Abstract
Familial hemiplegic migraine type 2 (FHM-2) and common types of migraine show phenotypic similarities which may indicate a common neurobiological background. The nitric oxide-cyclic guanosine monophosphate (NO-cGMP) pathway plays a crucial role in migraine pathophysiology. Therefore, we tested the hypothesis that ATP1A2 mutations in patients with FHM-2 are associated with hypersensitivity to NO-cGMP pathway. Eight FHM-2 patients with R202Q, R763C, V138A and L764P mutations and nine healthy controls received intravenous infusions of 0.5 μgkg−1 min−1 glyceryl trinitrate (GTN) over 20 min. We recorded the following variables: headache intensity on a verbal rating scale; mean flow velocity in the middle cerebral artery (VmeanMCA) by transcranial Doppler; diameter of the superficial temporal artery (STA) by ultrasound. The primary end-points were differences in incidence of migraine headache and area under the curve (AUC) for headache score during an immediate phase (0-120 min) and a delayed phase (2-14 h) after start of infusion. We found no difference in the incidence of reported migraine between FHM-2 patients, 25% (two out of eight), and controls, 0% (0 out of nine) (95% confidence interval −0.06, 0.56) (P = 0.21). The AUCheadache in the immediate (P = 0.37) and delayed (P = 0.09) phase was not different between patients and controls. The GTN infusion resulted in a biphasic response in patients. During the immediate phase, the median peak headache occurred at 30 min and tended to be higher in patients, 1 (0, 3.8), than in controls, 0 (0, 1) (P = 0.056). During the delayed phase, the median peak headache occurred 4 h after the start of the infusion and was significantly higher in patients, 2.5 (0, 3), than in controls, 0 (0, 0) (P = 0.046). We found no difference in the AUCVmeanMCA (P = 0.77) or AUCSTA (P = 0.53) between FHM-2 patients and controls. GTN infusion failed to induce more migraine in FHM-2 patients than in controls. The pathophysiological pathways underlying migraine headache in FHM-2 may be different from the common types of migraine.
Introduction
Familial hemiplegic migraine type 2 (FHM-2) is a rare, dominantly inherited subtype of migraine with aura (MA) (1). FHM-2 is associated with mutations in the ATP1A2 gene encoding the α2 subunit of a Na+, K+ ATPase (2, 3), and > 20 different FHM-2 mutations have been identified; for a review see (4). The identification of the mutated FHM genes (5–7) has lead to the assumption that migraine may be a channelopathy, and stimulated research in the link between genotype and phenotype (8).
Migraine attacks in FHM-2 are characterized by fully reversible half-sided weakness preceding or accompanying a migrainous headache (9). Phenotypically, there are many clinical similarities between FHM-2 and MA and migraine without aura (MoA) (9). In a population-based study of genotyped FHM patients, > 60% of the FHM-2 patients had one or two other forms of migraine attacks (10). Although MA and MoA are not associated with any of the known FHM mutations (11–14), FHM and the common types of migraine may share common neurobiological pathways underlying the pathogenesis of migraine.
Activation of the nitric oxide–cyclic guanosine monophosphate (NO–cGMP) pathways plays a fundamental role in migraine pathophysiology (15), as shown by administration of glyceryl trinitrate (GTN), which has been shown to trigger migraine attacks indistinguishable from the usual attacks in migraine patients (16–18). Recently, we examined a group of genotyped FHM-1 patients using the GTN model of migraine (19). We found that the R583Q and C1369Y mutations of the CACNA1A gene were not associated with hypersensitivity of the NO–cGMP pathway. Whether this pathway is a likely mechanism of FHM-2 remain unknown. Clinically, the migraine features of FHM-1 and FHM-2 are not different, but the mutated genes may lead to different functional consequences. It is therefore possible that the sensitivity to migraine trigger GTN may be different between patients with FHM-1 and FHM-2.
In the present study, we tested the hypothesis that ATP1A2 mutations in a genotyped group of FHM-2 patients might be associated with hypersensitivity to the NO–cGMP pathway.
Design and methods
Eight FHM-2 patients with R202Q, R763C, V138A and L764P mutations [5 M/3 F, mean age 45 years (range 19–59 years)] (Table 1) and nine healthy volunteers [5 M/4 F, mean age 33 years (range 24–49 years)] were recruited. The study was part of the EUROHEAD project (http://www.eurohead.org), and patients were recruited and examined in both Denmark and Italy, using the same methods. The measurements in Italy were conducted by an experienced member of the Danish research group (H.S.), to reduce interobserver variability.
Clinical characteristics of eight FHM-2 patients with R202Q, R763C, V138A and L764P mutations

FHM-2, familial hemiplegic migraine type 2.
We recruited seven of 13 patients (55%) with known ATP1A2 mutations from The Danish population-based cohort (10) and one patient from the Department of Neurology, Misericordia Hospital, Grosseto and University of Milan, Italy.
Inclusion criteria for the patients were a diagnosis of FHM-2 according to the criteria of the International Headache Society (IHS) (1), with a known mutation in the ATP1A2 gene. Exclusion criteria for the patients were: any daily medication apart from oral contraceptives; serious somatic or psychiatric diseases. The control healthy subjects did not have a history of migraine or any other type of headache (except episodic tension-type headache less than once a month). None of the healthy controls reported a family history of migraine.
The study was approved by the Ethics Committees of the County of Copenhagen (KA 04088) and County of Grosseto (731/CESF) and was undertaken in accordance with the Helsinki Declaration of 1964, as revised in Edinburgh in 2000. All subjects gave informed consent to participate in the study.
Experimental design
The study design was non-randomized, controlled and single-blinded. The laboratory technicians performing the measurements in Denmark were blinded with respect to the subject status as patient or control. All subjects received a continuous intravenous infusion of 0.5 μg kg−1 min−1 GTN over 20 min. All subjects were informed that GTN might induce headache in some individuals, but the timing and type of headache were not discussed.
All subjects reported headache-free to the laboratory. The intake of coffee, tea, cocoa or other methylxanthine-containing foods or beverages was not allowed for the last 8 h before the start of the study, to avoid a possible effect on the cerebral blood flow. All procedures were performed in a quiet room at a temperature of 25°C. The subjects were placed in the supine position, and a venous catheter (Venflon®) was inserted into an antecubital vein. The participant then rested for 30 min before baseline measurements of blood pressure, heart rate and electrocardiogram were done and the infusion started, using a time- and volume-controlled infusion pump (Braun Perfusor, Melsungen, Germany). Headache intensity, mean blood flow velocity of the middle cerebral artery (VmeanMCA), diameter of the superficial temporal artery, end-tidal partial pressure of CO2 (PetCO2), adverse events and vital signs were recorded at T 10, and then every 10 min until 120 min after start of infusion. The subjects were discharged from the hospital after finishing the measurements and were asked to complete a headache diary every hour until 12 h after the discharge. The diary included headache characteristics and accompanying symptoms according to the IHS (1), any rescue medication taken and adverse events. Subjects were allowed to take rescue medication of their own choice at any time.
Headache intensity
Headache intensity was recorded on a verbal rating scale (VRS) from 0 to 10 (0, no headache; 1, a very mild headache (including a feeling of pressing or throbbing); 5, moderate headache; 10, worst imaginable headache) (20).
Cerebral haemodynamics
The mean velocity of blood flow in the middle cerebral artery (VmeanMCA) was recorded bilaterally by transcranial Doppler (TCD) with hand-held 2-MHz probes (Multidop X; DWL, Sipplingen, Germany), as previously described (21). To correct VMCA measurements for possible significant changes in End-tidal PCO2 (PetCO2), we recorded changes in PetCO2 simultaneously with the TCD measurements using an open mask that caused no respiratory resistance (ProPac Encore®; Welch Allyn Protocol, Beaverton, OR, USA).
Diameter of the superficial temporal artery
The diameter of the frontal branch of the superficial temporal artery (STA) was measured by a high-resolution ultrasonography unit (Dermascan C; Cortex Technology, Hadsund, Denmark: 20 MHz, bandwidth 15 MHz), as previously described (22).
Vital signs
Heart rate and blood pressure were measured every 10 min using an auto-inflatable cuff (ProPac Encore®; Welch Allyn Protocol). ECG (Cardiofax V, Nihon-Kohden; Shinjuku-ku, Tokyo, Japan) was monitored on an LCD screen and recorded on paper every 10 min.
Data analysis and statistical methods
All values are presented as mean ±
Sample size calculation was based on the detection of a difference in proportion of patients and controls reporting GTN-induced migraine attack during the delayed phase (2–14 h), at 5% significance with 80% power. We assumed that GTN would induce a migraine headache in approximately 80% of FHM-2 patients, as reported previously in MA and migraine-like headache in < 10% of healthy controls (23). We therefore estimated that eight subjects should be included in each group (24).
The area under the curve (AUC) was used as summary measure for analysing differences between the groups [headache score, VmeanMCA, diameter of STA, mean blood pressure (MAP), heart rate (HR) and PetCO2]. The AUC was calculated according to the trapezium rule (25). The primary end-points were differences in incidence of migraine headache and in the AUC for headache score (AUCheadache 0−120 min and AUCheadache 2h−14h), VmeanMCA (AUCVmeanMCA), STA (AUCSTA) and PetCO2 (AUCPetCO2) between groups. The secondary end-points were differences in the AUC for heart rate (AUCheart rate) and MAP (AUCMAP) between groups during the immediate phase and differences between baseline and peak responses (VmeanMCA, diameter of the STA and heart rate) within groups and between groups at time of peak response (headache, VmeanMCA, diameter of the STA and heart rate).
Statistical analysis was performed using an unpaired, two-way t-test except for headache scores, where data are presented as medians and quartiles and tested with Mann–Whitney test. The prevalence of migraine and other adverse events between the groups was compared with Fisher's exact test. All analyses were performed with SPSS for Windows 14.0 (Chicago, IL, USA). Five per cent (P < 0.05) was chosen as the level of significance.
Results
All 17 subjects completed the study, and all subjects were headache-free at baseline (−10 min). The VmeanMCA recordings showed no differences between the sides at baseline (P > 0.05) and therefore the average of the two sides was used in the statistical calculations.
There was no difference between the groups at baseline for any other variables (Table 2).
Baseline values (mean ±

Groups compared with an unpaired t-test.
FHM-2, familial hemiplegic migraine type 2.
In one control subject we could find a reliable Doppler signal only from the MCA on one side of the head, which was then used in the calculations. There were 16 missing Doppler recordings in the patient group (n = 5) and two in the control group (n = 2), because of difficulties localizing the correct vessel. There were four missing recordings of the STA in one patient, mainly because of lack of time as this patient was examined in Italy without the help of the usual laboratory technicians. There was one missing value for the heart rate in the control group (50 min), and the CO2 recordings were not done in two patients because the mask caused discomfort.
Migraine attacks
No difference was found in the incidence of reported migraine between FHM-2 patients, 25% (2/8), and controls, 0% (0/9) [95% confidence interval (CI) −0.06, 0.56] (P = 0.21).
Patient 6 (Table 1) reported peak headache intensity in the immediate phase 20 min after start of the GTN infusion (4 on the VRS) and a peak headache intensity in the delayed phase 4 h after start of the infusion (3 on the VRS). The patient was not pain-free at any time after the infusion. The headache was described as a bilateral, throbbing pain, associated with aggravation by physical activity, mild nausea, phonophobia and photophobia, and thus fulfilled the IHS criteria during both the immediate and the delayed phase (1). No aura was reported by this patient.
Patient 7 (Table 1) reported to the laboratory headache-free and scored 0 on the VRS at baseline (−10 min). However, just before the start of the infusion (0 min), the patient reported a mild sensation of pressure in the head, but no headache (1 on the VRS). This patient reported peak headache intensity in the immediate phase 30 min after start of the GTN infusion (7 on the VRS), and a peak headache intensity in the delayed phase 12–14 h after start of the infusion (10 on the VRS). The patient was not pain-free at any time after the infusion. The headache was described as a bilateral, constant pain, but without any associated features in the immediate phase. During the delayed phase (7.2 h after start of infusion), the patient experienced a visual aura with fortification spectra (duration of 30 min), followed by hemiplegia of the left arm and leg (with a duration of approximately 20 min, starting in the arm and then the leg). Before and during the aura, the patient had bilateral constant headache associated with aggravation through physical activity, mild nausea, phonophobia and photophobia, and thus fulfilled the IHS criteria for hemiplegic migraine, apart from the fact that the headache was present before the onset of hemiplegic aura symptoms (1). The patient took 50 mg diclofenac and went to sleep for 3.5 h. The aura symptoms had vanished as the patient woke up again, but the headache had worsened (VRS 10), and the patient took Ibuprofen, 600 mg. The patient reported that the symptoms of the hemiplegic attack were similar to her usual attacks.
Non-migraine headache
During the immediate phase (0–120 min), four patients and three control subjects reported headache (Table 3). There was no difference in the AUCheadache 0−120 min between patients, 10 (0, 384) and controls, 0 (0, 20) (P = 0.37). The median peak headache occurred at 30 min and tended to be higher in patients, 1 (0, 3.8), than in controls, 0 (0, 1) (P = 0.056) (Fig. 1).
Number of patients and controls reporting headache and migraine headache

Groups compared with Fisher's exact test.
FHM-2, familial hemiplegic migraine type 2; ICHD, International Classification of Headache Disorders.

Headache scores on a verbal rating scale (VRS) during immediate (0–120 min) and delayed phases (2–14 h) after start of the glyceryl trinitrate (GTN) infusion in eight patients with familial hemiplegic migraine type 2 (FHM-2) and nine controls. There was no difference in the AUC between patients and controls during immediate (P = 0.37) and delayed phases (P = 0.09) following the GTN infusion. Thick lines in figure are median pain scores.
During the delayed phase (2–14 h), five patients and two controls reported headache (Table 3). No difference was found in the AUCheadache 2h−14h between FHM-2 patients, 6.5 (0, 12.3), and controls, 0 (0, 0) (P = 0.09) (Fig. 1). Median peak headache occurred 4 h after the start of the infusion and was significantly higher in patients, 2.5 (0, 3), than in controls, 0 (0, 0) (P = 0.046) (Fig. 1).
Middle cerebral artery mean blood flow velocity
There was no difference in the AUCVmeanMCA between FHM-2 patients and controls during the immediate phase (P = 0.77) (Fig. 2). The mean peak change in VmeanMCA compared with baseline occurred at 20 min, and was −22.0 ± 6.9% in the patient group and −20.0 ± 9.8% in the control group. The mean difference between patients and controls at 20 min was −2.0% (95% CI −8.5, 4.5) (P = 0.53). There was no difference in PetCO2 (0–120 min) between patients and controls (P = 0.51).

Mean flow velocities in the middle cerebral arteries (VMCA) measured by transcranial Doppler ultrasonography following glyceryl trinitrate (GTN) infusion. There were no differences between familial hemiplegic migraine type 2 (FHM-2) patients (filled squares) and controls (squares) following GTN infusion during the immediate phase (0–120 min) (P = 0.77). Thick lines in figure shows mean values.
Superficial temporal artery
There was no difference in the AUCSTA between FHM-2 patients and controls during the immediate phase (P = 0.53) (Fig. 3). The peak change in the STA diameter compared with baseline occurred at 20 min and was 32 ± 11% in the patient group and 52 ± 22% in the control group. The mean difference in response between patients and controls at 20 min was −19.5% (95% CI −37.4, −1.64) (P = 0.035).
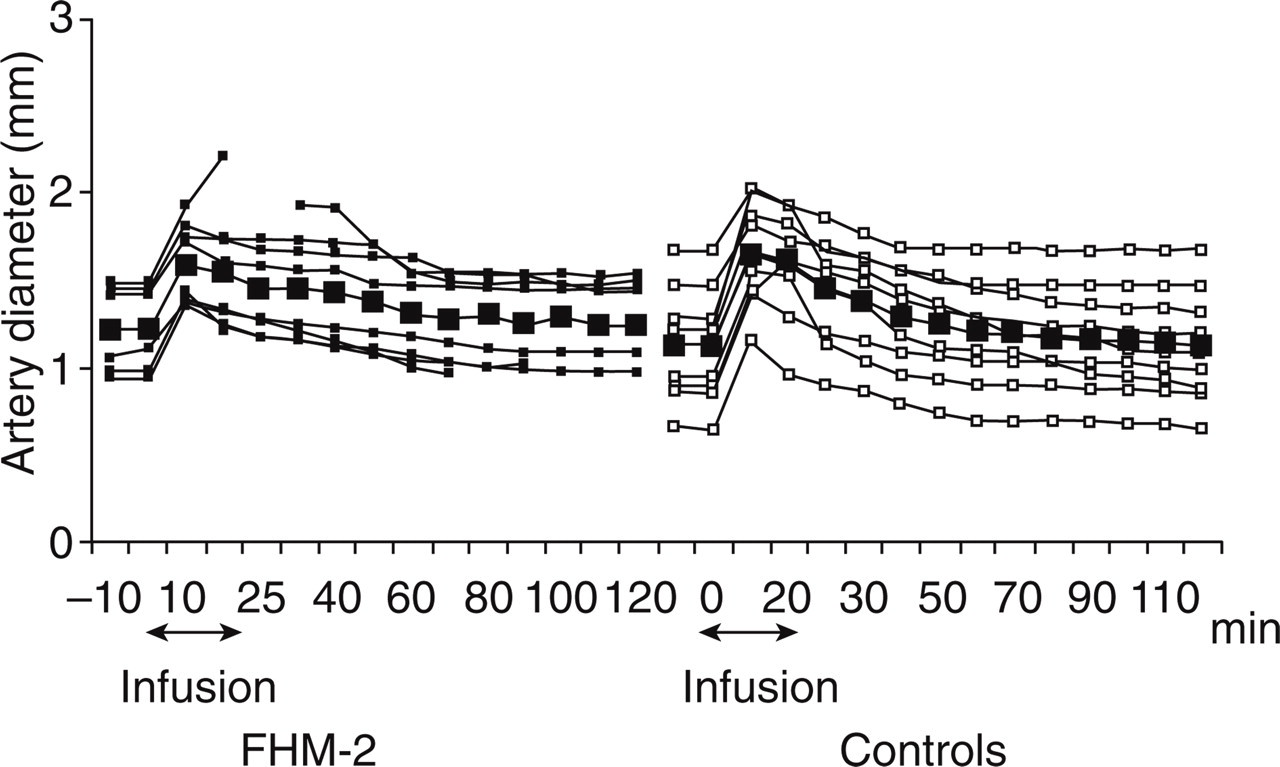
Diameter of the superficial temporal artery (STA) measured by high-resolution ultrasonography. During immediate phase (AUC0−120 min) there was no difference in the diameter of STA between familial hemiplegic migraine type 2 (FHM-2) patients (filled squares) and controls (squares) (P = 0.53). Thick lines in figure show mean values.
Mean arterial blood pressure and heart rate
We found no difference in the AUCMAP 0−120 min (P = 0.35) or AUCHR 0−120 min (P = 0.11) between patients and controls during the immediate phase (Fig. 4). There was a significant difference in heart rate between baseline and the peak response at 10 min in both FHM patients (P = 0.044) and controls (P = 0.025).

Change from baseline in mean arterial blood pressure (MAP) (squares) and heart rate (triangles) during and after infusion of glyceryl trinitrate (GTN) (patients, transparent; controls, filled). Mean arterial blood pressure did not differ between patients and controls (AUC0−120 min, P = 0.35). Peak decrease in MAP occurred 20 min after start of the infusion and the mean was −15.9 ± 6.8% in patients and −13.8 ± 3.9% in controls, and showed no difference between the groups (P = 0.52). There was no difference in AUCHR 0−120 min between familial hemiplegic migraine type 2 (FHM-2) patients and controls (P = 0.11). There was a significant difference in heart rate between baseline and the peak response at 10 min in both FHM patients (P = 0.044) and controls (P = 0.025). The mean increase in heart rate (HR) at 10 min was 9.5 ± 12.3% in the patient group and 15.5 ± 16.8% in the control group (P = 0.43).
Adverse events
During the immediate period (0–120 min), one patient and no controls reported flushing (P = 0.47); two patients and five controls reported palpitations (P = 0.33); and four patients and one control reported heat sensation (P = 0.13) (Fisher's exact test).
Discussion
This is the first study on FHM-2 patients using the GTN migraine provocation model to explore the functional consequences of the R202Q, R763C, V138A and L764P gene mutations in FHM-2 patients. The major finding of the present study is that activation of the NO–cGMP pathway failed to induce more migraine aura or migraine headache in FHM-2 patients than in healthy volunteers. This finding is in sharp contrast to previous findings in patients with common types of migraine. The question is how to reconcile this apparent reduced sensitivity to a known migraine trigger with the FHM-2 genotype?
A common denominator for all tested FHM-2 mutations is a slowed or reduced activity of the α2 Na+, K+-ATPase, which has been termed functional haploinsufficiency (3). Reduced activity of the Na+, K+-ATPase may reduce the gradients required to drive the astrocytic glutamate transporter (26), and haploinsuffiency of the α2 Na+, K+-ATPase may thus reduce the removal of glutamate and lower the threshold for cortical spreading depression (CSD) (27). Pharmacological inactivation of the Na+ K+-ATPase (similar to the effects of FHM-2 mutations) causes CSD-like depolarization (28), and stimulation of the NO–cGMP pathway has been shown to inhibit Na+K+-ATPase activity (29, 30). Experiments in rats have shown that CSD in both the cortex (31) and hippocampus (32) may activate the trigeminovascular system, thereby linking migraine aura and the triggering of migraine headache (33). Collectively, these data indicate that FHM-2 patients might show a reduced threshold for CSD and, thus, induction of aura and increased susceptibility to migraine headache.
The present data have shown, however, that GTN failed to induce more migraine aura in the FHM-2 patients than in healthy controls. This result corresponds to findings in patients with migraine with typical aura, where GTN rarely (18) or never (23) induces aura, and is in concordance with recent data from FHM-1 patients (19).
It is well established that MA and MoA patients share a common hypersensitivity to activation of the NO–cGMP pathway, because infusion of GTN causes migraine or migraine-like headache in about 50% of MA patients and up to 80% of MoA patients (15, 17, 18, 34). We would therefore expect the headache response after GTN in FHM-2 to be similar to these figures, because spontaneous migraine headache in FHM is similar to MA and MoA (1, 9).
Surprisingly, we found no differences in the prevalence of migraine attacks fulfilling the IHS criteria between FHM-2 and controls. GTN infusion induced migraine in only 25% of FHM-2 patients, which is much lower than that seen in both MoA and MA.
Six patients had migraine comorbidity of MoA (n = 2) and MA (n = 4). This co-occurrence could be a determinant for NO hypersensitivity, but to study this would require two groups of patients: one with known mutations and coexisting MA or MoA vs. ‘pure’ FHM-2 patients. Such a study would be highly relevant, but difficult to set up due to the rarity of these patients.
Based on the present results, we can not exclude a small hypersensitivity to NO in FHM-2 patients with co-occurring MoA and MA, but this effect is probably too small to be clinically relevant.
We found no differences in area under the headache curve and vascular variables between FHM-2 and controls. The FHM-2 group did, however, report a biphasic headache response after GTN infusion. Moreover, the median peak headache intensity was higher in patients than in controls during the immediate and the delayed phases. Although the delayed response has occasionally been reported in healthy volunteers after GTN (35), we can not rule out that FHM-2 patients may be more sensitive to the GTN provocation than healthy volunteers.
In the present study, one patient reported hemiplegic aura and migraine headache after GTN. This patient came twice to the laboratory for the experiments. At the first visit she was excluded because of tension-type like headache before the experiment. During the second visit she reported headache-free to the laboratory but complained of a pressing sensation in the head (1 on the VRS), just before infusion. This patient reported that she was going through a difficult period of her life (changing job) and was quite stressed. We can therefore not entirely rule out the possibility that the reported attack was in fact a spontaneous one. Another possibility is that this particular mutation is associated with hypersensitivity of the NO–cGMP pathway. To clarify whether the hemiplegic aura could be triggered again, we could have repeated the GTN infusion as previously described in migraine with typical aura (18), but decided against it because the strain of inducing a hemiplegic attack is considerable. The two patients reporting migraine (as well as a third patient not reporting migraine) took non-steroidal anti-inflammatory drugs as rescue medication for the headache. This treatment might affect the results by reducing the headache (36), but is unlikely to affect our results, as the majority of our patients did not need rescue medication.
The FHM-2 patients had very infrequent hemiplegic attacks (Table 1), but a large study, applying GTN sublingually, found no relationship between sensitivity to NO and frequency of MA attacks (34). FHM-2 is a subtype of MA (1), and the low attack incidence is therefore unlikely to have affected our results.
In summary, FHM-2 patients do not develop migraine attacks after GTN, but we can not rule out that FHM-2 patients are slightly more sensitive to GTN than healthy volunteers. Pathophysiological pathways underlying migraine headache in FHM-2 patients may thus be different from the pathways in patients with the common types of migraine.
Footnotes
Acknowledgements
The authors thank all participating FHM-2 patients, healthy volunteers, lab technicians Kirsten Brunsgaard and Lene Elkjær for their dedicated and excellent assistance, Henrik Schytz, MD for helping with the recordings and Massimo Alessandri, MD for useful comments. The study was supported by grants from the University of Copenhagen, the A.P. M⊘ller Foundation for Advancement of Medical Science, the Cool Sorption Foundation, The Danish Medical Association Research Fund, Danish Headache Society, Ms Else Torp and Flemming Jensen Foundation, Ms Lily Benthine Lund Foundation, Jacob Madsen and Olga Madsen Foundation, The Lundbeck Foundation through the Center for Neurovascular Signalling (LUCENS) and the European Community—EUROHEAD (LSHM-CT-2004-504837).
