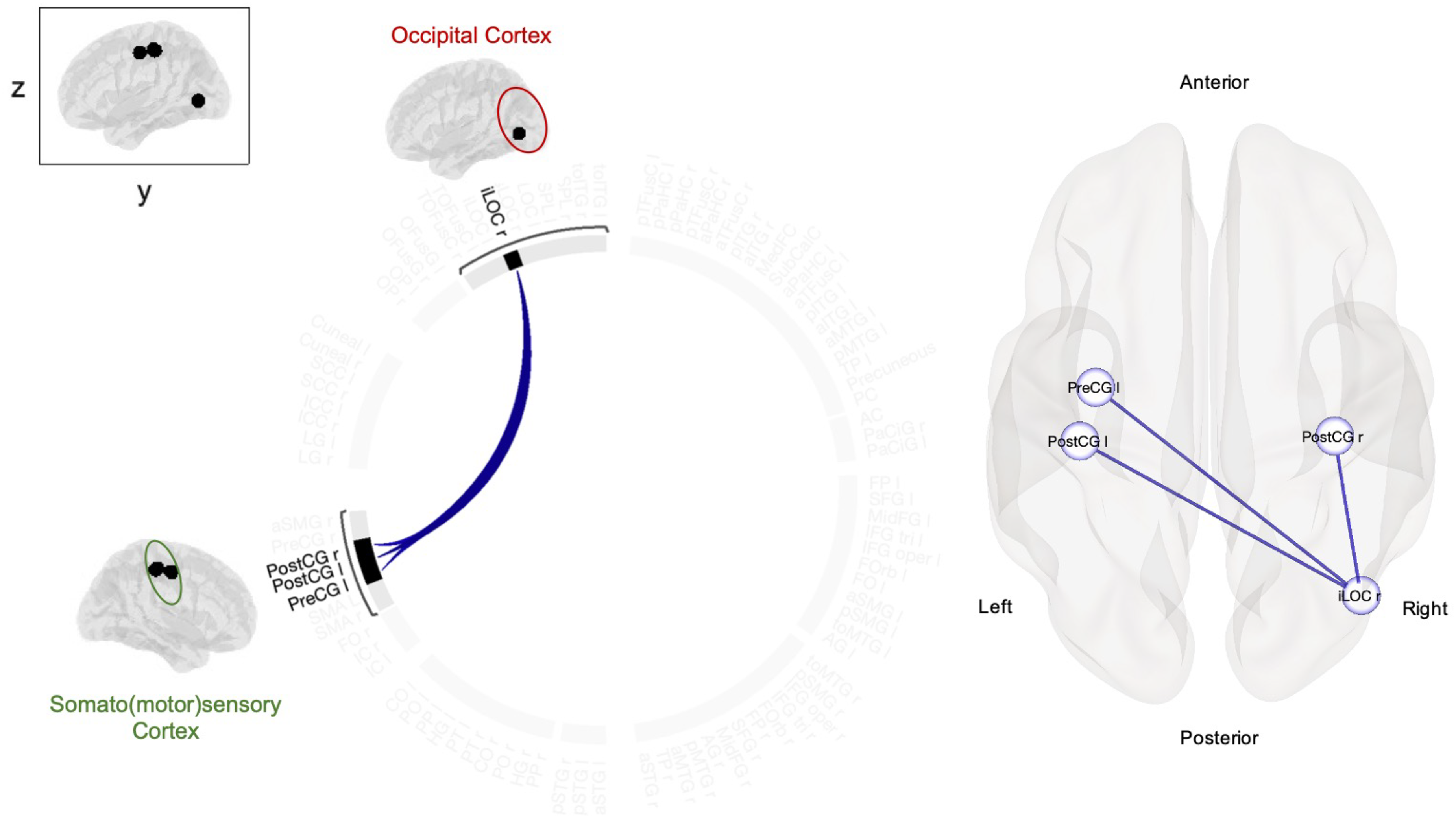
Changes in cortical functional connectivity after 3-month treatment with galcanezumab in all participants (N = 36). Using ROI-to-ROI approach, attenuated functional connectivity was found between the right lateral occipital cortex (inferior division) and regions corresponding to the primary somatosensory and motor cortices (bilateral postcentral gyrus and left precentral gyrus). The same findings are visualized as a connectome ring (left), and in MNI space (right). Results are corrected for multiple comparisons and reported at pFDR < 0.05. Lines between the regions indicate negative/decreased functional connections. Note. R, Right; L Left; MNI, Montreal Neurological Institute; ROI, region-of-interest. Regions: iLOC, Lateral Occipital Cortex, inferior division; PreCG, Precentral Gyrus; PostCG, Postcentral Gyrus.
Decreased resting-state functional connectivity between regions after 3-month treatment with galcanezumab in all participants (ROI-to-ROI).

Note. R, right; L, left; FDR, false discovery rate; ROI, region-of-interest. Results were corrected for multiple comparisons at pFDR < 0.05.
Differences in functional connectivity at baseline and after three months of treatment in the responders and non-responders
At baseline, no significant differences were found in functional connectivity between the responder and non-responder groups.
After three months of treatment, in the responder group, ROI-to-ROI analysis revealed decreased functional connectivity within three clusters, mainly between the visual (occipital and temporal lobes), primary somatosensory and motor cortical regions. More specifically, significantly decreased functional connectivity was found between the following regions: [Cluster 1]: 1) between the right inferior temporal gyrus (temporooccipital part) and the right lateral occipital cortex (superior and inferior division) and right occipital fusiform gyrus; 2) between the left temporal fusiform cortex (posterior division) and the bilateral lateral occipital cortex (inferior division); 3) between the bilateral parahippocampal gyrus and the left lateral occipital cortex (inferior division); [Cluster 2]: 4) between the right lateral occipital cortex (inferior division) and the bilateral temporal occipital fusiform cortex; 5) between the right lateral occipital cortex (superior division) and the right lateral occipital cortex (inferior division); 6) between the left lateral occipital cortex (inferior division) and the right occipital pole and right temporal occipital fusiform cortex; [Cluster 3]: 7) between the right insular cortex and the left occipital pole; 8) between the left precentral gyrus and the bilateral temporal occipital fusiform cortex and right lateral occipital cortex (inferior division); 9) between the right precentral gyrus and the bilateral temporal occipital fusiform cortex; 10) between the right postcentral gyrus and the right lateral occipital cortex (inferior division); and 11) between the right precentral gyrus and the left lateral occipital cortex (inferior division). Significant increase in functional connectivity was not found after treatment. Results are summarized in Table 3, Figure 2, and Figure 3. In the non-responder group, no results survived the correction for multiple comparisons.

Changes in cortical functional connectivity after 3-month treatment with galcanezumab in responders (n = 19). Using ROI-to-ROI approach, responders to treatment showed attenuated functional connectivity between regions corresponding to the visual (occipital and temporal lobes), primary somatosensory, motor, and insular cortices. The same findings are visualized as a connectome ring (left), and in MNI space (right). Results are corrected for multiple comparisons and reported at pFDR < 0.05. Lines between the regions indicate negative/decreased functional connections. Different shades of connections represent T-values (see Table 3), and darker shade indicates lower connections. Note. R, Right; L Left; MNI, Montreal Neurological Institute; ROI, region-of-interest.
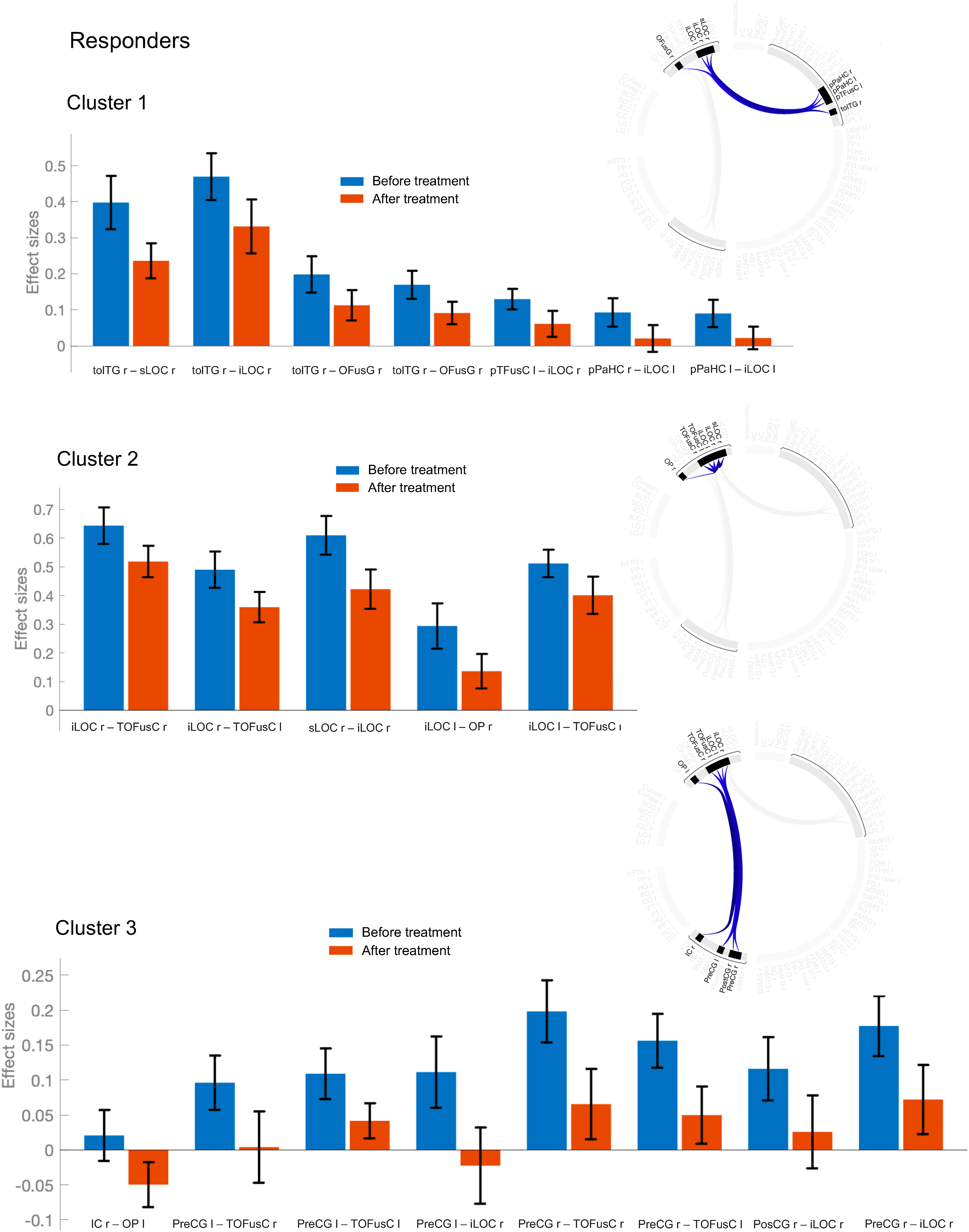
Mean functional connectivity values before and after 3-month treatment with galcanezumab in responders (n = 19). Bar graphs indicate mean connectivity values (Fisher's Z-transformed correlation coefficients) and 90% confidence intervals for the ROI-to-ROI pairings within the 3 significant clusters (clusters are visualized as connectome rings on the right). Note. R, Right; L Left; ROI, region-of-interest.
Decreased resting-state functional connectivity between regions in treatment responders after 3-month treatment with galcanezumab (ROI-to-ROI).

Note. R, right; L, left; FDR, false discovery rate; ROI, region-of-interest. Results are corrected for multiple comparisons at pFDR < 0.05.
Discussion
Present findings
We report that three months after treatment initiation, galcanezumab attenuated resting-state functional connections between regions of the occipital and somato(motor)sensory cortices (including the precentral and postcentral gyri) in treatment responders with high-frequency episodic or chronic migraine. In addition to the somato(motor)sensory cortices, reduced connectivity was found between the insula and several occipital and temporo-occipital regions as well as reduction in the intra-connectivity within the visual network. In contrast, in the non-responders, no significant changes were seen. Since galcanezumab likely prevents the activation of meningeal nociceptors, we conclude that a reduction in the magnitude of nociceptive signals that originate in the dura is sufficient to allow the cortex to return to a more normal and less excitable state. Indeed, attenuations in functional connections were seen between cortical regions where existing data report hyperexcitability in migraine patients. This may explain photophobia, allodynia, disease chronification, and shape pain perception and subjective headache experience in patients.
Functional connectivity attenuation in response to treatment: Pain and visual processing, multisensory integration
According to most electrophysiological and functional MRI studies, patients with migraine show greater activity in both the somatosensory and visual cortices (6,9,48,50–). Previous studies revealed that in migraine patients, the visual cortex is hyperresponsive to light exposure, and that this hyperresponsiveness is further intensified by simultaneous activation of pain fibers in the ophthalmic division of the trigeminal nerve (51,52). These observations have raised the possibility that the migraine brain may be modulated by somatosensory and visual stimuli, and potentially auditory and olfactory (4,53). Consequently, it may not be surprising that attenuation of functional connections between areas of the cortex that process the perception of pain/headache and vision was found only in the treatment responders.
Although less commonly discussed, the insular cortex functions as multi-modal sensory integrator for processing of visual input and generation of visual perception (54,–57). Specifically, interoceptive insular processing (that is, processing of signals from the body to understand its internal state) is capable of impacting visual consciousness and awareness (58,–60). Thus, it is reasonable to propose that the multimodal sensory integration process may be dysfunctional in migraine patients (61,–64) – to the extent that it heightens excitability and leads to chronic sensory amplification in the so-called ‘migraine brain’ (65). Functional connectivity depends on the excitability state of different brain areas (i.e., normal, sensitized or decreased state) and the synchrony between them (66). Decreased connectivity implies that the activity (Blood-Oxygen-Level Dependent [BOLD] signal) is less correlated (anti-correlated) between these cortical regions (66,67). This could possibly suggest a less excitable state after treatment in a sense that these regions became less synchronized (compared to the baseline) in the treatment responders. Hence, a reduction in functional connectivity may indicate a return to a more normal brain state, where the overcommunication is stabilized (i.e., regions are not simultaneously activated or deactivated) or, alternatively, reflect a new, more efficient way of processing, where certain brain regions (visual, sensory and motor cortices) are less reliant on strong functional relationships with one another, and act more independently. This assumption is also based on studies showing association between functional connectivity and neuronal excitability in different pain, neurological and psychiatric conditions, including fibromyalgia, epilepsy, autism, and depression (68,–71) where a decrease in functional connectivity was accompanied by a significant improvement. Lastly and importantly, it should be noted that this interpretation mainly relies on the crucial assumption that the cortex is hyperexcitable in migraine patients (7,8,72,74–), and with our approach, we could only elaborate on a potential decrease in hyperconnectivity during rest.
Modulation of cortical functions and connections by galcanezumab
The effect of galcanezumab on occipital cortex activity/responsivity to visual/photic stimulation has been recently reported in people with migraine (75,76). Interestingly, these studies showed that a three-month treatment with galcanezumab restored dysfunctional occipital reactivity to repetitive visual stimulation (black and white checkerboard patterns). More recently, we showed that treatment with galcanezumab reduced (a) cortical thickness in the somato(motor)sensory cortex of treatment responders but not non-responders (26), and (b) the incidence of headache following the occurrence of some premonitory symptoms, aura and exposure to triggers in responders (23). Mechanistically, we proposed that the observed cortical thinning was driven by reduced dendritic complexity (and an associated decrease in excitatory synapses) (3) and shrinkage of cortical neurons upon their return to a more normal (or lower) level of excitability and responsivity (77) following the selective inhibition of meningeal nociceptors by galcanezumab. Notably, both our present and previous MRI findings presented changes in regions of the primary somatosensory and motor cortices (i.e., precentral and postcentral gyri). This overlap between decreased cortical thickness and decreased functional connections could highlight the role of the somatomotor cortex in treatment responsivity to anti-CGRP mAbs. These findings also support the view that reduction of nociceptive signals from the meninges to the brain (78,79) is likely to decrease excitability and enhance sensory processing and sensorimotor integration in the brain (3,61).
The current findings that, after three months of treatment with galcanezumab, altered functional connectivity was observed selectively and preferentially between regions of the primary somatosensory, motor, and visual cortices (and visual-temporal cortex), raise the possibility that the interaction between other cortical areas known to be affected by migraine may require a much longer time (12 months of treatment) to ‘recover’ from their hyperexcitable state than the connections between the somatosensory, motor, and visual cortices. It is also important to note that thalamic trigeminovascular neurons (neurons that receive and process nociceptive signals from the meninges) project directly to the visual, sensory, and motor cortices (80,–82), and that activation in the trigeminal-thalamic-cortical pathway could be inhibited via blockade with galcanezumab (83). Another possibility is the previously demonstrated particularly high impact of galcanezumab treatment on two main domains: the sensory perception of headache and photophobia (23). In the responders, we reported that the number of migraine days per month decreased from a median value of 46.9% at baseline, to 20.9% after one month of treatment, 16.1% after two months, and 17.3% after three months, and that the incidence of photophobia decreased from a median value of 64.2% at baseline, to 33.3% after one month, 42.8% after two months, and 0 after three months of treatment (23).
Of note, according to recent studies, the efficacy of erenumab was also associated with functional connectivity changes and improvement of visual regions in patients with episodic or chronic migraine (29,30). Additionally, when exposed to pain (using gaseous ammonia or thermal stimulation), these patients exhibited changes (related to the drug's impact on headache pain) in neural networks that are involved in pain modulation and perception, and migraine generation and chronification (30,–32). Further (task-based) fMRI studies are needed to explore how light- or pain-induced hyperexcitability in the visual and somatosensory cortices could change after treatment and affect brain connectivity by levering the psychophysiological interaction (PPI) method (84).
Caveats
Limitations of the current study included: (i) Duration of treatment: Functional changes were investigated three months after treatment initiation, and a much longer treatment period may be necessary to fully restore alterations in brain functions and to evaluate cortical recovery. (ii) Sample size: This fMRI study was based on a small cohort of responders and non-responders (although in line with other treatment-29,–32), and the results should be interpreted with caution. Our migraine sample was also heterogeneous, including patients with high-frequency episodic and chronic migraine, and with and without aura. Further studies with larger sample sizes and longer treatment periods are needed to confirm these results, and to investigate the potential differences between these subgroups. (iii) Sex differences in response to treatment: biological sex differences could not be investigated due to the sample size and the female predominance in the study (migraine predominantly affects women with a ratio of approximately 3:1) (85). (iv) Lack of healthy control group: Our study design was to evaluate the effects of one variable - drug treatment - in a blinded fashion, and we did not evaluate the difference between disease states. Inclusion of a healthy control group could serve as a baseline comparison for individuals with migraine and would contribute to a better understanding of the posttreatment changes compared to the baseline. (v) Correlations with clinical characteristics: Associations between the fMRI findings and changes in clinical characteristics of patients were not evaluated since the current study design was not optimized to implement that. (vi) High-frequency episodic vs. chronic migraine: The present study included patients with high-frequency episodic or chronic migraine. Notably, studies have shown that the treatment needs for high-frequency episodic and chronic migraine seem to have similarities (86,87), and in our previous study, no baseline differences were found in cortical thickness between the two groups (26). However, more studies are needed considering that the 15-day threshold to distinguish them may not accurately represent the variations in migraine functional disabilities (88). (vii) Atlas based ROI-to-ROI analysis approach: The regions used in our connectivity analyses were from the Harvard-Oxford brain atlas (default atlas in CONN software), which is anatomically defined rather than functionally (89). By using this atlas, we were able to minimize between-atlas variability, but some of these regions are quite large and may have functionally defined subregions. As an example, the anterior insula or the face area of the somatosensory cortex are important regions in migraine, but they are not specified in this atlas. It is also plausible that studies using regions from different atlases (or manually defined ROIs) would show different results. (viii) Direction of the connections: Our findings present only correlated activities (i.e., BOLD signals) between different brain regions (66,67), and they do not provide information regarding the directions of the connections or whether they are unidirectional/bidirectional. Future studies should apply effective connectivity modelling.
Conclusion
Consistent with our previous findings on reduced cortical thickness in galcanezumab responders, in the current study, we report a widespread attenuation in functional connectivity between regions of multimodal reactivity, sensory integration, and pain modulations after three months of treatment. Based on these findings, we propose that the reduction in nociceptive signals that are likely to originate in the meninges is sufficient for resetting cortical (mainly visual and sensorimotor) excitability and functionality. Specifically, the observed changes could reflect reduced or desensitized co-activations as a result of decreased cortical responsivity, or reduction in the compensatory mechanisms needed to stabilize the level of cortical excitability before the treatment started.
Key findings
Functional connectivity changes were found after three months of treatment with galcanezumab among patients with high-frequency episodic or chronic migraine. In the treatment responders, reduced functional connectivity was observed between the somatosensory, motor, and insular cortices, and several visual regions after treatment. Our findings suggest that reduction in nociceptive signals allows the cortex to return to a more normal and less excitable state indicated by decreased synchrony/co-activation between these regions.
Footnotes
Data availability statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.A. received honoraria for consulting from Allergan/AbbVie, Amgen, Biohaven, Eli Lilly, Impel NeuroPharma, Novartis, Satsuma, Supernus, Theranica, Percept.
R.B. is the John Hedley-Whyte Professor of Anesthesia and Neuroscience at the Beth Israel Deaconess Medical Center and Harvard Medical School. He has received research support from the NIH: R01 NS094198-01A1, R37 NS079678, R01NS095655, R01 NS104296, R21 NS106345, Allergan/AbbVie, Teva, Dr Ready, Eli Lilly, Trigemina and the Migraine Research Foundation. He is a reviewer for NINDS, holds stock options in AllayLamp and Percept; serves as consultant, advisory board member, or has received honoraria from: Alder, AbbVie, Allergan, Amgen, Autonomic Technologies, Avanir, Biohaven, CGRP diagnostic, Dr Reddy's Laboratory, ElectroCore, Eli Lilly, GlaxoSmithKline, Merck, Pernix, Theranica, Teva, and Trigemina. CME fees from Healthlogix, Medlogix, WebMD/Medscape, and Patents 9061025, 11732265.1, 10806890, US2021-0015908, WO21007165, US2021-0128724, WO21005497. BIDMC owns patent US 10,766,952 B2. Other authors declare that they have no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Parts of this study were funded by Eli Lilly, the National Institutes of Health (R37 NS079678, RO1 NS094198, RO1 NS106345), and the Anesthesia Department at Beth Israel Deaconess Medical Center. Galcanezumab was provided by Eli Lilly.
