Abstract
Familial hemiplegic migraine type 1 (FHM-1) is a dominantly inherited subtype of migraine with aura and transient hemiplegia associated with mutations in the CACNA1A gene. FHM-1 shares many phenotypical similarities with common types of migraine, indicating common neurobiological pathways. Experimental studies have established that activation of the nitric oxide-cyclic guanosine monophosphate (NO-cGMP) pathway plays a crucial role in migraine pathophysiology. Therefore, we tested the hypothesis that CACNA1A mutations in patients with FHM-1 are associated with hypersensitivity to NO-cGMP pathway. We included eight FHM-1 patients with R583Q and C1369Y mutations and nine healthy controls, who received intravenous infusions of 0.5 μg kg−1 min−1 glyceryl trinitrate (GTN) over 20 min. We recorded: headache intensity on a verbal rating scale; mean flow velocity in the middle cerebral artery (VmeanMCA) by transcranial Doppler; diameter of the superficial temporal artery (STA) by Dermascan. One patient reported migraine without aura 5 h after start of the GTN infusion. No aura was reported. The AUCheadache in the immediate phase was more pronounced in patients than in controls (P = 0.01). In the 14 h following GTN infusion, there was no difference in the AUCheadache between patients and controls (P = 0.17). We found no difference in the AUCVmeanMCA (P = 0.12) or AUCSTA (P = 0.71) between FHM-1 patients and controls. None of the control persons reported migraine-like headache. FHM-1 patients do not show hypersensitivity of the NO-cGMP pathway, as characteristically seen in migraine patients with and without aura. This indicates that the pathophysiological pathways underlying migraine headache in FHM-1 may be different from the common types of migraine.
Introduction
Familial hemiplegic migraine (FHM) is a rare, dominantly inherited subtype of migraine with aura (1). FHM is phenotypically characterized by fully reversible half-sided weakness and other aura symptoms preceding or accompanying a migrainous headache (2, 3). FHM type 1 (FHM-1) is caused by missense mutations in the CACNA1A gene on chromosome 19p13, encoding the α1A subunit of calcium channels (4), FHM type 2 (FHM-2) is caused by mutations in the ATP1A2 gene encoding the α2 subunit of a Na+, K+ ATPase (5, 6) and FHM type 3 (FHM-3) is caused by mutations in the SCN1A gene encoding a neuronal voltage gated sodium channel (7). FHM-1 and FHM 2 are caused by several different mutations (8–16); in FHM-3, however, only two mutations have been described so far.
FHM-1 has many clinical similarities to migraine with (MA) and without aura (MoA) (17), and an epidemiological study of a population-based FHM cohort has shown that 65% of the FHM patients had MA and/or MoA (17, 18). However, it has been reported that MA and MoA are not associated with any of the known FHM mutations (19–22). Nevertheless, it could be expected that the clinical similarities between FHM, MA and MoA (17) would be caused by common neurobiological pathways underlying pathophysiological mechanisms.
The nitric oxide–cyclic guanosine monophosphate (NO–cGMP) pathway plays a fundamental role in migraine pathophysiology (23–25), and the glyceryl trinitrate (GTN) model of migraine has become established as a very robust and reproducible way of triggering typical attacks indistinguishable from the patient's usual attacks (24, 26, 27). This hypersensitivity to NO might be a shared feature of both MoA/MA and FHM, and could be the basis of the frequent co-occurrence of these disorders.
The aim of the present study was to test the hypothesis that CACNA1A mutations in a genotyped group of FHM-1 patients are associated with hypersensitivity to NO, and that FHM-1 would thus share a disturbance of the NO–cGMP pathway with MA and MoA.
Methods
Eight FHM-1 patients with R583Q and C1369Y mutations [2M/6F, mean age 40 years (range 27–57 years)] (Table 1) and nine healthy controls [5M/4F, mean age 33 years (range 24–49 years)] were recruited. Inclusion criteria for the patients were a diagnosis of FHM-1, with a known mutation in the CACNA1A gene according to the criteria of the International Headache Society (IHS) (3). The patients were recruited from a Danish population-based sample of FHM patients earlier reported (28, 29). This cohort consisted of 20 FHM-1 patients with known mutations. All patients were contacted and asked to participate in the study. Ten out of 20 patients declined participation for unspecified reasons, and two of the remaining 10 patients were not eligible for participation because of known cerebrovascular or cardiovascular disease. Thus, we were able to recruit eight out of 20 patients (40%) from the Danish population-based cohort. The most frequent CACNA1A mutation (T666M) (2, 30, 31) was not present in any of the participating patients, but R583Q, the second most frequent mutation (10, 32) and the most prevalent Danish FHM-1 mutation (29) was represented.
Clinical characteristics of eight FHM-1 patients with R583Q and C1369Y mutations
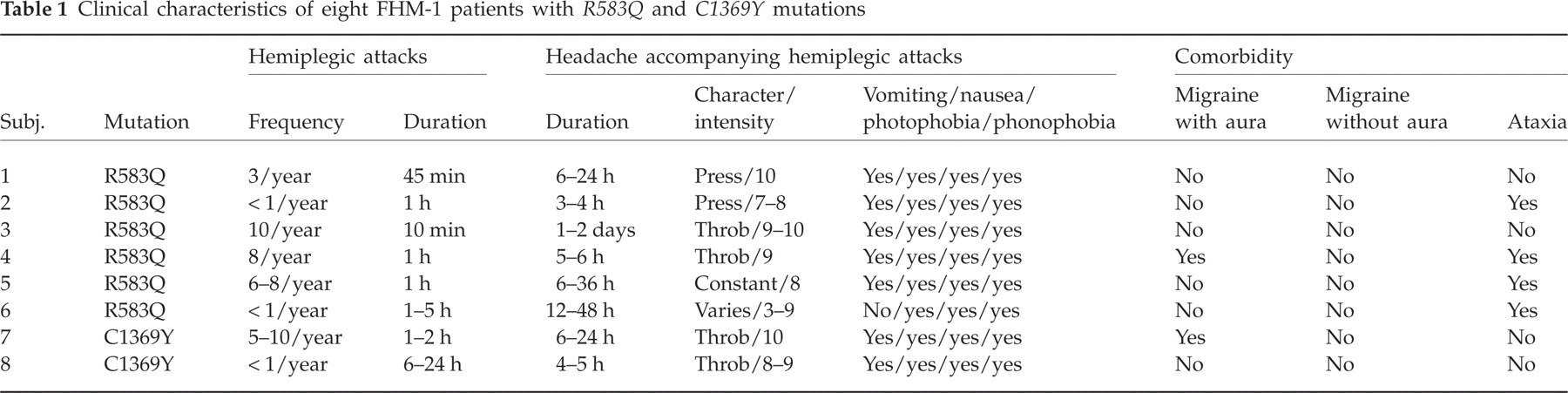
Exclusion criteria for the patients were: any daily medication apart from oral contraceptives; serious somatic or psychiatric diseases. The control healthy subjects did not have a history of migraine or any other type of headache (except episodic tension-type headache for < 1 month). The study was approved by the Ethics Committee of the County of Copenhagen (KA 04088) and was undertaken in accordance with the Helsinki Declaration of 1964, as revised in Edinburgh in 2000. All subjects gave informed consent to participate. The study was registered at ClinicalTrials.gov (åØåNCT00257985).
Experimental design
The study design was balanced and single-blinded. The laboratoty technicians performing the measurements were blinded with respect to patients and controls. All subjects received a continuous intravenous infusion of 0.5 μg kg−1 min−1 GTN over 20 min. The subjects were informed that GTN might induce headache in some individuals, but the timing or the type of headache were not discussed.
All subjects reported to the laboratory at 08.00 h headache-free. The intake of coffee, tea, cocoa or other methylxanthine-containing foods or beverages was not allowed for the last 8 h before the start of the study, to avoid a possible effect on cerebral blood flow. All procedures were performed in a quiet room at a temperature of 25°C. The subjects were placed in the supine position, and a venous catheter (Venflon®) was inserted into an antecubital vein. The participant then rested for 30 min before baseline measurements of blood pressure, heart rate and ECG were done and the infusion started, using a time- and volume-controlled infusion pump (Braun Perfusor, Melsungen, Germany). Headache intensity, middle cerebral artery mean blood flow velocity (VmeanMCA), superficial temporal artery diameter, end-tidal partial pressure of CO2 (PetCO2), adverse events and vital signs were recorded at T −10, and then every 10 min until 120 min after start of infusion. The subjects were discharged from the hospital after finishing the measurements and were asked to complete a headache diary every hour until 12 h after discharge. The diary included headache characteristics and accompanying symptoms according to the IHS (3), any rescue medication taken and adverse events. Subjects were allowed to take rescue medication of their own choice at any time.
Headache intensity
Headache intensity was recorded on a verbal rating scale (VRS) from 0 to 10 [0, no headache; 1, a very mild headache (including a feeling of pressing or throbbing); 5, moderate headache; 10, worst imaginable headache] (24).
Cerebral haemodynamics
The mean velocity of blood flow in the middle cerebral artery (VmeanMCA) was recorded bilaterally by transcranial Doppler (TCD) with hand-held 2-MHz probes (Multidop X; DWL, Sipplingen, Germany), as previously described (33). All recordings were done by the same skilled technician (L.E.). To correct the VMCA measurements for significant changes in PetCO2, changes were recorded in PetCO2 simultaneously with the TCD measurements using an open mask that caused no respiratory resistance (ProPac Encore®; Welch Allyn Protocol, Beaverton, OR, USA) (33).
Diameter of the superficial temporal artery
The diameter of the frontal branch of the superficial temporal artery (STA) was measured by a high-resolution ultrasonography unit (Dermascan C; Cortex Technology, Hadsund, Denmark; 20 MHz, bandwidth 15 MHz) as previously described (34).
Vital signs
Heart rate and blood pressure were measured every 10 min using an auto-inflatable cuff (ProPac Encore®; Welch Allyn Protocol). ECG (Cardiofax V; Nihon-Kohden, Shinjuku-ku, Tokyo, Japan) was monitored on an LCD screen and recorded on paper every 10 min.
Data analysis and statistical methods
All values are presented as mean ±
Calculation of sample size was based on the detection of a difference between the proportion of patients and controls reporting GTN-induced migraine attack during delayed phase (2–14 h), at 5% significance with 80% power. We assumed that GTN would induce a migraine headache in approximately 80% of FHM-1 patients, as reported previously in common types of migraine (24, 27) and migraine-like headache in < 10% of healthy controls (35, 36). We estimated that eight subjects should be included in each group (37), but it was planned to increase the sample size, in case more patients could be recruited.
The area under the curve (AUC) was used as summary measure for analysing differences between the groups [headache score, VmeanMCA, diameter of STA, mean blood pressure (MAP), heart rate (HR) and PetCO2]. The AUC was calculated according to the trapezium rule (38).
The primary end-points were differences in incidence of migraine headache and AUC for headache score (AUCheadache 0−120 min and AUCheadache 2 h−14 h), VmeanMCA (AUCVmeanMCA), STA (AUCSTA) and PetCO2 (AUCPetCO2) between groups. The secondary end-points were differences in the AUC for heart rate (AUCheart rate) and MAP (AUCMAP) between groups during the immediate phase and differences between baseline and peak responses (VmeanMCA, diameter of the STA and heart rate) within groups and between groups at time of peak response (headache, VmeanMCA, diameter of the STA and heart rate).
Statistical analysis was performed using an unpaired, two-way t-test except headache scores, where data are presented as medians and quartiles and tested with the Mann–Whitney test. The incidence of migraine and other adverse events between the groups was compared with Fisher's exact test.
All analyses were performed with SPSS for Windows 14.0 (SPSS Inc., Chicago, IL, USA). Five percent (P < 0.05) was chosen as the level of significance.
Results
All 17 subjects completed the study, and all subjects were headache free at baseline. The VmeanMCA recordings showed no differences between the sides at baseline (P > 0.05). Therefore, VmeanMCA of the right and left side were grouped and the average of the two was calculated. There were no differences between the groups at baseline for any other variables (Table 2).
Baseline values (±

Groups compared with an unpaired t-test.
In one control subject we were only able to find a reliable Doppler signal from the MCA on one side of the head. There was one missing value for the heart rate measurements in one control subject (50 min) and three CO2 recordings were missing from two subjects in the control group (−10, 0 and 50 min).
Aura
The GTN infusion did not induce an aura in any of the patients.
Headache
One patient (patient 4, Table 1), but no controls reported a delayed headache fulfilling the criteria for MoA according to the IHS criteria (3). This patient reported peak headache intensity (5 on the VRS) at 7 h after start of the infusion. The headache was described as a bifrontal and retrobulbar pressing pain, associated with aggravation during physical activity, mild nausea and photophobia. The patient reported that the headache was similar to the usual migraine headache.
This patient suffered from migraine with typical aura attacks in addition to hemiplegic attacks. The incidence of reported migraine was not different in the two groups, with 12.5% (one out of eight) in the patient group, and 0% (0 out of nine) in the control group [95% confidence interval (CI) −0.11, 0.36; P = 0.47].
During the immediate phase (0–120 min) seven patients and three control subjects reported headache (Table 3). The AUCheadache 0−120 min in the patient group, 50 (28.7, 57.5), was significantly greater than in the control group, 0 (0, 20; P = 0.008). The peak headache occurred at 20 min, and the median peak headache in the patient group, 1.5 (0.3, 2), was higher than in the control group, 0 (0, 1; P = 0.046) (Fig. 1).
Number of patients and controls reporting headache and migraine headache

Groups compared with Fisher's exact test.
ICHD, International Classification of Headache Disorders.
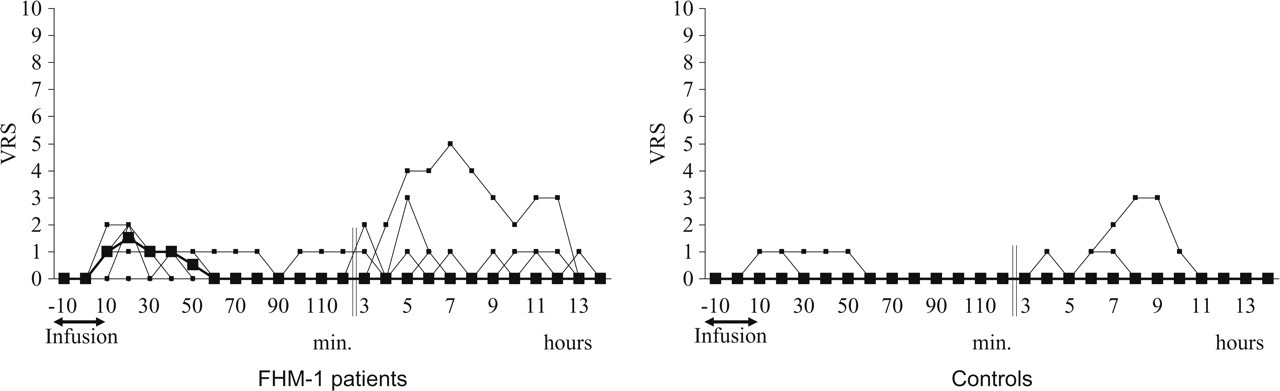
Individual and median headache scores on a verbal rating scale (VRS) during immediate (0–120 min) and delayed phases (2–14 h) after start of the glyceryl trinitrate (GTN) infusion in eight patients with familial hemiplegic migraine type 1 (FHM-1) and nine controls. There were significantly higher pain responses during the immediate phase in the patient group compared with the control group (P = 0.008). There were no differences between patients and controls in the 14 h following the GTN infusion (P = 0.167). Thick lines in figure are median pain scores.
During the delayed phase (2–14 h), five patients and two controls reported headache (Table 3), in all cases without any associated symptoms such as nausea, photophobia or phonophobia. We found no difference in the AUCheadache 2 h-14 h between FHM-1 patients, 2 (0, 5.4) and controls, 0 (0, 0; P = 0.167) (Fig. 2). The median headache was 0 for all time points in the 2–14 h period.

Individual and mean flow velocities in the middle cerebral arteries (VMCA) assessed by transcranial Doppler ultrasonography. There were no differences between patients and controls following glyceryl trinitrate (GTN) infusion during the immediate phase (0–120 min) (P = 0.115). Thick lines in figure mean values.
Middle cerebral artery mean blood flow velocity
There was no difference in the AUCVmeanMCA between FHM-1 patients (7617 ± 1001) and controls (8281 ± 969) during the immediate phase (P = 0.115) (Fig. 3). The mean peak decrease in VmeanMCA compared with baseline occurred at 20 min, and was −21.1 ± 9% in the patient group and −20.0 ± 10% in the control group. The mean difference between patients and controls at 20 min was −1.1% (95% CI −7.6, 5.4; P = 0.74). There were no differences in the PetCO2 recordings (0–120 min) during TCD scans between FHM patients and controls (P = 0.37).

Individual and mean diameters of the superficial temporal artery (STA) assessed by high-resolution ultrasonography. During immediate phase (AUC 0−120min) measurements of the diameter of STA showed no difference between patients (red) and controls (blue) (P = 0.71). Thick lines in figure show mean values.
Superficial temporal artery
There was no difference in the AUCSTA between FHM-1 patients (159 ± 25) and controls (165 ± 39) during the immediate phase (P = 0.71) (Fig. 4). The peak increase in the STA diameter compared with baseline occurred at 10 min, and was 45 ± 17% in the patient group and 52 ± 22% in the control group. The mean difference in response between patients and controls at 10 min was −7.1% (95% CI −27, 13; P = 0.46).

Mean percent change from baseline in mean arterial blood pressure (MAP) (squares) and heart rate (HR) (triangles) during and after infusion of glyceryl trinitrate (GTN) (patients, filled; controls, blank). There were no differences in mean arterial blood pressure (AUC 0−120min, P = 0.76) or HR (AUCHR 0−120min (P = 0.11) between patients and controls. Peak decrease in MAP occurred 20 min after start of the infusion and the mean was −13.1 ± 5.1% in patients and −14.8 ± 2.8% in controls, compared with baseline. There was a significant difference in HR between baseline and the peak response at 20 min in both familial hemiplegic migraine (FHM) patients (P = 0.011) and controls (P = 0.017). The mean increase in HR at 20 min was 9.9 ± 8.2% in the patient group and 11.8 ± 13.5% in the control group (P = 0.74).
Mean arterial blood pressure and heart rate
We found no difference in the AUCMAP (P = 0.76) or AUCHR 0−120min (P = 0.11) between patients and controls during the immediate phase. There was a significant increase in HR between baseline and the peak response at 20 min in FHM patients (P = 0.011) and at 10 min in controls (P = 0.017).
Adverse events
During the immediate period (0–120 min), two patients and 0 controls reported flushing (P = 0.20); one patient and five controls reported palpitations (P = 0.13); and one control reported heat sensation (P = 1.0).
Discussion
This is the first study using the GTN migraine provocation model to explore the functional consequences of the R583Q and C1369Y gene mutations in FHM-1 patients. The major finding of the present study was that activation of the NO–cGMP pathway failed to induce migraine aura or migraine headache in patients with FHM-1. This finding is in sharp contrast to results in migraine patients with and without aura, where GTN induces migraine in 50–80% of patients (26, 27, 36).
GTN failed to induce a migraine aura
Cortical spreading depression (CSD), discovered by Leão (39), has been linked to migraine aura pathogenesis in observational (40), animal (41) and human studies (42–44). Cerebral blood flow (CBF) studies during hemiplegic aura showed a spreading cortical hypoperfusion (45), similar to MA (46), which suggests that CSD is the most likely mechanism of hemiplegic aura (47, 48). It still remains unresolved, why migraine patients are more susceptible to CSD. In the case of FHM, animal studies of CACNA1A knock-in mice carrying the human R192Q mutation showed increased susceptibility to CSD (49). It has been proposed that CACNA1A mutations lead to increased release of glutamate, and thus increased susceptibility to CSD (50).
The molecular mechanisms of the initiation and propagation of CSD are not fully understood. However, animal studies have reported that CSD is associated with the release of NO (51, 52), and NO has also been linked to the modulation of the calcium entry through P/Q type calcium channels (53), and the transduction between neuronal activity and increased CBF after CSD (54). Furthermore, Read and colleagues have showed that GTN stimulates the release of NO in response to CSD (52) and that CSD increases the levels of cGMP in the cortex and brainstem (55). These findings suggest that the NO–cGMP pathway could be importantly involved in the pathogenesis of migraine aura. The i.v. GTN model was used by Christiansen et al. (36) in a study attempting to trigger migraine aura in 12 patients with pure MA, i.e. without any coexisting MoA. The study showed that 50% of the patients developed migraine headache, but none developed migraine aura. In another study of 21 patients with MA, i.v. GTN induced reproducible aura in one patient (27), and in a study of 22 patients, sublingually applied GTN induced aura in three patients (56).
Collectively, these data suggest that GTN may be able to induce aura in few migraine patients, i.e. with a relatively low rate of aura induction. The genotype of FHM-1 may be associated with a decreased CSD threshold, and it might therefore be expected that GTN would be able to induce aura in some of the FHM-1 patients. GTN failed, however, to induce migraine aura in this population-based cohort of Danish FHM-1 patents with the R5583Q and C1369Y mutations. Thus, our data suggest that the NO–cGMP pathway is not involved in the pathogenesis of migraine aura in FHM-1 patients with the R583Q/C1369Y mutations.
GTN failed to induce migraine headache
Experimental studies in migraineurs have demonstrated that the NO–cGMP pathway plays an important role in triggering (23–25, 27, 36, 57) and maintaining (58) migraine headache. Interestingly, the study by Christiansen et al. (36) has shown that although GTN failed to induce aura, 50% of MA patients developed migraine headache. This indicates that the NO–cGMP neurobiological pathway is involved in triggering migraine headache in patients with MA. Since FHM-1 and MA patients share clinical features such as non-hemiplegic aura symptoms, a similar headache phase and similar associated symptoms (17), we hypothesized that GTN infusion would induce a migraine headache in most FHM-1 patients. The present study has shown, however, that GTN failed to trigger more migraine headache in FHM-1 patients than in healthy controls, and the reported pain intensity during the delayed phase was not different between the groups. This is in sharp contrast to earlier findings, where exogenous NO has been found to cause more episodes of migraine headache in migraine patients than in controls (26, 27), and also to induce a more pronounced headache intensity response in migraine patients than in healthy control subjects (23, 36). The present results therefore suggest that the R583Q/C1369Y mutations do not cause hypersensitivity to GTN and consequently seem to affect neurobiological pathways other than those in MA and MoA.
Two out of eight FHM-1 patients had both FHM-1 and MA and only one of these, with the R583Q mutation, developed delayed headache fulfilling the criteria for MoA. This is similar to the placebo rate of migraine induction in a study, where one out of 10 MoA patients developed a migraine attack after placebo (24). Interestingly, family members (n = 5) of this patient with the same mutation but without known coexisting common types of migraine did not develop migraine.
In MoA patients, the sensitivity to i.v. GTN seemed not to depend on the frequency of spontaneously occurring migraine attacks (59), whereas in a large study, applying GTN sublingually, increased sensitivity to NO has been linked to increased attack frequency in MoA patients (56). This study also examined MA patients, where no relationship was found between sensitivity to NO and frequency of attacks. Because FHM-1 is a subtype of MA, and the FHM-1 patient experiencing the migraine attack after GTN suffered from MA, it seems that the difference in attack frequency is unlikely to affect our conclusion.
In the light of these surprising findings, one might suggest that neurobiological pathways responsible for migraine headache in coexisting MA are distinct from pathways responsible for migraine headache in FHM-1 patients with the R583Q/C1369Y mutations.
In line with previous studies on migraine patients (23), the FHM-1 patients developed more immediate headache (0–2 h) than controls. Arterial dilation may cause headache (60), and GTN infusion causes a more pronounced dilation of extra- and intracerebral arteries in migraine patients than in controls (61). In contrast, we found no differences in VmeanMCA, or the diameter of the STA between FHM-1 and controls. This could indicate that FHM-1 may not share the arterial hypersensitivity to NO that has been suggested for MoA patients (61). It also shows that the difference in immediate headache between FHM-1 and controls is unlikely to be caused by vasodilation.
Surprisingly few controls developed headache, compared with our earlier studies using the NO–cGMP model. The incidence of immediate headache in the control group, however, is similar to a large study by Sances et al. (56). Kinetic studies in the R593Q mutation (62) have shown that FHM mutated human CaV2.1 channels display an increased open probability, thus allowing FHM-1 channels to carry larger Ca2+ fluxes than in the wild type (63). Animal studies on knock-out rats for the Cav2.1 calcium channel have indicated that the P/Q-type calcium channels may have a pronociceptive role in inflammatory and neuropathic pain states (64). Based on these data, it could plausibly be suggested that the more pronounced immediate headache in the FHM patients may be due to the pronociceptive effect of the gain-of-function phenotype known from the R539Q mutation.
Methodological considerations
Epidemiological studies have shown that FHM is a very rare disease, with a prevalence of approximately 0.005% (28). We were able to recruit eight out of 20 patients (40%) from the Danish population-based cohort. We applied a single blinded design to avoid the risk of losing patients to follow-up in case of crossover design. Based on previous GTN studies and our hypothesis that FHM-1, MA and MoA share the hypersensitivity towards NO, our power calculation showed that eight subjects in each group would be enough to show statistical difference with respect to reported migraine attacks. The study, however, showed a non-significant difference in migraine induction between the two groups of only 12%. This difference is hardly clinically relevant, but we cannot exclude a small effect.
The present study suggests that FHM-1 patients do not show hypersensitivity of the NO–cGMP pathway, as characteristically seen in MoA and MA. Furthermore, the present data indicate that pathophysiological pathways underlying migraine headache in FHM-1 may be different from the common types of migraine (MA and MoA). Our material does not allow a separate evaluation of each mutation. Further studies are warranted to examine this, and explore whether FHM-2 and FHM-3 also differ from the common types of migraine.
Footnotes
Acknowledgements
The authors thank all participating FHM-1 patients, and laboratory technicians Kirsten Brunsgaard and Lene Elkjær for their dedicated and excellent assistance. The authors wish to thank Associate Professor Lene Theil Skovgaard (Department of Biostatistics, University of Copenhagen) for statistical advice. The study was supported by the University of Copenhagen, the A.P. M⊘ller Foundation for advancement of medical science, the Cool Sorption Foundation, The Danish Medical Association Research Fund, Danish Headache Society, Ms Else Torp and Flemming Jensen Foundation, Ms Lily Benthine Lund Foundation, Jacob Madsen and Olga Madsen Foundation, deCODE genetics, The Lundbeck Foundation for Neurovascular Signalling (LUCENS) and the European Community [EUROHEAD (LSHM-CT-2004-504837)].
